Abstract
The purpose of this study was to examine the safety and efficacy of sorafenib in Chinese patients with unresectable hepatocellular carcinoma. Data of 338 Chinese patients from the Global Investigation of therapeutic DEcisions in hepatocellular carcinoma and Of its treatment with sorafeNib study database were included. Patients were divided into those who received and did not receive sorafenib prior to surgical resection and those with and without portal vein tumor thrombosis. In the non-surgery group, the median survival was 302 days (95% confidence interval: 244–371), and the median time from diagnosis to death was 428 days (95% confidence interval: 352–556); in the surgery group, half of the patients survived for 345 days and the median time from diagnosis to death was 1000 days (95% confidence interval: 750–2816). Median progression-free survival and median time to progression were not different between the two groups. Median overall survival was 360 days (95% confidence interval: 309–435) in the non–portal vein tumor thrombosis group and 240 days (95% confidence interval: 181–296) in the portal vein tumor thrombosis group; median time between hepatocellular carcinoma diagnosis and death was 750 days (95% confidence interval: 472–1000) and 420 days (95% confidence interval: 252–567), respectively, in the two groups. Median progression-free survival was 209 days (95% confidence interval: 166–264) for patients without portal vein tumor thrombosis and 154 days (95% confidence interval: 112–202) for patients with portal vein tumor thrombosis; median time to progression was 295 days (95% confidence interval: 209–463) and 221 days, respectively. Adverse events were generally comparable regardless of prior surgery and portal vein tumor thrombosis status. We thus conclude that earlier administration of sorafenib may result in improved outcomes in patients with unresectable hepatocellular carcinoma and portal vein tumor thrombosis.
Introduction
Hepatocellular carcinoma (HCC) is the second most common cause of cancer mortality worldwide. 1 More than 75% of cases occur in the Asia-Pacific region, more than 50% of HCC cases from this region occur in China, and approximately 383,000 patients residing in China die from HCC each year.1,2 Sorafenib is a multi-kinase inhibitor used for the treatment of unresectable HCC. 3 Studies have examined sorafenib alone4,5 and in combination with transarterial chemoembolization (TACE).6–10 A recently published meta-analysis of randomized controlled trials (RCTs) concluded that treatment significantly improved overall survival (OS) and time to progression (TTP) of disease in patients with advanced HCC. 11 However, studies have varied with respect to disease stage, interventions, and dosing.
The Global Investigation of therapeutic DEcisions in HCC and Of its treatment with sorafeNib (GIDEON) 3 was a global, non-interventional, surveillance study that examined the safety of sorafenib for the treatment of unresectable HCC. Different from the other phase-III clinical trials4,7,12 that mostly included patients with Child–Pugh A liver disease, the study also included late-stage patients with Child–Pugh B or C liver function. The study, reflecting a growing interest in real-world outcomes of HCC patients treated with sorafenib, has generated a large database from a broad population with unresectable HCC. The first interim analysis of GIDEON data 13 showed global and regional differences in patient characteristics, disease causes, and practice patterns. The second interim analysis, 3 consistent with the first analysis, showed that the overall safety profile and dosing strategy were similar across Child–Pugh classes and that safety findings were comparable irrespective of initial sorafenib dose or Barcelona Clinical Liver Cancer (BCLC) stage. The results also showed that the initial treatment of unresectable HCC varied by country and region.3,13 Although TACE is widely used for the treatment of HCC, in the Asia-Pacific region, surgery remains the primary treatment method for resectable HCC. 14 However, in a large proportion of patients with HCC, the disease is not resectable.
In China, sorafenib is not widely available, and because it is not covered by insurance, it is rarely used as first-line treatment, and majority of BCLC-C stage HCC patients, including those with portal vein tumor thrombosis (PVTT), do not receive it as first-line therapy. This is different than proposed by international guidelines. For example, the American Association for the Study of the Liver Diseases/Barcelona Clinic for Liver Cancer (AASLD/BCLC) Staging System and Treatment Guidelines consider patients with PVTT to have stage C disease, and the only proposed treatment option is sorafenib.11,15 HCC with PVTT has a poor prognosis (3-year survival of approximately 17%), and the optimal treatment for HCC with PVTT remains unclear.16,17 In China, the HCC clinical guidelines recommend that BCLC-B or BCLC-C patients may choose surgery as first-line treatment. Furthermore, the administration of sorafenib for HCC with PVTT after surgery is also performed, even though not currently recommended in Western guidelines.15,18
This study is a sub-analysis of the GIDEON study data examining the safety and efficacy of sorafenib in Chinese patients with prior surgical resection and those with PVTT.
