Abstract
Primary chemotherapy treatment response monitoring in advanced epithelial ovarian cancer (EOC) is currently based on CT-imaging and serum CA125 values. Serum HE4 profile during first line chemotherapy has not been previously studied. We evaluated the HE4 profile during first line chemotherapy after primary (PDS) and interval debulking surgery (IDS). In total, 49 FIGO stage III/IV EOC patients were included in the study. 22 patients underwent PDS and 27 patients neoadjuvant chemotherapy (NACT) followed by IDS. Serial HE4 and CA125 serum samples were taken during first line chemotherapy. The association of postoperative tumor markers to surgery outcome, primary therapy outcome and progression free survival (PFS) were determined. The lowest HE4 and CA125 values during chemotherapy were compared to primary therapy outcome and PFS. The postoperative HE4 was associated to residual tumor after surgery (p = 0.0001), primary therapy outcome (p = 0.004) and PFS (p = 0.03) in all patients (n = 40). The postoperative CA125 was associated to PFS after IDS (n = 26, p = 0.006), but not after PDS. In multivariate analysis with FIGO stage (III/IV), residual tumor (0/>0) and postoperative CA125, the postoperative HE4 was the only statistically significant prognostic variable predicting PFS. Both HE4 and CA125 nadir corresponded to primary therapy outcome (HE4 p < 0.0001, CA125 p < 0.0001) and PFS (HE4 p = 0.009, CA125 p < 0.0001). HE4 is a promising candidate for EOC response monitoring. In our study, the performance of HE4 in response monitoring of first line chemotherapy was comparable to that of CA125. Of the postoperative values, only HE4 was statistically significantly associated to primary therapy outcome.
Keywords
Introduction
Epithelial ovarian cancer (EOC) is typically diagnosed in advanced Federation of Gynecology and Obstetrics (FIGO) stage III–IV due to lack of symptoms in the earlier stages. 1 The relative 5-year survival rate was 46% in the United States between 2005 and 2011, but the prognosis of advanced-stage EOC is even poorer compared with that of localized disease. 2 Primary debulking surgery (PDS) and platinum-based postoperative adjuvant chemotherapy are the current standard treatment for EOC. 1 Neoadjuvant chemotherapy (NACT) followed by interval debulking surgery (IDS) can be alternatively offered to the primarily unresectable and inoperable advanced EOC patients. 3 Age, FIGO stage, residual tumor in debulking surgery and platinum sensitivity are the most important prognostic factors in EOC.1,4
The serum tumor marker CA125 and computed tomography (CT) imaging are currently used for primary therapy response evaluation in EOC.5,6 CA125 is a MUC16 gene coded glycoprotein, which is physiologically expressed in pleural surface, airway epithelium, lacrimal glands, endometrium, and endocervix. 7 Malignancies, especially EOC, can exhibit increased serum CA125 levels. 8 Low CA125 levels before PDS have been associated with optimal surgery outcome, 9 but contradictory results have also been presented. 10 After PDS, rapid CA125 normalization,11,12 CA125 halftime, 13 and CA125 nadir during adjuvant chemotherapy14,15 are associated with patient’s outcome. In NACT-treated patients, CA125 levels preceding IDS have been presented to correspond with IDS outcome, 16 and CA125 regression during NACT to associate with overall survival (OS). 17
Although CA125 is generally used in treatment response evaluation, 6 there are restrictions in its use during primary therapy. After abdominal surgery, postoperative CA125 is an unreliable indicator of residual tumor.18,19 Human epididymis protein 4 (HE4) is a promising novel serum tumor marker in EOC diagnostics. 20 It is a glycosylated polypeptide encoded by a gene, also known as WFDC2, located on chromosome 20. 21 In healthy adults, HE4 is expressed mainly in the trachea and salivary glands. 22 The expression increases with age, renal insufficiency, and smoking,23,24 whereas high body mass index (BMI) decreases HE4 levels. 23 In addition to EOC, lung adenocarcinoma, breast cancer, pancreatic cancer, endometrial cancer, and bladder cancer also exhibit high HE4 expression. 22
Preoperative serum HE4 is suggested to associate with PDS outcome. 25 The study by Angioli et al. 26 concluded that the combination of preoperative HE4 and CA125 was a better predictor of PDS outcome than either of these tumor markers alone. In our previous study, we showed that in patients treated with NACT followed by IDS, substantial serum HE4 decreases during NACT were associated with better OS. 27 In the study by Angioli et al., 28 HE4 values after PDS predicted platinum sensitivity. No studies on HE4 values after IDS have been published. Currently, the use of HE4 at different phases of EOC treatment has not been clarified.
