Abstract
It is currently unclear as to which patients with node-negative gastric cancer can benefit from adjuvant chemotherapy. This study aimed to develop a prognostic model based on patient-, tumor-, and host-related factors to stratify high-risk patients eligible for adjuvant therapy. Correlations of clinicopathological and hematological features with overall survival were analyzed using a Cox model. A score to identify risk classes was derived from hazard ratios in multivariate analysis. In all, 436 patients with node-negative gastric cancer (stage pT1–4aN0M0) were analyzed in this study. Multivariate analysis showed that age, depth of invasion, and neutrophil–lymphocyte ratio were independent prognostic indicators of overall survival, and a prognostic model was developed using these significant factors. Patients were stratified into three risk groups with significant differences in the 3-year survival rates (98.5%, 91.6%, and 70.7%, respectively; p < 0.001) according to their scores. The prognostic model improved the predictive accuracy of postoperative 3-year survival rate by 7% when compared with the pathological T stage. A model based on age, depth of invasion, and neutrophil–lymphocyte ratio is more effective than traditional staging systems in predicting the prognosis of node-negative gastric cancer. High-risk patients could be considered for adjuvant therapy.
Introduction
Gastric cancer is the fourth most commonly diagnosed cancer and the second leading cause of cancer death worldwide. 1 Surgery remains the main treatment modality for gastric cancer. Despite curative resection, an appreciable proportion of patients develop recurrence and die of the disease, even those with node-negative gastric cancer. 2 Recent studies reported that postoperative adjuvant chemotherapy could improve the outcome of gastric cancer patients.3–5 However, it is unclear whether patients without nodal involvement can benefit from adjuvant chemotherapy because of inconsistent results from published studies.4,5 Currently, clinical practice guidelines for adjuvant treatment are based on tumor, node, metastasis (TNM) staging. However, for patients without nodal involvement, this is still not sufficient. In the CLASSIC study, the majority of patients (96%) in the pN0 subgroup presented with serosa invasion (T4a), yet the data indicated that adjuvant chemotherapy would not improve 3-year disease-free survival. 4 These results implied that in unselected patients with T4aN0M0 gastric cancer, the potential benefit of chemotherapy is limited and underscores the critical need for quantifying risk by combining TNM stage and other prognostic factors to select patients at high risk and to guide treatment decision-making.
Lymph node metastasis is the most important independent predictor of survival for patients undergoing radical gastrectomy. 6 Compared with node-positive cancer, node-negative gastric cancer has fewer aggressive biological properties, and the predictive factors are not consistent between them.7,8 Recently, many studies have shown that several tumor-related factors, such as depth of tumor invasion, lymph node micrometastases, and tumor size, were associated with the prognosis of patients with node-negative gastric cancer.9,10 However, research has increasingly showed that the outcome of patients is determined not only by the intrinsic properties of tumor cells but also by the characteristics of the host’s reaction to the tumor.11,12 Among these factors, systemic inflammatory factors in particular have gained notable interest due to their convenience in clinical application. It is reported that systemic inflammatory markers such as neutrophil–lymphocyte ratio (NLR), platelet–lymphocyte ratio (PLR), and fibrinogen are useful prognostic indicators for gastric cancer.13–15 However, for patients without nodal involvement, it is not clear whether inflammatory markers offer the same prognostic value. In addition, the prognostic models are generally used for predicting outcome in gastric cancer,16,17 but there is no model in frequent use for risk stratification in node-negative patients.
The aim of this study was to identify the role of patient-, tumor-, and host-related factors in predicting the outcome of patients with node-negative gastric cancer, with particular focus on the effect of systemic inflammatory markers on patient prognosis. Furthermore, a prognostic model based on multivariate analysis was developed, which may contribute to the implementation of personalized therapy based on risk stratification in patients without lymph node involvement.
Materials and methods
Patients
A total of 1494 patients who underwent gastrectomy in the First Hospital of China Medical University between January 2007 and December 2013 were retrospectively reviewed in this study. Of these, 436 patients had no lymph node metastasis and met the following criteria: (1) underwent D2 lymphadenectomy with more than 15 lymph nodes examined, (2) histologically confirmed T1–4aN0M0 gastric cancer according to the 7th Edition of Union for International Cancer Control (UICC) TNM system, 18 and (3) complete follow-up information and laboratory data available. Patients with double-cancer history or with evidence of active infection or inflammatory disease were excluded. And patients who died of non-cancer-related cause were also excluded. This study was approved by the institutional review board, and all patients provided written informed consent before enrollment.