The two study subgroups of interest were patients with unresectable HCC and those with PVTT, in which the use of sorafenib would likely have been indicated as first-line treatment following Western guidelines.
Materials and methods
Study population
The protocol and documentation were approved by the relevant institutional review boards, ethics committees, and health authorities.
The GIDEON study was an international prospective, open-label, multi-centered, non-interventional study of patients with unresectable HCC who had indications for systemic therapy and received sorafenib. The duration of observation was from the initiation of sorafenib therapy to withdrawal from the study, last follow-up, or death. The GIDEON study protocol (NCT00812175) is available at https://www.clinicaltrials.gov/ct2/show/NCT00812175.
Inclusion criteria were as follows: (1) histology/cytology or imaging examination such as multidimensional dynamic computed tomography (CT) or magnetic resonance imaging (MRI)-confirmed unresectable HCC, (2) indication for systemic therapy and was willing to receive sorafenib, (3) expected survival time more than 8 weeks, (4) provided informed consent, (4) clinician in charge was able to submit the completed Case Report Forms, and (6) clinicians were willing to undergo inspections aimed at verifying the accuracy of their raw and submitted data. Exclusion criteria were consistent with the sorafenib (Nexavar®) package information and the criteria of the local treatment center. 19
Sorafenib administration and study procedures
The indications for sorafenib therapy were consistent with recommendations on the use of sorafenib in local settings, and the dose of sorafenib and the duration of sorafenib therapy were determined by the local physicians based on the specific conditions of individual patients.
Observations were made and recorded from the initial patient visit to withdrawal from the study, follow-up, or death. During this period, patients were seen once every 8 weeks or at a frequency determined by the clinicians in charge. The interval between two visits was no shorter than 6 weeks and no longer than 12 weeks. Information recorded at each visit included body weight, blood pressure, Eastern Cooperative Oncology Group (ECOG) scores, Child–Pugh grade, information on sorafenib therapy, adverse events (AEs), and concomitant use of drugs and other therapies for HCC. After discontinuation of sorafenib, patient visits were continued every 8 weeks, and in addition to the above information, α-fetoprotein (AFP) level and AEs within 30 days after therapy discontinuation were also recorded. When a patient died or withdrew from the study for any reason, data from the last visit were used in the analysis.
Information related to sorafenib therapy included the dose of sorafenib, duration of sorafenib therapy, and date of and reasons for suspension or reduction in dose of sorafenib therapy.
Efficacy evaluation
End points for therapeutic efficacy were based on the Response Evaluation Criteria in Solid Tumors (RECIST) and included OS, TTP of disease, progression-free survival (PFS), remission rate (RR), and stable disease (SD) rate. Parameters used for HCC evaluation were determined by the clinicians in charge on the basis of CT or other imaging findings. CT or other imaging examinations were performed at baseline and once every 8 weeks or at a frequency determined by the clinicians in charge.
Safety evaluation
Safety-related parameters were used for the evaluation of overall tolerance. The Common Terminology Criteria for Adverse Events Version 3.0 grading system for AEs was used. The relationship between AEs reported by clinicians and patients and the drugs used, severity of AEs, measures taken in response to AEs, and outcomes were recorded. The Child–Pugh class and ECOG scores were also recorded for safety evaluation. Severe adverse events (SAEs), regardless of whether they were related to sorafenib therapy, were also recorded.
Statistical analysis
Outcomes regarding efficacy were reported based on data from the intent-to-treat (ITT) population. Parameters related to the therapeutic efficacy of sorafenib included OS, PFS, TTP, and best overall response. The Kaplan–Meier (KM) method was used for the evaluation of OS, PFS, and TTP, and the corresponding KM curves were shown. Safety outcomes were computed based on data from the safety set. All statistical analyses were performed with SAS software version 9.3 (SAS Institute Inc., Cary, NC, USA). Descriptive statistics were computed for demographic, clinical, efficacy, and safety data. Continuous variables were presented as the mean ± standard deviation (SD) or median (range); categorical variables were presented as count and percentage.
