Abstract
Renal cell carcinoma, the most common neoplasm of adult kidney, accounts for about 3% of adult malignancies and is usually highly resistant to conventional therapy. MicroRNAs are a class of small non-coding RNAs, which have been previously shown to promote malignant initiation and progression. In this study, we focused our attention on miR-21, a well described oncomiR commonly upregulated in cancer. Using a cohort of 99 primary renal cell carcinoma samples, we showed that miR-21 expression in cancer tissues was higher than in adjacent non-tumor tissues whereas no significant difference was observed with stages, grades, and metastatic outcome. In vitro, miR-21 was also overexpressed in renal carcinoma cell lines compared to HK-2 human proximal tubule epithelial cell line. Moreover, using Boyden chambers and western blot techniques, we also showed that miR-21 overexpression increased migratory, invasive, proliferative, and anti-apoptotic signaling pathways whereas opposite results were observed using an anti-miR-21-based silencing strategy. Finally, we assessed the role of miR-21 in mediating renal cell carcinoma chemoresistance and further showed that miR-21 silencing significantly (1) increased chemosensitivity of paclitaxel, 5-fluorouracil, oxaliplatin, and dovitinib; (2) decreased expression of multi-drug resistance genes; and (4) increased SLC22A1/OCT1, SLC22A2/OCT2, and SLC31A1/CTR1 platinum influx transporter expression. In conclusion, our results showed that miR-21 is a key actor of renal cancer progression and plays an important role in the resistance to chemotherapeutic drugs. In renal cell carcinoma, targeting miR-21 is a potential new therapeutic strategy to improve chemotherapy efficacy and consequently patient outcome.
Introduction
Renal cell carcinoma (RCC) corresponds to 3% of all adult malignant tumors. Clear RCC (cRCC) is the main histological subtype and accounts for 70%–75% of all RCC cases. 1 cRCCs are highly resistant to conventional cytotoxic, radiation, and hormone therapies, and nephrectomy is the only therapeutic option to cure early and local cRCCs. At diagnosis, 20%–30% of RCC patients have metastastic disease and one third develop metastasis after surgery. Targeted therapies are first-line treatments for advanced RCC with limited success, as the median survival of patients is only 1.5 years. 2 Hence, deciphering molecular mechanisms underlying renal tumor progression and chemoresistance is urgently needed to develop new therapeutic strategies to cure cRCC.
MicroRNAs (miRNAs) are a class of non-coding small RNAs of approximatively 22 nucleotides in length. They usually bind to the 3′ untranslated region (3′-UTR) of target gene messenger RNAs (mRNAs) and thereby repress their translation and/or induce their degradation. MiRNAs are major regulators of gene expression and control numerous biological processes such as differentiation, proliferation, and apoptosis. 3 Specific miRNA expression patterns have been observed in the initiation and progression of various diseases including cancer. Accumulating evidence suggests that miRNAs are also associated with resistance to anti-cancer drugs.
MiR-21 is the most commonly upregulated miRNA in both solid and hematological malignancies 4 and represents one of the earliest miRNAs identified as an “OncomiR.” Previous studies have demonstrated that the target genes of miR-21 (PTEN, PDCD4, TIMP3) are tumor suppressor genes. 5 MiR-21 is also involved in the resistance to conventional chemotherapies (paclitaxel, 5-fluorouracil (5-FU))6,7 and targeted therapies such as trastuzumab, gefitinib, and sorafenib.8–10
The purpose of this article was to validate the overexpression of miR-21 in cRCC patient tissues and examine the roles of miR-21 upregulation on renal cancer cell properties and drug resistance.
Materials and methods
Clinical specimens
In total, 99 primary cRCC samples with healthy tissues matched (n = 66) or not (n = 33) were collected after surgery and stored in the Tumor Bank and Tissue Collection of the Department of pathology of Lille Hospital (see Table 1). A written consent form was obtained from each patient, and the study was approved by the scientific committee of the institution “Tumorothèque du CHRU de Lille” (approval no. CSTMT078).