Laboratory and clinical data collection
Routine laboratory measurements including complete blood count, blood coagulation, and liver function tests were carried out within 7 days before surgery. Complete blood counts were measured with an automated hematological blood analyzer (Sysmex XE-5000; Sysmex Corporation, Kobe, Japan), blood coagulation tests (reference range, 2–4 g/L) were measured with an autoanalyzer (STA-R Evolution; Diagnostica Stago, Asnières sur Seine, France), and liver function tests were analyzed with an autoanalyzer (Hitachi 7600-210; Hitachi Co., Tokyo, Japan).
The NLR was defined as the absolute neutrophil count (ANC) divided by the absolute lymphocyte count (ALC). The PLR was defined as the absolute platelet count divided by the ALC. The cut-off values for NLR and PLR were 2.51 (75th percentile) and 110 (50th percentile), respectively.
The potential prognostic factors for gastric cancer were as follows: age (<65 versus ≥65 years), gender (male versus female), tumor size (≤3.5 versus >3.5 cm), tumor location (proximal versus distal), Lauren type (intestinal versus diffuse versus mixed), depth of invasion (T1 versus T2 versus T3 versus T4), histological grade (well and moderately differentiated versus poorly and undifferentiated), lymphovascular invasion (negative versus positive), white blood cell count (≤upper limit of normal (ULN) versus >ULN), ANC (≤ULN versus >ULN), ALC (<lower limit of normal (LLN) versus ≥LLN), platelet count (≤ULN versus >ULN), NLR (<2.51 versus ≥2.51), PLR (<110 versus ≥110), hemoglobin level (<LLN of female versus ≥LLN of female), albumin (<LLN versus ≥LLN), and plasma fibrinogen (≤ULN versus >ULN).
Statistical analysis
Correlations between clinicopathological parameters or systemic inflammatory markers and overall survival (OS) were assessed by univariate and multivariate analyses using a Cox proportional hazard model. All significant variables in the univariate analysis were entered into a multivariate analysis using the forward stepwise method. Survival curves were assembled using the Kaplan–Meier method, and the two-tailed log-rank test was used to assess the differences between the curves. A prognostic model was constructed by all significant factors in multivariate analysis. Risk scores were assigned to each variable based on the hazard ratios (HRs) in the multivariate analysis. The coefficients (logarithm of HR) were divided by the smallest value (2.372), and the results were rounded to 0.5 multiples. 19 The sum of the single scores produced the overall prognostic score for each patient. p-Values of <0.05 were considered statistically significant, and all the tests were two-sided. Statistical analysis was carried out using SPSS 19.0 (SPSS Inc., Chicago, IL, USA).
Results
Clinicopathological characteristics and prognostic significance
In all, 436 patients with node-negative gastric cancer were eligible for analysis. Patient demographics and tumor characteristics are listed in Table 1. The median age was 58 years with a range of 26–82 years. A total of 312 patients (71.6%) were males and 124 (28.4%) were females. The median tumor size was 3.5 cm (range, 0.3–12.0 cm). A total of 389 patients (89.2%) had tumors in the proximal stomach, 102 patients (23.4%) had T4a tumors, and 292 patients (67.0%) had poorly differentiated disease. Totally, 207 patients (47.5%) had intestinal-type tumor as opposed to diffuse type (n = 142, 32.6%) or mixed type (n = 87, 20.0%). The median follow-up time was 29 months (range, 6–67 months). The overall 3-year survival rates were 91.4%.
Patient characteristics and univariate analysis for overall survival (N = 436).

HR: hazard ratio; CI: confidence interval; G1: well differentiated; G2: moderately differentiated; G3: poorly differentiated; G4: undifferentiated.
The relationship between clinicopathological characteristics and OS of the enrolled patients is shown in Table 1. Univariate analysis demonstrated that age, tumor size, tumor location, Lauren type, and depth of invasion had prognostic significance.
Correlation between inflammatory parameters and survival
The median NLR was 1.77 (range, 0.44–45.21), and the median PLR was 110 (range, 36–741) in the group of 436 patients. Patients were distributed into equal quartiles according to the 25th, 50th, and 75th percentiles of NLR and PLR. Table 2 demonstrates the HRs for death and 95% confidence intervals (CIs) for each NLR and PLR quartile. According to the results, the 75th NLR and 50th PLR percentiles were the optimal values to distinguish high-risk groups and were thus used as cut-off values in multivariate analysis. Kaplan–Meier curves showed that higher NLR and PLR values were significantly associated with poor prognosis (Figures 1 and 2). The relationship between other systemic inflammation markers and OS in patients with node-negative gastric cancer is also shown in Table 2. Lower lymphocyte and hemoglobin levels were associated with higher risk of death, whereas white blood cells, neutrophils, platelets, fibrinogen, and albumin had no prognostic significance.
