Abstract
For tumor to grow beyond 1–2 mm3 size, tumor recruits new blood vessels referred as angiogenesis; therefore, targeting angiogenesis can be a promising strategy to suppress cancer progression. In this study, in order to develop a good angiogenesis model, we investigated effect of Dalton’s lymphoma on angiogenesis and further monitored the role of melatonin on regulation of angiogenesis. To evaluate angiogenesis, endothelial cells were isolated from main thoracic aorta and cultured in vitro in the presence or absence of Dalton’s lymphoma supplemented with or without melatonin to monitor their role on its proliferation and migration, a hallmark of angiogenesis. Chick chorioallantoic membrane as well as mice mesentery which allows in vivo studies of tumor angiogenesis and testing of anti-angiogenic molecules was used to validate the in vitro analysis. To further extend our understanding about the regulation of the angiogenesis, we evaluated expression of tissue inhibitor of metalloproteinases 3, vascular endothelial growth factor, vascular endothelial growth factor receptor, and fibroblast growth factor in Dalton’s lymphoma cells and mesentery by semiquantitative and quantitative reverse transcription polymerase chain reaction analysis. Dalton’s lymphoma ascites induced significant increase in endothelial cell proliferation, migration, and sprouting of the tertiary branching in chorioallantoic membrane and mesentery of Dalton’s lymphoma–bearing mice, whereas melatonin treatment led to their inhibition in a dose-dependent manner. Semiquantitative and quantitative reverse transcription polymerase chain reaction analysis of melatonin-treated Dalton’s lymphoma cells and mesentery tissue clearly demonstrated restoration of angiogenesis-related genes tissue inhibitor of metalloproteinases 3 and reduction of vascular endothelial growth factor, vascular endothelial growth factor receptor, and fibroblast growth factor messenger RNA expression. Taken together, our results strongly demonstrate that Dalton’s lymphoma provides pro-angiogenic environment leading to significant increase in angiogenesis, and further melatonin treatment reduced the Dalton’s lymphoma ascites–induced angiogenesis implying that Dalton’s lymphoma can serve as a very good model to study angiogenesis as well as for screening of drugs that can target angiogenesis.
Keywords
Introduction
Angiogenesis is a physiological process that leads to formation of new blood vessels from pre-existing ones and rarely occurs in normal adults except in female endometrium and during wound healing. However, it is a fundamental step and increases with disease progression in many disease conditions such as cancer metastasis, rheumatoid arthritis, and some ocular diseases.1,2 As tumor increases in size, cancer cells undergo necrosis due to deficiency of oxygen and nutrient resulting in a condition called hypoxia; therefore, for tumor to grow beyond a certain size, new blood vessels develop by angiogenesis.3,4 The key features of tumor progression are production of growth factors and modulation of the stromal environment for their own growth and benefits. 5 The cells of tumor stroma including fibroblasts, pericytes, and infiltrating immune cells such as macrophages play major role in promoting new blood vessel formation, particularly, by inducing endothelial cell proliferation, thereby supporting tumor growth and metastasis.6,7 With the existing understanding about the role of angiogenesis in the field of tumor growth and progression, targeting angiogenesis can be a potential strategy of anticancer therapy.8,9
The two major limitations in drug development for cancer are high genetic instability in cancer cells and development of drug resistance.9,10 The recruited endothelial cells at tumor vasculature are genetically more stable than cancer cells and are also relatively less susceptible to develop drug resistance; therefore, they are a better target for combating tumor growth.3,8,9 The process of angiogenesis is complex, and the inner monolayer of endothelial cells in blood vessels continuously senses the micro-environment and plays a major role in sprouting of new blood vessels. It involves endothelium proliferation, migration, and degradation of extracellular matrix (ECM). 11 The entire process of angiogenic growth is regulated by a balance between pro- and anti-angiogenic molecules secreted by the surrounding cells. The pro-angiogenic factors such as vascular endothelial growth factor (VEGF), fibroblast growth factors (FGF), transforming growth factor-alpha (TGF-α) and transforming growth factor-beta (TGF-β), tumor necrosis factor-alpha (TNF-α), angiogenin, angiopoitin-1 and -2, and interleukin-1 and -8 overcome the anti-angiogenic factors such as thrombospondin-1 (TSP-1), angiostatin, endostatin, vasostatin, interleukin-12, tissue inhibitor of metalloproteinase 3 (TIMP3), and E-cadherin (CDH1).1,12,13
The angiogenesis-related studies are mostly carried out in vitro using human umbilical vein endothelial cells (HUVECs). HUVECs which originate from umbilical veins are considerably different from the arteries/capillaries in the cancer tissue. It is well known that the endothelial cells show enormous heterogeneity depending upon tissue origin. 14 In addition, study of anti-angiogenic activity of any therapeutic regimen commonly is being done by the use of an artificial system comprising VEGF as mitogen on HUVEC proliferation, migration, and capillary formation. 15
In addition, chick chorioallantoic membrane (CAM) assay has been widely used as an in vivo model to study the pro-angiogenic and anti-angiogenic activities of various agents, for example, hormones, cytokines, growth factors, drugs, tumor milieu, and implanted grafts. 16 CAM is an extra embryonic membrane formed by fusion of chorionic and allantoic membranes. Its formation starts from 4th day and completely develops at the 10th day during chick embryo development. It has a very dense capillary network which provides gas and nutrient exchange to the chick embryo until hatching.17,18 This assay system is simple, low cost, reproducible, and reliable to study angiogenesis as the immune system of chick embryo is not fully developed until day 10 and the conditions for graft rejection have also not been established. Though developing chick embryo provides the natural immune-deficient environment. It is very different from the natural tumor environment.11,19
Dalton’s lymphoma (DL) is a murine non-Hodgkin’s T-cell lymphoma and a good model to study various parameters of cancer development, signaling mechanisms, and ultimately for screening of drugs for treatment.20,21 In this study, we have used DL model to evaluate angiogenesis due to ease in maintenance for in vivo as well as in in vitro study. Dalton’s lymphoma ascites (DLA) isolated from the peritoneum of DL-bearing mice provides conditions mimicking tumor micro-environment for in vitro assay which makes study easier and provides reproducible results.7,20,21 Furthermore, the mice mesentery vasculature network is reported to be an ideal system for angiogenic growth measurements. Transparency of thin membranous mesenteric window provides a good assay system for studying the micro-vascularization network to be assessed in in vivo condition.22,23 The assay allows quantitative assessment of density, capillary sprouting, and network pattern formation, thereby enabling statistical analyses. It is easy to exteriorize small-gut mesentery from mice abdominal cavity. Another advantage of the assay is that the mesentery is the only true quantitative assay for evaluation of normally vascularized tissue.23–25
Melatonin is a secreted product of pineal gland and plays major role in circadian and seasonal rhythms. 26 Melatonin shows oncostatic effect by regulating the expression and activity of various signaling molecules secreted by cancer and proximal cells.19,27 Studies reported that melatonin has paracrine interaction with VEGF signaling and matrix metalloproteinase-2 (MMP-2) and matrix metalloproteinase-9 (MMP-9) activity to inhibit HUVEC proliferation. Anti-angiogenic activity of melatonin has also been reported based on the in vitro studies on HUVEC.15,28 In this study, to circumvent the limitations of HUVECs, we have isolated endothelial cells of main thoracic aorta to study the effect of melatonin on DL-induced endothelial cell proliferation which was further validated in CAM assay.