Association between miR-21 expression and clinicopathological characteristics of cRCC patients.

cRCC: clear renal cell carcinoma.
Cell lines and culture conditions
HK-2 human proximal tubule epithelial cell line, ACHN, and 786-O human renal cancer cell lines were purchased from American Type Culture Collection (ATCC). RCC10 and RCC4 human renal cancer cell lines were gifted by Dr D Bernard (Inserm U1052—CNRS UMR5286, Centre de Recherche en Cancérologie de Lyon, France).11,12 ACHN, 786-O, and RCC10/RCC4 cells were cultured respectively in Minimum Essential Medium (MEM), Dulbecco’s Modified Eagle’s Medium (DMEM), and RPMI-1640 medium supplemented with 10% of heat-inactivated fetal bovine serum (FBS), 1% of
Tissue and cell RNA extraction
For human formalin-fixed samples, total RNA was obtained using the RecoverAll™ Total Nucleic Acid Isolation Kit (Ambion). Total RNA was extracted from cell samples with the miRNeasy Mini Kit (Qiagen).
Quantitative reverse transcription polymerase chain reaction
Mature miRNA expression
Retrotranscription was done on 5 ng of total RNA with TaqMan probe (hsa-miR-21: 000397; RNU48: 001006; RNU6B: 001093 (Applied Biosystems)) according to TaqMan MicroRNA Reverse Transcription Kit protocol (Applied Biosystems). The quantitative polymerase chain reaction (qPCR) reaction was performed using TaqMan Gene expression Master Mix (Applied Biosystems) following the manufacturer’s protocol using the CFX96 Real-Time PCR System (Bio-Rad). The miRNA expression data were normalized to the expression of RNU6B for tissue samples and to the expression of RNU48 for cell samples. Expression levels of mature miR-21 were calculated based on the comparative threshold cycle method (2−ΔΔCT). 13
Gene expression
Retrotranscription was done on 1 µg of total RNA with High-capacity cDNA Reverse Transcription Kit (Applied Biosystems). Expression levels of ABCC1–6, SLC22A1, SLC22A2, and SLC31A1 were analyzed using TaqMan expression assay (Thermo Fisher Scientific). The qPCR reaction was performed using TaqMan Gene Expression Master Mix (Applied Biosystems) following the manufacturer’s protocol using the CFX96 Real-Time PCR System (Bio-Rad). The mRNA expression data were normalized to the expression of peptidylprolyl isomerase A (PPIA; also known as cyclophilin A). Expression levels of mature miR-21 were calculated based on the comparative threshold cycle method (2−ΔΔCT). 13
In situ hybridization
In situ hybridization of miR-21 was performed using double DIG-labeled LNA probes (Exiqon) as described in Heetal.’s and Livak et al.’s study. 14
Modulating miR-21 expression in renal cancer cells
In order to upregulate miR-21 expression and dowregulate miR-21 expression in renal cancer cells, pre-miR hsa-miR-21-5p (Ambion) and miRCURY LNA Inhibitor hsa-miR-21-5p (Exiqon) were transfected, respectively. Control cells were transfected with pre-miR miRNA Precursor Molecules-Negative Control#2 (Ambion) or with miRCURY LNA Power Inhibitor Control (Exiqon). Reverse transfection was done with Lipofectamine RNAiMAX Reagent (Invitrogen) according to the manufacturer’s instruction.
In vitro migration and invasion assays
Invasion and migration assays were carried out as described previously. 15 Briefly, migration and invasion were assayed using BD BioCoat Control Insert and BD Biocoat Matrigel Invasion Chamber, respectively, with 8-µm pores (BD Biosciences). 5 × 104 cells were seeded and transfected in the upper chamber in serum-free medium while medium with 10% FBS was added into the lower chamber as chemoattractant. After incubation for 24 h, the cells that did not migrate or invade through the pores were carefully wiped out with cotton swab. Then, the cells located on the lower surface of the chamber were fixed with Diff-Quick Kit (Medion Diagnostics), stained with 4′,6-diamidino-2-phenylindole (DAPI), and counted.
