Abstract
Increasing evidence has indicated that the splicing factor hnRNPA2B1 plays a direct role in cancer development, progression, gene expression, and signal transduction. Previous studies have shown that knocking down hnRNPA2B1 in breast cancer cells induces apoptosis, but the mechanism and other functions of hnRNPA2B1 in breast cancer are unknown. The goal of this study was to investigate the biological function, clinical significance, and mechanism of hnRNPA2B1 in breast cancer. The expression of hnRNPA2B1 in 92 breast cancer and adjacent normal tissue pairs was analyzed by immunohistochemical staining. Stable clones exhibiting knockdown of hnRNPA2B1 via small hairpin RNA expression were generated using RNA interference technology in breast cancer cell lines. The effects of hnRNPA2B1 on cell proliferation were examined by MTT and EdU assay, and cellular apoptosis and the cell cycle were examined by flow cytometry. A nude mouse xenograft model was established to elucidate the function of hnRNPA2B1 in tumorigenesis in vivo. The role of hnRNPA2B1 in signaling pathways was investigated in vitro. Our data revealed that hnRNPA2B1 was overexpressed in breast cancer tissue specimens and cell lines. Knockdown of hnRNPA2B1 reduced breast cancer cell proliferation, induced apoptosis, and prolonged the S phase of the cell cycle in vitro. In addition, hnRNPA2B1 knockdown suppressed subcutaneous tumorigenicity in vivo. On a molecular level, hnRNPA2B1 knockdown decreased signal transducer and activator of transcription 3 and extracellular-signal-regulated kinase 1/2 phosphorylation. We concluded that hnRNPA2B1 promotes the tumorigenic potential of breast cancer cells, MCF-7 and MDA-MB-231, through the extracellular-signal-regulated kinase 1/2 or signal transducer and activator of transcription 3 pathway, which may serve as a target for future therapies.
Keywords
Introduction
Breast cancer is the most common malignancy and the second leading cause of cancer death in women worldwide. 1 Unfortunately, approximately one in eight women is likely to be diagnosed with breast cancer during her lifetime, and advanced breast cancers are currently incurable. 2 Thus, the clarification of molecular mechanisms underlying breast cancer progression is essential to reveal molecular targets for the development of therapeutics. 3
Recently, breast cancer progression and its response to therapy have been found to be linked to a number of differentially spliced oncogenes and tumor suppressors.4–6 The process of alternative splicing is broadly misregulated in cancers and contributes to the transformed phenotypes of cancer cells. 7 Moreover, several components of the spliceosome harbor mutations predicted to be driver mutations in several cancers suggesting that splicing factors are important players in cancer development.8,9 However, the acknowledge of splicing factors in cancer development and progression remains limited.10,11
Heterogeneous nuclear ribonucleoproteins (hnRNPs) are abundant RNA-binding proteins expressed in most human tissues. hnRNPs bind to nascent transcripts and are involved in messenger RNA (mRNA) biogenesis, DNA repair, telomere biogenesis, cell signaling, and the regulation of gene expression. 12 Increasing evidences suggest that some hnRNPs play a direct role in tumor development and progression. 13 The hnRNPA/B family is a key class within the hnRNP family of proteins. Previous studies have found that hnRNPA2B1 is overexpressed in lung and breast cancers.14,15 Knockdown of hnRNPA2B1 in breast cancer cells induces apoptosis, and hnRNPA2B1 plays an important role as a driver oncogene in glioblastoma development and acts as a predictor of glioblastoma patient survival.16,17 Recent studies have found that hnRNPA2B1 modulates hypoxia via alternative splicing of glycolytic pyruvate kinase isozyme 2 (PKM2) enzyme in cancer cells.18,19 However, the contribution of hnRNPA2B1 to the carcinogenic process is unclear yet.
Our study showed that hnRNPA2B1 is overexpressed in breast cancer tissue specimens and cell lines. Furthermore, we found that hnRNPA2B1increases the tumorigenic potential of cells by regulating signal transducer and activator of transcription 3 (STAT3) and extracellular-signal-regulated kinase 1/2 (ERK1/2) signaling pathway. Our study thus identifies a potential therapeutic target for human breast cancer.