Results
Study population
A total of 338 patients were eligible for ITT classification; however; 7 patients were excluded because they did not attend evaluation after enrollment (4 patients failed screening and 3 patients were not treated with the study drug). Thus, data of 331 eligible patients who met the inclusion criteria were assessed for safety (safety population).
Patient baseline demographic and clinical characteristics
Baseline demographic and clinical characteristics of patients in the ITT set categorized by whether they had prior surgery or PVTT are summarized in Table 1. A total of 32.8% of patients had prior surgery, and this group showed a tendency of earlier HCC stage than non-surgery patients. Surgical patients had a lower frequency of vascular invasion (12.6% vs 27.8%), PVTT (17.1% vs 42.3%), tumor node metastasis (TNM) stage III or IV (56.7% vs 87.2%), BCLC stage C or D (52.3% vs 74.9%), Child–Pugh score B or C (7.2% vs 19.8%), and Cancer of the Liver Italian Program (CLIP) score 4–6 (3.6% vs 20.7%), but a higher frequency of prior loco-regional therapy (82.9% vs 68.7%) and tumor confined to the liver (55.9% vs 35.7%). There were no differences of age, sex, history of hepatitis B, extrahepatic disease, lymph node metastases, number of lesions in the liver, or ECOG performance status between the two groups.
Patient baseline (at study entry) demographic and clinical characteristics.

PEI: Percutaneous ethanol injection; RFA: radiofrequency ablation; TACE: transarterial chemoembolization; ECOG PS: Eastern Cooperative Oncology Group Scale of Performance Status; TNM: tumor node metastasis; BCLC: Barcelona Clinic Liver Cancer; CLIP: Cancer of the Liver Italian Program; HCC: hepatocellular carcinoma.
Age is presented as mean ± standard deviation and (range). Other variables are presented as number and (%).
Some data were missing or could not be found: 2 for age; 32 for TNM stage; 33 for BCLC stage; 35 for Child–Pugh score; 53 for CLIP score; 14 for portal vein thrombosis; 28 for number of liver lesions.
Concomitant therapy refers to using at least two therapies for local–regional anti-cancer treatment.
Patients with PVTT, in comparison to those without PVTT, had a lower frequency of prior surgery (16.5% vs 42.6%), prior loco-regional therapy (65.2% vs 78.5%), tumor confined to the liver (19.1% vs 54.5%), and CLIP score 0 or 1 (4.3% vs 50.7%), but a higher frequency of vascular invasion (60% vs 3.8%), TNM stage III or IV (94.8% vs 69.9%), BCLC stage C or D (95.7% vs 53.1%), and CLIP score 4–6 (40.9% vs 1.9%). There were no differences in age, sex, history of hepatitis B, extrahepatic tumor spread, lymph node metastases, Child–Pugh score, number of lesions in the liver, or ECOG performance status between the two groups (Table 1).
Changes in disease characteristics from the time of diagnosis to study entry
Disease characteristics at initial diagnosis of HCC and at study entry (baseline) are presented in Table 2. Compared with the time of diagnosis, HCC had progressed and liver function tests had worsened by the time of study entry. Thus, regardless of surgical history by the time sorafenib was started, the proportion of patients with a more advanced TNM stage IV, BCLC stage C or D, and multi-nodular plus extensive tumor morphology had increased. In general, all of the clinical characteristics of the no-surgery group at initial diagnosis were worse compared to the prior surgery group.
Disease characteristics at initial diagnosis of HCC and at start of sorafenib.