Western blotting
Total cellular extracts and western blotting were performed as previously described 11 using specific primary antibodies (PDCD4 (Cell Signaling Technology), cyclin D1 (Cell Signaling Technology), Cdk4 (Santa Cruz Biotechnology), Bax (Santa Cruz Biotechnology), Bcl-xL (Santa Cruz Biotechnology), and β-actin (Sigma-Aldrich)). Peroxidase-conjugated secondary antibodies (Sigma-Aldrich) were used and immunoreactive bands were visualized using the West Pico Chemoluminescent Substrate (Thermo Fisher Scientific, Pierce). Chemiluminescence was visualized using LAS4000 apparatus (Fujifilm). Three independent experiments were performed.
Cytotoxicity assay
Cells were transfected onto 96-well plates at a density of 104 cells per well. After 24 h of transfection, the medium was replaced with fresh medium containing paclitaxel, 5-FU, oxaliplatin, or dovitinib (Euromedex) at the determined concentration (range: 10 nM–20 µM) and incubated for 72 h at 37°C. The viability of cells was determined using the “Celltiter96® Aqueous Non Radioactive Cell Proliferation Assay” Kit (MTS; Promega) as previously described. 16 At least three independent experiments were performed.
Statistical analysis
Data are presented as mean ± standard error of the mean (SEM). Statistical analyses were performed with GraphPad Prism software (GraphPad Software). Differences between multiple groups were assessed by one-way analysis of variance (ANOVA) followed by Bonferroni’s multiple comparison test. Statistical differences between each two groups were determined by the Student’s t test. Differences were considered significant when p < 0.05.
Results
MiR-21 is overexpressed in cRCC
First, by quantitative reverse transcription PCR (qRT-PCR), we have examined miR-21 expression in 66 paired cRCC tumor and matched adjacent non-tumor tissues. MiR-21 expression was (1) detected in all studied tissues, (2) upregulated in 61 (92.4%) tumors compared to the paired healthy tissues, and (3) downregulated in only five tumors (7.6%) (Figure 1(a)). MiR-21 expression was significantly upregulated in cRCC compared to the normal renal tissues (14.48 ± 2.6 vs 1.48 ± 2, respectively; p < 0.001; Figure 1(b)). Overexpression of miR-21 was confirmed by in situ hybridization assay in which the staining signal of miR-21 was very weak or absent in non-tumor counterpart and strong in cRCC tissues (Figure 1(c)). Then, in a cohort of 99 cRCC tumors, we found no relationship with miR-21 expression, age, and gender (p > 0.05). In addition in this cohort, we showed no significant difference between different stages (pT1, pT2, and pT3) and nuclear Fürhman grades (II, III, and IV) (Table 1). Lastly, miR-21 expression in primary tumors was not a predictive factor of metastastic disease since no significant difference was observed between cRCC patients free of metastasis over 5 years after surgery (n = 26) and cRCC patients developing metastasis in 5 years (n = 27; p = 0.66; Table 1). In conclusion, in our cohort, we showed that miR-21 is overexpressed in cRCC tissues independently of tumor stage and grade.

MiR-21 is significantly upregulated in cRCC human tissues compared to the paired healthy tissue. MiR-21 expression in the tumor and adjacent renal healthy tissues of a cohort of 66 cRCC patients was determined by qRT-PCR. RNU6B was used as an internal control. (a) MiR-21 levels in tumors of individual cRCC patients are shown in relation to miR-21 expression in matched normal renal tissue. (b) The average expression of miR-21 in cRCC is shown in relation to the value obtained for normal kidney tissues (***p < 0.001). (c) In situ hybridization of miR-21 shows an overexpression in tumor tissues relative to healthy tissues.