Materials and methods
Breast cancer specimens
Breast cancer tissue samples and adjacent normal tissue samples were collected after obtaining informed consent from the patients. All procedures were performed under institutional review board-approved protocols. A total of 77 human breast tissue samples were obtained from breast cancer patients who underwent surgical resection in the First Affiliated Hospital of Third Military Medical University (Chongqing, China), from March 2015 to January 2016. Meanwhile, 15 normal human breast tissue specimens were also collected peripheral tissue surrounding the tumor lesions. The fresh tissue samples were immediately snap-frozen and stored in liquid nitrogen until use.
Immunohistochemistry
The tissue specimens were sectioned using the Ultra Sensitive TMS-P kit (#SP-9002; Zymed, San Diago, CA, USA) according to the manufacturer’s instructions. The sections were stained with an anti-hnRNPA2B1 antibody (1:100; Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) overnight at 4°C. The degree of immunostaining was reviewed and scored semiquantitatively by two independent observers. The staining index was calculated as the product of the proportion of positively stained tumor cells and the staining intensity. The former was scored as follows: 1 (<25%), 2 (25%–49%), and 3 (⩾50%). Staining intensity was graded as follows: 1 (weak), 2 (moderate), and 3 (strong). A staining index (SI = distribution score + intensity score) was established for each tissue and was scored between 1 and 9. An SI < 3 was considered as low hnRNPA2B1 expression, 3 ⩽ SI < 6 as medium expression, and SI ⩾ 6 as high expression.
Breast cancer cell lines and culture conditions
All cell lines used in this study (MCF-10A, MCF-7, and MDA-MB-231) were purchased from American Type Culture Collection (ATCC). The MCF-10A and MCF-7 cell lines were grown in Dulbecco’s Modified Eagle Medium (DMEM), and the MDA-MB-231 cell line was grown in PRMI-1640. The medium was supplemented with 10% fetal bovine serum (FBS) and 1% penicillin–streptomycin solution. All cell lines were incubated in a 5% CO2 environment at 37°C. The cells were detached from tissue culture flasks by digestion with 0.125% trypsin-ethylenediaminetetraacetic acid (EDTA).
hnRNPA2B1 knockdown and stable transfection
RNA interference technology was used to knockdown hnRNPA2B1 expression. Tumor cell lines were trypsinized and seeded into six-well plates at a density of 2×105 cells and were grown overnight. Transfection was performed on 80%–90% confluent cells using FuGENE® HD transfection reagent (Hoffmann-La Roche, Ltd., Basel, Switzerland) according to the manufacturer’s instructions with an hnRNPA2B1 small hairpin RNA (shRNA) vector. Forty-eight hours after transfection, the cells were switched to a complete medium containing puromycin dihydrochloride (Sigma-Aldrich Co., St. Louis, MO, USA; 10 µg/mL for MCF-7 cell lines and 8 µg/mL for MDA-MB-231) for 2 weeks. The subclone cells were then expanded into puromycin-resistant sublines. The stable sublines were transfected with the hnRNPA2B1-specific pLKO.1-shRNA vector (Table 1), identified as MCF-7/H1, MCF-7/H2, MCF-7/H3, 231/H1, 231/H2, and 231/H3, respectively.
Primers and sequence of hnRNPA2B1/shRNA.

shRNA: small hairpin RNA; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
RNA isolation and quantitative real-time polymerase chain reaction
Total RNA was isolated from the cell lines using the TRIzol reagent (TaKaRa, Dalian, China) according to the manufacturer’s instructions. RNA was quantified using a spectrophotometer at 260 and 280 nm (A260/280) and reverse-transcribed into complementary DNA (cDNA) using a PrimeScript™ RT reagent kit (TaKaRa). Quantitative real-time polymerase chain reaction (qRT-PCR) was performed to determine hnRNPA2B1 expression using a SYBR Green RT-PCR kit (TaKaRa). All experiments were performed in triplicate and were repeated at least twice. The amount of amplified target sequence was calculated using the 2−ΔΔCT method and Light Cycle 480 SW1.5 software (Roche).