The main emphasis of our study was to focus on the effect of DL on angiogenesis and dose-dependent inhibitory potential of melatonin. DL or DLA-induced endothelial cell proliferation and migration were studied in vitro using endothelial cells isolated from main thoracic aorta, whereas growth and inhibition of tertiary sprouting of blood vessel were evaluated in vivo in CAM and mesentery window of mice with growing DL. Results from reverse transcription polymerase chain reaction (RT-PCR) and quantitative RT-PCR (qRT-PCR) analyses of angiogenesis-related genes in DL cells or in mesenteric tissue with increased sprouting of micro-vessels in DL-bearing mice suggested that DL-induced angiogenesis was regulated at transcriptional level. The messenger RNA (mRNA) expression of VEGF and FGF was upregulated in DL cells and vascular endothelial growth factor receptor (VEGFR) in mesenteric tissue, whereas expression of TIMP3 mRNA was found to be downregulated in both DL and mesenteric tissue. Melatonin treatment also led to restoration of gene expression in DL and mesenteric tissue almost to normal levels.
Material and methods
Chemicals
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT); Dulbecco’s Modified Eagle’s Medium (DMEM) media; melatonin; dexamethasone; fetal bovine serum (FBS); primers for VEGF, VEGFR, FGF, TIMP3, glyceraldehyde 3-phosphate dehydrogenase (GAPDH), and β-actin; and antibiotics were purchased form Sigma-Aldrich (India). Methanol and crystal violet were purchased from HiMedia (India). SYBR Green PCR Master Mix was purchased from Applied Biosystems by Life Technologies (UK). Rabbit polyclonal anti-CD31 and Alexa-568-labeled goat anti-rabbit antibodies were purchased by Abcam (UK). Human lung adenocarcinoma A549 and cervical carcinoma SiHa cells were purchased from National Centre for Cell Science (NCCS, India). DL cell line was obtained from Prof. Ajit Sodhi, Banaras Hindu University (BHU, India). For CAM assay, fertilized chick embryos were purchased from Keggfarms Pvt. Ltd. (India).
Animals
The animal experiments were carried out with BALB/c mice aged 3–4 months of either sex obtained from the Animal House Facility of Department of Zoology, University of Delhi, India. In a polypropylene cage, 4–5 animals were housed, provided with a standard pellet diet, water ad libitum, and maintained under standard laboratory conditions of temperature and humidity with an alternating 12-h light/dark cycle in accordance with the guidelines laid by Animal Ethics Committee, University of Delhi, and protocols approved by the CPCSEA, India.
Maintenance of DL and collection of ascitic fluid (DLA)
Dalton lymphoma (DL) cells were maintained in the peritoneum of BALB/c mice by serial passaging of tumor cells via intraperitoneal (IP) injection as described previously.7,21 Briefly, 1 mL of 1 × 106 cells/mL of DL cell suspension (in phosphate-buffered saline (PBS) was injected in the peritoneal cavity of mice and was left to grow for 18–21 days. Increased body weight and abdominal swelling signified the development of DL, which was clearly visible from 10 to 11 days post transplantation, and these mice survived for 20 ± 3 days.
To get DLA, the peritoneal lavage was drawn from mice having fully grown tumor (15–18 days) and was centrifuged at 5000 r/min for 10 min at 4°C. Supernatant was collected and filtered through 0.45 µm membrane filter. Collected DLA was aliquoted and stored at −20°C until used in experiments.
Preparation of condition media of A549 and SiHa cells
The A549 and SiHa cells were obtained from national tissue repository, NCCS (India). The cells were cultured in DMEM supplemented with 10% FBS, gentamycin (20 mg/mL), streptomycin (100 mg/mL), and penicillin (100 IU) in a CO2 incubator at 37°C in a humidified atmosphere with 5% CO2; 80% confluent of exponentially growing cells was sub-cultured and used in the experiments.
In 1 mL of complete DMEM having 10% FBS, 1 × 106 cells/well were plated in six-well plate overnight in a CO2 incubator. Next day, the cells were washed with PBS two times and re-fed with 500 µL of incomplete DMEM. After 24 h of incubation, the condition media (CM) were collected, centrifuged at 10,000 r/min at 4°C, filtered through 0.45 µ membrane filter, and stored at −20°C to be used later as source of angiogenic factor.
MTT assay
The anti-proliferative effect of melatonin against DL cells was determined by the MTT assay described earlier with slight modification. 15 Briefly, 1 × 104 cells/well (DL) were grown in medium alone and in the presence of melatonin (1 and 5 mM; 100 mM stock in 17% ethanol) or dexamethasone (5 µg/mL) for different time durations (0–96 h) in CO2 incubator at 37°C with 70%–80% humidity; 4 h prior to termination, 20 µL MTT solution (5 mg/mL in PBS) was added into each well. For termination, cells were collected in microfuge tubes and centrifuged at 5000 r/min for 5 min, and the medium was removed and washed with PBS and 100 µL of dimethyl sulfoxide (DMSO) was added to the pellet to dissolve the formazan crystal formed in the cells. Finally, optical density was measured at 570 nm using an micro-plate reader (Biotek, USA).11,29
Isolation of endothelial cells from main thoracic aorta from BALB/c mice using 0.2% collagenase enzyme
The endothelial cells were isolated from main thoracic aorta following protocol described earlier with few modifications.30–32 Main aorta was dissected out, stripped of extraneous fatty tissue, and washed with PBS (having 3× gentamycin (20 mg/mL), streptomycin (100 mg/mL), and penicillin (100 IU)). Washed artery was placed in cold PBS (having 3× gentamycin (20 mg/mL), streptomycin (100 mg/mL), and penicillin (100 IU)) for 2 h at 4°C in a cold room to reduce the contamination. To dissociate endothelial cells, the artery was first drained off any PBS trapped, cut laterally, and incubated in 0.23% collagenase in DMEM at 37°C in an incubator for 35–45 min in a microfuge tube with intermittent shaking at every 10 min. The enzyme suspension with dissociated cells was collected in a falcon, debris was removed, and cells were rinsed with DMEM two times. Finally, 1 × 105 cells/well were plated to form a monolayer of endothelial cells in 24-well plate in DMEM containing 20% FBS, gentamycin (20 mg/mL), streptomycin (100 mg/mL), and penicillin (100 IU) and were incubated in a CO2 incubator at 37°C and 70%–80% humidity. Media were replaced after every 2 days.
Characterization of endothelial cells by immunostaining with CD31
Endothelial cells isolated from thoracic aorta (1 × 105) were plated on sterilized cover glasses in a 12-well plate and were allowed to adhere for 4 h. Culture media from 0 and 72 h grown cells were removed, washed with PBS three times, and fixed with 3% paraformaldehyde for 10 min at room temperature. The fixed cells were again washed with PBS and permeabilized with cold 0.5% Nonidet P-40 for 1 min, washed, and then blocked with blocking solution (5% goat serum in
Endothelial cell proliferation assay
The endothelial cells harvested from main thoracic aorta of BALB/c mice, 1 × 104 cells/well, were divided in two groups: group I control and group II grown in complete medium alone and in the presence of 25% DLA, respectively, for 48 h. After 48 h, the DLA-treated cells were further divided into two groups: one group (II-a) was no more supplemented with DLA and the other group (II-b) was again supplemented with 25% DLA for 72 h. Different doses of melatonin (1 and 5 mM) or dexamethasone (5 µg/mL) were given to all the three groups (Figure 3) after 48 h for next 72 h and proliferation of cells was analyzed using crystal violet (0.02% aqueous solution). 28 Absorbance was measured at 540 nm using microplate reader (Biotek, USA).
