Abstract
Inorganic polyphosphate is a linear polymer containing tens to hundreds of orthophosphate residues linked by high-energy phosphoanhydride bonds. Polyphosphate has been recognized as a potent anti-metastasis reagent. However, the molecular mechanism underlying polyphosphate action on cancer cells is poorly understood. In this study, we investigated the involvement of polyphosphate in radio-sensitivity using a human non–small cell lung cancer cell line, H1299. We found that polyphosphate treatment decreases cellular adenosine triphosphate levels, suggesting a disruption of energy metabolism. We also found that the induction of DNA double-strand breaks was enhanced in polyphosphate-treated cells after X-ray irradiation and colony formation assay revealed that cell survival decreased compared with that of the control groups. These findings suggest that polyphosphate is a promising radio-sensitizer for cancer cells. Therefore, we hypothesized that polyphosphate treatment disrupts adenosine triphosphate–mediated energy transfer for cellular survival and DNA repair, thereby reducing the cellular capability to resist X-ray irradiation.
Keywords
Introduction
Non–small cell lung cancer (NSCLC) is one of the most common causes of cancer-related death around the world. 1 The most common postoperative therapy against NSCLC is platinum chemotherapy combined with radiation therapy. 2 In spite of the recent breakthroughs in medical technology, the 5-year survival rate of patients with stage III–IV NSCLC treated with a combination of radiotherapy and chemotherapy is 10%–20%.3,4 More than 50% of NSCLCs involve p53 mutations, 5 which is a negative prognostic factor for radiotherapy and chemotherapy.6,7 In this study, we used the p53-null NSCLC cell line H1299 to check whether inorganic polyphosphate (polyP) can be used as a radio-sensitizer.
PolyP is a polymer containing tens or hundreds of orthophosphate (Pi) residues linked by high-energy phosphoanhydride bonds. PolyPs exist in all living organisms, including bacteria and fungi, and in smaller amounts in plants and animals.8,9 Although polyP function in mammalian cells is poorly understood, many important physiological functions have been clarified recently. For example, polyP mediates astroglial signal transduction via calcium-dependent P2Y purinergic receptor signaling in mammalian brain 10 and promotes osteocalcification in both mouse and human osteoblastic cells.11–14 PolyP also blocks tumor metastasis by inhibiting basic fibroblast growth factor (b-FGF)-induced proliferation and ERK/p38 activation in melanoma cells 15 and induces apoptosis in human plasma cells. 16 The exopolyphosphatase activity of human protein h-prune (the DHH superfamily), which is overexpressed in metastatic cancers, is also modulated by polyP. 17 This highlights the important role of polyP not only in physiological functions but also in therapeutic applications. PolyP also influences the mitochondrial ion transport system via modulation of the mitochondrial permeability transition pore (mPTP).18–21 It is therefore clear that polyP plays a key role in cell death and in controlling mitochondrial metabolism.22,23
Radiation therapy is a powerful approach to eliminate tumor tissue. After irradiation with high-energy photon beams such as linac 6 MV X-rays, DNA double-strand breaks (DSBs) are induced along radiation particle, which is the essential damage for dictating cell survival. 24 Histone H2AX surrounding the DSBs is rapidly phosphorylated on serine 139 (gamma-H2AX foci) by ataxia-telangiectasia mutated (ATM), ATM and Rad3-related (ATR), and DNA-dependent protein kinase (DNA-PK). 25 Phosphorylation of H2AX is one of the initial responses to irradiation and triggers the signal transduction associated with DNA repair, cell proliferation, and cell death. 26
Because surviving tumor cells after irradiation adversely affect patient prognosis, an effective radio-sensitizer is essential for radiation therapy.
To study the effect of polyP on radio-sensitivity, we used a human NSCLC cell line, H1299.
Materials and methods
Cell culture and reagents
The human NSCLC cell line, H1299, was purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). The cells were maintained in Dulbecco’s Modified Eagle’s Medium (DMEM; Sigma–Aldrich Co., St. Louis, MO, USA) supplemented with 10% fetal bovine serum (FBS; Cansera International Inc., Toronto, ON, Canada), 100 unit/mL penicillin, and 100 µg/mL streptomycin (Sigma–Aldrich Co., St. Louis) at 37°C in a humidified atmosphere containing 5% CO2. PolyP (the average chain length was 120 residues) was kindly provided by Dr Shiba (RegeneTiss Inc., Tokyo, Japan).