Patient characteristics and univariate analysis for overall survival (N = 436).
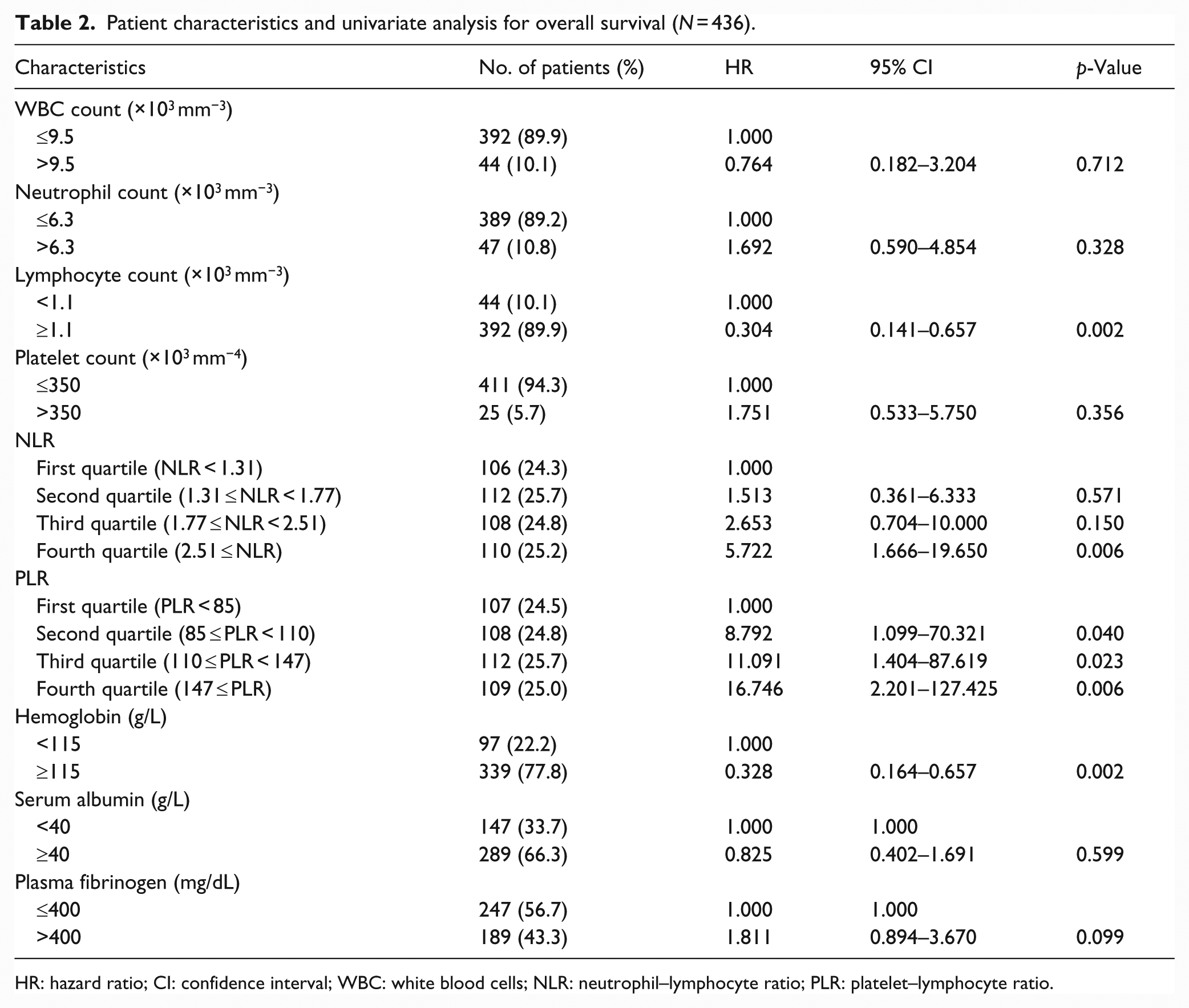
HR: hazard ratio; CI: confidence interval; WBC: white blood cells; NLR: neutrophil–lymphocyte ratio; PLR: platelet–lymphocyte ratio.