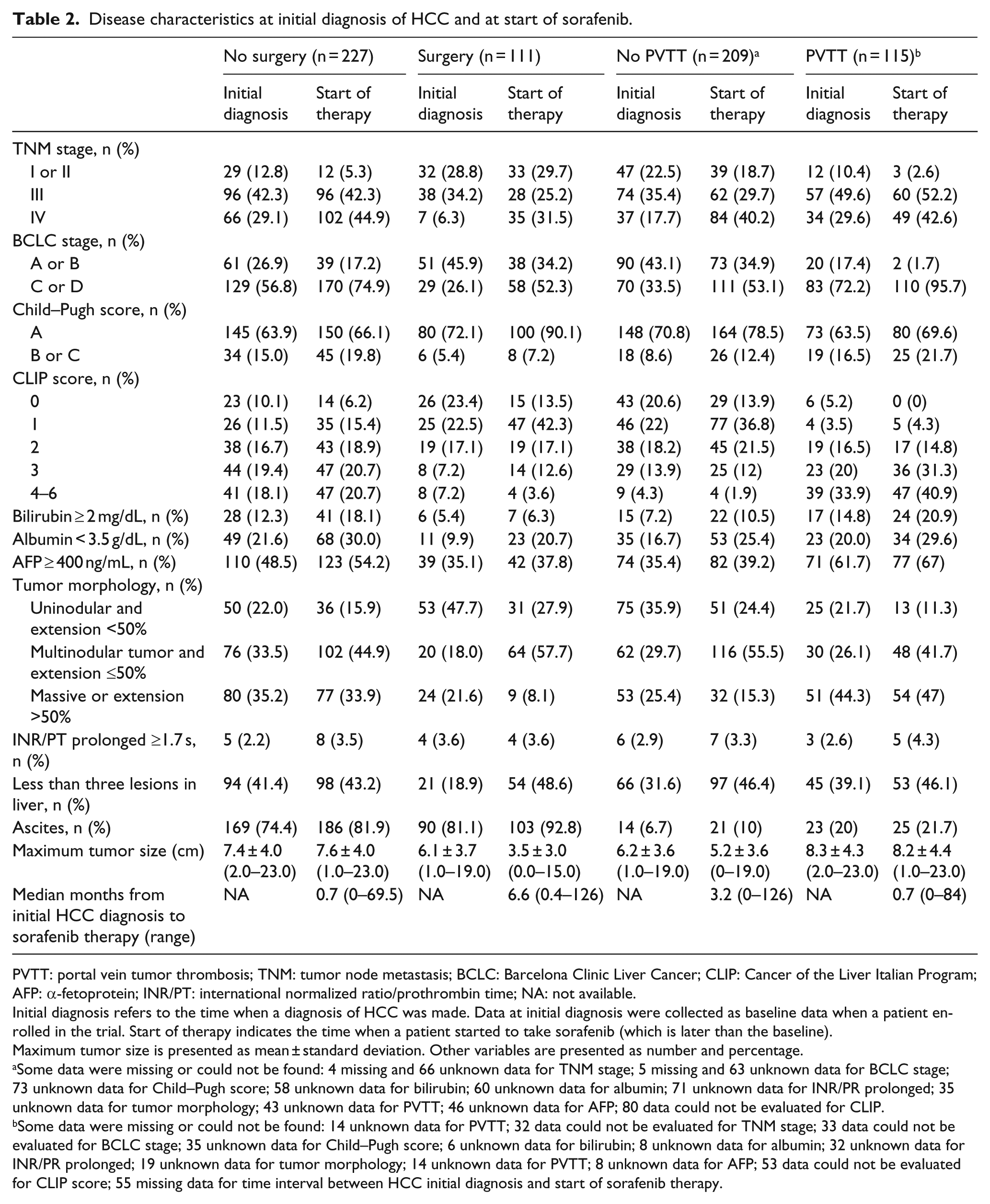
PVTT: portal vein tumor thrombosis; TNM: tumor node metastasis; BCLC: Barcelona Clinic Liver Cancer; CLIP: Cancer of the Liver Italian Program; AFP: α-fetoprotein; INR/PT: international normalized ratio/prothrombin time; NA: not available.
Initial diagnosis refers to the time when a diagnosis of HCC was made. Data at initial diagnosis were collected as baseline data when a patient enrolled in the trial. Start of therapy indicates the time when a patient started to take sorafenib (which is later than the baseline).
Maximum tumor size is presented as mean ± standard deviation. Other variables are presented as number and percentage.
Some data were missing or could not be found: 4 missing and 66 unknown data for TNM stage; 5 missing and 63 unknown data for BCLC stage; 73 unknown data for Child–Pugh score; 58 unknown data for bilirubin; 60 unknown data for albumin; 71 unknown data for INR/PR prolonged; 35 unknown data for tumor morphology; 43 unknown data for PVTT; 46 unknown data for AFP; 80 data could not be evaluated for CLIP.
