Abstract
Periostin (POSTN) is an extracellular matrix protein which is overexpressed in a variety of cancers and has been related to tumorigenesis of renal cell carcinoma. However, the involvement of POSTN in renal cell carcinoma migration, invasion, and their underlying mechanisms has not been established. In this study, renal cell carcinoma cell lines stably overexpressing POSTN were established using a lentiviral vector, and the effects of POSTN on renal cell carcinoma cell migration and invasion were investigated. POSTN overexpression increased the migration and invasion capabilities of renal cell carcinoma cell lines as well as activity of matrix metalloproteinase-2 and matrix metalloproteinase-9. Integrin αvβ3 and αvβ5 antibodies inhibited POSTN overexpression or recombinant POSTN-induced focal adhesion kinase activation, cell migration, and invasion. Furthermore, lentivirus-mediated focal adhesion kinase knockdown and c-Jun N-terminal kinase inhibitor reduced POSTN-enhanced phosphorylation of c-Jun N-terminal kinase, matrix metalloproteinase-9 and matrix metalloproteinase-2 expressions, cell migration, and invasion. Our research thus indicates that POSTN promotes renal cell carcinoma cell migration and invasion through interaction with integrins αvβ3 and αvβ5 and subsequent activation of the focal adhesion kinase/c-Jun N-terminal kinase pathway. These results suggest that POSTN plays a critical role in renal cell carcinoma metastasis and may represent a potential target for novel therapeutic approaches against renal cell carcinoma.
Keywords
Introduction
In recent years, because of an increase in the application of ultrasound in physical examinations, the number of incidentally diagnosed renal cell carcinomas (RCC) has increased in China.1,2 The clear cell subtype of renal cell carcinomas (ccRCC) is the most frequent and aggressive subtype of all RCC cases, accounting for more than 50% of cases in China. 1 Surgery remains the main treatment for ccRCC; however, it is not sufficiently effective for patients who present with advanced and metastatic disease relative to the patients who present with localized disease. 3 Therefore, a better understanding of the mechanisms involved in RCC metastasis is urgently required for the development of more effective therapeutic approaches.
Periostin (POSTN), also known as osteoblast-specific factor 2, is a 93-kDa-secreted protein and was originally found to promote cell adhesion and motility in osteoblastic cells. 4 The highly conserved N-terminal region of POSTN regulates cell function by binding to integrins while the variable C-terminal region regulates cell-matrix organization and interactions by binding extracellular matrix (ECM) proteins. 5 POSTN is present in many normal tissues and tissues under stress conditions and has been shown to be essential for the functioning of bone, tooth, and heart.6–8
Recently, overexpression of POSTN has been observed in a wide variety of cancers including colon cancer, 9 non-small cell lung cancer, 10 pancreatic cancer, 11 breast cancer, 12 gastric cancer, 13 and prostate cancer.14,15 POSTN is thought to increase cell survival, angiogenesis, epithelial-mesenchymal transition (EMT), invasion, and metastasis of carcinoma cells by binding to integrins, including αvβ3, αvβ5, and α6β4. 2 Depending on context, POSTN can activate specific integrin-mediated signaling pathways such as Akt/protein kinase B and focal adhesion kinase (FAK) signaling pathways, inducing different biological responses. 2 Our previous study showed that POSTN could induce EMT of prostate cancer cells through PI3K/Akt/GSK-3 beta signaling pathways. 16 In RCC, POSTN was elevated in stromal fibroblasts and enhanced ccRCC cell attachment. 17 However, the involvement of POSTN in other biological processes such as migration and invasion of RCC is still unclear. In this study, our goal was to clarify the effect of POSTN on migration and invasion of RCC cells and explore the underlying mechanism.
Materials and methods
Cell culture
The human RCC cell lines 786-O and ACHN were obtained from American Type Culture Collection (Manassas, VA, USA). All cells were cultured in Dulbecco’s Modified Eagle’s Media (Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS) and 1% penicillin–streptomycin at 37°C in 5% CO2.