Western blot
Total protein was extracted from cell lines and lyzed in radioimmunoprecipitation assay (RIPA) buffer (P0013B, Beyotime Institute of Biotechnology, Beijing, China) containing phenylmethanesulfonyl fluoride (PMSF; ST506, Beyotime Institute of Biotechnology) according to the manufacturer’s instructions. The protein samples were quantified using a Pierce® Bicinchoninic Acid (BCA) Protein Assay Kit (Thermo Scientific, Waltham, MA, USA). Equal amounts of each protein sample (50 µg) were separated with sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene difluoride (PVDF) membranes. The primary antibodies used were rabbit monoclonal anti-STAT3 (Epitomics, Inc. Burlingame, CA, USA; at a dilution of 1:1000), rabbit anti-phospho-STAT3 (Epitomics, Inc.; 1:1000), mouse anti-hnRNPA2B1 (Santa Cruz Biotechnology; 1:200), mouse anti-ERK-1/2 (Santa Cruz Biotechnology; 1:200), and rabbit anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH; Epitomics, Inc.; 1:1000). The secondary antibodies used were goat anti-rabbit IgG (MultiSciences, Hangzhou, China, 1:10,000) and goat anti-mouse IgG (MultiSciences, 1:10,000). The protein bands were revealed using a developing reagent (P0019, Beyotime Institute of Biotechnology, Jiangsu, China) and were photographed using a chemiluminescence imaging system (ChemiDoc XRS+, Bio-Rad, Hercules, CA, USA). Quantity One 4.6 computer software was used to quantify the integrated density of the bands. The results were normalized as a ratio of hnRNPA2B1 to an internal reference using Gaussian Model Trace.
Cell proliferation assays
The cell proliferation was assayed using MTT (Sigma-Aldrich Co., St. Louis, MO, USA) and EdU (RiboBio, Guangzhou, China) according to the manufacturer’s instructions. Cells were subcultured in 96-well plates at a density of 1×104 cells/wells in 100 µL. MTT was incubated with the cells for 24 h, 48 h, and 72 h after transfection, and its absorbance at a wavelength of 570 nm was measured using a standard microplate reader. EdU was incubated for 24 h until it reached the logarithmic growth phase. DNA content of the cells was stained with Hoechst 33342 for 30 min and visualized under a laser scanning confocal microscope (Leica TCS Sp5). Each assay was performed in triplicate and repeated independently for three times.
Cell cycle and apoptosis analysis
To analyze cell cycle distribution, 5 × 105 stable sublines and negative control cells were seeded and grown in a six-well plate. The cells were harvested when achieving approximately 85% confluence and then were fixed in precooled 70% ethanol overnight. On the next day, the fixed cells were suspended in 500 µL phosphate buffered saline (PBS) containing ribonuclease A (RNase A; 50 µg/mL) and propidium iodide (PI; 50 µg/mL) and were incubated for 30 min at 37°C in a water bath. Cell cycle distribution was analyzed using flow cytometry (FCM; FACSVantage SE, Becton Dickinson, New Jersey, USA). To assess apoptosis, the treated cells were harvested and incubated with reagents from the Annexin V-FITC apoptosis kit (BioVision. Inc., Palo Alto, CA, USA) according to the manufacturer’s protocol. Then, the cells were resuspended in 500 µL Annexin V binding buffer and stained with 5 µL Annexin V and PI in the dark at room temperature for 30 min. Annexin V positive cells were quantitated by FCM. All assays were repeated for three times in triplicate.
Tumorigenesis in nude mice
Animal studies were conducted to assess the tumorigenicity of hnRNPA2B1 in vivo. Negative control and stably transfected breast cancer cell lines (1 × 106) were washed twice and suspended in 1 mL PBS and then were orthotopically injected into the mammary fat pads of 6- to 8-week-old female nude mice. The mice were randomly divided into two groups (n = 3). Tumor size was measured every 2 days using a caliper, and the tumor volume (TV) was calculated using the formula TV = L × W × H × 0.52 where L, W, and H are the tumor dimensions in three perpendicular planes. The mice were sacrificed 15 days post-inoculation. The tumors were surgically removed, weighed, fixed in 4% polyformaldehyde, and stored at −80°C. All procedures were performed under the Laboratory Animal Welfare and Ethics Committee of the Third Military Medical University review board-approved protocols.
Statistical analysis
Quantitative data were expressed as the mean ± standard error of mean (SEM). Student’s t-test was used for two-group comparison, and the differences between more than two groups were assessed using the χ2 test. SPSS 22.0 software was used for statistical analysis. The difference was considered statistically significant if p < 0.05 or p < 0.01, as appropriate.