MiR-21 increases migration and invasion properties
Then, we investigated the expression level of miR-21 in human proximal tubule epithelial cell line HK-2 and in human cRCC cell lines. By qRT-PCR, we showed that miR-21 expression was significantly upregulated in 786-O, RCC4, RCC10, and ACHN cancer cell lines compared to HK-2 cells (p < 0.05 and p < 0.001; Figure 2(a)). To determine the role of miR-21 in kidney cancer cell properties, pre-miR-21 or anti-miR-21 (LNA-modified ASO) were transfected into cRCC cell lines to either overexpress or inhibit miR-21, respectively (Figure 2(b) and (c). The role of miR-21 in cell migration was assessed using Boyden chambers. We observed significant enhanced migration in ACHN and 786-O cell lines transfected with the pre-miR-21 (4.2- and 3.8-fold, respectively; Figure 3(a)). By contrast, in cell lines transfected with the LNA-anti-miR-21, a significant reduced migration was observed compared to control cells transfected with a LNA-control (27% and 24% inhibition, p < 0.01; Figure 3(b)). Invasion assays showed that miR-21 overexpression significantly enhanced invasiveness of ACHN and 786-O cells (10.8- and 12.4-fold, respectively; p < 0.05; Figure 3(c)), whereas blocking miR-21 activity lowered invasiveness by 30% (p < 0.01; Figure 3(d)).
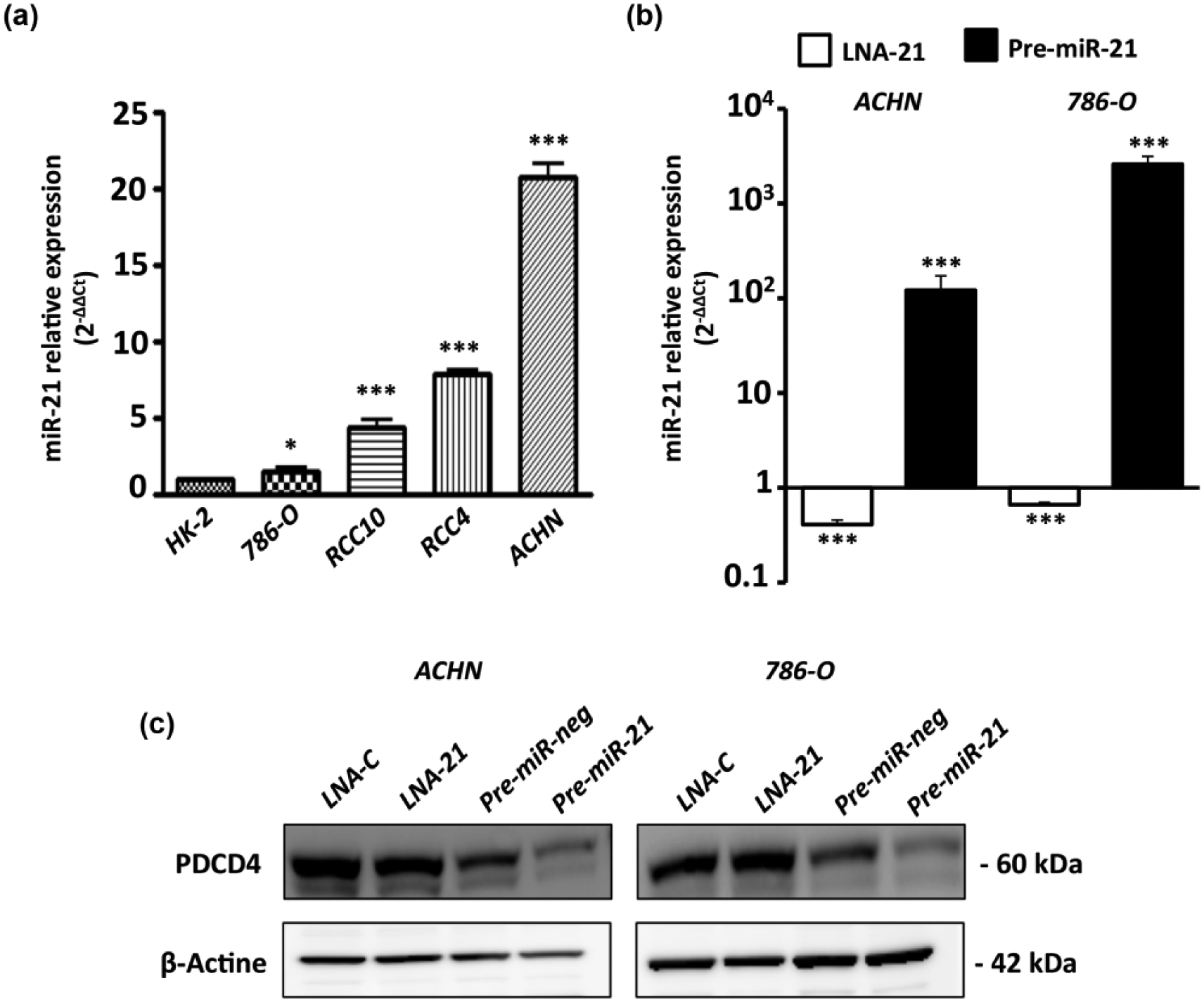
MiR-21 is overexpressed in cRCC cell lines and its expression can be modulated by pre-miR-21 or LNA-anti-miR-21 transfection. (a) MiR-21 expression in renal cancer cells 786-O, RCC10, RCC4, and ACHN compared to normal renal epithelial cells HK-2. RNU48 was used as an internal control. (b–c) ACHN and 786-O cells were transfected with 10 nM of pre-miR-21, pre-miR-Neg, LNA-C, or LNA-anti-miR-21. After 24 h of transfection, relative miR-21 or PDCD4 expression was measured by qPCR and western blotting, respectively. Values are mean ± SEM and represent at least three separate experiments (*p < 0.05 and ***p < 0.001).

