Abstract
The aim of this study is to detect the accumulation status of organochlorine pesticides in breast cancer patients and to explore the relationship between organochlorine pesticides contamination and breast cancer development. We conducted a hospital-based case–control study in 56 patients with breast cancer and 46 patients with benign breast disease. We detected the accumulation level of several organochlorine pesticides products (β-hexachlorocyclohexane, γ-hexachlorocyclohexane, polychlorinated biphenyls-28, polychlorinated biphenyls-52, pentachlorothioanisole, and pp′-dichlorodiphenyldichloroethane) in breast adipose tissues of all 102 patients using gas chromatography. Thereafter, we examined the expression status of estrogen receptor, progesterone receptor, human epidermal growth factor receptor-2 (HER2), and Ki-67 in 56 breast cancer cases by immunohistochemistry. In addition, we analyzed the risk of breast cancer in those patients with organochlorine pesticides contamination using a logistic regression model. Our data showed that breast cancer patients suffered high accumulation levels of pp′-dichlorodiphenyldichloroethane and polychlorinated biphenyls-52. However, the concentrations of pp′-dichlorodiphenyldichloroethane and polychlorinated biphenyls-52 were not related to clinicopathologic parameters of breast cancer. Further logistic regression analysis showed polychlorinated biphenyls-52 and pp′-dichlorodiphenyldichloroethane were risk factors for breast cancer. Our results provide new evidence on etiology of breast cancer.
Introduction
Breast cancer is the most common malignancy among women worldwide. It is also the primary cause of cancer-related death among women globally, responsible for more than 40,000 deaths each year in the United States and 69,500 deaths in China.1,2 The known factors contributing to breast cancer development are many and varied, including genetic, epigenetic, and environmental abnormalities.3,4 Numerous studies have established that estrogen plays an important role in the initiation and progression of breast cancers. 5 However, as the cancer progresses, tumor cells acquire growth autonomy and no longer require estrogen, resulting in resistant to antiestrogens such as tamoxifen.6,7
Organochlorine (OC) pesticides have been widely used since the 1940s, which brought about a huge agricultural benefit, but at the same time also did harm to the environment. Since the 1980s, most countries have banned the production of OC pesticides. However, certain OC pesticides (e.g. dicofol) that are used to prevent malaria are still not forbidden in some developing countries. OC compounds can be kept for a long time through the food chain due to the slow degradation and may be accumulated in a variety of biota including humans. These man-made chemicals are environmentally persistent and may be detected in human adipose tissue, blood, and breast milk.8,9
Accumulating evidence showed a possible association between OC pesticides exposure and breast cancer risk. 10 In 1993, Wolff’s group first detected high concentrations of OC residues in blood serum of breast cancer patients. 9 Since then, emerging epidemiological reports have confirmed that OC pesticides are potential risk factors for breast cancer.3,11–16 It has been revealed that OC pesticides can produce endocrine-disrupting effects in humans. As environmental estrogens (xenoestrogens), they are associated with a significant increase in breast cancer risk.17,18 However, the clinicopathologic characteristics of OC pesticides–related breast cancer are still unknown. In addition, the contamination status of OC pesticides in breast cancer patients from modern China remains to be fully elucidated.
In this study, we performed a hospital-based case–control study to determine the contamination status of OC pesticides in breast cancer patients from Qingdao, a second-tier city in eastern China. A panel of degradation products of OC pesticides, including β-hexachlorocyclohexane (β-HCH), γ-hexachlorocyclohexane (γ-HCH), polychlorinated biphenyls-28 (PCB28), polychlorinated biphenyls-52 (PCB52), pentachlorothioanisole (PCTA), pp′-dichlorodiphenyldichloroethane (pp′-DDE, metabolite of pp′-dichlorodiphenyltrichloroethane (DDT)), were tested in adipose tissue samples of breast cancer patients using gas chromatography. Thereafter, the relationship between accumulation levels of OC compounds and clinicopathologic parameters of breast cancer was carefully analyzed. The aim of this study is to evaluate the correlation between OC pesticides exposure and breast cancer risk in modern China.