Results
hnRNPA2B1 expression was significantly upregulated in human breast cancer tissues and cell lines
We first examined hnRNPA2B1 expression in 77 cases of tumor-node-metastasis (TNM) stage I–IV breast cancer and 15 normal breast specimens by immunohistochemistry (Figure 1(a)–(f)). We observed high expression (between 33.3% and 80%) of hnRNPA2B1 in breast cancer specimens of stage I–IV. In contrast, all normal breast samples exhibited low expression of hnRNPA2B1 (Table 2). As for the mean SI of hnRNPA2B1 expression in the four groups, hnRNPA2B1 expression was significantly higher in three breast cancer groups (p < 0.001) compared to the normal control group. However, no significant difference was observed among three breast cancer groups. Next, we assessed hnRNPA2B1 expression in two breast cancer cell lines—MDA-MB-231 and MCF-7. It was found that hnRNPA2B1 was highly expressed in these two cell lines compared with the non-tumorigenic epithelial cell line MCF-10A (Figure 1(g)). These data demonstrated the obvious correlation between hnRNPA2B1 expression and tumorigenic potential.

hnRNPA2B1 expression in breast cancer cell lines and tissue specimens. (a) Immunohistochemistry analysis shows hnRNPA2B1 expression in normal breast samples; (b)–(f) hnRNPA2B1 expression in stage I–IV clinic breast cancer samples; scale bar, 200 µm. (g) hnRNPA2B1 protein expression in cell lines: MDA-MB-231, MCF-7, and MCF-10A.
Summarized data of 92 cases.

SI: staining index; SEM: standard error of mean; TNM: tumor-node-metastasis.
p-value of the mean SI between normal and stage I to stage III–IV was <0.001.
Knockdown of hnRNPA2B1 inhibits the proliferation of breast cancer cell lines
The marked correlation between hnRNPA2B1 expression and clinicopathological characteristics prompted us to further examine whether it plays a role in tumorigenesis. To this end, the MCF-7 and MDA-MB-231 cell lines were infected with hnRNPA2B1 knockdown or empty plasmids constructs. The expression of hnRNPA2B1 was detected by qRT-PCR and western blotting (Figure 2(a) and (b)). Three pairs of shRNA were evaluated for knockdown efficiency. We chose stably transfected cell lines (one each for MCF-7/H1 and 231/H2) for subsequent experiments due to their higher knockdown efficiency. MTT assay performed at different time points revealed that the downregulation of hnRNPA2B1 inhibited the proliferation of both the MCF-7 and MDA-MB-231 cell lines (Figure 2(c)). Furthermore, Edu assay confirmed these results and revealed that the decreased expression of hnRNPA2B1 suppressed cell proliferation in both the MCF-7 and MDA-MB-231 cell lines (Figure 2(d)).

Knockdown of hnRNPA2B1 inhibits the proliferation of breast cancer cell lines. (a) and (b) Knockdown of hnRNPA2B1 with three shRNAs (H1,H2, and H3) in MCF-7 and MDA-MB-231cells was verified by quantitative RT-PCR and western blotting. CONTROL, negative control shRNA. (c) MTT and (d) EdU incorporation assays showed that cell growth was inhibited by knockdown of hnRNPA2B1 in MCF-7 and MDA-MB-231cells. The results are expressed as the means ± SEM of three independent experiments in triplicate.
Knockdown of hnRNPA2B1 causes S cell cycle arrest and apoptosis
To elucidate the mechanisms underlying tumor cell growth inhibition by hnRNPA2B1, its effects on apoptosis and cell cycle progression were studied by FCM. hnRNPA2B1-knockdown cells exhibited a significant increase in the S phase population compared with control cells. The percentage of S phase (DNA synthesis period) cells was increased in the MCF-7/H1 (38.28% ± 6.04%), 231/H1 (39.06% ± 1.78%), and 231/H2 (43.16% ± 1.09%) groups compared with the control groups (MCF-7/CONTROL: 24.38 ± 8.35; 231/CONTROL: 26.69 ± 3.18); however, there was no obvious difference between the MCF-7/H2 and MCF-7/CONTROL groups (Figure 3(a)). In addition, we observed that apoptosis of MCF-7/H1 (31.50% ± 2.95%) and 231/H2 (14.34% ± 2.70%) cells increased significantly compared to MCF-7/CONTROL (6.15% ± 1.01%) and 231/CONTROL (5.07% ± 0.22%) (Figure 3(b)) cells. These results suggest that hnRNPA2B1 knockdown causes S cell cycle arrest and apoptosis.