MiR-21 increases migration and invasion properties in ACHN and 786-O cells. ACHN and 786-O transfected with 10 nM of (a and c) pre-miR-21, pre-miR-Neg, and (b and d) LNA-anti-miR-21, or LNA-C. Then, cell migration and invasion were evaluated using 24-well Boyden chambers coated or not with Matrigel® with 10% fetal bovine serum as chemoattractant. After 24 h, the number of migrating (a and b) and invasive (c and d) cells was determined. Values are mean ± SEM and represent at least three separate experiments (*p < 0.05, **p < 0.01, and ***p < 0.001).
Roles of miR-21 in intracellular signaling
Having shown that miR-21 promotes cell migration and invasion, we studied the impact of miR-21 overexpression on the major intracellular signaling pathways by western blotting. In miR-21 overexpressing cells compared to cells tansfected with pre-miR-neg, no impact was observed on cyclin D1 expression but expression of its kinase partner Cdk4 increased (Figure 4(a)). In contrast, in LNA-21 transfected cells compared to LNA-C transfected cells, cyclin D1 and Cdk4 expression decreased (Figure 4(b)). These data showed that miR-21 expression is involved in the regulation of cell cycle progression. MiR-21 expression in ACHN and 786-O cells was also associated with cell survival since Bax pro-apoptotic marker expression was decreased, whereas Bcl-xL anti-apoptotic marker expression was increased resulting in increased of Bcl-xL/Bax ratio suggesting an increase of apoptosis resistance in miR-21 expressing cells (Figure 4(a)). Opposite results were observed with the LNA-anti-miR-21 strategy (Figure 4(b)). Altogether, these results show that miR-21 promotes renal cancer cell migration, invasion, proliferation, and resistance to apoptosis in renal cancer cells.