Endothelial cell migration: wound-healing assay
Migration of endothelial cells was analyzed by method described earlier with slight modification.5,15 Endothelial cells isolated from thoracic main aorta were seeded in 12-well plate in DMEM containing 20% FBS and were allowed to reach full confluence. A line of cells was scraped away in each well with the help of sterile pipette tip. Wells were washed with PBS to remove detached and dead cells. Melatonin (1 or 5 mM) or dexamethasone (5 µg/mL; +ve control) was added in the presence and absence of DLA (25%) in respective wells (as shown in Figure 4(a)) and were photographed at different time points under inverted microscope (4×) to assess cell migration.
Chorioallantoic membrane assay
CAM model allows in vivo system to study tumor angiogenesis and can be utilized for testing of anti-angiogenic molecules. The methodology was described elsewhere and was followed with slight modification.2,11,17,33 On 4th day after fertilization, a window was formed, and the eggs were incubated at 37°C. The tumor factors such as ascites (DLA; freshly isolated from mice having 18- to -21-day-old tumor), cancer cell condition media (CM of A549 or SiHa), or cancer cells (DL) were added in control embryos and drug-treated embryos, which were supplemented with drugs 2 h prior to tumor cells or tumor factors on sterile Whatman filter paper discs. The window was again sealed and incubated for 96 h at 37°C, with 60%–70% relative humidity in an incubator. After final 96-h incubation, CAMs were fixed with 4% formalin for 15 min at room temperature. Fixed CAM was cut, separated from the egg, and was spread properly to be photographed under stereo zoom microscope (Nikon) and were analyzed with the help of NIS element software. Counting of primary, secondary, and tertiary blood vessels was done to analyze the effect of DLA and treatment on angiogenesis.
Schedule of in vivo melatonin treatment and sample collection
For the experiment, 8- to 10-week-old mice of either sex weighing 25–30 g were used. The animals were randomly distributed into three groups each having six mice per group per cage. Group I was kept as control without any treatment. Group II and group III were injected with 1 × 106 DL cells/mL IP. To evaluate anti-angiogenic activity of melatonin, group III mice were given melatonin treatment at a dose of 50 µg/g/body weight simultaneously with DL transplantation in evening after 6 p.m. for consecutively 10 days. On day 21, post DL transplantation, all the mice from each group were sacrificed by cervical dislocation. Peritoneum and mesentery were exteriorized immediately after sacrificing the animal, washed in chilled PBS, spread on wax plate, and photographed under stereo zoom microscope following the method described by Norrby 34 in 2006 with slight modifications.
Histological study
Furthermore, the histological study of exteriorized peritoneum was performed by hematoxylin and eosin (H&E) staining. The peritoneum was isolated, washed with chilled PBS, and fixed using Bouin’s fixative followed by gradual dehydration and parafilm wax embedding. Tissue sectioning of 5–6 µm was done using microtome. Finally, the sections were stained with H&E. 35 The sections were micro-photographed and analyzed by NIS element software.
Semiquantitative RT-PCR and qRT-PCR analysis of VEGF, FGF, and TIMP3 mRNA expression in DL cells and VEGFR and TIMP3 in mice mesentery
The effect of melatonin on expression of VEGF, FGF, and TIMP3 genes in the DL cells was measured by RNA isolation from the normal thymic cells (isolated from mice), untreated DL cells, and DL cells treated with melatonin (1 mM for 24 h), TIMP3, and VEGFR genes in stromal cells retrieved from mesentery of group I, II, and III, followed by first-strand complementary DNA (cDNA) synthesis, and then, gene-specific PCR was done using forward and reverse primers enlisted in Table 1 to obtain PCR amplicons, and GAPDH was taken as internal control. 29 In the real-time reactions, fold change of each transcript was calculated by the comparative ΔΔCT method and was normalized with β-actin.
List of primers used in RT- and qRT-PCR.

VEGF: vascular endothelial growth factor; TIMP3: tissue inhibitor of metalloproteinase 3; FGF: fibroblast growth factor; VEGFR: vascular endothelial growth factor receptor; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Statistical analysis
Data shown are representative images and difference among groups of cells was analyzed using Student’s t-test. The p value of 0.05 was considered statistically significant (*p ≤ 0.05, **p ≤ 0.01, and ***p≤ 0.001). All experiments were done in triplicate and repeated three times unless otherwise stated.
Results
DLA induces endothelial cell proliferation
Endothelial cells are precursor for the formation of neo-vasculature, and proliferation of endothelial cells is the initial and essential step in sprouting of new blood vessels or angiogenesis in tumor environment. 13 In order to study the effect of DLA on angiogenesis and role of melatonin, endothelial cells were isolated from the main thoracic aorta, made single cell suspension, plated in 24-well culture plate, and were allowed to grow as monolayer in DMEM with 20% FBS. The endothelial cell proliferation was monitored from 24 to 72 h (Figure 1). To characterize the cells isolated from aorta, immunostaining with endothelial cell–specific marker CD31 was performed. As shown in Figure 1(a), the isolated cells showed significant proliferation in in vitro culture (Figure 1(a)) and were also CD31 positive (Figure 1(b)). Next, to evaluate the effect of DLA on endothelial cell proliferation, 4-day cultured endothelial cells were fed with 25% DLA and further incubated for 72 h. We observed that DLA induced significant increase in endothelial cell proliferation when compared to control endothelial cells that were not exposed to DLA (Figure 2(a)), suggesting that DLA must have factors leading to endothelial cell proliferation. The number of endothelial cells increased up to 2.3 fold (Figure 2(a)).

In vitro proliferation and CD31 immunostaining of aortic endothelial cells. (a) Endothelial cells harvested from main thoracic aorta were seeded in 96-well culture plates (1 × 104 cells/well) in medium supplemented with 20% FBS and incubated for 0–72 h. (a(i)) The cells were fixed with chilled methanol, stained with crystal violet, and photographed. (a(ii)) The same stained cells were solubilized in DMSO to measure the absorbance for graphical presentation of proliferation. (b) Photomicrograph of endothelial cells showing expression of CD31 surface marker by immunostaining with anti-CD31 antibody and secondary antibody tagged with Alexa568. Nuclei are counterstained by DAPI.

Effect of (a) DL-ascitic fluid (DLA) or (b) melatonin on endothelial cells. Endothelial cells harvested from main thoracic aorta were seeded in 96-well culture plates (1 × 104 cells/well) in a medium supplemented with 20% FBS for 24 h. Then, media were aspirated and replaced with fresh media supplemented with 20% FBS containing 25% freshly isolated DLA (from 14- to 15-day-old DL-bearing BALB/c mice) or (b) increasing concentration of melatonin and further incubated for 72 h. (a(i)) Representative photomicrograph of control and DLA-treated endothelial cells. (a(ii)) Graphical presentation of crystal violet assay for the same groups. (b) Graphical presentation of dose kinetics study of melatonin on endothelial cells (**p < 0.01; ###p < 0.001 vs control).
Melatonin inhibits DLA-induced endothelial cell proliferation
To evaluate role of melatonin on DLA-induced endothelial cell proliferation, based on previous studies 15 and dose kinetics of melatonin on normal endothelial cells (Figure 2(b)), pharmacological concentration of melatonin (1 mM) and an higher dose of 5 mM were used for treatment. As described in section “Material and methods,” the cells were divided into three groups. Group I—endothelial cells without DLA; Group IIa—endothelial cells induced with DLA for 48 h, washed, and treated with melatonin for 72 h; and Group IIb—endothelial cells grown in DLA for 48 h, washed, and treated with different concentrations of melatonin for 72 h in the presence of DLA. A volume of 1 mM concentration of melatonin showed cytostatic effect, whereas 5 mM concentration showed growth inhibitory effect on group I control cells. The Group IIa cells (induced with DLA for initial 48 h only) showed high sensitivity toward melatonin, and significant apoptosis was observed in a dose- and time-dependent manner. Melatonin also induced apoptosis in Group IIb cells in dose-dependent manner. However, the effect was less pronounced in the presence of DLA. A volume of 5 µg/mL dexamethasone was taken as positive control in all conditions (Figure 3).