Lentiviral infections and creation of stable cell lines
Lentiviral particles encoding POSTN cDNA (Lv-POSTN) were constructed as described previously. 16 For stable overexpression of POSTN, 786-O and ACHN cells were transfected with Lv-POSTN for 24 h, and complete medium containing 5 mg/mL puromycin was then used to select stably transfected cells. The empty lentiviral vector was used as negative control (Lv-NC). Stable cells overexpressing POSTN were obtained after 3 weeks, and the expression level of POSTN was determined by western blotting and real-time polymerase chain reaction (PCR).
For knockdown of FAK, cells were transiently transfected with FAK short hairpin RNA (shRNA) (human) lentiviral particles (Santa Cruz Biotechnology, Dallas, TX, USA) according to the manufacturer’s protocol.
Real-time PCR
Total RNA was isolated using the Qiagen RNeasy Mini Kit (Qiagen, Hilden, Germany) and reverse transcription was performed using SuperScript III First-strand Synthesis SuperMix (Invitrogen) according to the manufacturer’s instructions. Real-time PCR was performed on the Applied Biosystems StepOnePlus system (Foster City, CA, USA) using the following primers: POSTN, 5′-GAGCTTTACAACGGGCAAATAC-3′ (sense) and 5′-CTCCCTTGCTTACTCCCTTTC-3′ (antisense); β-actin, 5′-CACTCTTCCAGCCTTCCTTC-3′(sense) and 5′-GTACAGGTCTTTGCGGATGT-3′ (antisense). Each measurement was repeated at least three times, and all measurements were normalized to the corresponding β-actin content values using the 2−ΔΔCt method.
Western blotting
Cells were lysed in radioimmunoprecipitation assay (RIPA) lysis buffer and 10 µg of protein was subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis as described. 16 After transfer to polyvinylidene difluoride membrane (Millipore, Billerica, MA, USA) and incubation with appropriate primary and secondary antibodies, protein bands were visualized using enhanced chemiluminescence reagents (Pierce Biotechnology, Rockford, IL, USA). The following primary antibodies were used: anti-POSTN (Santa Cruz), anti-p-FAK, anti-FAK, anti-p-JNK, anti-JNK, anti-p-AKT, anti-AKT (all from Cell Signaling Technology, Danvers, MA, USA), anti-MMP-2 (Novus Biologicals, Littleton, CO, USA), anti-MMP-9 (Abcam plc., Cambridge, MA, USA), and anti-β-actin (Santa Cruz).
Wound-healing migration assay
RCC cells were plated on six-well tissue culture plates and grown to subconfluence (90%) in serum-containing medium. A scratch wound was then made in the monolayer using a pipette tip. The debris was removed and the cells were cultured in serum-free media. After incubation for an additional 24 h, cell migration was analyzed in six different microscopic fields and calculated as the percentage of wound healing.
Cell invasion assay
The invasion capability of RCC cells was determined using transwell assays. In brief, 5 × 104 cells were harvested in serum-free media and plated into the upper chamber of Matrigel invasion chambers (BD Biocoat, Bedford, MA, USA). Medium containing 10% FBS was applied to the lower chamber as chemoattractant. In some experiments, cells were treated with antibodies against αVβ3 and αVβ5 (10 µg/mL; Millipore), and inhibitor of c-Jun N-terminal kinase (JNK; SP600125, 10 µM; Santa Cruz). After 24-h incubation, the invading cells were fixed in 4% paraformaldehyde and stained with 1% toluidine blue. Cell numbers were quantified by counting in 10 random microscopic fields.