In this study, we evaluated the usefulness of novel serum marker HE4 during first-line chemotherapy of advanced EOC. Our patient cohort consisted of stage III/IV EOC patients operated upfront or after NACT. The performance of HE4 in EOC monitoring was studied by comparing postoperative values to residual tumor after surgery and to treatment outcome.
Materials and methods
Patients
We recruited 89 patients with suspected ovarian malignancy admitted to the Department of Obstetrics and Gynecology, Turku University Hospital, Finland, between December 2009 and March 2014. This prospective study (ClinicalTrials.gov Identifier: NCT01276574) was designed to assess fluorodeoxyglucose-positron emission tomography/computed tomography (18F-FDG-PET/CT) imaging and serum tumor markers in EOC staging and chemotherapy response evaluation. The study protocol was approved by the local ethical committee. Study exclusion criteria were medical history of diabetes or previous cancer. For the current analysis, 49 patients with newly diagnosed advanced stage III to IV ovarian cancer were included. The patient characteristics are presented in detail in Table 1. The inclusion criteria for the current analysis were (1) PDS or IDS performed, (2) elevated serum HE4 (>70 pmol/L) or CA125 (>35 U/mL) at the time of diagnosis, and (3) at least three measurements of serum HE4 or CA125 during postoperative primary chemotherapy cycles.
Patient characteristics.

PDS: primary debulking surgery; NACT: neoadjuvant chemotherapy; IDS: interval debulking surgery; FIGO: Federation of Gynecology and Obstetrics; NACT: neoadjuvant chemotherapy.
Primary therapy and response evaluation
The selection of patients for PDS or NACT followed by IDS was made by an experienced gynecologic oncologist during the diagnostic laparoscopy or laparotomy. The Fagotti scoring system 29 and preoperative 18F-FDG-PET/CT imaging results were also utilized in the operability assessment. If the patient was considered primarily operable, the diagnostic procedure was converted into debulking surgery. After diagnostic laparoscopy, primarily inoperable and unresectable patients were referred to NACT followed by IDS. One patient with EOC diagnosis confirmed by pleural effusion cytology was directly referred to NACT due to recurrent dyspnea and extensive pleural effusion.
The study patients received standard postoperative platinum-based adjuvant chemotherapy consisting of carboplatin/paclitaxel. Seven patients received also 7.5–15 mg/kg bevacizumab in combination with primary chemotherapy cycles. Three patients received liposomal doxorubicin or docetaxel instead of paclitaxel, and three patients received only carboplatin due to reduced general condition. The distribution of neoadjuvant and adjuvant chemotherapy cycles is presented in Table 1. The primary therapy outcome was defined with contrast-enhanced CT imaging and serum CA125 value at the end of primary therapy according to the standard response evaluation criteria.5,6 Seven patients had progressive disease (PD) and proceeded to second-line chemotherapy. At the end of first-line therapy, 42 patients had either partial response (PR) or complete response (CR). During the follow-up, a clinical examination and CA125 measurement were performed every 3 months. The disease progression was defined as CA125 >70 U/mL confirmed twice or the presence of any new lesions in CT or 18F-FDG-PET/CT.
