Abstract
In this study, we evaluated the prognostic potential and functional regulation of human nature antisense, brain-derived neurotrophic factor antisense, in non–small cell lung cancer. Non–small cell lung cancer carcinoma and adjacent non-carcinoma lung tissues were extracted from 151 patients. Their endogenous brain-derived neurotrophic factor antisense expression levels were compared by quantitative reverse transcription polymerase chain reaction. Clinical relevance between endogenous brain-derived neurotrophic factor antisense expression level and patients’ clinicopathological variances or overall survival was analyzed. The potential of brain-derived neurotrophic factor antisense being an independent prognostic factor in non–small cell lung cancer was also evaluated. In in vitro non–small cell lung cancer cell lines, brain-derived neurotrophic factor antisense was upregulated through forced overexpression. The effects of brain-derived neurotrophic factor antisense upregulation on non–small cell lung cancer in vitro survival, proliferation, and migration were evaluated by viability, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide, and transwell assays. Brain-derived neurotrophic factor antisense is lowly expressed in non–small cell lung cancer carcinoma tissues and further downregulated in late-stage carcinomas. Brain-derived neurotrophic factor antisense downregulation was closely associated with non–small cell lung cancer patients’ advanced tumor, lymph node, metastasis stage, and positive status of lymph node metastasis, and confirmed to be an independent prognostic factor for patients’ poor overall survival. In non–small cell lung cancer A549 and H226 cell lines, forced overexpression of brain-derived neurotrophic factor antisense did not alter cancer cell viability but had significantly tumor suppressive effect in inhibiting in vitro non–small cell lung cancer proliferation and migration. Endogenous brain-derived neurotrophic factor antisense in non–small cell lung cancer carcinoma could be a potential biomarker for predicting patients’ prognosis. Overexpressing brain-derived neurotrophic factor antisense may also have a therapeutic potential in inhibiting non–small cell lung cancer tumor growth.
Keywords
Introduction
Human lung cancer is one of the most common and most malign carcinomas for both male and female patients in the world. In the United States, lasted estimation showed that there are approximately quarter million new cases of lung cancer every year. 1 Among them, more than 150,000 patients died of lung cancer every year. 1 In China, due to lack of regulation on public smoking and deteriorating air quality, the number of newly diagnosed lung cancer patients is growing exponentially, as lung cancer claims more than 300,000 lives every year.2,3 The major subtype of human lung cancer is non–small cell lung cancer (NSCLC), which accounts for more than 80% of all lung cancer cases.4,5 For decades, significant progress had been made in seeking early-diagnosis methods/reagents and developing efficient treatment plans and post-surgery cares for patients with NSCLC.4,6–8 Yet, the exact mechanisms contributing to pathogenesis and development of lung cancer are largely unknown.
During the past decade, emerging evidence demonstrates that a group of long noncoding RNAs complementary to their endogenous sense counterparts, or nature antisense transcripts, may play critical roles in regulating human cancers by post-transcriptionally modulating messenger RNA (mRNA) stability and translatability.9,10 For example, HIF-AS, a nature antisense to hypoxia-inducible factor 1-alpha (HIF-1α), was shown to be upregulated, inversely regulating HIF-1α and acting as prognostic factors in breast cancer and renal cancer.11–13 Also, an antisense RNA transcript at the INK4b/ARF/INK4a locus, or ANRIL, was demonstrated to reversely inhibit its cognate sense transcripts to induce cancer proliferation in human cancers, such as acute lymphoblastic leukemia, prostate cancer, and glioma.14–16
Brain-derived neurotrophic factor (BDNF) is widely considered as a neuronal transcriptional factor regulating various aspects of neurogenesis and neuron growth in the human nerve system.17,18 The antisense of BDNF, BDNF-AS, was shown to reversely associate with BDNF expression 10 ; thus, it functions as an anti-BDNF factor in neural regulation.19–21 In the recent decade, emerging evidence demonstrates that BDNF is also an active tumor regulator, most likely acting as an oncogene, in various human cancers, such as ovarian cancer, colon cancer, or bladder cancer.22–24 In human lung cancer of NSCLC, BDNF and its receptor tropomyosin receptor kinase B (TrkB) were shown to be overexpressed in tumor tissues and closely associated with poor prognosis of NSCLC patients.25,26 However, the expression profile and mechanism of BDNF-AS have never been described in NSCLC.