Kaplan–Meier survival curves stratified by NLR (75th percentile).

Kaplan–Meier survival curves stratified by PLR (50th percentile).
Multivariate analysis and prognostic model
In multivariate analysis, age, depth of invasion, and NLR were identified as independent factors of OS after controlling for the other factors (Table 3). Risk scores were assigned to each variable based on the final Cox proportional hazard model, and a prognostic score was developed for each individual patient. Patients were stratified into three risk groups: low risk of death (0–1.0 scores, n = 200), intermediate risk of death (1.5–2.5 scores, n = 162), and high risk of death (3.0–4.5 scores, n = 74). The 3-year survival rates for the low-, intermediate-, and high-risk groups were 98.5%, 91.6%, and 70.7%, respectively (p < 0.001). Kaplan–Meier curves for the risk groups are shown in Figure 3. The prognostic model improved the accuracy of predicting death at 3 years compared with pathological T stage (area under the receiver operating characteristic (ROC) curve 0.80 versus 0.73; Figure 4).
Multivariate analysis for overall survival and prognostic score of patients with gastric cancer.

HR: hazard ratio; CI: confidence interval; NLR: neutrophil–lymphocyte ratio.

Survival curves based on risk groups for patients with node-negative gastric cancer.
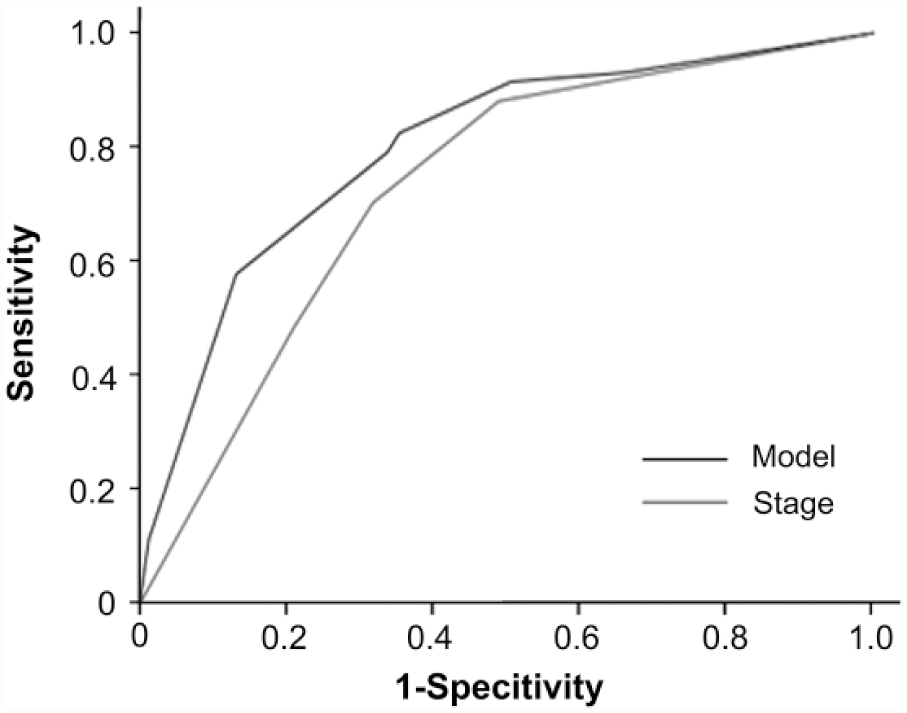
ROC plot of 3-year survival rate based on established model and TNM stage for patients with node-negative gastric cancer.
Discussion
Although patients with node-negative gastric cancer have better clinical outcome than those with lymph node metastasis, they also experience locoregional recurrence, peritoneal seeding, or hematogenous spread.2,8 Currently, for patients with node-negative gastric cancer, adjuvant therapy is recommended for T3/T4 tumors or T2 tumors with high-risk features according to the National Comprehensive Cancer Network guidelines. However, the role of adjuvant therapy is controversial for these patients based on the data from subgroup analyses in the CLASSIC and ACTS-GC studies.4,5 Therefore, accurate estimates of prognosis and potential benefit from adjuvant therapy are important considerations when making treatment decisions for patients with node-negative gastric cancer. Thus, in this study, we identified clinically useful prognostic indicators including patient-, tumor- and host-related factors in node-negative patients who underwent gastrectomy with D2 lymphadenectomy and established a prognostic model. The model discriminated patients into three risk groups with significant differences in the 3-year survival rates. Compared with the traditional pT stage, the model increased the predictive accuracy by 7.0%.
