Abstract
Gastric cancer is one of the most common and aggressive malignancies. Both bacterial virulence factors and host chronic inflammation are thought to promote gastric cancer development. In this study, we investigated the potential involvement of follicular helper T cells in gastric cancer. Functions of follicular helper T subsets were examined in
Introduction
Approximately 50% of the world population is infected with
However, infection by high-risk
Recently, the participation of follicular helper T (Tfh) cells in cancer is being increasingly recognized. The germinal center (GC) Tfh cell is the key cell type in the formation and maintenance of the GC, which allows B cell proliferation, somatic hypermutation, and plasmablast/plasma cell generation.15,16 In the peripheral blood, the CD4+CXCR5+ T cells, but not CD4+CXCR5− T cells, are capable of inducing naive B cell plasmablast differentiation and long-lasting humoral immunity and are considered GC Tfh-equivalent.17–19 Studies have also found that peripheral Tfh cells were composed of Th1-like, Th2-like, and Th17-like subsets, based on their mutually exclusive IFN-γ, IL-4, and IL-17 expression.
18
These results suggest a possibility that in addition to enhancing B cell responses, Tfh cells could also modulate immunity through cytokine production. Interestingly, favorable outcomes were found in breast cancers with high Tfh infiltration in the tumor.
20
Similarly, impaired Tfh cell function was associated with progression of hepatitis B virus (HBV)-related hepatocellular carcinoma.
21
These studies demonstrated that Tfh cells contributed to anti-tumor immunity. In gastric cancer development, the roles of Tfh cells and Tfh subsets are still unclear. Given that gastric cancer is associated with chronic Th1-type inflammation, the involvement of different Tfh subsets in reducing and/or increasing the risk of gastric cancer in
Materials and methods
Subjects
A total of 16
Flow cytometry
Fluorescently labeled monoclonal anti-human antibodies, including CD3, CD4, CXCR5, CD45RA, CD45RO, IFN-γ, IL-4, IL-17, CXCR3, CCR6, and IL-10, as well as corresponding isotype controls, were purchased from BioLegend (San Diego, CA, USA). PBMCs were stimulated with 2 µg/mL of Staphylococcal enterotoxin B (SEB) for 24 h at 37°C in 5% CO2. GolgiStop and GolgiPlug (BD Pharmingen, Franklin Lakes, NJ, USA) were added to PBMCs 5 h before staining. The PBMCs were then washed in fluorescence-activated cell sorting (FACS) buffer (2% fetal bovine serum (FBS) in phosphate-buffered saline (PBS)) and treated with surface antibodies and Dead Cell Stain Aqua (Invitrogen, San Diego, CA, USA) for 30 min at 4°C. Excess surface antibodies were removed by two washings in FACS buffer, and the PBMCs were fixed and permeabilized in CytoFix/CytoPerm (BD Pharmingen). Intracellular antibodies were dissolved in 1× Perm Wash (BD Pharmingen) and incubated with PBMCs for 30 min at 4°C. The cells were then washed twice in 1× Perm Wash and analyzed in BD FACSCanto cytometer (Franklin Lakes, NJ, USA).
For live sorting of Th1–Tfh, Th2–Tfh, Th17–Tfh, and CD19+IgD+CD27− naive B cells, PBMCs were surface stained with CD3, CD4, CXCR5, CXCR3, and CCR6 or with CD19, IgD, and CD27 for the fractionation of Tfh cells or the isolation of B cells, respectively. Excess antibodies were removed by washing twice in FAS buffer and sorted in BD FACSAria cell sorter (Franklin Lakes, NJ, USA).
Tfh–B cell coculture
Sorted Tfh cells and B cells were cocultured at 1 × 105 cells per mL each, in the presence of 2 µg/mL SEB, under 37°C and 5% CO2. Cell supernatant was harvested and replaced for every 3 days until day 12. In some experiments, Th1–Tfh cells were added at 1 × 105 to 5 × 105 cells per mL. A volume of 10 µg/mL IFN-γ-neutralizing antibody (NIB42) or corresponding isotype controls were added to the coculture as indicated.