In this study, we used quantitative reverse transcription polymerase chain reaction (qRT-PCR) to evaluate BDNF-AS gene expression in NSCLC carcinoma tissues. The clinical relevance of BDNF-AS expression in predicting NSCLC patients’ clinicopathological variances and overall survival (OS) was then investigated. Moreover, in NSCLC cell lines, BDNF-AS was upregulated through forced overexpression to evaluate its functional effect on NSCLC in vitro proliferation and migration.
Materials and methods
Ethic approval
All experimental designs in our study were approved by the Human Research and Ethic Committee at the Second Affiliated Hospital of Soochow University in Suzhou, China. Consent forms were signed by all patients who enrolled in the study. All clinical protocols were conducted in accordance with the Declaration of Helsinki and general guidelines for medical practice in People’s Republic of China.
Patients
A total number of 151 patients diagnosed with NSCLC with either adenocarcinoma or squamous cell carcinoma were enrolled in our study between June 2012 and July 2014. Classification on patient’s tumor, lymph node, metastasis (TNM) stage was performed according to the seventh edition of American Joint Committee on Cancer. 27 All participating patients underwent surgery to remove tumor at the Departments of Thoracic Surgery and First Surgery. Samples of NSCLC carcinoma tissues and their adjacent non-carcinoma (control) lung epithelial tissues were extracted and immediately snap-frozen in liquid nitrogen. Clinical samples were then stored at −80°C prior to RNA extraction.
RNA extraction and qRT-PCR
RNA extraction was performed using a Trizol assay (Thermo Fisher Scientific, USA) according to the manufacturer’s recommendation. Complementary DNA sequences were then reversely transcribed from RNA products using a TaqMan Reverse Transcription Kit (Applied Biosystems, USA). A Brilliant SYBR Green II qRT-PCR kit (Stratagene, USA) was performed on an ABI automated 7900 HT Fast Real-Time PCR System (Applied Biosystems) to probe the gene expression of natural antisense BDNF-AS in both NSCLC clinical samples and NSCLC cell lines. Relative gene expression levels of BDNF-AS were then normalized to the gene expression level of house keeping gene of U6 small nuclear RNA (U6 sRNA) in control samples using the 2−ΔΔCt algorithms.
NSCLC cell lines
A normal human bronchial lung epithelium cell line (NuLi-1) and NSCLC carcinoma cell lines, A549, H226, H596, H510A, H920, H522, H2991, SK-MES-1, and SW900 cells, were all purchased from China Center for Type Culture Collection (CCTCC, China). All cells were cultured in six-well plates in RPMI 1640 medium (MilliporeSigma, China), supplemented with 10% fetal bovine serum (FBS; MilliporeSigma) and antibiotics of penicillin (100 U/mL) and streptomycin (100 µg/mL; MilliporeSigma). Cells were passaged every 3–5 days and maintained in a controlled environment with 95% O2/5% CO2 at 37°C.
BDNF-AS overexpression assay
Full length of human natural antisense BDNF-AS was inserted into a mammalian overexpressing cassette pCDNA3/+ (Takara Biotechnology, China) to create a BDNF-AS overexpression plasmid, pcDNA/BDNFAS. An empty pCDNA3/+ cassette was used as a negative-control overexpression plasmid. NSCLC cell lines, A549 and H226 cells, were transfected with pcDNA/+ or pcDNA/BDNFAS for 24 h, followed by a 72-h selection procedure using 1 mg/mL G418. After that, healthy A549 and H226 cells were re-seeded in six-well plates and passaged for three times. Efficiency of transfection was evaluated by qRT-PCR.