Some data were missing or could not be found: 14 unknown data for PVTT; 32 data could not be evaluated for TNM stage; 33 data could not be evaluated for BCLC stage; 35 unknown data for Child–Pugh score; 6 unknown data for bilirubin; 8 unknown data for albumin; 32 unknown data for INR/PR prolonged; 19 unknown data for tumor morphology; 14 unknown data for PVTT; 8 unknown data for AFP; 53 data could not be evaluated for CLIP score; 55 missing data for time interval between HCC initial diagnosis and start of sorafenib therapy.
In patients with or without PVTT, TNM stage, BCLC stage, CLIP score, and the extent of tumor morphology had increased from the time of initial diagnosis to the start of sorafenib (Table 2). The median time interval between initial diagnosis of HCC and the start of sorafenib was longer in the surgery group than in the non-surgery group (6.6 months vs 0.7 months) and longer in the non-PVTT group than in the PVTT group (3.2 months vs 0.7 months; Table 2).
Efficacy assessment
Overall, there were 166 deaths (115 in the non-surgery group and 51 in the surgery group). KM survival curves for the surgery and non-surgery groups are shown in Figure 1. In the non-surgery group, the median survival time was 302 days (95% confidence interval (CI): 244–371); in the surgery group, half of the patients survived for 345 days. In the non-surgery group, the median time interval from initial diagnosis to death was 428 days (95% CI: 352–556); in the surgery group it was 1000 days (95% CI: 750–2816).

Kaplan–Meier curve of (a) overall survival and (b) time from initial diagnosis to death stratified by patients who had surgery (n = 227; red line) or no surgery (n = 111; blue line).
KM curves of PFS and TTP of 47 patients who had prior surgery and 50 without prior surgery are shown in Figure 2. By RECIST 1.0 criteria, the median PFS time in the surgery group was 206 days (95% CI: 149–232), and the median TTP was 221 days (95% CI: 146–463). In the non-surgery group, the median PFS time was 173 days (95% CI: 142–231), and the median TTP was 295 days. The PFS and TTP of the surgery group and the non-surgery group were not different. The results defined by the modified RECIST criteria were similar, although two additional progression events were identified with the modified criteria.
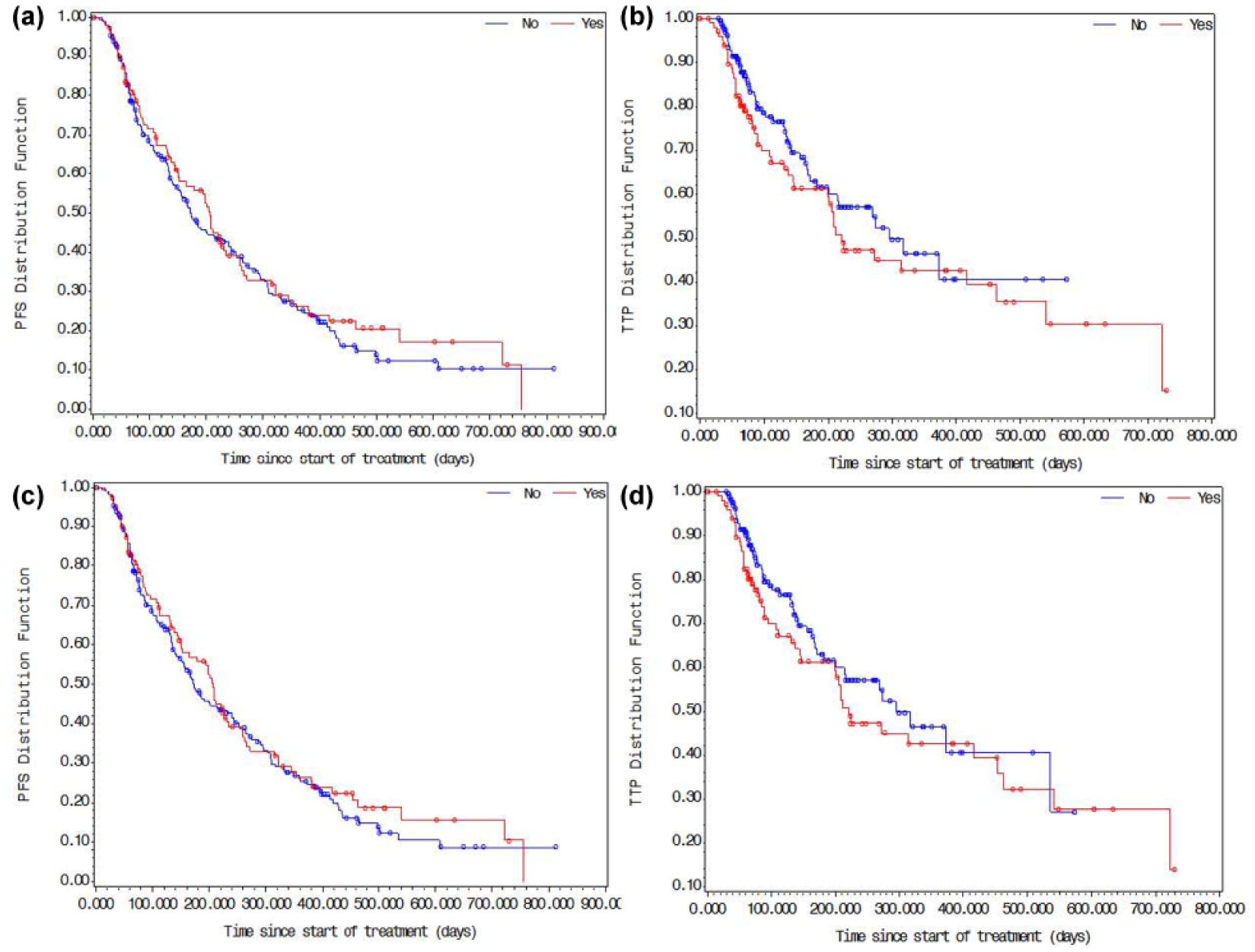
Kaplan–Meier curve of (a) progression-free survival (PFS) and (b) time-to-progression (TTP) defined by RECIST criteria. Red line indicates patients treated surgically and blue line indicates patients not treated surgically. (c and d) PFS and TTP defined by modified RECIST.