Enzyme-linked immunosorbent assay
Enzyme-linked immunosorbent assay (ELISA) for human immunoglobulin (Ig)M, IgG, and IgA was performed using commercial kits from Abcam (Cambridge, UK). ELISA for human IFN-γ, IL-4, and IL-17A was performed using commercial kits from eBioscience (Waltham, MA, USA). Values below the respective detection limit in each kit were assigned a value of zero.
Statistical analyses
For comparisons between two groups, Mann–Whitney test was performed on samples from different individuals, while Wilcoxon matched-pairs test was performed on samples from the same individual but were treated under different experimental conditions. For comparisons between multiple groups, one-way or two-way analysis of variance (ANOVA) was performed. Pearson correlation test was used to examine correlations. Two-tailed p value less than 0.05 was used considered to be significant. All tests were computed by GraphPad Prism (GraphPad Software, La Jolla, CA, USA).
Results
Tfh cells in gastric cancer patients were skewed to the Th1/Th17 type
In peripheral blood, 8.7% ± 2.4% CD4+ T cells in stage I/II and 9.7% ± 2.6% CD4+ T cells in stage III/IV gastric cancer patients expressed CXCR5 (Figure 1(a) and (b)), which was on par with that in non-cancer controls (NCs). The vast majority of CD4+CXCR5+ Tfh cells were CD45RO+ (Figure 1(c)), suggesting that they were memory T cells but not naive T cells.

Circulating Tfh cells in gastric cancer patients and non-cancer controls (NCs). (a) The CXCR5-expressing cells are gated in CD4+ T cells using an isotype control as a negative reference. All cells shown were live CD3+ T cells determined by Aqua Dead cell stain and appropriate FCS/SSC gates (not shown). (b) Percentages of circulating CD4+ T cells expressing CXCR5 in NCs, stages I-to-II gastric cancer individuals (I/II), and stages III-to-IV gastric cancer individuals (III/IV). One-way ANOVA followed by Tukey’s test (NS: not significant). (c) Expression of CD45RO by CD4+CXCR5+ T cells.
To determine their subset differentiation, the expression of IFN-γ (Th1 marker), IL-4 (Th2 marker), and IL-17 (Th17 marker) was examined in SEB-treated T cells. The frequencies of IFN-γ and IL-17 expressing CD4+CXCR5+ Tfh cells were significantly increased in gastric cancer patients than in NC (Figure 2(a)). Consistent with previous findings, 18 The IFN-γ, IL-4, and IL-17 expressing CD4+CXCR5+ Tfh cells were strongly concentrated in the CXCR3+CCR6−, the CXCR3−CCR6−, and the CXCR3−CCR6+ subsets (Figure 2(b)), respectively.

Circulating Tfh cell subset composition in gastric cancer patients and non-cancer controls (NCs). (a) Percentages of circulating CD4+CXCR5+ Tfh cells expressing IFN-γ, IL-4, or IL-17 after the PBMCs from gastric cancer patients (Cancer) and NCs were treated with 2 µg/mL SEB for 24 h. Mann–Whitney test was performed (*p < 0.5; **p < 0.01). (b) Expression of CXCR3 and CCR6 by circulating Tfh cells. (c) Secretion of IFN-γ, IL-4, or IL-17 after circulating Tfh cells were sorted according to CXCR3 and CCR6 expression and stimulated with 2 µg/mL SEB for 24 h. Data shown represent mean ± SD of three Cancer and three NC individuals.