Serum tumor marker analysis
The serum tumor markers HE4 and CA125 were measured at the time of diagnosis, before IDS, postoperatively before first adjuvant chemotherapy cycle and before every postoperative adjuvant chemotherapy cycle. Both tumor markers were also determined before each NACT cycle. Preoperative HE4 and CA125 values were defined as samples preceding PDS or IDS. Preoperatively, the sample closest to the surgery was chosen. Postoperatively, the sample closest to the onset of chemotherapy was chosen. HE4 and CA125 nadir values were defined as the lowest tumor marker values during postoperative primary chemotherapy.
The median time interval from preoperative HE4 samples to surgery was 1 day (range: 0–41 days), and that for CA125 was 1 day (range: 0–48 days). The median time interval from surgery to postoperative HE4 and CA125 sample analysis was 19 days (range: 4–33 days). After surgery, the median number of serum samples taken per patient was 6 (range: 3–10) for HE4 and 5 (range: 3–9) for CA125. In all, 11 patients did not have either preoperative or postoperative HE4, and three patients lacked preoperative or postoperative CA125. One patient lacked all HE4 samples after IDS. These patients were excluded from the analyses considering tumor marker change perioperatively, postoperative biomarkers or HE4 nadir values.
After 30-min incubation at room temperature, the blood samples in non-heparinized tubes were centrifuged for 15 min at 3000 r/min (800 g). Serum was stored at −20°C or −80°C in 2-mL cryotubes. HE4 concentrations (pmol/L) were manually analyzed by enzyme-linked immunosorbent assay (ELISA; Fujirebio Diagnostics Inc., Malvern, PA, USA) according to the manufacturer’s instructions. CA125 concentrations (U/mL) were analyzed by electro-chemiluminescence immunoassay (ECLIA; Modular E170 automatic analyzer, Roche Diagnostic GmbH, Mannheim, Germany).
Statistical analysis
The serum HE4 and CA125 percent change perioperatively was calculated using preoperative and postoperative tumor marker values. The tumor marker change, postoperative, and nadir values were compared with primary therapy outcome using cumulative logit model and to PFS using Cox regression. Postoperative tumor markers were also compared with residual tumor after surgery using linear models. The patients were divided into no macroscopic tumor (R = 0) and residual tumor > 0 (R > 0) groups according to the debulking surgery outcome. PFS was defined as the time from the diagnosis until the disease progression.
Tumor marker, patient subgroup (PDS or NACT), and interaction between these two variables were used as predictors. As the interactions were not statistically significant, the results are reported by all patients combined. The exception to this was the analysis of postoperative CA125 association to PFS, in which the interaction between PDS and NACT groups was observed. A fitted model in which both HE4 and CA125 values were used as predictor variables simultaneously was employed to study whether either predictor improved the ability to predict the response beyond the effect of the predictor. Finally, a multivariate analysis was performed with postoperative tumor markers, FIGO stage, and residual tumor in surgery as predictors of PFS.
Univariate associations between categorical predictor variables and PFS were studied using the log-rank test. Statistical analyses were performed using SAS for Windows version 9.4. p values <0.05 were considered statistically significant.
Results
Preoperative and postoperative CA125 and HE4 values
All the tumor marker values at various time points are presented in Table 2. HE4 at the time of diagnosis was not associated with PFS (p = 0.24), whereas lower CA125 at the time of diagnosis predicted longer PFS (p = 0.01, hazard ratio (HR) = 1.45, 95% confidence interval (CI) = 1.09–1.94). The tumor marker’s percent change in preoperative to postoperative values was not associated with primary therapy outcome or PFS. In our patient cohort, the FIGO stage did not predict primary therapy outcome (p = 0.26) or PFS (p = 0.28).
Serum HE4 and CA125 profiles during primary chemotherapy are presented in all patients and in the both subgroups according to the treatment strategy (PDS or NACT + IDS).