Viability assay
Viability or apoptosis of NSCLC cells was examined using a CellTiter-Glo Luminescent Cell Viability Assay (Promega, USA) according to the manufacturer’s recommendation. Briefly, in the culture of A549 or H226 cells, culture medium was mixed with CellTiter-Glo reagent for 15 min at room temperature. After that, cancer cell viability was characterized by measuring absorbance at 440 nm using a PowerWave XS spectrophotometer reader (BioTek, USA), according to the manufacturer’s recommendation.
NSCLC in vitro proliferation assay
In vitro proliferation of NSCLC cells was characterized using a Cell Proliferation Kit I (MilliporeSigma), according to the manufacturer’s recommendation. Briefly, A549 and H226 cells were plated in 96-well plates at the density of 1000 cells per well. NSCLC cells were allowed to proliferate for five consecutive days. At an interval of 24 h, culture medium (100 µL) was mixed with 15 µL staining reagent of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) for 4 h. After aspirating the culture medium, cell culture was treated with acidic isopropanol for solubilization. NSCLC cell proliferation rates were then characterized by measuring absorbance at 570 nm using a PowerWave XS spectrophotometer reader (BioTek), according to the manufacturer’s recommendation.
NSCLC in vitro migration assay
In vitro migration of NSCLC cells was characterized using a Transwell Migration Assay (Thermo Fisher Scientific), according to the manufacturer’s recommendation. Briefly, the upper chamber of transwell insert was coated with 0.1% gelatin (MilliporeSigma) overnight. A549 and H226 cells were then plated in the upper chamber in culture medium with 1% FBS at the density of 3000 cells per well, whereas the lower chamber was filled with normal culture medium (with 10% FBS) to function as a chemoattractant. After 24 h, the upper chamber was removed and culture medium aspirated from the lower chamber. NSCLC cells migrated into lower chambers were fixed by 70% methanol and immunostained with 0.1% crystal violet. NSCLC relative migration was characterized by measuring migrated NSCLC cells in each lower chamber and normalizing them to migrated NSCLC cell numbers under control conditions.
Statistical analysis
All experiments were repeated for at least times, and averaged data were shown as mean ± standard error of the mean (SEM). Statistical analysis was performed using a Windows-based SPSS software (version 12.0; SPSS, USA). For analysis regarding correlation between BDNF-AS and NSCLC patients’ clinicopathological variance, a chi-square test was used. For analysis regarding NSCLC patients’ OS, a Kaplan–Meier model with log-rank test was used. For analysis regarding correlation between NSCLC patients’ clinicopathological variance and their OS, cox regression models with both univariate and multivariate estimations, as well as hazard ratio (HR) at 95% confidence intervals (CIs), were used. For all other analyses, a two-tailed unpaired Student’s t test was used. In all cases, a p value smaller 0.05 was considered as significantly different.
Results
BDNF-AS is downregulated in NSCLC and associated with late-stage NSCLC tumors
We first examined the expression profile of nature antisense BDNF-AS in NSCLC tumors. Through surgical procedures, NSCLC carcinoma tissues and their adjacent non-carcinoma lung epithelial (control) tissues were obtained. Analysis of qRT-PCR showed that BDNF-AS was significantly downregulated in NSCLC carcinoma tissues, as compared to control tissues (Figure 1(a), *p < 0.05). In addition, NSCLC tumors were divided into two sub-groups, based on their clinical stages. Eighty-one tumors were graded as early-stage tumors (stage I/II), and the other 70 tumors were graded as late-stage tumors (stage III/IV). Subsequent qRT-PCR analysis demonstrated that BDNF-AS downregulation was further associated with late-stage tumors than early-stage tumors (Figure 1(b), *p < 0.05).

BDNF-AS is lowly expressed in NSCLC. Clinical samples, including NSCLC carcinoma tissues and their adjacent non-carcinoma lung epithelial (control) tissues, were surgically obtained from 151 NSCLC patients. (a) qRT-PCR was used to compare BDNF-AS expression level between control and carcinoma tissues (*p < 0.05). (b) NSCLC tumors were separated into two groups, early stage (I/II, n = 81) and late stage (III/IV, n = 71). qRT-PCR was used to further evaluate BDNF-AS gene expression level between control tissues, early-stage NSCLC carcinoma tissues, and late-stage NSCLC carcinoma tissues (*p < 0.05).