Th1–Tfh enhanced IgG antibody responses
The association between effector cytokine production and surface chemokine receptor expressions enabled us to fractionate total circulating Tfh cells into Th1-like, Th2-like, and Th17-like subsets. To examine the effects of Th1–Tfh and Th17–Tfh upregulation in gastric cancer patients, we examined their capacity in helping B cell antibody production. Naive CD19+IgD+CD27− B cells were cocultured with purified Th1–Tfh, Th2–Tfh, or Th17–Tfh cells in the presence of 2 µg/mL SEB for 12 days. The Ig content in the supernatant was examined for every 3 days by ELISA. A naive B cell–only culture was maintained as the negative control. We found that Th1–Tfh cells did not, by themselves, induce significant IgM, IgG, and IgA production from B cells, while both Th2–Tfh and Th17–Tfh induced significant amounts of IgM, IgG, and IgA (Figure 3(a)). Interestingly, when Th1–Tfh was added to Th2–Tfh and Th17–Tfh coculture with B cells, we observed a significant upregulation of IgG production, which was associated with the amount of Th1–Tfh added (Figure 3(b)). Given that the Th1–Tfh cells include IFN-γ-producing cells and that IFN-γ is a class switch factor for IgG,23,24 we suspected that the Th1–Tfh cells promoted IgG class switch through IFN-γ production. Indeed, neutralization of IFN-γ resulted in lowered IgG production (Figure 3(c)). Interestingly, the IgG isotype made up a larger percentage of total serum Ig in gastric cancer patients compared to healthy controls (Figure 3(d)). Together, these results demonstrated that Th1–Tfh themselves did not induce Ig production of B cells but could assist the IgG secretion in collaboration with other Tfh subsets through IFN-γ production.

Capacity of Tfh subsets in B cell help. (a) The Ig concentration in culture supernatant of naive CD19+IgD+CD27− B cell and Th1–Tfh, Th2–Tfh, or Th17–Tfh cell coculture (at 1 × 105 B + 1 × 105 T per mL). A volume of 2 µg/mL SEB was added and 100 µL supernatant was harvested for ELISA and replaced for every 3 days. Two-way ANOVA with Sidak’s multiple-comparison test was performed (**p < 0.01; ***p < 0.001). (b) The IgG concentration from the day 9 to day 12 supernatant of naive CD19+IgD+CD27− B cell and Th2–Tfh or Th17–Tfh coculture, without Th1–Tfh cells, or with Th1–Tfh supplemented at 1 × 105 (1:1 Th2/Th17 to Th1 ratio) to 5 × 105 (1:5 Th2/Th17 to Th1 ratio) cells per mL. RM one-way ANOVA with Tukey’s multiple-comparison test was performed (*p < 0.05; **p < 0.01; ***p < 0.001). (c) IgG production with isotype control (Ctrl Ab) or with anti-human IFN-γ neutralizing antibody (IFN-γ neut.), 10 µg/mL each. The IgG concentration from the day 9 to day 12 supernatant of naive CD19+IgD+CD27− B cells and all Tfh cells (unfractionated CD4+CXCR5+ T cells) was measured by ELISA. Wilcoxon matched-pairs test was performed (**p < 0.01). (d) Serum Ig isotype composition in NC and gastric cancer patients. Two-way ANOVA followed by Sidak’s test (*p < 0.05).