Knockdown of hnRNPA2B1 causes (a) S cell cycle arrest and (b) apoptosis.
Knockdown of hnRNPA2B1 suppresses tumorigenicity in vivo
It has been reported that increased expression of hnRNPA2B1 plays a crucial role in the process of tumor formation. To verify this finding, we investigated whether hnRNPA2B1 knockdown attenuated tumorigenicity in vivo. MCF-7/H1, 231/H2, and control cells were orthotopically transplanted into the mammary fat pads of nude mice. Then, tumors were assessed every 2 days, and were surgically removed and weighed 15 days after inoculation. Our results showed that hnRNPA2B1 knockdown significantly suppressed the growth of xenografted human breast cancer cells in nude mice compared with negative controls. TV and weight in the interference groups were markedly decreased compared to the negative control groups. Compared with controls, hnRNPA2B1 knockdown significantly suppressed the growth of xenografted human breast cancer cell lines in nude mice. Similar suppression of tumor growth by hnRNPA2B1 knockdown was observed for both xenografted MCF-7 and MDA-MB-231 (Figure 4) cell lines, suggesting that the observed effects are not cell line specific.
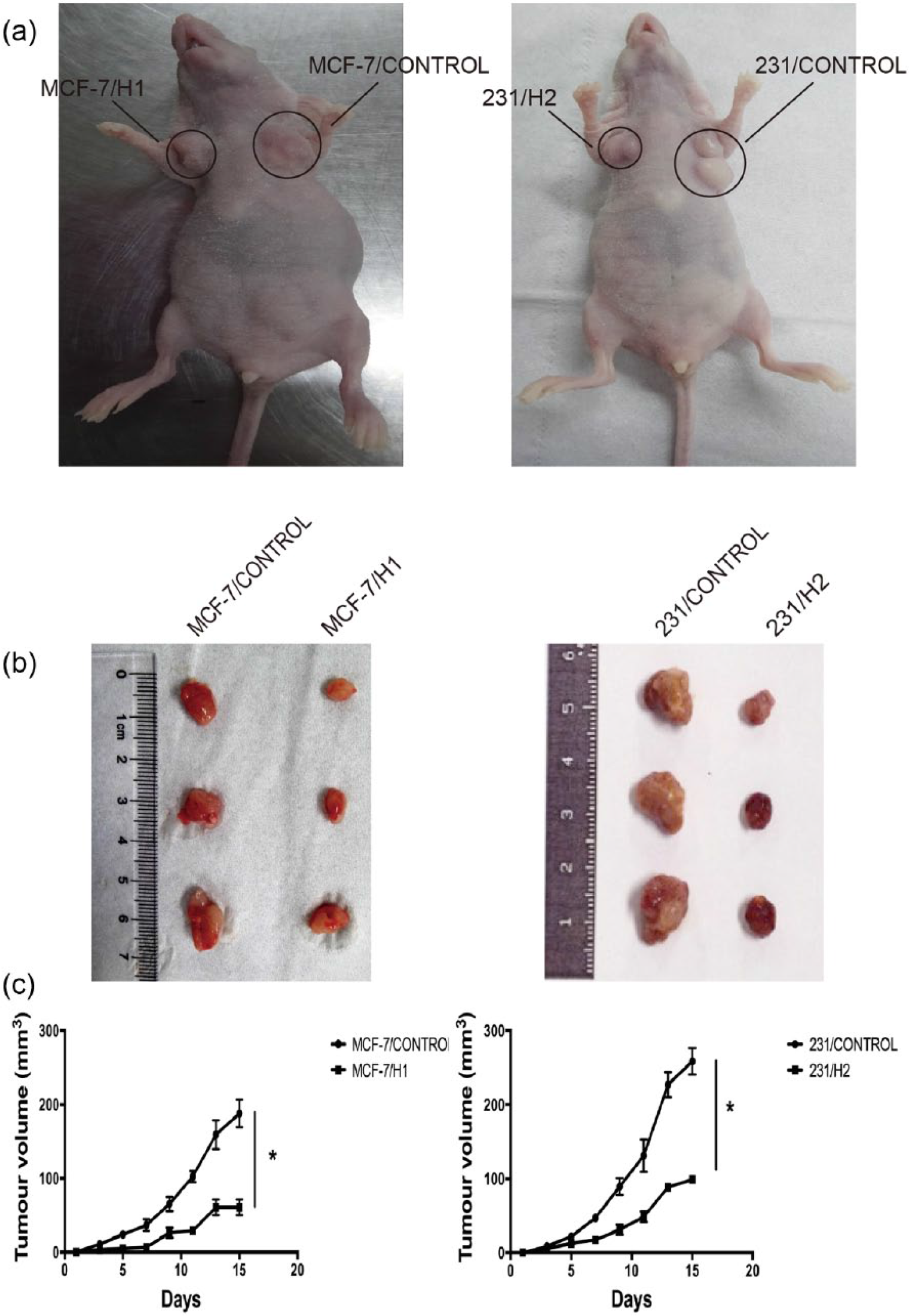
hnRNPA2B1 knockdown inhibits the tumor growth in vivo. (a) Tumor in nude mice 15 days after being orthotopically transplanted into the mammary fat pads, (b) tumor tissues obtained from xenograft model in nude mice. (c) Mice with established tumors were measured almost every 2 days and by a caliper and calculated using the formula.
hnRNPA2B1 knockdown suppressed the STAT3 and ERK1/2 signaling pathway
Previous studies have reported that abnormal activation of the STAT3 and ERK1/2 signaling pathway plays an important role in breast cancer pathogenesis. In this study, we explored whether hnRNPA2B1 had an effect on the STAT3 and ERK1/2 signaling pathway. We found that phospho-STAT3 and phospho-ERK1/2 expression was significantly decreased in hnRNPA2B1 knockdown cells, especially in MCF-7/H1 and 231/H2 cells, whereas the total STAT3 and ERK1/2 level did not change much in these cell lines. On the molecular level, hnRNPA2/B1 knockdown reduced the expression of phospho-STAT3 and phospho-ERK1/2 (Figure 5).