BDNF-AS downregulation is associated with NSCLC patients’ key clinicopathological variances
As we showed that BDNF-AS downregulation was associated with late-stage NSCLC tumors, we further evaluated its clinical relevance with NSCLC patients’ clinicopathological variances. In all, 151 NSCLC patients were separated into two groups, according to the medium expression level of endogenous BDNF-AS. One group of patients (n = 76) had low endogenous BDNF-AS than the medium value. The other group of patients (n = 75) had high endogenous BDNF-AS than the medium value. The clinicopathological variances were tallied for two groups of NSCLC patients and then compared with each other using the chi-square test (Table 1). It showed that some of the clinicopathological variances, such as patients’ age, sex, smoking history, tumor differentiation status, or NSCLC subtype, were similar between patients with low BDNF-AS and patients with high BDNF-AS. However, some of the key clinicopathological variances, including TNM stage and lymph node metastasis, were significantly different between patients with low BDNF-AS and patients with high BDNF-AS (Table 1, *p < 0.05).
Correlation between BDNF-AS expression and patients’ clinicopathological variances in NSCLC (N = 151).
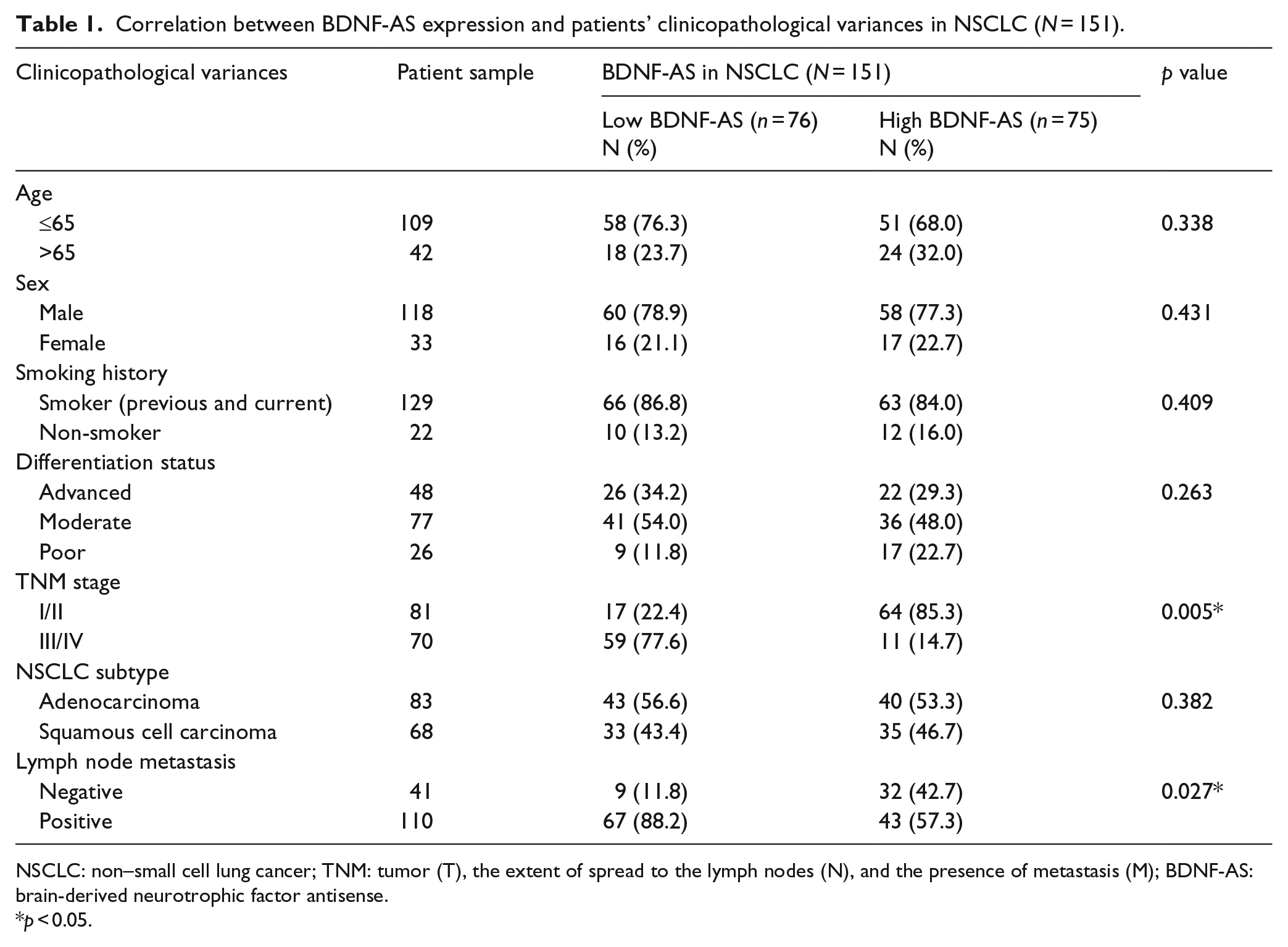
NSCLC: non–small cell lung cancer; TNM: tumor (T), the extent of spread to the lymph nodes (N), and the presence of metastasis (M); BDNF-AS: brain-derived neurotrophic factor antisense.
p < 0.05.
BDNF-AS downregulation is associated with NSCLC patients’ poor OS
As we showed that BDNF-AS downregulation was closely associated with NSCLC late-stage tumor and metastasis, we further evaluated its clinical relevance with NSCLC patients’ OS. Postoperative OS was followed up for two groups of NSCLC patients up to 45 months after surgery. It demonstrated that patients with low BDNF-AS had much worse OS than patients with high BDNF-AS (Figure 2, log-rank test, p < 0.005). We also evaluated the association between NSCLC patients’ clinicopathological variances