Impact of miR-21 expression on signaling pathways. Western blotting was performed on whole cell extracts obtained from ACHN and 786-O cells transfected 24 h with 10 nM of (a) pre-miR-21 and pre-miR-Neg or (b) LNA-C and LNA-anti-miR-21. Antibodies against cyclin D1, Cdk4, Bax, Bcl-xL, and β-actin were used.
MiR-21 promotes chemoresistance in cRCC
Since cRCCs are highly resistant to conventional cytotoxic chemotherapies and miR-21 is associated to chemoresistance in numerous cancers such as colon and pancreas, we investigated the impact of anti-miR-21 strategy on renal cancer cell responses to different anti-cancer agents including targeted therapies. First, we tested paclitaxel, a drug known to inhibit cell division by binding to tubulin and consequently blocking the disassembly of microtubules. Measurement of half maximal inhibitory concentration (IC50) showed an increased sensitivity of 786-O (LNA-C = 1.2 ± 0.13 µM vs LNA-21 = 0.29 ± 0.06 µM, p < 0.001; Figure 5(a)), RCC10 (LNA-C = 1.25 ± 0.13 µM vs LNA-21 = 0.53 ± 0.13 µM, p < 0.001; Figure 5(a)), and ACHN (LNA-C = 1.21 ± 0.09 µM vs LNA-21 = 0.59 ± 0.1 µM, p < 0.001; Figure S1) cells to paclitaxel. Similar results were obtained with (1) the fluoropyrimidine 5-FU, an antimetabolite drug (Figure 5(b); LNA-C = 22.6 ± 3.1 µM vs LNA-21 = 14.1 ± 2.5 µM in 786-O cells and LNA-C = 10.3 ± 1.8 µM vs LNA-21 = 5.7 ± 0.95 µM in RCC10 cells) and (2) oxaliplatin, a platinum-based chemotherapy drug resulting in the formation of platinum–DNA adducts which block DNA replication (LNA-C = 12.4 ± 0.63 µM vs LNA-21 = 5.8 ± 1.5 µM in 786-O cells (Figure 5(c)), LNA-C = 10.8 ± 0.21 µM vs LNA-21 = 1.27 ± 0.28 µM in RCC10 cells (Figure 5(c)), and LNA-C = 12.92 ± 0.77 µM vs LNA-21 = 7.02 ± 1.36 µM in ACHN cells (Figure S1)). Then, we tested sunitinib, a multi-targeted receptor tyrosine kinase (RTK) inhibitor used as first-line therapy in patients with advanced cRCC. Sensitivity of 786-O, RCC10, and ACHN cells to sunitinib was not impacted by LNA-21 silencing strategy (data not shown). In contrast, for the first time, we showed that LNA-21 chemosensitized 786-O and RCC10 cells to dovinitib, another multi-targeted RTK inhibitor (Figure 5(d); LNA-C = 22.3 ± 1.5 µM vs LNA-21 = 8.7 ± 1.1 µM and LNA-C = 22.4 ± 3.7 µM vs LNA-21 = 9.3 ± 3.1 µM, respectively). To better understand the impact of miR-21 on chemoresistance, we focused our attention on genes known to confer a multi-drug resistance (MDR) phenotype to cancer cells: family of adenosine triphosphate (ATP)-binding cassette (ABC) transporters. By qPCR, we showed that miR-21 silencing strategy decreased significantly ABCC3–6, ABCC2–6, and ABCC3 and ABCC5 expression in RCC10 (Figure 6(a)), 786-O (Figure 6(b)), and ACHN (Figure S2) cells, respectively. Furthermore, LNA-21 strategy increased significantly SLC22A1/OCT1, SLC22A2/OCT2, and SLC31A1/CTR1 platinum influx transporter expression. In conclusion, miR-21 is an actor in chemoresistance observed in cRCC tumors.