Effects of hnRNPA2/B1 knockdown on regulation of STAT3 and ERK1/2 expression in breast cancer cell.
Discussion
Increasing evidence has made clear that splicing factors play a major role in cancer development and progression. The hnRNPA/B protein families are overexpressed or downregulated in various cancers and likely account for the oncogenic effects of several types of cancer. Recent evidence has indicated that hnRNPA2B1 plays an important role in the development and progression of human cancers. For example, increased expression of hnRNPA2B1 protein was observed in pancreatic cancer cells and lung cancer and may act as a biomarker for early detection. 14 In glioblastoma, hnRNPA2B1 protein was overexpressed in glioma tissue specimens and was associated with advanced glioma grade. hnRNPA2B1 knockdown inhibited glioblastoma cell invasion, growth, and survival, acting as a novel oncogene and a potential target for glioblastoma therapy. Interestingly, it has been reported that hnRNPA2B1 might have an effect on chemotherapy. Inhibition of hnRNPA2B1 expression improved chemosensitivity to gemcitabine, 5-fluorouracil (5-FU), and oxaliplatin in a pancreatic cancer cell line. 20 In glioma cells, hnRNPA2/B1 knockdown has been shown to increase temozolomide (TMZ) sensitivity.
In recent years, hnRNPA2B1 has attracted increasing attention in breast cancer research. The role of hnRNPA2B1 in breast cancer development and progression has not been clearly defined. Studies have shown that mutations in BReast CAncer susceptibility gene 1 (BRCA1) predispose carriers to an increased risk of breast and ovarian cancers. hnRNPA2B1 is overexpressed in BRCA1-mutated breast and ovarian cancers, especially in BRCA1-deficient cells and tumors. 21 Furthermore, knockdown of hnRNPA2 in cancer cell lines is associated with increased p21 levels and downregulated BRCA1 levels. 22 Evidence has shown that hnRNPA2B1 induces epithelial-to-mesenchymal transition (EMT) in lung cancer. 23 It has also suggested that hnRNPA2B1 might contribute to the expression of the mesenchymal traits observed in BRCA1-associated tumors, such as breast and ovarian cancers. Our results supported the hypothesis that hnRNPA2B1 expression is essential for the maintenance and progression of breast cancer cells.