Materials and methods
Tissue samples
A total of 102 consecutive candidates (56 with breast cancer and 46 with adenosis) who accepted operation in three medical institutions (The Affiliated Hospital of Qingdao University, Qingdao Municipal Hospital, and Qingdao Central Hospital) during a 6-month period from 1 September 2015 to 29 February 2016 were enrolled in this study. All clinical and pathological documents, including patients’ age, tumor size, lymph node status, and histological type, were carefully reviewed. None of these patients received preoperative chemotherapy or radiotherapy. All fresh breast adipose tissues (5 g of tissue each case) were sampled at the time of surgery and immediately frozen and stored at −70°C in preparing for OC compounds detection. Ethical approval was obtained from the institutional review board (No. QDU201509213).
Gas chromatography
A panel of OC pesticides products (β-HCH, γ-HCH, PCB28, PCB52, PCTA, and pp′-DDE; Tianjin pesticide co., LTD, Tianjin, China) were quantified in the extracts by gas chromatography with micro-electron capture detection using an AGILENT 7890B Series GC system equipped with a 63Ni electron capture detector and a HP-5 Trace (30 m, 0.25 mm id, 0.25 Am film) column (Agilent, Palo Alto, CA, USA). System settings were injection temperature, 300°C; detector temperature, 300°C; helium gas flow rate, 1.0 mL/min; and oven temperature, 100°C. Briefly, the oven temperature was quickly increased to 160°C at 20°C/min, then to 250°C at 10°C/min, and held at 250°C for 3 min. Under the condition, the six OC pesticides products were detected successively (Figure 1). The accumulation level of OC pesticides was calculated using the retention time qualitative and peak area quantitative with an external standard method. The computational formula is Csa = Ssa/Sst × Cst (sa = sample, st = standard).

The chromatograms of six kinds of OC pesticide compounds. Under the condition of gas chromatography, the six kinds of OC pesticide compounds appear successively: β-HCH, γ-HCH, PCB28, PCB52, PCTA, and pp′-DDE.
Immunohistochemistry
For immunohistochemistry (IHC) staining, breast cancer samples were fixed in formalin and embedded in paraffin. Estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor-2 (HER2), and Ki-67 expressions were detected with a Leica Bond-Max automatic immunostainer (Leica, Bannockburn, IL, USA) according to the manufacture’s instruction. All antibodies (ER, Cat. # PA0009; PR, Cat.# PA0312; Ki-67, Cat.# PA0410) and Bond™ Oracle™ HER2 IHC System were purchased from Leica. After blocking of endogenous peroxidase with hydrogen peroxide diluted in phosphate buffer (volume concentration, 3%), heat-induced epitope retrieval (ethylenediaminetetraacetic acid–based buffer, pH = 9.0) was carried out for 25 min. Primary antibodies were incubated for 30 min at room temperature. A Leica Bond-Max avidin–biotin-free polymer system was used in the detection according to the company’s recommended procedure. The color was developed by reacting with diaminobenzidine and counterstained by a light hematoxylin. 19 ER and PR were defined as positive when more than 1% of tumor cells showed positive immunoreactivity. HER2 was defined as overexpression when more than 10% of contiguous and homogeneous tumor cells showed immunoreactivity. 20
Statistical analysis
Statistical analysis was performed using the SPSS statistical packages (version 19.0 for Microsoft Windows, La Jolla, CA, USA). All measurement data were expressed as mean ± standard error of the mean (SEM). Student’s t test (unpaired, two-tailed) was used to compare the difference between two groups (case group vs control group). Pearson’s chi-square test was used to analyze the numeration data, and Fisher’s exact test was used when there are data of certain theoretical frequency less than 5. Multivariable logistic regressive model was applied to evaluate the role of OC pesticides in breast cancer development. The p values of less than 0.05 were considered to be statistically significant.