Cell growth
To compare the growth of polyP-treated cells with that of control cells, 2500 cells were seeded in each well of 96-well plates. After 24 h, the cells were treated with culture medium supplemented with 1 mM sodium phosphate (pH 6.8) and 1 mM polyP. The cell growth was then monitored by MTS assay (CellTiter 96® AQueous One Solution Cell Proliferation Assay; Promega Corporation, Madison, WI, USA). Absorbance was measured at 490 nm using the Model 680 Microplate Reader (Bio-Rad Laboratories, Inc., Hercules, CA, USA).
Cell viability
Cell viability after X-ray irradiation was monitored using both the MTS and the colony formation assays. For the MTS assay, 2500 cells were seeded in each well of 96-well plates. After 24 h, the cells were treated with 1 mM sodium phosphate (pH 6.8)–containing culture medium and 1 mM polyP-containing culture medium. Then the cells were irradiated with X-rays (10 Gy), and their growth was assessed by MTS assay. The MTS assay was performed using CellTiter 96 AQueous One Solution Cell Proliferation Assay (Promega Corporation). Absorbance was measured at 490 nm by the Model 680 Microplate Reader (Bio-Rad Laboratories, Inc.). For colony formation assay, the cultured cells were seeded onto a culture flask (25 cm2 growth area) and incubated for 24 h (2 × 105 per flask). The cells were then treated with control and polyP medium overnight. The treated cells were then irradiated with X-rays (10 Gy) and were immediately trypsinized. Then 104–105 viable cells were re-plated onto a culture dish (60 mm in diameter) and were cultured in DMEM containing each treatment condition. After 14 days, the grown cells were fixed with methanol and stained with 2% of Giemsa solution (Kanto Chemical Co., Inc., Tokyo, Japan) to count the number of colonies per dish. The number counted was corrected using the plating efficiency of the non-treated cells. The plating efficiency of control cells was 60%.
Detection of DSBs
DSBs induced after irradiation were detected by immunofluorescence staining using an anti-γH2AX antibody (Abcam Plc, Cambridge, UK) as γ-H2AX foci. The cells grown on a glass bottom dish were treated with 1 mM sodium phosphate (pH 6.8)–containing culture medium and 1 mM polyP-containing culture medium for 16 h and then irradiated with 10 Gy of 6 MV X-rays. Thirty minutes or 3 h after irradiation, the cells were fixed with 4% paraformaldehyde solution. Immunofluorescence staining was performed according to the procedure in our previous report. 27 The fluorescent cells were imaged by fluorescence microscopy (BZ-9000; Keyence Corporation, Osaka, Japan). To evaluate the relative change in the number of the γ-H2AX foci, fluorescence intensity per nucleus was measured using ImageJ, which was provided free of charge by the Research Services Branch (RSB) of the National Institute of Mental Health (NIMH). 28
Measurement of adenosine triphosphate levels
The cells were seeded on a 96-well plate at a density of 3000 cells/well and cultured for 16 h. Then the cell culture medium was replaced by a fresh culture medium containing 1 mM sodium phosphate and 1 mM polyP. After 16 h of culturing, the cells were irradiated with 10 Gy of the X-ray. The changes in cellular adenosine triphosphate (ATP) levels after irradiation were measured using the CellTiter-Glo® 2.0 Assay kit (Promega Corporation) at the indicated time. Chemical luminescence was detected by AVRO MX (PerkinElmer, Inc., Waltham, MA, USA).
Irradiation condition
The cells cultured in the culture flask on a water-equivalent phantom with a 50-mm-thickness were irradiated with 10 Gy at the side of the bottom of the cell flask with 6 MV therapeutic X-rays (Mitsubishi Electric Co., Tokyo, Japan) at a dose rate of 250 MU/min at room temperature. The absorbed dose in water was determined according to the dose protocol of the Japanese Standard Dosimetry 12. 29
Statistical analysis
Statistical analyses were performed using IBM SPSS version 22 (IBM Corporation, Armonk, NY, USA).
Results
PolyP treatment does not affect cell growth
Because polyP can induce apoptosis in human plasma cells, 16 we first performed the MTS assay after polyP treatment to examine its effect on cell growth. When comparing the cell growth among treatment groups, the control cells (without treatment and sodium phosphate) and polyP-treated cells exhibited no significant difference at the indicated time (Figure 1).