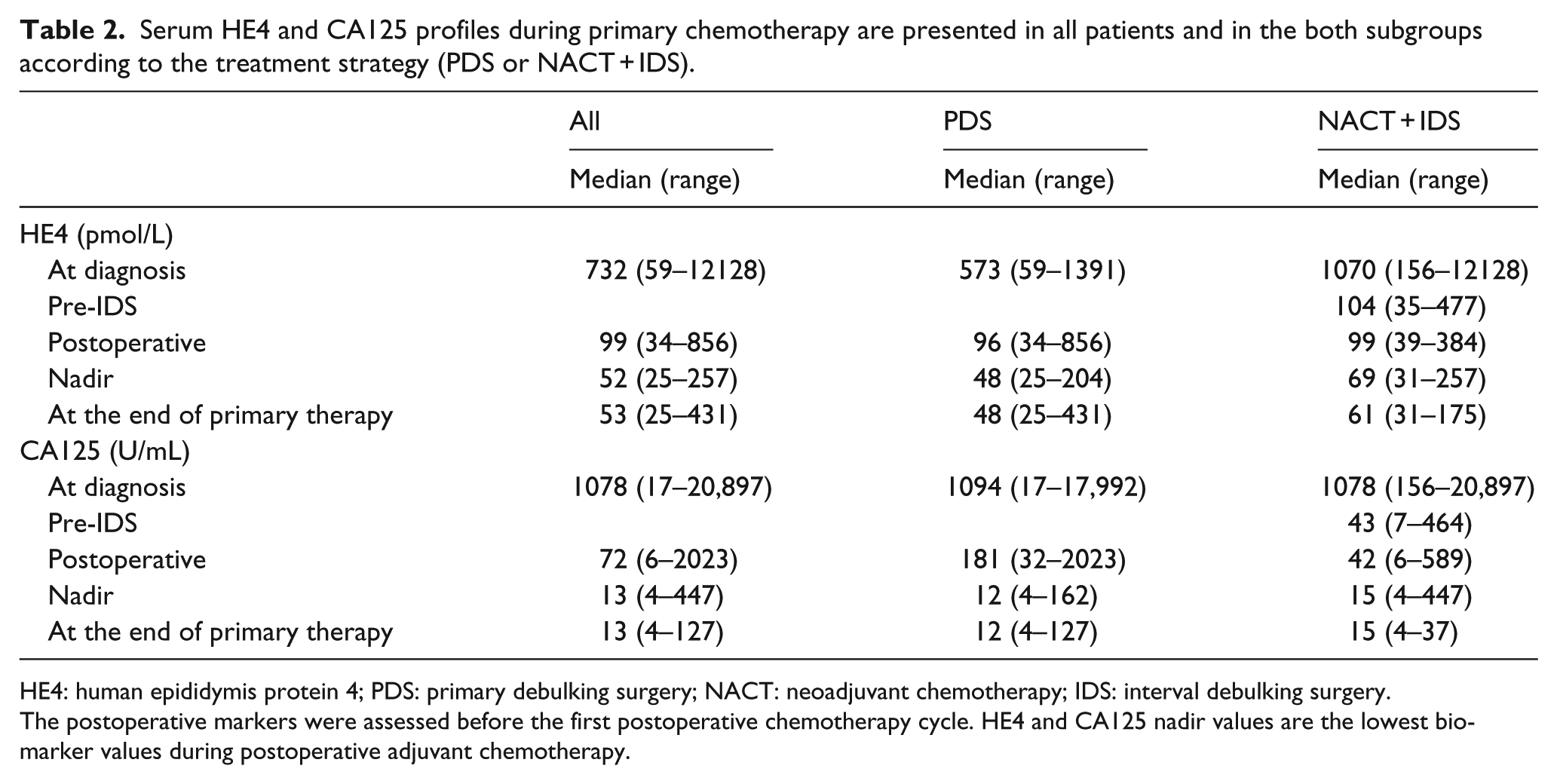
HE4: human epididymis protein 4; PDS: primary debulking surgery; NACT: neoadjuvant chemotherapy; IDS: interval debulking surgery.
The postoperative markers were assessed before the first postoperative chemotherapy cycle. HE4 and CA125 nadir values are the lowest biomarker values during postoperative adjuvant chemotherapy.