One study has revealed that hnRNPA2B1 is more frequently expressed in primary invasive breast cancers than in normal breast tissue. In our study, we reported that hnRNPA2B1 is preferentially expressed in advanced breast cancer. However, no significant difference between primary and middle-advanced breast cancer cells was observed, indicating that hnRNPA2B1 might be involved in the occurrence of breast cancer. Meanwhile, the higher level of hnRNPA2B1 mRNA expression in the two types of breast cancer cells compared with the non-tumorigenic breast cell line suggests that the hnRNPA2B1 expression level may also be linked to proliferation status, as previously reported 24 Moreover, our results revealed that hnRNPA2B1 knockdown significantly inhibits the proliferation of cells as well as the regulation of the cell cycle and apoptosis in vitro, while hnRNPA2B1 downregulation suppresses tumorigenicity in vivo. These results suggest that the overexpression of hnRNPA2B1 leads to the increasing tumorigenic potential of breast cancer.
In our research, hnRNPA2B1 was shown to contribute to the tumorigenic potential of breast cancer cells by changing the balance between growth and apoptosis. In other words, hnRNPA2B1 takes advantage of aberrant survival signaling to drive tumor development. Thus, the association between hnRNPA2B1 and the ERK signaling pathway attracted our attention because activation of the Ras-Raf-mitogen-activated protein kinase (MAPK)–ERK signaling pathway is one of the survival hallmarks of cancer cells, including many breast cancers. Ras or Raf mutations, the inactivation of Ras GTPases, or the activation of upstream growth factor receptors can aberrantly activate the Ras-Raf-MAPK–ERK signaling pathway.25,26 Some studies have suggested that hnRNPA2 activates the Ras-Raf-MEK-ERK pathway via its effect on A-Raf splicing in hepatocellular carcinoma (HCC). 27 Furthermore, in malignant melanoma, hnRNP K is a target of the MAPK pathway.28,29 Enhanced phosphorylation of ERK is frequently observed in malignant melanoma downstream of NRAS and BRAF. 30
Taken together, our experimental evidence suggests a possible role of the hnRNPA2B1–MAPK axis in the tumorigenic potential of breast cancer. Moreover, it is well known that hnRNPA2B1 plays a critical role in pre-mRNA splicing and may regulate the splicing of A-Raf in breast cancer. Thus, we examined whether hnRNPA2B1 might affect the activity of this pathway. We found that besides its oncogenic potential, hnRNPA2B1 was the strongest activator of the MAPK-ERK pathway as measured by ERK phosphorylation. This suggests that hnRNPA2B1 is required for the activation of this pathway in breast cancer.
Another signaling pathway, STAT3, has been implicated in the regulation of apoptosis and survival in tumors. 31 STAT3 is constitutively activated in all breast cancer subtypes, regarded as a convergent point of many signaling pathways and plays a major role in cell proliferation, apoptosis, angiogenesis, immune response, and metastasis in breast cancer. 32 However, the link between STAT3 and hnRNPA2B1 has not been studied. Based on these findings, we speculated that the role of hnRNPA2B1 in breast cancer may be associated with the STAT3 pathway. Knockdown of hnRNPA2B1 prevented STAT3 activation, suggesting that hnRNPA2B1 promotes tumorigenicity via the STAT3 pathway in breast cancer.
In conclusion, hnRNPA2B1 is overexpressed in human breast cancer tissues and cell lines. Furthermore, hnRNPA2B1 promotes tumor proliferation, regulation of the cell cycle and apoptosis and tumorigenicity in vivo. Our study provides novel insights into the mechanism of hnRNPA2B1 in breast cancer; hnRNPA2B1 promotes tumorigenicity via the ERK1/2 and STAT3 pathways. These findings on hnRNPA2B1 imply a useful therapeutic strategy to restrain the progression of breast cancer.
Footnotes
Acknowledgements
Y.H. performed all the experiments and drafted the manuscript. Z.S. assisted with Immunohistochemistry analysis. J.D. assisted with establishing hnRNPA2B1 knockdown cell lines. B.H. and W.Y. conceived of the study. H.W. assisted with date analysis. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
