Abstract
Heat shock protein 90 is induced in response to the cell stress. Its overexpression has been reported in many cancers with poor prognosis. It acts as a chaperone to the client proteins, especially the activated oncoproteins in malignancies to protect them from degradation. Heat shock protein 90 inhibition represented anti-cancer effects in many studies. Previous natural product–based compounds are limited by their association with target toxicities. BIIB021 is an orally available, fully synthetic novel small-molecule heat shock protein 90 inhibitor that has shown strong antitumor activities in a large number of preclinical models and is now under clinical investigation. This review will summarize its therapeutic effects and highlight the prospect of targeting heat shock protein 90 in the cancer therapy.
Introduction
Heat shock protein 90 (HSP90) is an essential component of the cellular shock response, which is also induced in response to the cell stress. 1 It is an ubiquitously expressed molecular chaperone that plays an important role in homeostasis by regulating the function and maturation of a number of the client proteins. 2 HSP90 is reported to be overexpressed in many malignant cancers linked with the poor prognosis.3–5 Many oncogenically activated proteins such as KIT, FLT3, HIF, AKT, and CDKs are chaperoned by HSP90. 6 The tyrosine kinase inhibitor resistance could be overcome by HSP90 inhibition, which suggests that the drug resistance in cancer may be dependent on the HSP90 function because of their abilities as the chaperones to protect the oncoproteins from degradation. 7 Given the important roles of HSP90 in oncology, there is no doubt that targeting HSP90 represents a broad perspective in cancer therapy.
BIIB021, formerly named CNF2024, is a fully synthetic HSP90 inhibitor. It binds to the adenosine triphosphate (ATP) binding pocket of HSP90, resulting in HSP90 chaperone dysfunction, which will induce client protein degradation and tumor growth inhibition. 8 The structure of this BIIB021 compound is different from previous natural product–based compounds such as geldanamycin and radicicol or their derivatives (Figure 1). BIIB021 is a novel small-molecule HSP90 inhibitor that can be administered orally. The previous studies about BIIB021 including its phase I and II clinical trials promised a new therapeutic chance for the treatment of hematopoietic malignancies and also solid tumors.

Different structures of BIIB021 and the previous natural product–based compounds.
Initial attempts to target HSP90 from natural products
Since the HSP90 is a novel therapeutic target in cancer, a number of preclinical and clinical studies were performed to evaluate the effects of a series of HSP90 inhibitors. Geldanamycin was originally isolated from
Radicicol is another natural product that binds the HSP90 and subsequently alters its function. It was isolated from the fungus
BIIB021 as a novel, fully synthetic, and orally active inhibitor of HSP90
Based on different chemical scaffolds, many novel synthetic HSP90 inhibitors are developed, and some of them are undergoing phase I/II clinical evaluation at present. Of them, the first one to enter the clinical evaluation is BIIB021, developed initially by Conforma Therapeutics (currently Biogen Idec) based on the purine scaffold through structure-based design. 29 Currently, there are many ongoing phase I/II trial studies about BIIB021 in cancers, including studies about advanced solid tumors, 30 human T-cell acute lymphoblastic leukemia, 31 primary effusion lymphoma, 32 Kaposi sarcoma, 33 gastrointestinal stromal tumors (GIST), 34 breast cancer, 35 Hodgkin’s lymphoma, 36 and T- and natural killer cell lymphomas. 37 BIIB021 was shown to bind competitively with geldanamycin in the ATP-binding site of HSP90 protein. However, it showed higher binding affinity for HSP90 in the binding affinity assay in the study by Lundgren et al. 38 The binding affinity for 17-AAG was 4.6 nmol/L, but it was only 1.7 nmol/L for BIIB021. 38 The X-ray diffraction (1.79 Å resolution) of BIIB021 binding HSP90 complex was also explored by researchers (Figure 2), which was shared in the protein data bank in Europe.39,40 Inhibiting HSP90 in cells induced the client proteins degraded via the proteasome pathway, which were dependent on HSP90 for proper activity and folding. 41 Zhang et al. 42 indicated that BIIB021 may have broader application against tumors with acquired multidrug resistance (MDR) or tumors located in organs protected by MDR proteins and Bcl-2 overexpressions, which are commonly encountered in current cancer therapy.