The patients with lower postoperative HE4 were more likely to have optimal surgery outcome (p < 0.0001, R2 = 0.36), better primary therapy outcome (p = 0.005, odds ratio (OR) = 0.01, 95% CI = 0.003–0.01) and longer PFS (p = 0.03, HR = 1.00, 95% CI = 1.00–1.01) in the combined cohort (n = 40). The postoperative HE4 values in different subgroups are presented in Figure 1. Similarly, the lower postoperative CA125 was associated with longer PFS in all the patients (p = 0.002, HR = 1.00, 95% CI = 1.001–1.004) and patients treated with NACT (p = 0.006, HR = 1.00, 95% CI = 1.00–1.01) but not with residual tumor in debulking surgery (p = 0.46) or primary therapy outcome (p = 0.80). In multivariate analysis, which also included FIGO stage, the postoperative HE4 was the only statistically significant prognostic variable predicting primary therapy outcome (p = 0.02, OR = 1.00, 95% CI = 1.00–1.01) and PFS (p = 0.04, HR = 1.003, 95% CI = 1.00–1.01) (Table 3).

Postoperative and nadir HE4 values during first-line chemotherapy. The postoperative HE4 and HE4 nadir values are presented (a) in the R = 0 and R > 0 groups, (b) in the primary therapy outcome groups, (c) in the R = 0 and R > 0 groups, and (d) in the primary therapy outcome groups.
Univariate and multivariate analyses were performed with FIGO stage (III/IV), residual tumor (R0/>0) and postoperative serum tumor markers HE4 and CA125.

HE4: human epididymis protein 4; FIGO: Federation of Gynecology and Obstetrics.
The postoperative HE4 was the only statistically independent prognostic variable in multivariate analysis.
Statistically significant variables.
The R = 0 patients had longer PFS than the R > 0 patients (p = 0.03, HR = 0.37, 95% CI = 0.15–0.89). The median PFS was 1.5 years (mean: 2.2 years, standard deviation (SD) = 0.4) in the R = 0 group and 1.2 years (mean: 1.2 years, SD = 0.1) in the R > 0 group. In PDS patients, the median PFS was 4.0 years (mean: 3.2 years, SD = 0.5) in the R = 0 group and 1.3 years (mean: 1.4 years, SD = 0.2) in the R > 0 group. In contrast, the median PFS was practically similar in all IDS patients regardless of surgical outcome, 0.9 years (mean: 0.9 years, SD = 0.1) in the R = 0 group and 0.7 years (mean: 0.9 years, SD = 0.1) in the R > 0 group.
HE4 and CA125 nadir values during first-line chemotherapy
The HE4 and CA125 nadir values are presented in Table 2, and HE4 nadir values in different subgroups are illustrated in Figure 1. The lower HE4 nadir values during postoperative chemotherapy were associated with better primary therapy outcome (p < 0.0001) and longer PFS (p = 0.009, HR = 1.01, 95% CI = 1.003–1.02) in all the patients (n = 48). Similarly, the CA125 nadir values also corresponded to primary therapy outcome (p < 0.0001) and PFS (p < 0.0001, HR = 1.01, 95% CI = 1.01–1.02). Both biomarkers together were significantly better predictors of primary therapy outcome than either HE4 or CA125 alone.
The time to reach nadir was similar for both tumor markers. The median times for HE4 nadir values to be reached from the date of debulking surgery were 119 days (range: 19–238 days) after PDS and 62 days (range: 5–149 days) after IDS. The median time for nadir CA125 values were 118 days (range: 40–259 days) and 69 days (range: 5–172 days) after PDS and IDS, respectively. In patients with CR, the median HE4 was 50 pmol/L (range: 25–122 pmol/L) at the end of the primary therapy.
Discussion
This study is the first to demonstrate that the postoperative serum HE4 is associated with residual tumor after debulking surgery, primary therapy outcome, and PFS in patients with advanced stage III/IV EOC. In addition, we showed that the HE4 nadir values during postoperative chemotherapy are associated with primary therapy outcome and PFS. HE4 increased also the predictability of CA125 when combined. The combination of CA125 and HE4 predicted outcome better than CA125 alone.