Effect of melatonin on endothelial cell proliferation. Endothelial cells isolated from main thoracic aorta were divided into three groups. In group I, the cells were incubated in medium alone, group IIa cells were grown in the presence of 25% DLA for 48 h, washed, and treated with drugs (melatonin 1 and 5 mM or dexamethasone 5 µg/mL) for 72 h. In group IIb, the cells were grown in the presence of 25% DLA for 48 h, washed, and treated with drugs (melatonin 1 and 5 mM or dexamethasone 5 µg/mL) in the presence of 25% DLA for further 72 h. After the treatment, the cells were fixed with chilled methanol, stained with crystal violet, and (a) photographed at 10× magnification. (b) Graphical representation of growth profile from crystal violet assay (*p ≤ 0.05, **p ≤ 0.01, and ***p ≤ 0.001).
Regulation of endothelial cell migration by melatonin
Migration of endothelial cell is an essential event for neovascularization during angiogenesis. Therefore, we focused on the anti-migratory effects of melatonin treatment on endothelial cells using wound-healing assay. As shown in Figure 4, the wound created was covered considerably within 72 h by the DLA-induced endothelial cells signifying cell migration. Melatonin treatment significantly inhibited the migration of endothelial cells in a dose-dependent manner. An amount of 5 µg/mL dexamethasone was taken as positive control (Figure 4).

Effect of melatonin on endothelial cell migration analyzed by the wound-healing assay. (a) Representative photomicrographs after 72 h of wound created with or without DLA in the presence or absence of drugs (melatonin 1 and 5 mM or dexamethasone 5 µg/mL). Pictures are shown at 40× magnification and migration distance was measured using Motic Image Plus 2.0 software. (b) Graphical representation of distance migrated by the endothelial cells in the presence of DL ascites with or without drugs. (Data presented as mean ± SEM and ***p ≤ 0.001 and ###p ≤ 0.001 for control vs DLA.)
Effect of DLA on angiogenesis in CAM
In in vitro study, we observed that DLA induced endothelial cell proliferation and melatonin treatment and significantly downregulated the DLA-induced endothelial cell proliferation. Next, we extended our investigation to monitor the effect of DLA on angiogenesis in vivo in CAM assay. Initially, we compared the effect of DLA and cancer cell (A549 and SiHa) conditioned media for angiogenic response in CAM; 50 µL of freshly isolated DL-ascitic (DLA) fluid was injected in 4-day-old fertilized chick egg and left to grow for another 4 days. The chorioallantoic membrane was analyzed for new blood vessel formation. As shown in Figure 5(a), DLA treatment to growing embryo resulted in increased sprouting of tertiary blood vessels suggesting the presence of pro-angiogenic factors in DLA. There was significant 2.25-fold increase in growth of tertiary vessel formation (control: DLA (mean ± standard error of mean (SEM)) 65 ± 4.8: DLA: 146.5 ± 4.5). In addition, there was a significant increase in the branch points (p = 0.0001). The secondary branches also increased but not to the significant level (Figure 5(b)). The DLA treatment did not affect the primary vessel development. Next, as shown in Figure 5(b), 24-h conditioned media of A549 and SiHa cells known to secrete pro-angiogenic factors also promoted tertiary branching of blood vessels in CAM of developing chick embryo to 1.4- and 1.04-fold increases in A549- and SiHa-conditioned media, respectively (Figure 5).
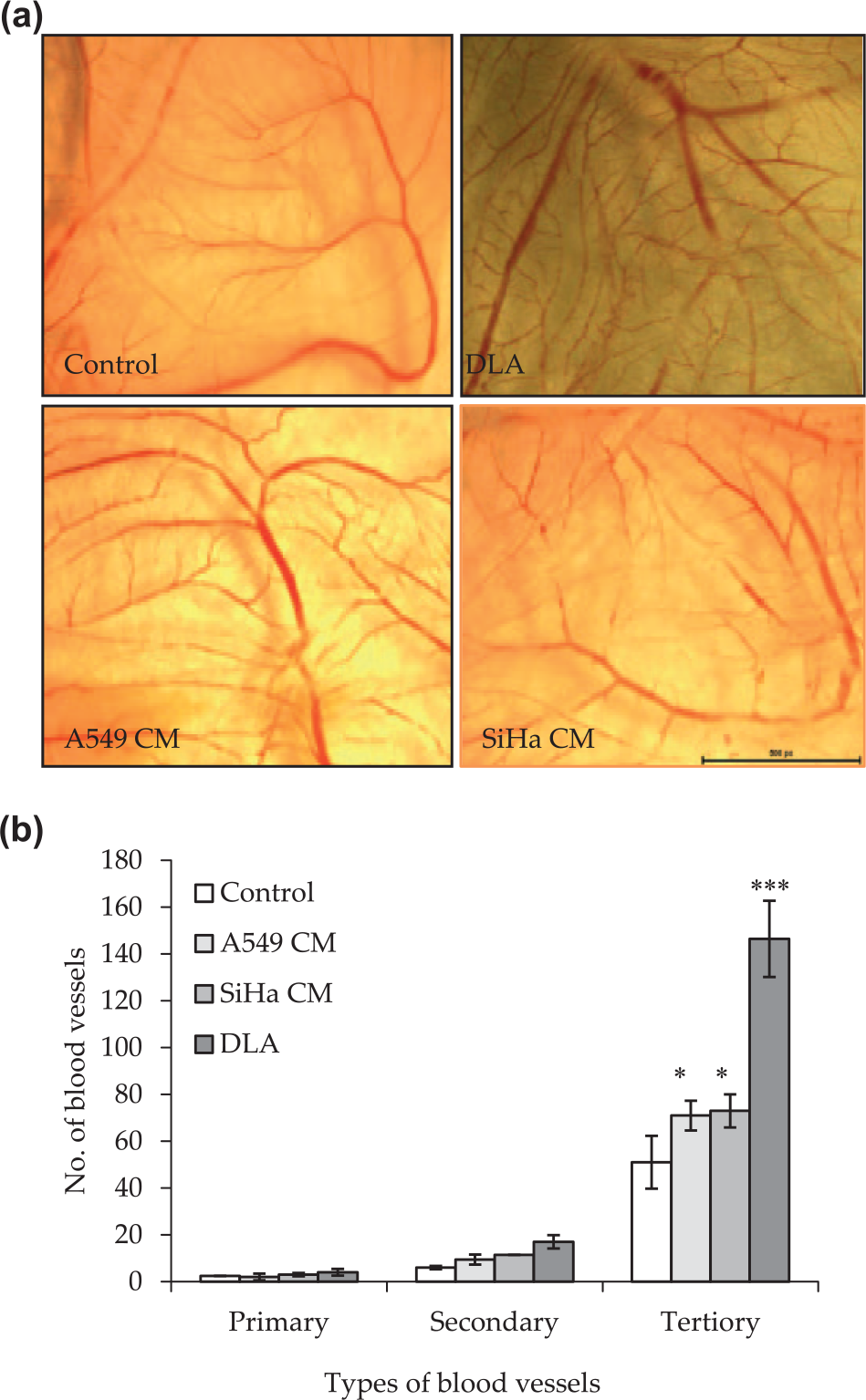
Effect of DLA and tumor cell–conditioned media from A549 and SiHa cells on CAM. (a) Photomicrograph showing control and DLA-, A549 CM–, and SiHa CM–treated CAM. (b) Graphical representation of number of micro-blood vessels formed after 96 h of incubation, quantified using NIS element software as described in section “Material and methods” (*p ≤ 0.05, **p ≤ 0.01, and ***p ≤ 0.001).