Results
Patients’ characteristics
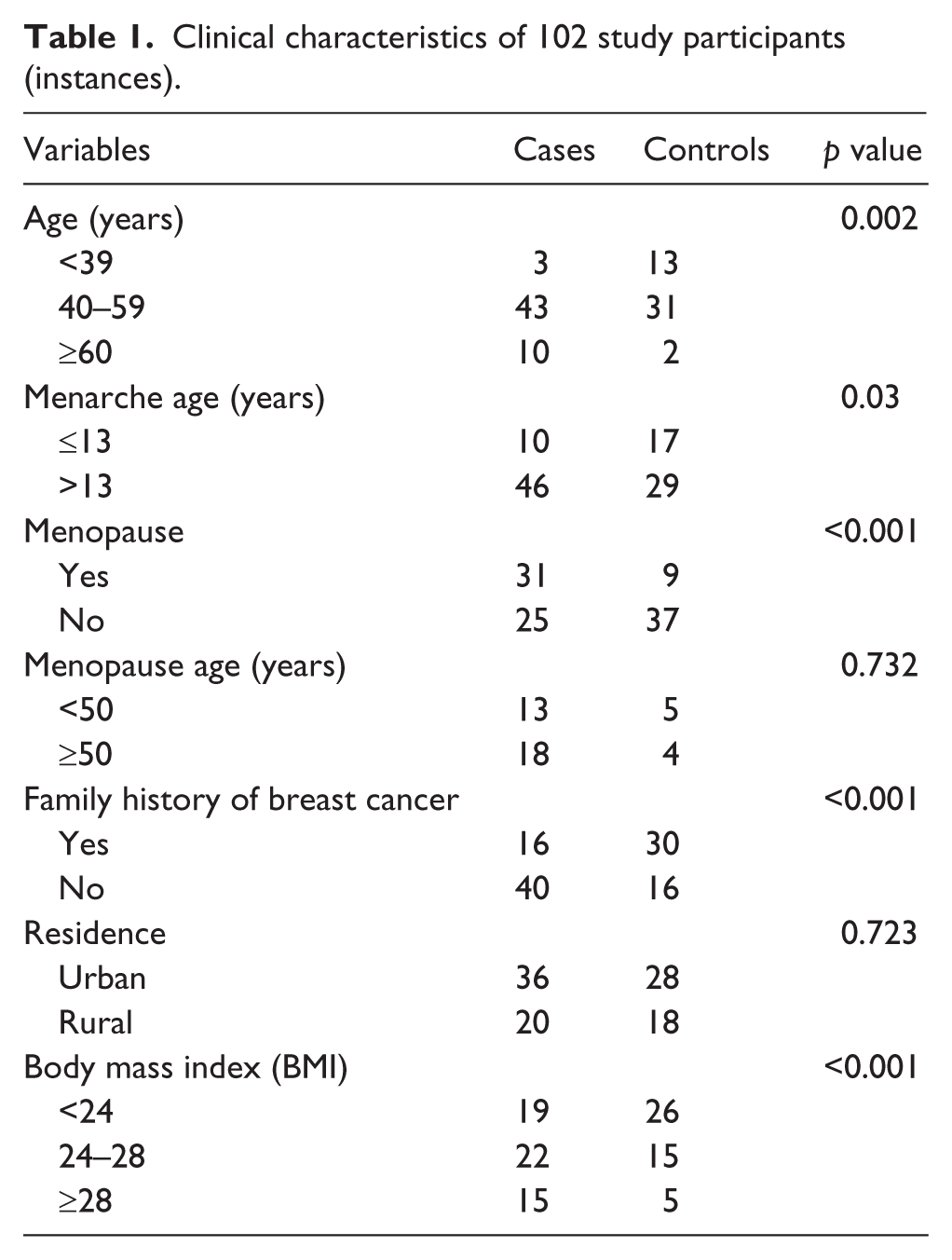
The characteristics of all study participants are summarized in Table 1. The prevalences of age, menopause, family history, and body mass index (BMI) were different between case group and control group. Among the 56 breast cancer cases, 53 were diagnosed as invasive carcinoma of no specific type, accounting for 94.6%. The other type was lobular breast carcinoma (three cases). The mean age of the 102 patients was 52.4 ± 10.6 years ranging from 24 to 80 years. The cases mainly focused on the age of 40–59 years (accounting for 76.8%). By comparison, the mean age of the controls was 44 ± 8.5 years ranging from 17 to 63 years which mainly distributed in the age range of 30–49 years (accounting for 76.1%). The mean BMI value was 25.59 ± 3.35 in cases and 24.11 ± 3.14 in controls, which showed a borderline difference (p = 0.054). The differences of menopause age and residence distribution between cases and controls were not significant.