Cell growth after treatment with polyP in comparison with that of non-treated and sodium phosphate–treated cells. The cell growth was monitored by MTS assay. The solid triangle (▲) indicates the cells treated with 1 mM sodium phosphate (pH 6.8); the solid square (■) designates the cells treated with 1 mM polyP; and the open circle (○) denotes the non-treated cells. The plots represent the mean ± SD of quintuplicate experiments.
PolyP affects cell viability after 10 Gy irradiation
Next, we investigated the effects of polyP treatment on cell viability after irradiation. Cell viability after 10 Gy irradiation was measured by the MTS and colony formation assays. From the result of the MTS assay until 3 days after irradiation, the cell viabilities were almost the same among the three groups (without treatment, sodium phosphate, and polyP-treated), although the cell growths in the all groups were depressed in comparison with those without irradiation (Figure 2(a)). On the contrary, decreased cell viability was observed in polyP-treated cells compared with that of the control groups by the colony formation assay (Figure 2(b)).

Cell viability after exposure to 10 Gy irradiation as measured by (a) MTS assay and (b) colony formation assay. The solid triangle (▲) indicates the cells treated with 1 mM sodium phosphate (pH 6.8), the solid square (■) designates the cells treated with 1 mM polyP, and the open circle (○) denotes the non-treated cells. The values represent the mean ± SD of quintuplicate experiments for the MTS assay and triplicate experiments for the colony formation assay. *p <0.05 were determined by Mann–Whitney U test in the colony formation assay.
Phosphorylation of histone H2AX was enhanced in polyP-treated cells
Serine 139 on histone H2AX is phosphorylated by DNA DSBs. 25 We measured the change in the number of phosphorylation of histone H2AX per cell nucleus after irradiation by observing fluorescent intensity. 30 Thirty minutes after irradiation, the intensity of γ-H2AX increased for all treatment groups, and there were no statistically significant differences among them. However, 3 h after irradiation, fluorescence intensity increased significantly for the polyP-treated cells compared with that of the control groups (Figure 3(a)). Since phosphorylation of H2AX depends on the number of DSBs,25,27–31 this result suggests that DSBs increased specifically in the polyP-treated cells after irradiation (Figure 3(b)).

The number of DSBs after exposure to 10 Gy irradiation, as detected by immunofluorescence staining using an anti-γH2AX antibody as the γ-H2AX foci at the time indicated. (a) Integrated fluorescence intensity per nucleus area and (b) immunofluorescence staining using an anti-γ-H2AX antibody at 3 h after irradiation. The experiments were performed independently in triplicate. p-Values were determined by Student’s t-test. ***p < 0.001; **p < 0.01; *p < 0.05.
PolyP reduces cellular ATP levels
Because polyP plays an important role in the mitochondrial activity as a modulator associated with mPTP opening, 23 we investigated the changes in the cellular ATP levels after polyP treatment. The ATP levels increased for all the treatment groups after irradiation, except for the polyP-treated cells, wherein the ATP levels were consistently lower compared with those of the control groups (Figure 4).
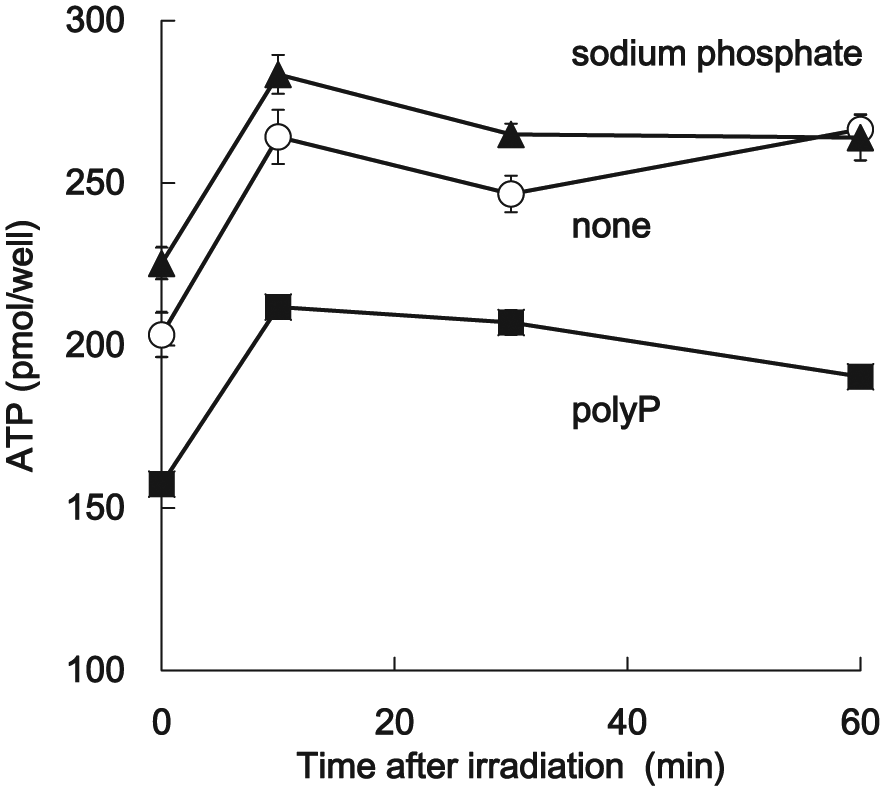
Assessment of the cellular ATP levels. The changes in the cellular content of ATP after X-ray irradiation were measured by CellTiter-Glo® 2.0 Assay kit (Promega Corporation). The solid triangle (▲) indicates the cells treated with 1 mM sodium phosphate (pH 6.8), the solid square (■) designates the cells treated with 1 mM polyP, and the open circle (○) denotes the non-treated cells. The values represent the mean ± SD of quintuplicate experiments.