Melatonin pretreatment suppressed DL- and DLA-induced angiogenesis in CAM
Furthermore, after establishing DLA-mediated angiogenesis in CAM, we sought to investigate the effect of melatonin on DLA-induced angiogenesis by giving 2 h pretreatment of melatonin (5 mM) or dexamethasone (5 µg/mL) on a disc before treating with DL cells or DLA to CAM. In Figure 6(a), quantification of images from various CAM assay showed anti-angiogenic activity of melatonin. It can be very clearly observed that groups which are treated with DL cells or DLA show relatively higher number and thicker blood vessels with more branching points, whereas the CAMs pretreated with melatonin before supplementing DL cells or DLA showed significant inhibition in branching and tertiary blood vessel development, which strongly suggested that melatonin treatment suppressed sprouting of tertiary vessels from the main vessels, a hallmark feature of tumor angiogenesis. Statistical analysis displayed 2.5- and 1.3-fold inhibition in vascularization in case of DLA and DL-induced angiogenesis, respectively. Dexamethasone was used as a positive control for anti-angiogenic activity (Figure 6(b)). The control groups with no treatment or treatment with only 5 mM melatonin showed normal vascularization with primary, secondary, and tertiary branching.
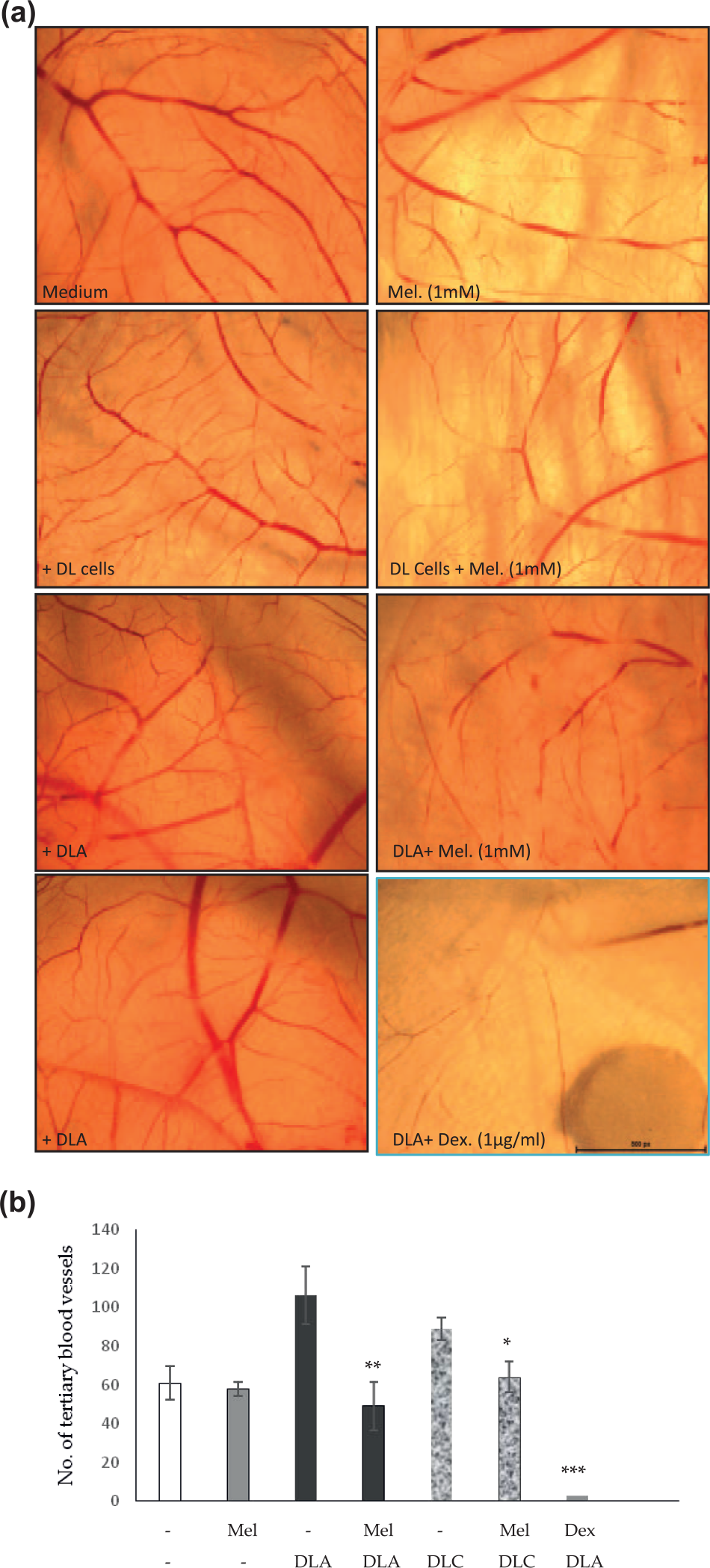
In vivo effect of melatonin on angiogenesis in chick embryo CAMs; 4-day-old developing embryos were pretreated with 50 µL of 5 mM/disc of melatonin or 12 µg/disc dexamethasone, and then, DL ascites were added. After 96 h of incubation, the CAM was harvested, photographed, counted, and plotted. (a) Photomicrograph of control groups including medium, DL cells (1 × 106), ascites, and 5 mM melatonin, and DL cells + melatonin, ascites + melatonin, and ascites + 12 µg/disc of dexamethasone were the experimental groups. (b) Graphical representation of number of tertiary blood vessels formed at 96 h of incubation of different control and pretreated chick embryo CAMs, and a number of blood vessels were quantified using NIS element software (*p ≤ 0.05, **p ≤ 0.01, and ***p ≤ 0.001).
Melatonin suppresses DL-induced angiogenesis in mice peritoneum
Many reports suggest that highly vascularized tumors are bad for survival and also results in recurrence. 36 DL can serve as reliable model for angiogenic experimentations because DL cells can be easily grown in mice abdominal cavity, and DL growth is related to the progression and enlargement of peritoneal micro-vessels. The mice were divided into three groups that included control without tumor, DL group, and melatonin-treated DL group. Melatonin (50 µg/g/body weight) was injected (IP) for 10 consecutive days starting simultaneously with DL transplantation. Peritoneal blood vessels were analyzed after 21 days post tumor transplant. Peritoneum of control mice exhibited very few micro-vessels, while the number of micro-vessels was found to be significantly high in DL-bearing mice. Melatonin treatment to DL mice resulted in considerable reduction in the development of new micro-vasculature (Figure 7(a)). Furthermore, the histological H&E-stained sections of control mice showed normal peritoneal histology. DL-bearing mice showed the symptoms of peritonitis, that is, the inflammation of peritoneum resulted from increased leukocytes infiltration clearly visible in the section at higher magnification (Figure 7(b)). The melatonin-treated mice showed reduction in peritonitis with less leukocytes infiltration and normal histoarchitecture. Increased leukocyte infiltration is the hallmark of more blood vessels with increased permeability.