and their OS. Univariate estimation showed that patients’ TNM stage (p = 0.003, HR (95% CI) = 1.211 (0.983–1.354)), lymph node metastasis (p = 0.023, HR (95% CI) = 1.844 (1.088–3.217)), and BDNF-AS expression level (p = 0.002, HR (95% CI) = 3.492 (2.774–5.912)) were significantly associated with their OS (Table 2, univariate estimation). Moreover, we used multivariate cox regression estimation to examine whether these three variances (TNM stage, lymph node metastasis, and BDNF-AS expression) may independently predict NSCLC patients’ OS. The estimation showed that, each of them, p = 0.009, HR (95% CI) = 0.987 (0.859–1.124) for TNM stage, p = 0.031, HR (95% CI) = 1.452 (0.876–2.453) for lymph node metastasis, and p = 0.004, HR (95% CI) = 2.894 (2.051–4.112) for BDNF-AS expression level, was capable of predicting patients’ OS independently (Table 2, multivariate estimation).

Association of BDNF-AS expression with NSCLC patients’ overall survival. Postoperative overall survivals were compared, using the Kaplan–Meier model along with log-rank test, between NSCLC patients with low BDNF-AS expression and NSCLC patients with high BDNF-AS expression (p < 0.005).
Cox regression estimations between clinicopathological variances and overall survival (OS) in NSCLC (N = 151).

NSCLC: non–small cell lung cancer; TNM: tumor (T), the extent of spread to the lymph nodes (N), and the presence of metastasis (M); HR: hazard ratio; CI: confidence interval; BDNF-AS: brain-derived neurotrophic factor antisense.
p < 0.05.
BDNF-AS is downregulated in NSCLC cell lines and its overexpression functionally inhibited NSCLC proliferation and migration
Finally, we evaluated the functional mechanisms of BDNF-AS in NSCLC. We used qRT-PCR to examine the expression profile of BDNF-AS in several NSCLC cell lines. It demonstrated that, as compared to the expression level in NuLi-1 cells, BDNF-AS was significantly downregulated in all evaluated NSCLC cell lines, including A549, H226, H596, H510A, H920, H522, H2991, SK-MES-1, and SW900 cells (Figure 3(a), *p < 0.05).