Discussion
In this study, we investigated the role of polyP in radio-sensitivity using the human NSCLC cell line, H1299. PolyP treatment showed no effect on cell growth (Figure 1). Despite the fact that the short-term cell survival was not altered by polyP treatment after irradiation (Figure 2(a)), the long-term survival of polyP-treated cells decreased compared with that of the control groups (Figure 2(b)). These results suggest that although polyP does not affect the early cellular response leading to cell death (including apoptosis; Figure 2(a)), it does induce a long-term effect leading to cell death involving the reproductive death (mitotic death). Indeed, the formation of DSBs increased specifically in the polyP-treated cells 3 h after irradiation (Figure 3(a)). Accompanying with the results of Figure 3(a) at 0.5 h that there were no differences in phosphorylated-H2AX between those three treatments, none (without treatment), sodium phosphate, and polyP, the accumulation of phosphorylated-H2AX in polyP-treated cells at 3 h after irradiation may be due to the inhibition of the DNA repair pathways under the condition of low ATP.
Figure 4 illustrates that polyP reduces the cellular ATP levels (Figure 4). PolyP acts as an ATP reservoir relative to the activity of polyP metabolic enzymes,8,9 and ATP is mainly produced by mitochondrial ATP synthase. 32 Therefore, we hypothesize that polyP might affect the mitochondrial ATP metabolism, such as ATP synthase activity, ATPase activity, and adenylate kinase activity. PolyP also plays an essential role in mitochondrial ion transport by regulating the mPTP, 33 while mPTP opening is strongly associated with cell death. 34 Furthermore, Elustondo et al. 23 reported that the composition of the ATPase C-subunit, polyhydroxybutyrate, and polyP is required for the mPTP opening. Mitochondrial ATP is also strongly associated with cell death, 35 and, consequently, the increased radio-sensitivity of polyP-treated cells would be due to a depletion of mitochondrial ATP. It is possible that polyP alters the ATP metabolism, which results in the reduction of DNA repair and which may lead to increased radio-sensitivity. Because cancer cells use ATP as an energy source for tumor progression, 36 the reduction in the ATP levels by polyP would lead to a depleted energy source, thereby neutralizing tumor progression.
Conclusion
To our knowledge, this is the first study to investigate the effect of polyP on cell survival after X-ray irradiation. Although polyP did not affect the growth of H1299 cells on its own, the viability of polyP-pretreated cells decreased compared with that of the control group, in addition to increasing DSBs. Furthermore, polyP reduced the cellular ATP levels. These findings suggest that polyP has the potential to increase cellular radio-sensitivity by controlling the mitochondrial ATP metabolism and interfering with DNA repair.
Although further investigation would be necessary to clarify the specific mechanisms underlying polyP action on the mitochondrial function, polyP is related to the inhibition of tumor metastasis, 15 as found in this study. Thus, polyP may be available as a sensitizer in radiotherapy by combining decreased tumor metastasis with depletion of energy source for tumor progression via the modulation of mitochondrial metabolism.
Footnotes
Acknowledgements
The authors thank Dr Evgeny Pavlov for giving them a precise and helpful advice.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid for Scientific Research, which was provided by the Ministry of Education, Culture, Sports, Science and Technology of Japan and a Grant-in-Aid for Scientific Research from the Faculty of Health Sciences, Hokkaido University, Japan.