Zymography assay
Matrix metalloproteinase (MMP)-2 and MMP-9 activities were detected using zymography assays. Cells were incubated with different treatments in serum-free media for 24 h, and the proteins in the conditioned medium were concentrated. Proteins (10 µg) were then loaded on a 10% polyacrylamide gel containing 0.1% gelatin (Sigma-Aldrich). After staining with 0.5% Coomassie blue R250 (Sigma-Aldrich, St. Louis, MO, USA) for 3 h, gels were destained overnight in 30% methanol and 10% glacial acetic acid.
Statistical analysis
Significance of differences was analyzed using Student’s
Results
Creation of stable RCC cell lines overexpressing POSTN
RCC 786-O and ACHN cells were transfected with lentivirus expressing POSTN complementary DNA (cDNA) and then selected by puromycin. Real-time PCR assays showed that the mRNA expression levels of POSTN in transfected 786-O and ACHN cells were significantly higher than in cells without lentivirus transfection or transfected with Lv-NC (Figure 1(a)). Similar results were also found by western blotting assay, in which POSTN protein levels were significantly increased after Lv-POSTN transfection (Figure 1(b)). These results indicated that RCC cell lines stably overexpressing POSTN had been established.

Creation of renal cell carcinoma (RCC) cell lines stably overexpressing POSTN. RCC 786-O and ACHN cells were transfected with lentivirus encoding POSTN cDNA and selected by puromycin exposure. (a) mRNA expression of POSTN was detected by real-time PCR. (b) Protein levels of POSTN were detected by western blotting. Values are mean ± SD of three independent experiments.
POSTN overexpression promoted RCC cell migration and invasion
Cell migration capacity of 786-O and ACHN cells transfected with or without POSTN were analyzed by wound-healing assays (Figure 2(a) and (b)). The result showing that POSTN-overexpressing RCC cells exhibited higher wound closure activity than control cells suggesting that POSTN enhanced the motility of RCC cells.

The effect of POSTN overexpression on migration of RCC cells. Migration of (a) 786-O and (b) ACHN cells transfected with or without POSTN were identified by wound-healing assays. Representative images are shown at the top. Values are mean ± SD of three independent experiments.