KM curves of OS and time from initial diagnosis to death in patients classified according to whether they had PVTT at the start of sorafenib therapy are shown in Figure 3. A total of 98 deaths occurred in the non-PVTT group and 63 in the PVTT group. The median OS was 360 days (95% CI: 309–435) in the non-PVTT group and 240 days (95% CI: 181–296) in the PVTT group. The median time interval between initial HCC diagnosis and death was 750 days (95% CI: 472–1000) in the non-PVTT group and 420 days (95% CI: 252–567) in the PVTT group.

Kaplan–Meier curve of (a) overall survival (OS) time and (b) from the time of initial diagnosis to death, stratified according to the presence of portal vein tumor thrombosis (PVTT) (red line; n = 115) or no PVTT (blue line; n = 209) at the start of sorafenib.
PFS and TTP of patients (n = 92) with or without PVTT analyzed according to the original and modified RECIST criteria are shown in Figure 4. The results were similar using the two versions, but one additional case was defined by the modified version. The median PFS time was 209 days (95% CI: 166–264) for patients without PVTT and 154 days (95% CI: 112–202) for patients with PVTT, and the median TTP was 295 days (95% CI: 209–463) for patients without PVTT and 221 days for patients with PVTT.

Kaplan–Meier curve of (a and c) progression-free survival (PFS) and (b and d) time to progression (TTP) from the start of sorafenib treatment defined by RECIST criteria of patients with (red line) or without (blue line) portal vein tumor thrombosis. Data in (a) and (b) were defined with the original RECIST criteria and data in (c) and (d) with the modified RECIST criteria.
Safety assessment
AEs are summarized in Table 3. AEs were generally comparable regardless of prior surgery and PVTT status, except that grade 3–4 AEs and AEs resulting in permanent discontinuation were slightly more frequent in patients with prior surgery than in those without prior surgery (9% vs 4.5% and 13.5% vs 6.4%, respectively). All-cause mortality was higher in patients with PVTT than in those without PVTT (57.3% vs 47.3%). Drug-related AEs and SAEs were comparable irrespective of surgical or PVTT status. The frequency of treatment-related deaths was about 20%, and the rate was similar regardless of surgery or PVTT status.
Treatment-related emergent adverse events and severe adverse events according to whether patients had surgery or portal vein tumor thrombosis.

AE: adverse event; SAE: severe adverse event.
A total of 14 data items could not be evaluated.