BDNF-AS is downregulated in NSCLC. (a) qRT-PCR was used to evaluate endogenous BDNF-AS expressions in NSCLC cell lines, including A549, H226, H596, H510A, H920, H522, H2991, SK-MES-1, and SW900 cells. Their expressions were compared to BDNF-AS expression in a normal human bronchial lung epithelium cell line (NuLi-1; *p < 0.05). (b) A549 and H226 cells were transfected with a BDNF-AS overexpression plasmid, pcDNA/BDNFAS, or a negative-control overexpression plasmid, pcDNA/+. After transfection was stabilized, endogenous BDNF-AS expression levels were evaluated by qRT-PCR between un-transfected cells (blank) and those transfected with pcDNA/+ or pcDNA/BDNFAS (*p < 0.05, Δp > 0.05, vs blank). (c) A CellTiter-Glo Luminescent Cell Viability Assay was performed to compare NSCLC viability between un-transfected A549 and H226 cells (blank) and those transfected with pcDNA/+ or pcDNA/BDNFAS. Absorbance was measured at an optical density of 440 nm (Δp > 0.05 vs blank).
We forced upregulation of BDNF-AS in NSCLC cell lines. A549 and H226 cells were transfected with a BDNF-AS overexpression plasmid, pcDNA/BDNFAS, or a negative-control plasmid pcDNA/+. After transfection was stabilized. Analysis of qRT-PCR demonstrated that, as compared to its expression level in un-transfected NSCLC cells (blank), endogenous BDNF-AS was not altered by pcDNA/+ but significantly upregulated by pcDNA/BDNFAS in both A549 and H226 cells (Figure 3(b), *p < 0.05, Δp > 0.05). In addition, cancer viability assay demonstrated that transfection-induced BDNF-AS upregulation did not induce apoptosis or cell death among NSCLC cells (Figure 3(c), Δp > 0.05).
Transfected A549 and H226 cells were then re-seeded in 96-well plates and evaluated by a proliferation assay for five consecutive days. It showed that, 72 h after the onset of assay, cancer proliferation was significantly reduced by BDNF-AS upregulation in both A549 and H226 cells (Figure 4(a), *p < 0.05). Moreover, transfected NSCLC cells were evaluated through a transwell migration assay. After 24 h, A549 and H226 cells migrated into the lower chamber of transwell assay were identified by immunohistochemistry. It showed that, in both A549 and H226 cells, much less NSCLC cells migrated into the lower chamber with BDNF-AS upregulation (Figure 4(b) and (c), left). Through quantitative evaluation, it showed that, in both A549 and H226 cells, relative migration capability was reduced by approximately 40% to 50% with BDNF-AS upregulation (Figure 4(b) and (c), right, *p < 0.05).

BDNF-AS regulates NSCLC proliferation and migration. (a) Transfected A549 and H226 cells were re-seeded in 96-well plates and evaluated through an in vitro proliferation assay (5 days). Proliferation rates were measured at absorbance of 570 nm every 24 h and compared between NSCLC cells transfected with pcDNA/+ and NSCLC cells transfected with pcDNA/BDNFAS (*p < 0.05). Transfected (b) A549 and (c) H226 cells were evaluated through an in vitro transwell migration assay; 24 h after the start of assay, NSCLC cells in the lower chambers were immunostained and imaged (left panels). Relative migration was evaluated, and compared between NSCLC cells transfected with pcDNA/+ and NSCLC cells transfected with pcDNA/BDNFAS (right panels, *p < 0.05).