The invasion capacity of 786-O and ACHN cells transfected with or without POSTN was then examined by transwell assay. As shown in Figure 3(a) and (b), invasive cells were increased after POSTN overexpression compared with the normal and Lv-NC transfection groups, indicating that POSTN promoted the invasive capacity of RCC cells. Moreover, we detected an effect of POSTN overexpression on MMP activity which has been closely correlated with degradation of basement membrane and invasion of cancer cells. 18 The results from zymography assays showed that activity of MMP-2 and MMP-9 was significantly increased compared with control cells (Figure 3(c) and (d)). This result suggested that enhanced activity of MMP may contribute to POSTN-induced RCC invasiveness.
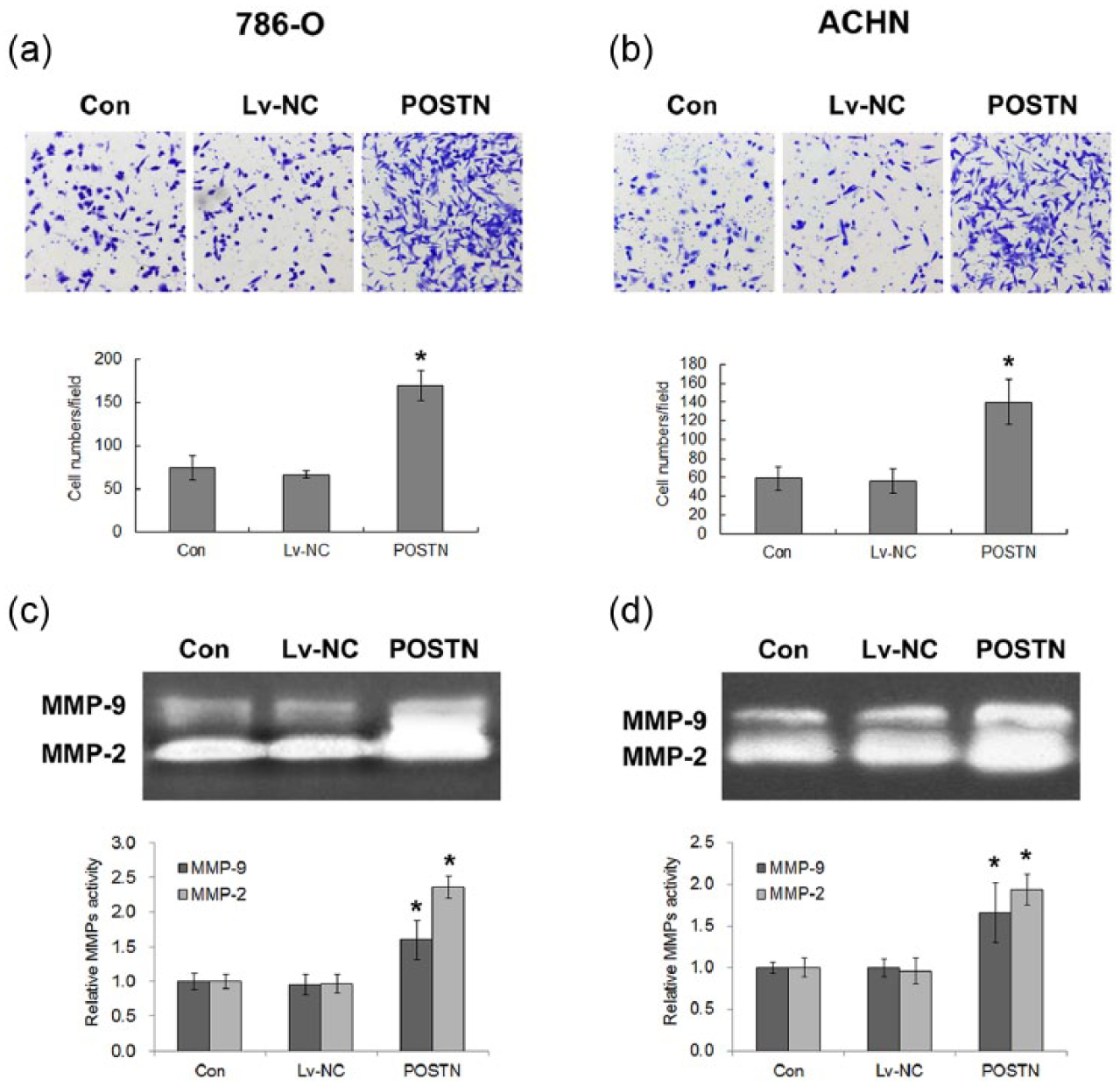
The effects of POSTN overexpression on the invasion and MMP activity of RCC cells. Invasion by (a) 786-O and (b) ACHN cells transfected with or without POSTN was detected by transwell assay. (c and d) MMP activity was demonstrated by zymography assay. Values are mean ± SD of at least three independent experiments.
αVβ3- and αVβ5-mediated POSTN-induced RCC cell migration and invasion
It has been reported that POSTN regulates smooth muscle cell migration through activation of the FAK pathway mediated by interaction with alphaV-integrins. 19 Here, we first determined whether the FAK pathway was activated by POSTN in RCC cells. As shown in Figure 4(a), low levels of FAK phosphorylation (p-FAK) were detected in both wild type and Lv-NC transfected 786-O cells. POSTN overexpression increased the phosphorylation level of FAK while not markedly affecting total levels of FAK. Integrins αVβ3 and αVβ5 have been identified as major potential receptors of POSTN in multiple cell types,20–22 but the interaction between POSTN and integrins in RCC cells has not yet been elucidated. To analyze the roles of integrins αVβ3 and αVβ5 on POSTN-mediated RCC cell migration and invasion, specific neutralizing antibodies were used to block both integrins. As shown in Figure 4(b), POSTN-induced upregulation of FAK phosphorylation was inhibited by treatment with either alone or combined αVβ3 and αVβ5 antibody. Moreover, MMP-2 and MMP-9 activity in 786-O cells was also significantly inhibited by αVβ3 or αVβ5 blocking antibody (Figure 4(c)). Migration and invasion abilities of 786-O cells treated with αVβ3 or αVβ5 antibody were also significantly lower than that in cells without antibody blocking conditions (Figure 4(d) and (e)).