Antagonism of miR-21 sensitizes cRCC cell lines to paclitaxel, 5-FU, oxaliplatin, and dovitinib. 786-O and RCC10 were transfected with 10 nM of LNA-C or LNA-anti-miR-21. After 24 h of transfection, 786-O and RCC10 cells were incubated with (a) paclitaxel, (b) 5-FU, (c) oxaliplatin, (d) or dovitinib. IC50 rates were measured after 72 h of drug treatment using MTS assay. Values are mean ± SEM and represent at least three separate experiments (*p < 0.05, ** p < 0.01, and ***p < 0.001).

Antagonism of miR-21 decreases expression of multi-drug resistant genes but increases expression of platinum influx transporters. (a) 786-O and (b) RCC10 were transfected with 10 nM of LNA-C or LNA-anti-miR-21. After 24 h of transfection, relative expression of ABCC1-6, SLC22A1, SLC22A2, and SLC31A1 was determined by qPCR. PPIA was used as an internal control. Values are mean ± SEM and represent at least three separate experiments (*p < 0.05 and *** p < 0.001).
Discussion
cRCC is the main histotype of kidney cancer, which is typically highly resistant to conventional systemic therapies. For metastatic cRCC, targeted therapies are standard treatments but the 5-year survival rate is dismal at less than 10%. 2 Identifying actors and deciphering molecular mechanisms that lead to tumor progression is an important step to develop new therapeutic strategies to cure cRCC. In this context, we focused our attention on miR-21, an oncogenic miRNA upregulated in many solid tumors such as breast, lung, pancreas, prostate, and stomach cancers. 4 Previous studies on RCC have shown that miR-21 is also overexpressed in clear cell and papillary subtypes of RCC compared with healthy kidney and benign renal tumors.17,18 Herein, we confirmed this overexpression (Figure 1(b)) and further showed that miR-21 expression in cRCC is not associated with stage, nuclear Fürhman grade, and patient outcome. Thus, miR-21 expression in primary tumors is of limited interest as a diagnostic or a prognostic biomarker and must be included in miRNA signature. 19
In this study, we confirmed the oncogenic potential of miR-21 by showing that ectopic expression of miR-21 in cRCC cells significantly increased their migration, invasion, proliferation, and anti-apoptotic properties. A study on more than 400 renal tumor samples showed the aberrant activation of phosphoinositide 3-kinase (PI3K)/Akt pathway suggested that inhibitor drugs of this pathway represent a valid therapeutic option in cRCC. 20 In line with this study, miR-21 silencing strategy is of potential interest since PTEN, a potent inhibitor of PI3K/Akt pathway, is a direct target of miR-21. Furthermore, we showed that the inhibition of miR-21 enhanced the sensitivity of cRCC cells not only to conventional genotoxic drugs (paclitaxel, 5-FU, and oxaliplatin) but also to dovinitib indicating a correlation between miR-21 expression and chemoresistance in cRCC. The inhibition of miR-21 also decreased expression of MDR genes (ABCC2–6) by a mechanism that remains to be deciphered. These findings are consistent with those of other solid tumors such as breast, pancreas, and colorectal cancer. In pancreatic cancer, cells resistant to 5-FU expressed high levels of miR-21 in comparison to sensitive primary cells. Overexpression of two miR-21 targets, PTEN and PDCD4, antagonized miR-21-induced resistance to 5-FU. 21 In HT-29 colon cancer cells, miR-21 mediated 5-FU chemoresistance by targeting directly the human mutS homolog 2 (hMSH2) and indirectly thymidine phosphorylase and dihydropyrimidine dehydrogenase, two enzymes involved in 5-FU metabolism. 7 In tongue squamous cell carcinoma and in A172 glioma cell line, miR-21 increased chemoresistance to cisplatin by targeting PDCD4 22 and FOXO1 to decrease the expression of pro-apoptotic factors (Bim and FASL) and cell cycle inhibitors (p27 and p21). 23 Herein, in cRCC cells, we showed that miR-21 expression was correlated to (1) chemoresistance to platinum-based therapy and (2) for the first time, decrease in SLC22A1/OCT1, SLC22A2/OCT2, and SLC31A1/CTR1 platinum influx transporter expression. MiR-21 is also involved in the resistance to targeted therapies, and different studies suggested that targeting miR-21 altered PI3K/Akt signaling pathway and constituted a novel therapeutic strategy for reversing resistance to targeted therapies such as gefitinib or sorafenib.9,10 Two previous studies have shown that miR-21 silencing enhanced the cytotoxic effect of sunitinib in glioblastoma and pancreatic cell lines24,25 but we observed no effect of this strategy on cRCC cell lines. For the first time, we showed that miR-21 was involved not only in dovitinib resistance but also in the regulation MDR gene expression likely through PTEN targeting since previous works have observed that hyperactivation of PI3K/Akt pathway was able to regulate expression of MDR genes, including ABCC2, ABCC4, and ABCC5 genes.26–28 MiR-21 appears as an important actor in MDR by regulating cell proliferation, cell cycle, apoptosis, expression of MDR, and DNA repair.