Tumor progression depends on tumor features, but is also influenced by the reaction of the host to malignancy. There is now evidence that the hallmarks of cancer comprise six biological capabilities; underlying these hallmarks are genome instability and inflammation. 20 This highlights the inflammatory response triggered by tumor cells, which plays a critical role in the occurrence and progression of malignancy. Moreover, research increasingly showed that a variety of systemic inflammatory factors are related to patient outcome in many tumors.11,12 However, the prognostic significance of inflammatory factors has not been sufficiently addressed in node-negative gastric cancer. In this study, we observed that among the examined inflammation-related factors, only NLR was significantly associated with survival after controlling for the clinicopathological parameters. Our data first confirmed the prognostic value of NLR in node-negative gastric cancer. This result essentially supports the previous study, which analyzed 377 gastric cancer patients with or without lymph node metastasis. 21 Increased NLR is the consequence of relative neutrophilia and lymphocytopenia, which reflects a non-specific response of the host to tumors. Relative neutrophilia increases the levels of pro-angiogenic factors, growth factors, and anti-apoptotic markers that stimulate angiogenesis and promote tumor growth. 22 Lymphocytopenia represents a decrease in the cell-mediated immune system, thus failing to counter the tumor growth. 23
Jiang et al. 21 demonstrated that PLR and C-reactive protein (CRP) are related to survival, but are not independent prognostic factors. Our multivariate analysis showed PLR and other hematological indexes were also not independent risk factors for patients with node-negative gastric cancer, although PLR had prognostic value in univariate analysis. It was reported that PLR was an independent predictor of postoperative complications but not survival. 21 The limitation of this study was that CRP was not involved in the analysis, as it is not a routine index for preoperative evaluation of gastric cancer patients. In contrast, NLR as a lower cost and routinely performed laboratory indicator is easy to continuously and dynamically monitor and is suitable for widespread use in the clinic.
In this study, multivariate analysis showed that age, depth of tumor invasion, and NLR were independent prognostic factors for node-negative gastric cancer. Based on the results of multivariate analysis, a prognostic model was developed. Several prognostic models are currently used to estimate the treatment benefits for individual patients with early breast cancer or stage II colon cancer.24,25 However, there was previously no available models to help predict survival and treatment benefits for node-negative gastric cancer (stage pT1–4aN0M0). Du et al. 26 defined a high-risk subgroup of T2N0 gastric cancer using a prognostic model based on postoperative pathological characteristics, suggesting adjuvant therapy may be considered for these patients. In this study, we combined patient-, tumor- and host-related factors to develop an integrated model, which classified the patients into different risk groups. The 3-year survival rates for the low-risk groups were 98.5%, thus these patients are unlikely to need adjuvant treatment and do not have to be subjected to the toxic effects of chemotherapy. In contrast, patients in the high-risk group had a low 3-year survival rates and might be good candidates for adjuvant therapy. Our data further demonstrated that integrated multiple factors to predict the outcome of patients could compensate for the limitation of the current staging systems and improve the predictive accuracy. Therefore, the current analysis suggests that when making treatment decisions, patient-, tumor-, and host-related factors need to be considered, and therapeutic strategies based on risk stratification should be explored in future clinical studies.
Our study has some limitations. First, it is a retrospective study and is not able to provide information about follow-up treatment. Second, some inflammatory markers such as CRP were not involved in the analysis, as they are not routine indicators for preoperative assessment. Third, the sample size of the study is relatively small; further studies are needed to confirm the feasibility of the model.
Conclusion
To our knowledge, this study is the first to confirm the role of NLR in predicting outcome of patients with node-negative gastric cancer and establish a prognostic model based on age, pathological T stage, and NLR. The model is more effective than traditional staging systems and easy to apply in clinical practice. Using this model, clinicians can accurately acquire the prognostic information for each patient, and it is suggested that high-risk patients might be considered for adjuvant treatment. Nevertheless, these findings need to be validated in additional cohorts, as well as further prospective randomized trials.
Footnotes
Acknowledgements
The authors thank the medical oncology staff at the First Hospital of China Medical University for their advice and assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (nos 81372547, 81372485, and 81172369), National Science and Technology Major Project of the Ministry of Science and Technology of China (no. 2013ZX09303002), and Science and Technology Plan Project of Liaoning Province (nos 2014225013 and 2014226033).