Discussion
In a previous clinical study, Okamura et al. 25 examined tumor samples from 102 NSCLC patients and found BDNF, along with its receptor TrkB, was highly expressed in tumor cells and closely correlated with aggressive malignance in NSCLC cells and poor prognosis of patients with NSCLC. These findings thus lead to our hypothesis that nature antisense of BDNF, BDNF-AS, may act oppositely as BDNF in NSCLC, as previous study suggested that BDNF-AS and BDNF were inversely correlated. 10 Thus, as the first step of our study, we used qRT-PCR to compare BDNF-AS expression between NSCLC carcinoma tissues and their adjacent non-carcinoma lung tissues in 151 NSCLC patients. And we discovered that BDNF-AS was significantly downregulated in NSCLC tumors than in normal lung tissues. This result is consistent with Okamura’s report, suggesting that BDNF-AS is reversely expressed as BDNF in NSCLC tumors.
As we investigated further into the expression profile of BDNF-AS in NSCLC, we found that BDNF-AS was further downregulated in late-stage NSCLC tumors than in early-stage NSCLC tumors. This result was further confirmed by our statistical analysis on patients’ clinicopathological variances, as the result of the chi-square test demonstrated that TNM stage was closely associated with differential BDNF-AS expression in NSCLC. Interestingly, in Okamura et al.’s 25 report, differential BDNF expression was not statistically associated with patients’ TNM stage. One possible explanation may be that, in our study, NSCLC patients had either subtype of adenocarcinoma or subtype of squamous cell carcinoma. Yet, in Okamura’s report, NSCLC patients with the third subtype, large cell neuroendocrine carcinoma, were also included in their analysis. Therefore, our study may suggest that clinical relevance of BDNF-AS is much significant within selective or defined NSCLC subtypes. However, the discrepancy between BDNF’s and BDNF-AS’s clinical relevance indicates that, though expression of BDNF and BDNF-AS may be closely (and reversely) associated with each other, other factors may complexly contribute to their prognostic properties in NSCLC.
Our analysis on NSCLC patients’ OS revealed that BDNF-AS downregulation is closely correlated with patients’ poor survival. This result is also consistent with Okamura’s report, which showed NSCLC patients with BDNF-positive tumors had worse OS than NSCLC patients with BDNF-negative tumors. It suggests that BDNF upregulation, or BDNF-AS downregulation, or both BDNF upregulation and BDNF-AS downregulation might be serving as potential biomarker to predict poor prognosis of patients with NSCLC.
Since we discovered that BDNF-AS was closely associated with late-stage NSCLC tumors, which is indicative of tumor development or proliferation, and with lymph node metastasis, which is indicative of migration, we thus further evaluated the function of BDNF-AS in regulating NSCLC cancer cell proliferation and migration in vitro. We transfected NSCLC cells with an overexpression plasmid to upregulate endogenous BDNF-AS in A549 and H226 cells. By examining cancer cell viability, we discovered that transfection-induced BDNF-AS upregulation did not cause apoptosis or cell death among NSCLC cells. This is an interesting observation, as it was demonstrated that inhibition of BDNF or application of TrkB antagonists induced cancer cell apoptosis in hepatocellular carcinoma. 28 However, as we mentioned earlier, this discrepancy may further highlight the notion that much more complex mechanisms may be involved in the (inverse) association and functions between BDNF and BDNF-AS in human NSCLC.
On the contrary, we discovered that BDNF-AS overexpression had tumor suppressive effect in NSCLC tumor cells by significantly inhibiting cancer proliferation and migration in A549 and H226 cells. Functional role of BDNF-AS was recently shown to be an active pro-neuronal and anti-apoptosis factor in neural populations.20,21 However, the novelty of this study is that our report is the first one to reveal a regulatory mechanism of BDNF-AS in NSCLC, or any other human cancers.
Conclusion
In conclusion, our report demonstrated that nature antisense BDNF-AS was downregulated in both NSCLC human tumors and NSCLC cell lines. Low expression of BDNF-AS is closely correlated with patients’ key clinicopathological variances and poor OS, making it a potential prognostic biomarker for NSCLC diagnosis. Furthermore, we demonstrated for the first time that overexpression of BDNF-AS had an anti-tumor effect in inhibiting NSCLC growth and metastasis, making it a potential therapeutic target for NSCLC treatment.
Footnotes
Acknowledgements
The authors M.S. and Z.X. contributed equally to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