The X-ray diffraction (1.79 Å resolution) of BIIB021 binding HSP90 complex. The views from the (a) front, (b) side, and (c) top of the complex were shown.
Nuclear factor-κB (NF-κB) is constitutively active in many cancers involved in the cellular survival and proliferation. Similar to other HSP90 inhibitors, BIIB021 is also reported to block the constitutive NF-κB activity in cancers 32 and induce apoptosis by disrupting the p53-MDM2 balance in leukemia. 31 Moreover, He et al. 43 proved for the first time that BIIB021 could also induce autophagic response as evidenced by the formation of autophagosome in chronic myeloid leukemia (CML), which is related to the Akt-mTOR-Ulk1 signaling pathway. Besides, this chemical compound was also reported to sensitize esophageal squamous cell carcinoma and head and neck squamous cell carcinoma to radiation.44,45 It showed the enhanced reduction of the radio-responsive proteins with the increased apoptotic ratio. This suggests that the synthetic and bio-available HSP90 inhibitor affects multiple pathways involved in tumor development and progression either as a monotherapy or a radiosensitizer.44,45 Additionally, besides the direct cytotoxic effects on tumor cells induced by HSP90 inhibition itself, targeting HSP90 may block immune checkpoints as a complementary strategy from previous study. 22 Immune therapy is considered the most promising method in cancer therapy at present. Indeed, the immune checkpoint blockade provides broad and diverse opportunities in the war against cancer, though it is not so applicable to all the cancers and every patient.46–48 In conclusion, it seems that further aspects regarding this agent will be revealed in anti-cancer effects (Figure 3).
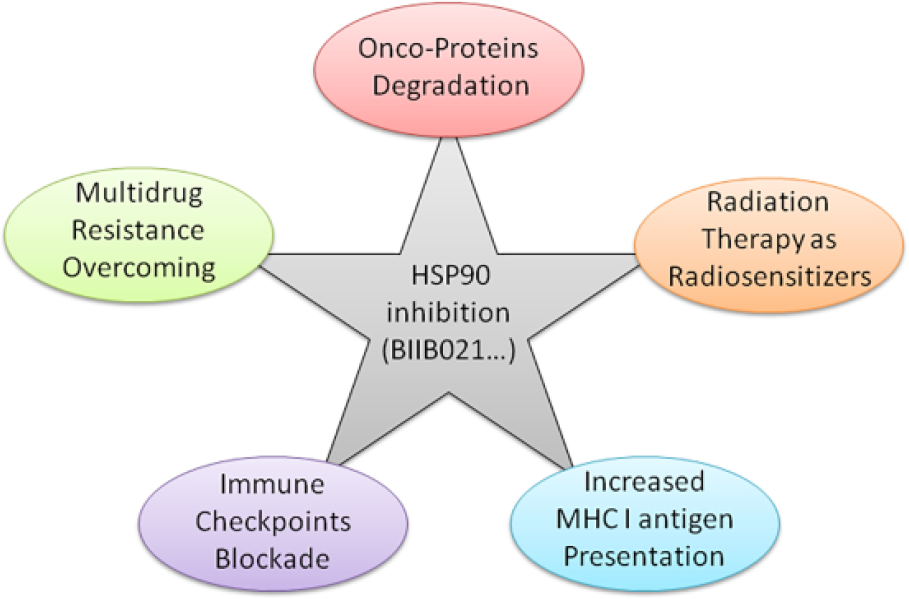
Multiple mechanisms of the HSP90 inhibition in cancer therapy.
HSP90 addiction in cancer cells leads to its therapeutic selectivity
In normal cells, cell growth and division are controlled by multiple signal pathways, which need a sensitive modulation system. These normal cells start to proliferate only when they are required, which are usually activated by various growth factor stimulations. However, in cancer cells, the growth-promoting signaling pathways are constitutively “On,” which induced growth forever as the cancer character, even without the extracellular growth stimulations. As mentioned earlier, many cancer promoting factors such as the mutated kinases work with HSP90 dependently. 49 This means that the cancer cell proliferation is tightly related to its HSP90 functions, which may explain why inhibition of HSP90 by some specific inhibitors suppresses the growth and proliferation of cancer cells. Although the normal cells also require HSP90, its dependency is much lower than the rapid proliferated cancer cells. In fact, the study by Yao et al. 49 indeed proved that the more cells proliferate rapidly, the more they may depend on HSP90 activity. Recently, Zong et al. 50 also reported that the more malignant cancer cells showed higher sensitivities to the HSP90 inhibition in acute myeloid leukemia (AML). Different from such leukemia cells, the mononuclear cells from cord blood and normal peripheral blood leukocytes represent only minor toxicity. Here, we may consider that cancer cells rigorously need HSP90 function (which is called HSP90 oncogene addiction), while the normal cells depend on HSP90 modestly. This suggests cancer cells require higher amounts of HSP90, which is more heavily occupied with its clients in cancer cells. Furthermore, the higher affinity conformation of HSP90 and clients in cancer cells would also make it as the “Achilles hell” in the cancer defense war. 51 These may be the reason why HSP90 in cancer cells is more sensitive to HSP90 inhibitors than in normal cells (Figure 4).

HSP90 addiction in cancer cells leads to its therapeutic selectivity.
Conclusion
Purine-based HSP90 inhibitors are the first class of fully synthetic agents developed and have been explored well in many preclinical studies. BIIB021 as the first one to enter clinic trials in particular has shown preclinical anti-cancer activity well in a variety of cancers. Since HSP90 was identified as a chaperone highly expressed in cancers, the critical assessment of multi-pharmacological mechanisms for this agent and some other novelly developed HSP90 inhibitors will hopefully provide a new avenue to the cancer therapy.
Footnotes
Acknowledgements
L.Y., W.Z., B.Z., and C.X. wrote the manuscript. D.W. conceived and wrote the manuscript. L.Y., W.Z., and B.Z. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