The cancer stem cell (CSC) model suggests that a small subset of cancer cells possess stem cell properties and are responsible for tumor initiation, growth, metastasis, therapy resistance, relapse, and poor prognosis. 29 Epithelial–mesenchymal transition (EMT) process is also involved in CSC formation in numerous cancers. It was demonstrated that miR-21 overexpression facilitates sphere formation and induces EMT in numerous cancers including cRCC.30–32 Our preliminary results showed that miR-21 knockdown decreased the expression of mesenchymal markers in 786-O and RCC10 cells (data not shown). Furthermore, carcinoma-associated fibroblasts (CAFs), which are activated fibroblasts, communicated with cancer cells to promote tumor progression and metastasis. 33 Xu et al. 34 have shown that, by coculture experiments, CAFs stimulated tumor cell proliferation and migration and induced resistance to everolimus in cRCC cells. MiR-21 is one of known actors in fibroblast activation,3,35 and anti-miR-21 strategy will not only target cRCC tumor cells but also the tumor microenvironment.
In conclusion, miR-21, as a masteroncomiR, profoundly influences renal tumorigenesis in a multifaceted way (tumor growth, differentiation, apoptosis, chemoresistance, and stemness). The implication of miR-21 in resistance to both conventional cytotoxic agents and targeted therapies highlights the possible clinical application of miR-21 inhibition. Interestingly, new therapeutic approaches are emerging in particular with the development of nanoparticles for sequential co-delivery of miRNA inhibitors and chemotherapeutic compounds to enhance anti-cancer efficacy. 36 In cRCC, miR-21 silencing is a promising strategy to improve chemotherapy efficacy and will open new therapeutic options to clinicians to ameliorate patient’s care.
Footnotes
Acknowledgements
The authors thank M Samyn (Institut de Pathologie, CHRU, Lille), M.H. Gevaert, and R. Siminsky (Department d’Histologie, Faculté de Médecine, Université de Lille 2) for their excellent technical help. Renal cell lines RCC4 and RCC10 were gifted by Dr D Bernard (Inserm U1052—CNRS UMR5286, Centre de Recherche en Cancérologie de Lyon, France). The authors also thank “Tumorothèque C2R de Lille” (under supervision of Pr M.C. Copin, Centre de Biologie-Pathologie, CHRU de Lille).
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinski declaration and its later amendments or comparable ethical standards.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from SIRIC ONCOLille (Grant INCa-DGOS-Inserm 6041 to S.A.), from Contrat de Plan Etat Région (CPER; Cancer 2007-2013 to I.V.S.), from la Ligue Nationale Contre le Cancer (comité du Nord to S.A.), and from the Conseil Régional du Nord Pas de Calais to M.P., C.C., and N.P. K.G. is the recipient of a doctoral fellowship from “Ligue Nationale Contre le Cancer,” “Région Nord-Pas-de-Calais,” and “CHRU de Lille.”