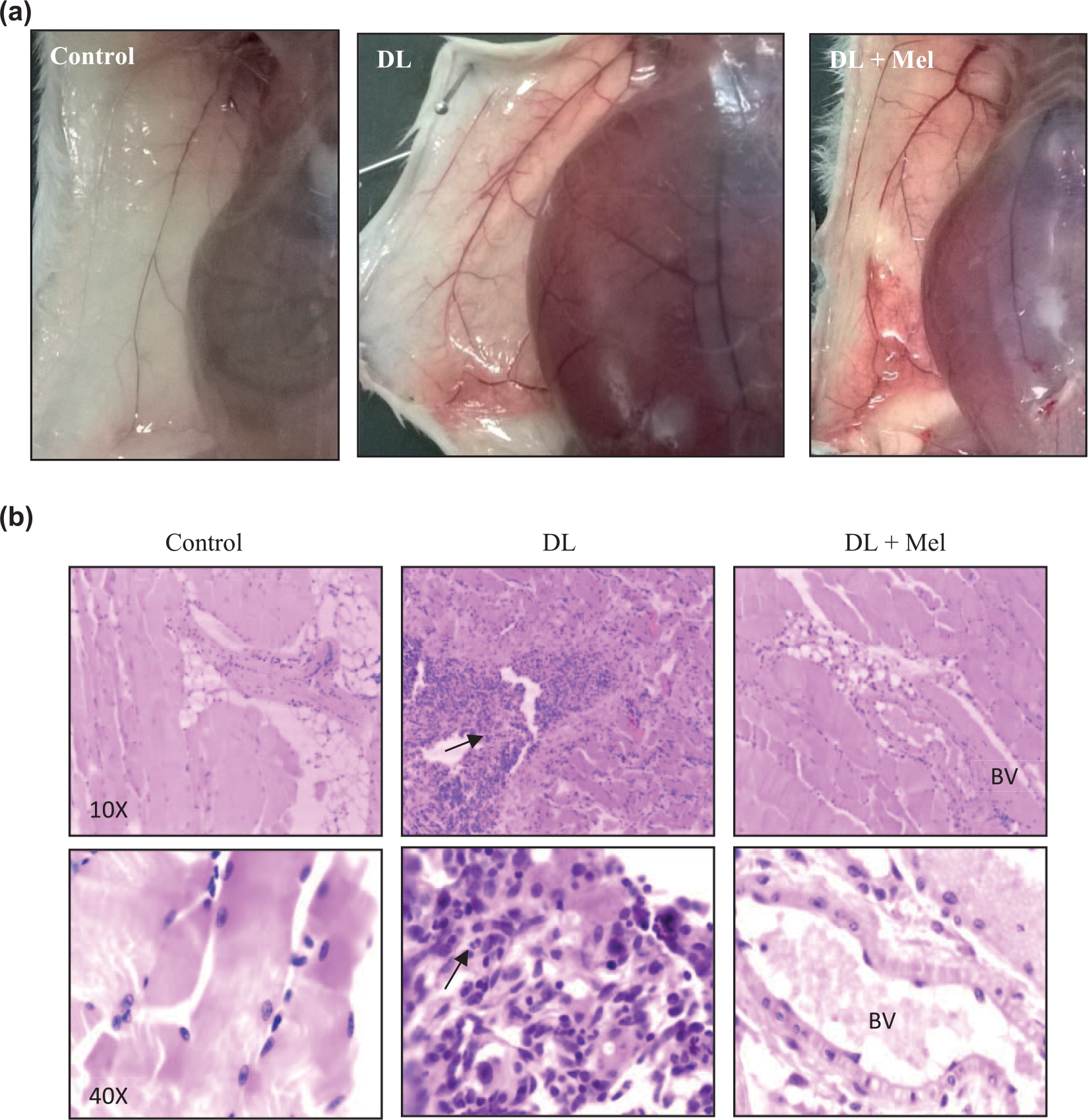
Effect of melatonin on DL-induced angiogenesis in mice peritoneum. (a) Anatomy and (b) photomicrographs of H&E-stained section of peritoneum from control and DL- and melatonin-treated mice. DL-transplanted mice were given simultaneous treatment of melatonin (50 µg/g body weight; intraperitoneal) and were continued for 10 days consecutively. The peritoneum was collected on day 21 of tumor transplantation. Control section of peritoneum revealed normal tissue architecture. Inflammation of peritoneum due to an increase in leukocyte infiltration in circulation as well as in between the loose connective tissue layer was observed in case of tumor (arrow indicates the infiltrating cells and BV represents blood vessels. (b) Melatonin-treated group showed preserved histoarchitecture of peritoneum. Magnification is 100× and 400×.
Effect of melatonin on DL-induced angiogenesis in mesentery perivasculature of mice
The mice mesentery was exteriorized from all the three groups (described in earlier section) after 21 days post tumor transplantation. Reports suggest that adult normal mice have 40–50 thin mesentery windows which are surrounded by fat tissue. Whole mount microscopic view of mesentery window from control mice showed avascular central part and very few capillaries present toward intestinal tissue. The translucent fatty part of the membranous mesentery in normal mice had no or very little vascularization. In DL-bearing mice, crowded network of capillaries originating from the pre-existing mesenteric blood vessels were developed. The capillaries were more on both intestinal side and in perivascular fat tissue (Figure 8). The melatonin-treated mice showed no capillaries in the perivascular fat tissue as well as there were very few capillaries on the intestinal tissue as compared to DL-bearing mice (Figure 8).

Effect of melatonin on angiogenic growth in mesentery. DL-transplanted mice were given simultaneous treatment of melatonin (50 µg/g body weight; intraperitoneal) and were continued for 10 consecutive days. Peritoneum and mesentery from control and DL- and melatonin-treated mice were photographed using stereo zoom microscope at 0.8× magnification on day 21 post DL transplantation. Black arrows represent perivascular fatty tissue full of micro-vessels.
Melatonin differentially regulates VEGF, VEGFR, FGF, and TIMP3 mRNA expression in DL cells and in mesenteric tissue
Induction and suppression of pro-angiogenic and anti-angiogenic genes are the key markers of angiogenic switch in cancer progression and survival. The imbalance between these genes causes activation of endothelial cell proliferation. In order to study the involvement of these genes in DLA-induced angiogenesis and its subsequent modulation by melatonin, DL cells (2 × 106 cells/well) were grown for 24 h with or without melatonin (1 mM) or dexamethasone (5 µg/mL) in six-well plate. mRNA expression of VEGF, FGF, and TIMP3 was analyzed by semiquantitative RT-PCR as well as by qRT-PCR. As DL cells are spontaneous T-cell lymphoma, we used normal thymic cells from BALB/c mice as control. There was significant decrease in expression of TIMP3 in DL cells as compared to normal thymic cells. When DL cells were treated with melatonin (1 mM for 24 h), the expression of TIMP3 increased in comparison to untreated DL cells (Figure 9(a)). Pro-angiogenic VEGF expression is reported to be higher in many cancer cells; therefore, we also investigated the expression of VEGF in DL cells. There was marked increase in VEGF mRNA expression in DL cells than in normal thymic cells. Melatonin (1 mM for 24 h) treatment led to suppression of VEGF expression (Figure 9(a)). Similarly, FGF mRNA expression in DL cells was also found to be more than normal thymic cells, which decreased with melatonin treatment (Figure 9(a)). In the mouse model, mesentery is the thinnest tissue showing change in perivascular angiogenic growth in the presence of tumor. So, we also analyzed the mesentery of normal, DL-bearing, and melatonin-treated DL mice for expression of VEGFR and TIMP3 mRNA. Melatonin treatment significantly reduced the VEGFR expression, whereas TIMP3 expression was found to be increased significantly with melatonin treatment (Figure 9(c)). The qRT-PCR results further corroborated RT-PCR data. There was significant increase in VEGF and FGF expression in DL cells as compared to normal thymic cells, whereas the TIMP3 levels were relatively low. With melatonin treatment the expression of VEGF and FGF reduced significantly (p ≥ 0.001), which was very much comparable with the control and dexamethasone groups (Figure 9(b(i)) and (b(ii))). On the contrary the melatonin treatment resulted in increase in the expression of TIMP3 to a significant level (p ≥ 0.05) (Figure 9(b(iii))). The mesentery tissue also showed high VEGFR expression in DL-bearing groups (p ≥ 0.001) and melatonin caused significant reduction in VEGFR expression (p ≥ 0.01) almost to that of control (Figure 9(d(i))). The TIMP3 expression, which was reduced in DL-bearing mice (p ≥ 0.05), was found to be induced to a significant level (p ≥ 0.001) (Figure 9(d(ii))) with melatonin treatment. Change in pro and anti- angiogenic genes VEGF, VEGFR, FGF and TIMP3 mRNA expression in DL cells and mesentery with melatonin treatment suggested that melatonin may regulate angiogenesis at transcriptional level in both in vitro and in vivo conditions.