The time to onset of AEs after beginning sorafenib was generally comparable regardless of surgical history and PVTT status (Figures 5 and 6). In both patient groups, AEs occurred most frequently within 30 days of starting sorafenib and then declined markedly.
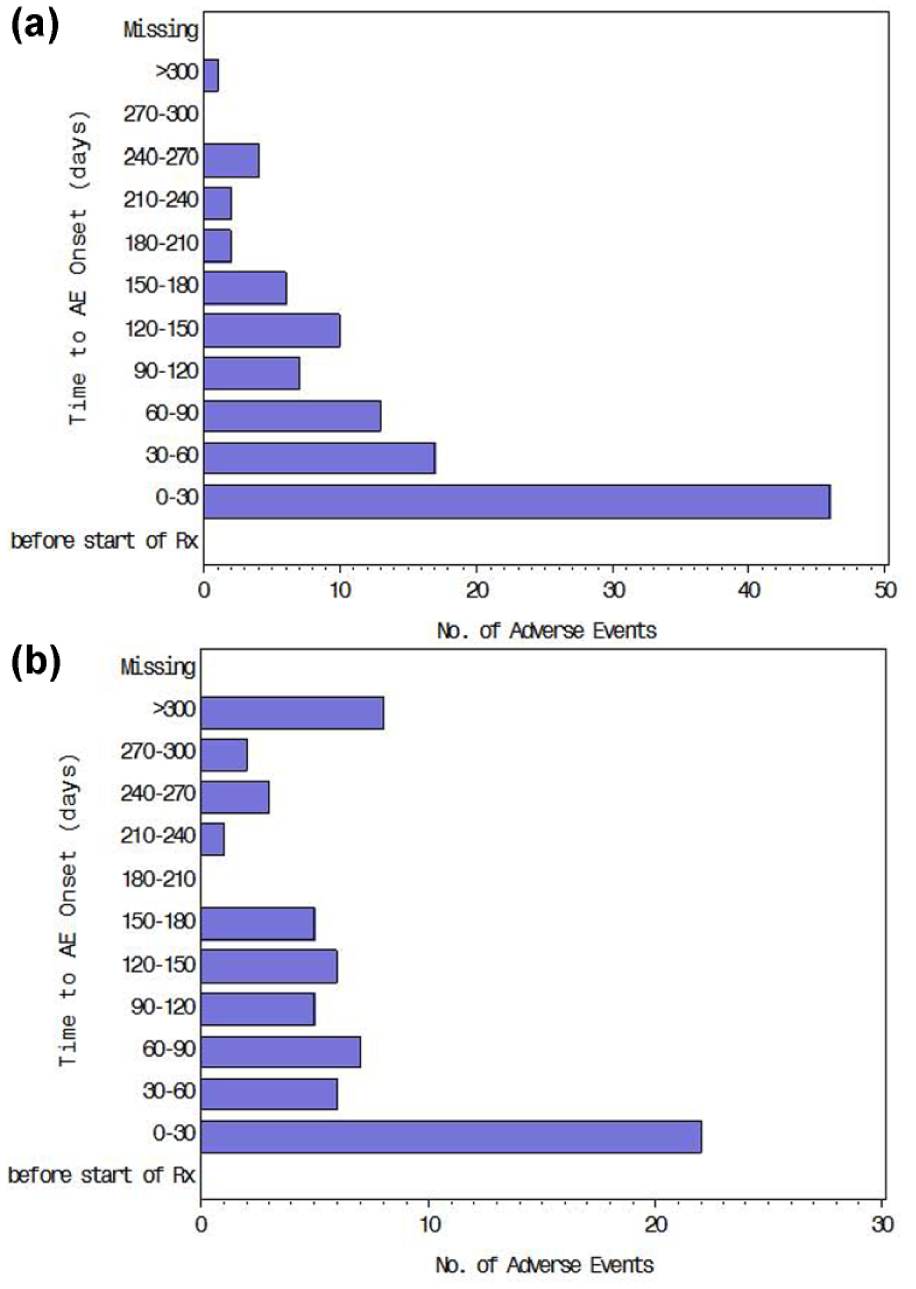
Time to onset of adverse events (AEs) after start of sorafenib in patients (a) without prior surgery (n = 220) and (b) with prior surgery (n = 111).