POSTN activation of FAK and promotion of cell invasion through αVβ3 and αVβ5. (a) The effects of POSTN overexpression on FAK phosphorylation were demonstrated by western blotting. (b) 786-O cells transfected with or without POSTN were incubated with antibodies against integrins αVβ3 and/or αVβ5. FAK phosphorylation was detected by western blotting. (c) MMP activity was demonstrated by zymography assay. (d) Cell migration was identified by wound-healing assays. (e) Invasion ability was detected by transwell assay. (f) 786-O cells were incubated with 10 µg/mL recombinant human Periostin (rPOSTN) and antibodies against integrins αVβ3 and/or αVβ5. FAK phosphorylation was detected by western blotting. (g) Cell migration and (h) invasion ability were identified by wound-healing assays and transwell assay. Values are mean ± SD of at least three independent experiments.
To directly confirm that integrin αVβ3 or αVβ5 mediated the function of POSTN, recombinant human POSTN was used to treat 786-O cells. As shown in Figure 4(f)–(h), exogenous POSTN also induced upregulation of FAK phosphorylation and enhanced the motility and invasion capacity of RCC cells. These functions of POSTN were inhibited by either alone or combining αVβ3 and αVβ5 blocking antibody treatment. These results indicated that integrins αVβ3 and αVβ5.mediated POSTN-induced activation of FAK and MMPs and subsequently cell migration and invasion.
POSTN promoted RCC cell migration and invasion through the FAK/JNK pathway
FAK is linked with cell migration and invasion by activated multiple downstream signaling factors including JNK and Akt.23,24 To determine whether the FAK/JNK pathway was also involved in POSTN-induced RCC cell migration and invasion, we examined the effects of lentiviral-mediated FAK interference and JNK inhibitor SP600125 on POSTN function. As shown in Figure 5(a), POSTN overexpression increased phosphorylated JNK levels, whereas FAK knockdown attenuated the increase, which suggested that POSTN activated JNK signaling through FAK. Meanwhile, we found no significant change in phosphorylated Akt levels, suggesting that Akt signaling was not a major downstream signal of POSTN and FAK in RCC cells. We also detected the effect of POSTN on the protein levels of MMP-2 and MMP-9, and found that POSTN overexpression increased these two MMPs levels. Furthermore, FAK knockdown or JNK inhibitor attenuated the MMP levels, abrogated MMP activities (Figure 5(b)), cell migration (Figure 5(c)), and invasion (Figure 5(d)), which were all enhanced by POSTN. These results indicated that POSTN promoted RCC cell migration and invasion through the FAK/JNK pathway.

POSTN promotion of RCC cell invasion through the FAK/JNK pathway. The 786-O cells stably expressing POSTN or control vector were transfected with FAK shRNA or incubated with JNK inhibitor SP600125. (a) Western blotting shows the levels of phosphorylated and total FAK, JNK, and AKT, and the levels of MMP-9 and MMP-2. (b) MMP activity was demonstrated by zymography assay. (c) Cell migration was identified by wound-healing assay. (d) Cell invasion was detected by transwell assay. Values are mean ± SD of at least three independent experiments.