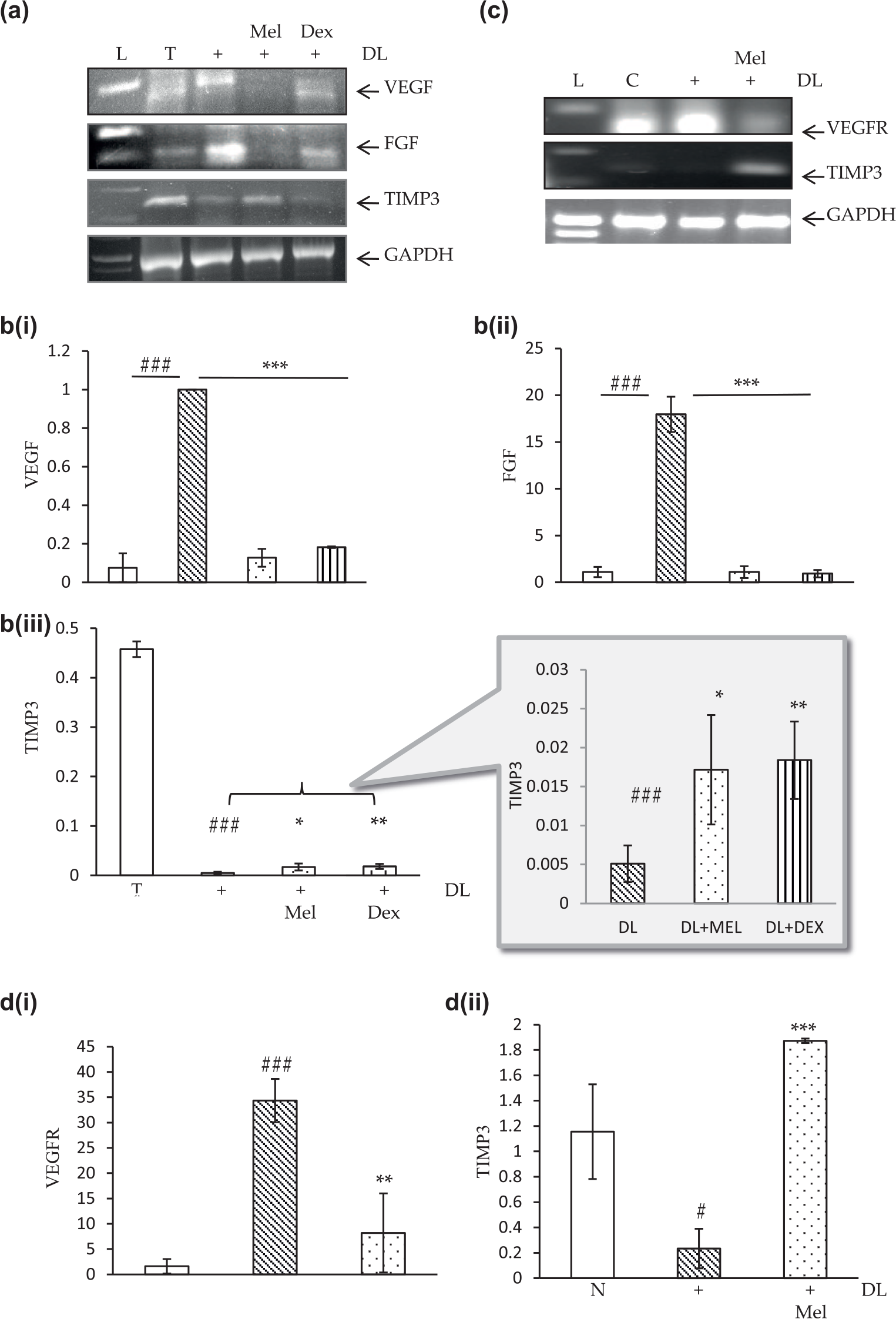
Effect of melatonin on expression of VEGF, FGF, and TIMP3 in DL cells and VEGFR and TIMP3 in mesentery. (a) Gel electrophoresis photomicrograph of RT-PCR products and (b) qRT-PCR analysis of DL cells (2 × 106 cells/mL) grown with or without melatonin (24 h) for mRNA expression of VEGF, FGF, and TIMP3 genes. (c) Gel electrophoresis photomicrograph with RT-PCR products and (d) qRT-PCR analysis of mesentery tissue obtained from control, DL-bearing, and DL-bearing melatonin-treated mice for expression of VEGFR and TIMP3 mRNA. mRNA was analyzed by semiquantitative RT-PCR and qRT-PCR as described in section “Materials and methods.” GAPDH was taken as endogenous control for RT-PCR. Fold change of the target mRNAs was normalized with that of β-actin in qRT-PCR. Values are indicated as the mean ± SD. (b(i), b(ii), b(iii), d(i), and d(ii)) Graphs showing fold change in mRNA expression of various genes.
Discussion
This study clearly demonstrates that DL can be one of the ideal angiogenesis models for both in vitro and in vivo studies. We further investigated the dose-dependent effect of melatonin on DL-induced angiogenesis.
Initially, we characterized the aortic endothelial cells with the help of endothelial cells–specific marker CD31 and also checked the normal growth profile of endothelial cells (Figure 1). After that, we observed that DLA induced significantly increased proliferation of endothelial cells isolated freshly from main thoracic aorta of mice. There was increased sprouting of tertiary micro-vessels upon incubation with DL cells or DLA in CAM as well as in mice mesentery window with in vivo DL growth. In addition, RT-PCR and qRT-PCR analyses of angiogenesis-related genes in DL cells and target mesenteric tissues revealed that regulation of angiogenesis takes place at transcriptional level.
Angiogenesis in tumor micro-environment has been found to be strongly correlated with advanced-stage disease and poor prognosis. By inducing proliferation and migration of endothelial cells, sprouting of new blood vessels takes place, which provide nutrition and oxygen supply to growing tumors that is being regulated by various factors released by the growing tumors.1,3 In normal condition, endothelial cell proliferation and migration rates are very low, but it increases radically in tumor mico-environment. 8 Therefore, it is a mandate to develop proper understanding of tumor-induced angiogenesis and its therapeutic target for inhibiting angiogenesis-mediated tumor growth.