In our patient cohort, CA125 but not HE4 at the time of diagnosis was associated with PFS. Preoperative serum CA125 has been reported to correspond to PDS outcome16,30–32 although contradictory results have also been presented.10,33 Perioperative CA125 changes have also been associated with PFS. 34 Our previous study focused on HE4 in NACT response evaluation. We presented similar results showing that HE4 at diagnosis was not a prognostic variable, whereas HE4 after NACT was a prognostic variable. 27 In this study, we also evaluated the perioperative tumor marker change, but it was not associated with surgery outcome, primary therapy outcome or PFS.
Previously, no reliable indicator of residual tumor after debulking surgery has been presented. Serum CA125 levels increase even after explorative abdominal surgery 19 due to incision and healing of the peritoneum and omentum. 35 Even surgery for non-malignant indication (i.e. atherosclerosis) can cause increased CA125 values.18,36 CA125 levels are the highest during the second week after abdominal surgery. 36 Chi et al. 37 presented that CT results after debulking surgery correlate with the surgeon’s estimation of residual tumor in only 52% of their study patients. One explanation is that the surgeon’s assessment is always subjective. Postoperatively, a reliable serum tumor marker could be a simple and non-invasive method to measure tumor burden before initiation of chemotherapy. In our patient cohort, the postoperative CA125 was not associated with debulking surgery outcome, which is consistent with earlier studies.18,38 However, our results suggest that R = 0 patients had lower postoperative serum HE4 levels compared with the R > 0 group. This finding suggests that HE4 is a more reliable indicator of the residual tumor than CA125.
Serum HE4 levels differ in platinum-resistant and platinum-sensitive patients during postoperative chemotherapy after PDS. 28 The results of this study indicate that HE4 nadir values correspond to primary therapy outcome and PFS. To the best of our knowledge, no other previous studies on the significance of HE4 nadir during postoperative chemotherapy have been published. In our patient cohort, serum CA125 nadir values were also associated with primary therapy outcome and PFS. This finding is consistent with earlier studies.14,15 As the combination of HE4 and CA125 predicted outcome better than either alone, HE4 nadir combined with CA125 could be practical in clinical use to identify patients with the highest risk of relapse.
FIGO stage is a well-known prognostic factor in EOC. 1 In our study population, FIGO stage did not correspond to PFS. This finding may be attributed to the fact that all our patients represented advanced stage III-IV EOC. Our previous observation from 18F-FDG-PET/CT suggests 39 that in many patients, the FIGO stage might underestimate the true extent of the patients’ disease. Therefore, it is not unexpected that we did not detect any survival differences between the patients when FIGO stages III and IV were compared.
The strength of our study is the prospective study design and standardized treatment and evaluation strategy. The limitation of our study is our relatively small patient cohort, which included both PDS and NACT patients. This limitation may interfere with the evaluation of outcome. However, we did not find evidence of the serum tumor markers acting dissimilarly in our interaction analysis. Another weakness of this study is that our residual tumor assessment is based exclusively on surgeon’s evaluation and was not verified with post-surgery imaging. Our results nevertheless suggest that HE4 is not equally sensitive as CA125 to other possible confounding non-malignant factors. Third, limitation of our study is that when evaluating the postoperative biomarkers, the sample date range was quite broad (4–33 days). However, the majority of the serum samples were obtained approximately 19 days after debulking surgery.
To conclude, our results indicate that HE4 is a promising candidate for EOC treatment response evaluation. We demonstrate that the postoperative HE4 is a prognostic factor in patients with advanced EOC and might be a more reliable indicator of residual tumor than CA125. HE4 appears to be as useful and reliable as CA125 as a tumor marker of treatment response evaluation during EOC primary therapy. Further studies with larger patient cohorts considering the topic are warranted.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Funding
This study was financially supported by the Clinical Research (EVO) fund of the Turku University Hospital, a grant from the Turku University Foundation and a grant from the Finnish Cultural Foundation.
Informed consent
Informed consent was obtained from all individual participants included in the study.