Th1–Tfh prevented regulatory B cell development
Recently, the crucial role of B cells in suppressing T cell inflammation has been increasingly recognized in autoimmune disorders, infectious diseases, and tumors.25,26 Several subsets of B cells, including CD24hiCD38hi B cells, B10 cells, and plasmablasts, have shown IL-10 secretion capacity and regulatory activity in numerous studies. These cells are collectively termed as regulatory B cells (Bregs).27–30 Interestingly,
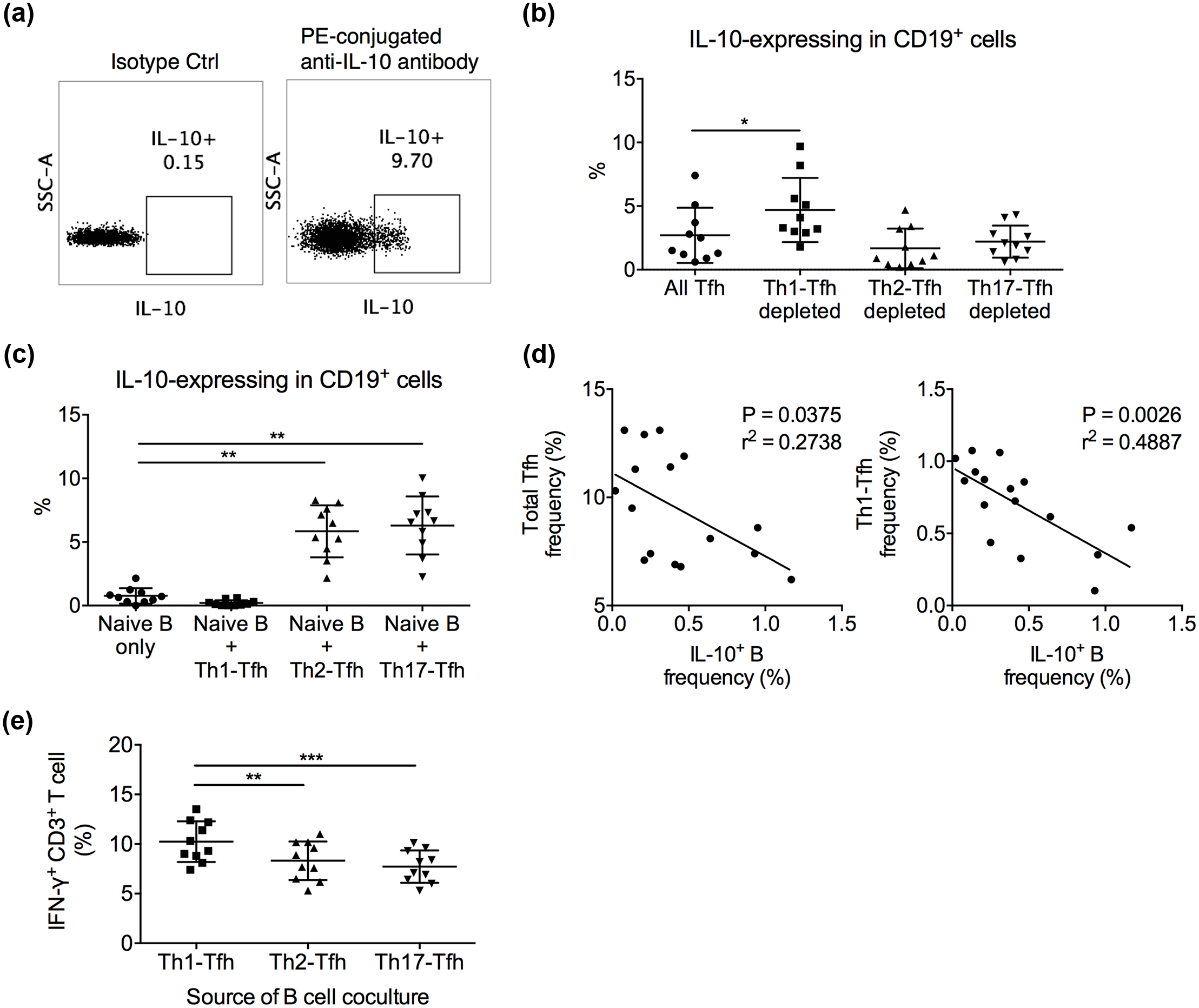
Capacity of Tfh subsets in inducing regulatory B cells. (a) The gating of IL-10-expressing B cells was set using an isotype control of anti-human IL-10 antibody as the negative gate. Cells shown were pre-gated on live CD3−CD19+ lymphocytes in the PBMC of one gastric cancer individual. (b) Naive CD19+IgD+CD27− B cells were cocultured with whole Tfh cells, or Th1-Tfh-, Th2-Tfh-, or Th17-Tfh-depleted Tfh cells at 1 × 105 B + 1 × 105 T per mL with 2 µg/mL SEB. IL-10 expression in B cells was examined by flow cytometry at day 12. Friedman test was followed by Dunn’s multiple-comparison test (*p < 0.05). (c) Naive CD19+IgD+CD27− B cells were cultured alone or with purified Th1–Tfh, Th2–Tfh, or Th17–Tfh cells at 1 × 105 B + 1 × 105 T per mL with 2 µg/mL SEB. IL-10 expression in B cells was examined by flow cytometry at day 12. Friedman test was followed by Dunn’s multiple-comparison test (**p < 0.01). (d) The frequency of IL-10-expressing B cells was inversely correlated with the frequency of total Tfh cells and the frequency of Th1–Tfh cells (calculated by multiplying the frequency of Th1-like cells in Tfh (Figure 2(a)) with the frequency of Tfh in CD4+ T cells (Figure 1(b)) in the peripheral blood of gastric cancer patients. A line of best fit is shown. Pearson correlation test was performed. (e) Anti-CD3/CD28 (2 µg/mL each)-stimulated CD3+ T cells cocultured with CD19+ B cells harvested from the Th1–Tfh coculture presented higher IFN-γ secretion than CD3+ T cells with B cells from Th2–Tfh or Th17–Tfh cocultures. Friedman test was followed by Dunn’s multiple-comparison test (**p < 0.01; ***p < 0.001).
Low Th1–Tfh frequency was associated with better survival
The gastric cancer patients were followed-up for 2 years after receiving curative resection. The months of survival from the day of surgery to tumor recurrence or death were recorded. We found that low frequency of Th1–Tfh cells was associated with better survival. The frequency of total Tfh cells, Th2–Tfh cells, or Th17–Tfh cells was not correlated with patient survival time after surgery (Figure 5).