Discussion
POSTN is an ECM protein which is involved in a variety of biological processes including tumorigenesis by interacting with other ECM proteins or binding to cellular integrins. 2 POSTN upregulation was observed in high-grade RCC tumors and was significantly associated with poor overall survival.25,26 However, the relationship between POSTN and RCC metastasis, as well as the underlying mechanism, has not been clearly identified. In this study, we found that lentiviral-mediated POSTN overexpression activated the FAK/JNK signaling pathway via interaction with integrins αVβ3 and αVβ5, further increasing MMP activity and promoting RCC cell migration and invasion.
Consistent with previous studies in other cancers,20,27,28 our results showed that POSTN was also involved in motility and invasion of RCC. These data, combined with a recent report that extracellular POSTN could enhance A498 ccRCC cell attachment, 17 suggest that POSTN plays a critical role in the multistep cascade process of cancer metastasis. Invasion of basement membrane and ECM is necessary for metastasis of RCC, which depends on degradation of these components mainly by MMP. In this study, we found that POSTN overexpression significantly increased the enzyme activity of MMP-2 and MMP-9, which likely contributes to RCC cells migration and invasion.
Binding to integrins and subsequent activation of intracellular pathways is an important mechanism by which POSTN exerts its pro-tumorigenic effect. 2 Multiple integrins, including αvβ3, αvβ5, α6β4, and α5β1, have been reported to interact with POSTN in different cell types. Our results showed that αVβ3 or αVβ5 antibody blocked POSTN-promoted MMP activity, migration, and invasion abilities of RCC cells, demonstrating that αVβ3 or αVβ5 was involved in the effect of POSTN in RCC. These integrins mediate POSTN functions in epithelial ovarian carcinoma 20 and were also previously observed to be expressed in RCC. 29 Meanwhile, we found that αVβ3 or αVβ5 antibody inhibited POSTN-induced phosphorylation of FAK, which is a tyrosine kinase always activated by signaling from integrins. 30 Similar to POSTN overexpression, exogenous POSTN also promoted cell migration and invasion, and induced phosphorylation of FAK, which were inhibited by αVβ3 or αVβ5 antibody. These results verified the interaction of POSTN and these integrins more directly.
FAK is a nonreceptor tyrosine kinase and is one of most prominent components of integrin signaling. 30 Integrin activates FAK by inducing conformational changes and auto-phosphorylation of FAK. Our results showed that POSTN was unable to significantly enhance cell migration and invasion when FAK was downregulated. These data indicated that an integrin (αVβ3/αVβ5)/FAK pathway was required for POSTN to promote migration and invasion of RCC.
FAK contributes to cancer development through diverse molecular signaling pathways. For example, the extracellular signal–regulated kinases (ERK) pathway is required for FAK to maintain the growth or cell motility of at least some tumor cells.31,32 FAK suppresses apoptosis by binding to a receptor-interacting protein, which is then sequestered from the death-receptor complex. 33 FAK also regulates cell invasion through JNK signaling and MMP-mediated matrix degradation. 32 Although FAK may mediate cell migration and invasion through different signaling pathways, here, JNK activity was shown to be involved in both POSTN-induced cell migration and invasion in RCC cells. FAK knockdown or JNK inhibitor attenuated the MMP levels, which were enhanced by POSTN, suggesting that MMP-9 and MMP-2 are downstream factors of POSTN/FAK/JNK signaling, and POSTN may promote migration and invasion in RCC cells through upregulation of MMP expression. In addition, our previous study 16 showed that POSTN could induce EMT and cell invasion through PI3K/Akt signaling in prostate cancer cells; however, POSTN-activated Akt was not observed in RCC cells, suggesting that the function of POSTN is dependent on cell type.
In conclusion, this study demonstrates that POSTN promoted migration and invasion of RCC cells through interaction with integrins αvβ3 and αvβ5 and subsequent activation of the FAK/JNK pathway. This finding may provide new prospects for developing novel therapeutic approaches against RCC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was funded by the National Natural Science Foundation of China (Nos 81201620 and 81372316) and ChenGuang Program of Shanghai Municipal Education Commission (N158554).