Usually, in most of the angiogenesis-related studies, HUVECs are used as source of endothelial cells, probably because of the ease of procurement and its high proliferative and capillary forming ability. However, several reports suggest that endothelial cells show tremendous heterogeneity depending upon tissue they are located. 14 Moreover, HUVECs are not from arteries rather they are from human umbilical vein, which are very different from endothelial cells in the adult blood vessels. 30 Therefore, freshly isolated endothelial cells from micro-vessels present in adult tissues are physiologically more relevant than endothelial cells passaged several times or commercial cell lines like HUVECs. 32 Thus, to establish DL as a model system for angiogenesis and to overcome the limitations of the existing models, we isolated endothelial cells from main thoracic aorta of mice and cultured them in vitro in the presence or absence of DLA. Increased endothelial cell proliferation in the presence of DLA strongly indicated the presence of pro-angiogenic factors in DLA (Figure 2). This was further validated by increased sprouting of tertiary vessels in CAM, both with DL cells and DLA (Figures 5 and 6). Tumor growth greatly influences nearby stroma leading to angiogenesis. It is noteworthy that growing DL in the abdominal cavity of mice resulted in significant change in the peritoneum and mesentery tissue. DL-bearing mice significantly developed more number of peritoneal micro-blood vessels as compared to the normal control mice. Similar effect of DL growth has been observed previously on peritoneal vasculature. 7 In our study, we found that peritoneum showed not only increase in number of micro-vessels but also showed the symptoms of peritonitis. Enormous leukocyte infiltration was seen in peritoneal sections of DL-bearing mice. Histological sections of peritoneum of DL-bearing mice also showed more number and bigger sized blood vessels as compared to the normal mice. Mesentery assay which is an ideal system to study physiological measurements of angiogenesis in mice was also evaluated in control and DL mice.22,34 In control mice that has 40–50 thin membranous mesentery windows, covered by fatty tissue with normally no blood vessels, DL-bearing mice showed capillary formation at perivascular fatty tissue, originating from pre-existing blood vessels. The intestinal periphery also showed increased number of blood vessel branching, whereas the center of window remained devoid of vessels (Figure 8). These results thus strongly suggest that DL can induce angiogenesis and it can serve as a good model for studying angiogenesis both in in vitro and in vivo condition. Moreover, our dosage study of melatonin in DL and suppression of angiogenesis in this model further proved its advantage in screening the pharmaceutical drug or treatment regime by targeting angiogenesis.
As a potential strategy for targeting angiogenesis, we found that melatonin significantly inhibited the growth of DL cells in dose- and time-dependent manner (data not shown). As observed in adenocarcinoma, hepatocarcinoma, and cervical carcinoma previously,27,37,38 melatonin treatment resulted in growth inhibition of DL cells at 1 and 5 mM concentrations. Apart from DL, endothelial cells also showed that melatonin may have cytostatic effect. Where 1 mM (pharmaceutical dose) of melatonin is not toxic to the normal endothelial cells and the 2.5 and 5 mM are cytostatic doses (Figure 2(b)).
Melatonin not only affects the cancer cell growth but also reduces the tumor-induced angiogenesis by inhibiting endothelial cell proliferation and migration (Figures 3 and 4) One of the most interesting aspects of this study is that the DLA represents actual tumor environment, and therefore, the treatment strategies in particular melatonin in this study have been monitored in real environment. However, in most of the earlier studies for anti-angiogenic activity of melatonin on endothelial cells, VEGF was used as a mitogen on HUVECs which represents artificial condition.13,19 We checked the effect of melatonin on DLA-induced proliferation of endothelial cells obtained from main thoracic aorta of mice which normally is exposed to the tumor (DL) growing in peritoneum as ascites. There was a significant reduction in DLA-induced endothelial cell proliferation suggesting dose-dependent anti-angiogenic activity of melatonin. Melatonin showed angiostatic effect at lower concentration (1 mM) and apoptosis at higher concentration (5 mM) in proliferating endothelial cells (Figure 3). The marked inhibition in sprouting of tertiary vessels induced in vivo in CAM assay with DLA or DL cells (Figure 6) significantly validated in vitro results on DL-induced endothelial cell proliferation.
Endothelial cell migration increases the rate of new blood vessel formation, and several previous reports suggest the importance of endothelial cell migration in cancer micro-vessel development.2,5,39 Hence, we also tested the role of melatonin on endothelial cells migration to further ascertain the efficacy of melatonin on angiogenesis. Results from our study indicated that there was a significant reduction in DLA-induced migration of endothelial cells with melatonin treatment (Figure 4), interestingly suggesting anti-metastasis potential of melatonin as well.
With melatonin treatment, the peritoneum of DL-bearing mice showed fewer blood vessels and normal histological architecture with reduced “peritonitis” and less leukocyte infiltration. Mesentery vasculature network was also restored showing marked reduction in capillary branching on intestinal side as well as at perivascular fatty tissue (Figures 7 and 8).
New blood vessel development, which provides vital nutrients to the growing tumor, is regulated by intricate balance of pro- and anti-angiogenic factors such as the VEGF family genes, FGF-1, FGF-2, placental growth factor (PGF), TGF-β1, thymidine phosphorylase, pleiotrophin, and adrenomedullin as reported in the case of breast cancer.9,12,40 Many of these factors are expressed preferentially at different stages of tumor development, though some factors such as VEGF expression occurs throughout all the stages of tumor. It has been demonstrated that VEGF produced from tumor cells is essential for the progression of breast cancer and may function in both paracrine and autocrine manners to allow tumor growth and progression by inducing angiogenesis. 39 FGF also contributes in endothelial cell activation, whereas TIMP3 is an endogenous inhibitor that suppresses MMPs and reduces the disintegration of matrix to inhibit endothelial and cancer cell migration.29,36 In this study, significant angiogenic effect of DLA suggests that it might induce various growth factors to promote neovascularization. Furthermore, RT-PCR and qRT-PCR analyses indeed revealed that DL expressed relatively more VEGF and FGF in comparison to normal thymic cells contributing to increased endothelial cell proliferation and vessel development. However, relative lower expression of TIMP3 in DL cells than in normal thymic cells implied pro-angiogenic effect. Evaluation of melatonin treatment to DL cells for 24 h resulted in downregulation of VEGF and FGF expression; however, it induced TIMP3 expression (Figure 9(a) and (b)). Similarly, the exteriorized mice mesenteries of DL mice showed increased VEGFR and reduced TIMP3 expression which upon melatonin treatment was found to be restored showing suppression of VEGFR and increased expression of TIMP3 (Figure 9(c) and (d)). These results clearly demonstrate that melatonin-mediated anti-angiogenic effect was regulated at transcriptional level by altering the expression of VEGF, FGF, VEGFR, as well as TIMP3. Increase in TIMP3 mRNA expression upon melatonin treatment in DL as well as in mesenteric tissue of DL mice also suggests anti-metastasis potential of melatonin.
These results further strengthen that DL can be a good model to study angiogenesis followed by evaluation of expression of genes involved in angiogenesis, and finally, a good system to find angiogenesis-mediated tumor treatment regime.
Conclusion
In this study, we have discussed about the possibility of DL as an efficient model for in vitro and in vivo analyses of angiogenesis in cancer and other angiogenesis-related diseases. This study very convincingly established that DL led to increase in endothelial cell proliferation, migration, and sprouting of tertiary blood vessels involving regulation of expression of genes like VEGF, VEGFR, FGF, and TIMP3 at transcriptional level. Second, for the first time, it was shown that anti-tumor activity of melatonin against DL in mice involved multiple mechanisms including strong anti-angiogenic activity and inhibition of endothelial migration along with direct inhibition of tumor cell proliferation. The anti-angiogenic and anti-metastasis effects of melatonin were regulated at transcription level. Today, the investigators are interested in finding the drugs, which has the anti-tumor as well as anti-angiogenic potential to reduce the risk of invasion and metastasis, for which a good angiogenesis model is required and DL can serve as efficacious model.
Footnotes
Acknowledgements
The authors are thankful to Prof. Namita Agrawal for critical scientific discussion and providing microscopy facility and Prof. Umesh Rai for providing qRT-PCR facility.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by research grants to Dr Anju Shrivastava from University of Delhi (R&D Research Grant) and DST-PURSE. Ms Rani Kumari and Ms Kavita Rawat are JRF of University Grants Commission, New Delhi.