Clinical characteristics of 102 study participants (instances).
High concentrations of pp′-DDE and PCB52 in breast cancer patients
The gas chromatography showed that all the six OC compounds could be detected in both case group and control group. The positive detection rates of β-HCH, γ-HCH, PCB28, PCB52, PCTA, and pp′-DDE in all participants were 86%, 72%, 5%, 90%, 34%, and 92%, respectively. In addition, the mean concentrations of the six compounds were 0.8921, 0.4913, 0.001, 2.3831, 0.0573, 0.9250 µg/kg, respectively. Considering the low positive rate and low concentration of PCB28, it was not included in further analysis. Moreover, we found breast cancer cases bear high concentrations of two OC compounds: pp′-DDE and PCB52. In comparison with controls, cases showed higher concentrations of pp′-DDE (p < 0.001) and PCB52 (p = 0.044). However, the concentrations of the other three compounds, γ-HCH, β-HCH, and PCTA, were similar in cases and controls (Figure 2, Table S1).

The scatter dot plot of six kinds of OC pesticides in case and control group. We compare the accumulation levels of six kinds of OC pesticide products in case group and control group. Only concentrations of pp′-DDE (p < 0.001) and PCB52 (p = 0.044) are different between cases and controls.
Next, we investigated the relationship between OC compounds accumulation and clinicopathologic parameters of breast cancer. We found that most of these parameters were unrelated to concentrations of pp′-DDE and PCB52 (Table 2). Interestingly, it seemed that pp′-DDE level was related to menopause (p = 0.046) and age (p = 0.016).
Correlation between concentrations of OC compounds and clinicopathologic characteristics of breast cancer (instances).

OC: organochlorine; pp′-DDE: pp′-dichlorodiphenyldichloroethane; PCB52: polychlorinated biphenyls-52; ER: estrogen receptor; PR: progesterone receptor.
The cutoff value is the mean concentration in cases.
HER2 status was validated by fluorescence in situ hybridization.
The pp′-DDE and PCB52 are risk factors for breast cancer
The two OC compounds (pp′-DDE and PCB52) and five potential risk factors for breast cancer (age, menarche age, menopause, BMI, and family history) were brought into a multiple logistic regression model (Table 3). We found that high concentrations of pp′-DDE (p = 0.005, odds ratio (OR): 0.129, 95% confidence interval (CI): 0.031–0.542) and PCB52 (p = 0.038, OR: 0.656, 95% CI: 0.441–0.976) were significantly associated with an increased risk of breast cancer. However, the other five potential risk factors were not significantly related to breast cancer. Thus, pp′-DDE and PCB52 are two independent risk factors for breast cancer.
Logistic regression analysis of OC pesticides and breast cancer.