Time to adverse event (AE) onset after beginning sorafenib in patients (a) without portal vein tumor thrombosis (n = 207) and (b) with portal vein tumor thrombosis (n = 110).
Discussion
This study specifically evaluated patients who had undergone surgery before the start of sorafenib therapy and patients who had HCC with associated PVTT, both of which comprise more than one-third of the Chinese patients with unresectable HCC. The results showed that patients who received surgery had less advanced disease as compared to those who did not receive surgery. More advanced disease with conditions such as vascular invasion or HCC extending beyond the liver could have precluded surgery. Also, patients with PVTT had more advanced disease than patients without PVTT. The results also showed that the proportion of patients with disease progression during the period from the time of diagnosis to beginning sorafenib was greater in the surgery group than in the non-surgery group.
The median OS was comparable between patients with and without prior surgery. However, the time from initial diagnosis to death was longer in patients with prior surgery, and this may be in part attributed to less advanced disease at initial diagnosis. The GIDEON data showed a median OS of 13.6 months for Child–Pugh A patients.3,13 In our study, 90% of patients in the surgery group were Child–Pugh A at start of sorafenib (66.1% in the no-surgery group). Given that our OS data include Child–Pugh B and C patients as well, the results show no remarkable deviation from the global GIDEON results, suggesting the efficacy of sorafenib in terms of OS is comparable regardless of prior surgical history. This in combination with the AE data suggests that sorafenib should not be denied in patients with more advanced stage disease, which is the stage for the fear of greater toxicity.
A number of studies have examined sorafenib administered post surgery. Wang et al. 20 treated patients who had undergone surgical resection of HCC with sorafenib for 4 months postoperatively and reported that the time to recurrence and recurrence rates were significantly less in sorafenib-treated patients. Zhang et al. 21 recently reported that sorafenib did not significantly prolong recurrence-free survival or reduce the recurrence rate, but did significantly prolonged OS.
Therapies available for patients with PVTT include surgical resection, TACE, and radiotherapy, and while studies have suggested that hepatic resection may result in better long-term survival, outcomes are still poor. 22 Surgical resection of HCC with PVTT is more common in Asia than in other regions of the world, 14 and overall median survival of patients with PVTT ranges from 9 to 33 months based on the degree of invasion.23,24 The current analysis of patients treated with sorafenib indicated that PVTT resulted in shorter OS and shorter time from initial diagnosis to death as compared to patients without PVTT. An RCT performed in a Western population found that the median survival of patients with portal invasion treated with sorafenib was 8.1 months as compared to 4.9 months in patients not treated. 25 In an RCT conducted in an Asian population, survival of patients with PVTT and/or extrahepatic spread was 5.6 months as compared to 4.1 months in patients who did not receive sorafenib. 26
Common AEs associated with sorafenib are hand–foot skin reactions, diarrhea, fatigue, and lipase elevations, and hand–foot skin reactions and diarrhea are the most common that result in dose reduction, though they rarely lead to discontinuation of treatment.3,7,27 Study has shown that sorafenib is not associated with increased AEs when administered prior to hepatic resection. 28 In our study, the most frequent AE onset time was <30 days, and AEs were generally comparable regardless of prior surgical history and PVTT status, and the frequencies were comparable to those reported in the second interim analysis of the GIDEON study. 3
There are limitations to this study. First, an open-label registry observational study lacks a randomized control arm and therefore does not provide the same robust level of evidence as do RCTs. Second, the study was carried out at multiple tertiary care centers in China, so there may be many variations with respect to items such as classification of disease stage or severity (including interpretation of imaging studies), indications for surgery or medical treatment and treatment regimens, assessment and reporting of AEs, and thoroughness and duration of follow-up. Also, whether sorafenib was administered according to a continuous or an interrupted schedule was not assessed.
In summary, approximately one-third of Chinese unresectable HCC patients had prior surgery and one-third had PVTT before starting sorafenib, and the safety profile of sorafenib was similar regardless of prior surgical history or the presence of PVTT and comparable to the results of the GIDEON study. In China, sorafenib is generally administered to patients with advanced disease with surgery, and other treatments are preferred when the disease is less advanced. Earlier initiation of sorafenib prior to further deterioration of liver function and disease progression may lead to better outcomes than other treatments.
Footnotes
Ethical approval
The protocol and documentation were approved by the relevant institutional review boards, ethics committees, and health authorities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