Gastric cancer patient survival and Tfh subset frequency. The disease status of all gastric cancer subjects in this study was monitored for 2 years. The number of months of disease-free survival from the date of tumor resection was recorded and correlated with the peripheral blood frequency of total Tfh, Th1–Tfh, Th2–Tfh, or Th17–Tfh cells. Pearson correlation test was performed.
Discussion
In this study, we demonstrated a dysregulation of Tfh cells in gastric cancer patients, characterized by an overrepresentation of the IFN-γ-expressing Th1–Tfh subset and the IL-17-expressing Th17–Tfh subset. Interestingly, the frequency of total Tfh cells and Th1–Tfh cells was negatively correlated with the disease-free survival time of gastric cancer patients after tumor resection. These results are consistent with earlier findings that IFN-γ-mediated immunity was a driving factor in gastric atrophy and suggested that dysregulated Tfh cells contributed to cancer development.
We also demonstrated the novel finding that although Th1–Tfh cells by themselves were ineffective at helping B cells to mount a robust IgG response, they could promote the IgG response in collaboration with other Tfh subsets through IFN-γ production in a cell number–dependent manner. Neutralization of IFN-γ reverted this effect. The role of humoral responses in the clinical outcome of
Immunoregulatory functions mediated by IL-10-secreting B cells are crucial in suppressing excessive inflammation in multiple autoimmune disorders.
36
In cancer, several studies suggested that regulatory functions mediated by B cells possibly contributed to disease progression. Significant infiltration of B cells was demonstrated in multiple human solid tumors.
37
In a murine breast cancer model, tumor-evoked regulatory B cells possibly promoted the conversion of resting CD4+ T cells to Foxp3+ regulatory T (Treg) cells, which promoted lung metastasis.
38
Regulatory B cells also accelerated the growth of hepatocellular carcinoma through the CD40/CD40L signaling.
39
In contrast, regulatory B cells appeared to protect
Footnotes
Acknowledgements
H.Z., R.Y., P.Z., and X.Y. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Basic and Advanced Technology Research Funding of Kaifeng City (1508001).