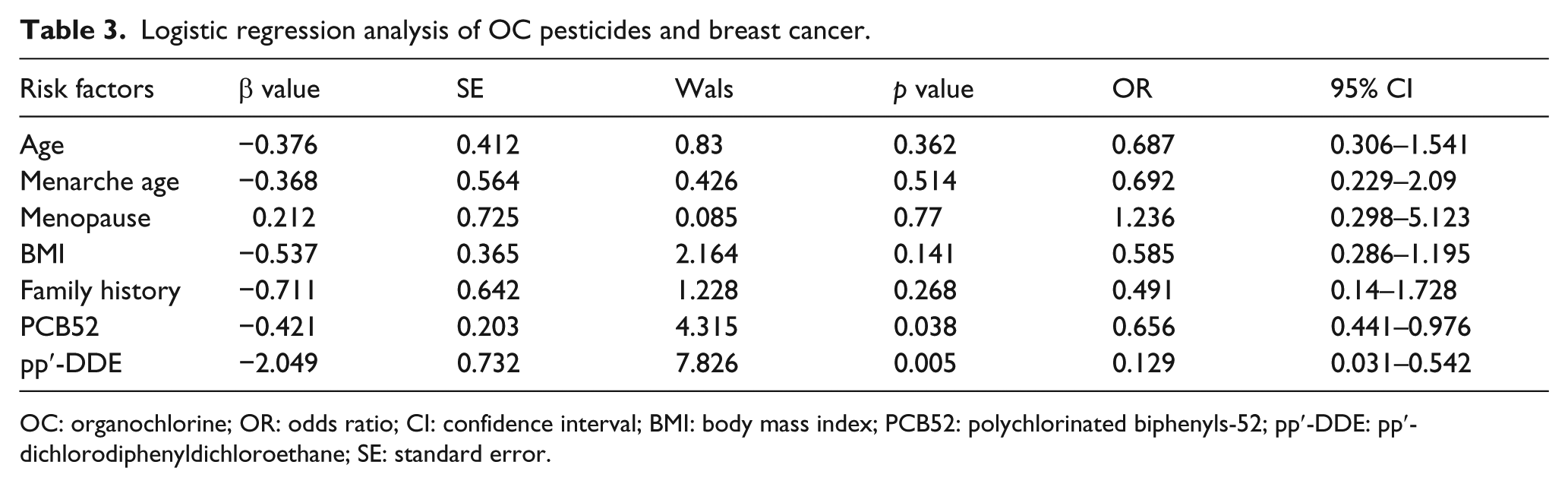
OC: organochlorine; OR: odds ratio; CI: confidence interval; BMI: body mass index; PCB52: polychlorinated biphenyls-52; pp′-DDE: pp′-dichlorodiphenyldichloroethane; SE: standard error.
Discussion
Breast cancer is a globally recognizable major public health problem, and the cause of breast cancer has not yet been fully understood. It has been reported that OC pesticides can mimic the environmental carcinogens by disrupting homeostasis of hormones with an estrogen-like effect.3,11–16,21 In this study, we found that pp′-DDE and PCB52, two metabolites of OC pesticides, were accumulated in the adipose tissues of breast cancer patients in China. Nonetheless, the multiple logistic analysis showed that pp′-DDE and PCB52 were risk factors for breast cancer. However, this study failed to demonstrate the relationship between accumulation concentrations of OC pesticides products and clinicopathologic parameters of breast cancer.
Our study showed breast cancer patients bear high concentrations of pp′-DDE and PCB52. This result indicates that OC pesticides exposure may increase the risk of breast cancer. The metabolites of OC pesticides may act on ER, interfere with endocrine hormones (affecting hormone synthesis, transport, or metabolism), or act as cancer promoters or inducers of cytochrome P450 enzymes, possibly leading to the formation of genotoxic DNA adducts. 21 Interestingly, for the first time, our study demonstrated that OC concentrations were unrelated to clinicopathologic parameters of breast cancer, including ER status. Based on this result, we speculate ER-related signaling is not involved in OC pesticides–induced breast cancer development. More samples are needed to further validate this finding.
Over the past decades, a number of studies have mentioned the association between OC pesticides exposure and breast cancer. However, up to now, these results are still in dispute.14,22 Earlier studies showed that DDT and its metabolite DDE accumulations in breast adipose tissues were closely related to breast cancer.10,23 However, other reports showed that DDE and DDT were not associated with breast cancer.24,25 In addition, the effects of PCBs on breast cancer are also contentious.26,27 We consider these different results are attributed to various factors, including differences in region discrepancy, in study designs, in biological matrices used to estimate OC pesticides exposure, and in target populations with highly varied historical and different exposure levels to OC pesticides and distinct races, age groups, and/or dietary patterns.16,17
In conclusion, our study demonstrated that the products of OC pesticides pp′-DDE and PCB52 are risk factors for breast cancer. The detection rates of the two kinds of OC compounds are more than 90% in breast cancer cases. This study provides a new way for breast cancer prevention, that is, prohibition of OC pesticides may help to reduce the risk of breast cancer.
Footnotes
Acknowledgements
The authors are grateful to all staffs of Qingdao Municipal Center for Disease Control & Prevention for their kind help.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the grant from the National Natural Science Foundation of China (No. 81502272) and the grant from Applied Fundamental Research of Qingdao government (No. 13-1-4-141-jch).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
