Abstract
The purpose of this study was to investigate the relationship between the expression of stem-cell markers nestin and cluster of differentiation 146 with clinicopathological characteristics in breast cancer and to determine whether a prognostic impact of nestin and CD146 expression exists regarding occurrence of disease relapse in breast cancer. A total of 141 patients who were histologically diagnosed with breast cancer and underwent radical operations from November 2006 to October 2013 in Laiko General Hospital, National and Kapodistrian University of Athens, were enrolled in the study. CD146 and nestin protein expression were evaluated using immunohistochemistry. Nestin expression was observed in 18.4% (26/141) of the cases, while CD146 expression was observed in 35.5% (50/141) of the cases. Nestin expression is significantly higher in younger patients with breast cancer. Nestin and CD146 expression were not correlated with the tumor size and the presence of lymph node metastasis. On the contrary, a significantly higher expression of nestin and CD146 was observed with triple-negative cancers (p < 0.0001 for both markers), low differentiated tumors (p = 0.021 for nestin and p = 0.008 for CD146), and increased Ki-67 expression (p = 0.007 for nestin and p < 0.0001 for CD146). The nestin-positive group of patients and the CD146–positive group of patients presented significantly higher rates of disease recurrence (log-rank test, p = 0.022 for nestin and p = 0.003 for CD146) with a distant metastasis, 30 months after the primary treatment. CD146 but not nestin, however, predicted independently (p = 0.047) disease recurrence. Nestin and CD146 are expressed in breast cancer cells with highly aggressive potency. They might contribute to disease relapse in breast cancer by activating the epithelial–mesenchymal transition pathway and assist tumor neovascularization.
Keywords
Introduction
Breast cancer is the most common type of cancer among western women and is the second leading cause of cancer death. 1 Although the majority of these patients are diagnosed with early stage disease demonstrating an excellent overall 5-year survival rate, many of these women will experience a disease recurrence, and it is not the primary tumor, but its metastases at distant sites that are the main cause of their death during the initial years after diagnosis. 2 The risk of breast cancer recurrence following therapy 2 shows that treated patients still maintain a subset of tumorigenic cancer stem cells (CSCs) and underlines the necessity for the development of new stem-cell-targeted therapies as future therapeutics urgently needed for individual patients who are at the highest risk of developing metastases after standard therapies fail.
CSCs frequently also called tumor-initiating cells are stem-like cells that constitute a subpopulation of self-sustaining tumorigenic cells within tumors with indefinite potential to self-renew and maintain the tumor3–8 and have been demonstrated to contribute to chemo-resistance in cancer therapeutics and further disease progression. CSCs can differentiate and give rise to non-tumorigenic progeny contributing to the genetic heterogeneity within tumors, whereas breast cancer stem cells (BCSCs) undergoing genetic and epigenetic modifications do not behave as or arise from classic stem cells. New experimental approaches have provided perspective and insight that BCSCs and non-stem breast cancer cells (NSBCCs) develop and remain in a dynamic balance that is dependent of interleukin 6 (IL6) 9 and is associated with the epithelial–mesenchymal transition (EMT). More recently, substantial investigation has implicated that cells undergoing EMT and tumor-initiating cells share common features, cell surface markers, and expression profiles, whereas forced EMT produces mammary cells with stem cell and tumorigenic characteristics in distinct tumor types that can ultimately effect patient prognosis. 10 These findings reveal the potential plasticity of these tumor cells suggesting that EMT-associated transcription factor networks could reprogram cancer cells into CSCs. 8 Interestingly, another hypothesis suggests that metastasis in tumors occurs after cells in a primary epithelial malignancy undergo an EMT. 10 The association between EMT and CSCs is also relevant to chemo-resistance, as cells undergoing EMT are more resistant to chemotherapeutics, whereas the features of CSCs are relevant to cancer therapeutics as tumors relapse even after an effective primary response with various chemotherapeutic approaches. 9
Nestin, an intermediate filament protein which is transiently expressed in undifferentiated cells, including newly synthesized blood vessels,11,12 was one of the two markers used in this study. Furthermore, nestin is expressed in proliferating progenitor cells in various embryonic tissues, while it also appears as an important molecular marker for neural stem cells.13–15 Studies have recently shown that nestin is also expressed in tumor cells and proliferating microvascular endothelial cells,16,17 while it is also preferentially expressed in triple-negative and basal-like breast carcinomas. 15
The second marker used in this study was cluster of differentiation 146 (CD146), also known as melanoma cell-adhesion molecule (MCAM), and cell surface glycoprotein (MUC18) that was first identified as a melanoma-specific cell-adhesion molecule 18 and has been demonstrated as a marker for tumor angiogenesis 19 and endothelial cell migration.20–22 Moreover, CD146 is a unique activator of EMT and is significantly associated with triple-negative cancers. In epithelial breast cancer cells, overexpression of CD146 down-regulated epithelial markers and up-regulated mesenchymal markers significantly promoted cell migration and invasion and induced CSC-like properties. 23
On this background, the aim of this study was to determine whether nestin and CD146 are correlated with aggressive clinical features and which is the impact, if any exists, on the time that disease relapse occurs.
Materials and methods
Patients and tissues specimens
This study has been approved by the research ethics committee of the National and Kapodistrian University, Athens Medical School, Laiko General Hospital. Written informed consent was obtained from all patients included in this study. Medical research charities from the families of the patients provided the funding that supported this study.
We identified from the files of the 1st and 2nd Departments of Propedeutic Surgery in Laiko General Hospital in Athens, Greece, a total of 141 patients who were histologically diagnosed with breast cancer and had underwent surgery from November 2006 to October 2013 (study group). The inclusion criteria of this study were the following: (a) patients who underwent intended curative operations and (b) resected specimens with a precise histopathological examination. As a control group, we included benign breast samples obtained from the same patients who had undergone the breast biopsy during that time.
Clinical information
Specific clinical information was recorded for all of the cases including age of patients at the time of diagnosis of cancer and details of the breast disease such as Nottingham grade, tumor/node/metastasis (TNM) stage, hormone receptors (estrogen receptor (ER) and progesterone receptor) status, human epidermal growth factor receptor 2 (HER2)/neu status, and the presence of chemotherapy and/or radiotherapy. Statistical correlations between these clinicopathological features and the expression of nestin and or CD146 were examined. The time of disease recurrence—defined either as local recurrence or the presence of a distant metastasis—was measured in the first 5 years after the primary operation, performed with curative intention.
Immunohistochemistry
Staining was done on regular 4-µm sections of formalin-fixed and paraffin-embedded tumor tissue. Immuno-histochemistry was performed on tissue sections with the standard biotin–avidin complex technique using two monoclonal antibodies against nestin and CD146. Nestin expression was assessed using a mouse monoclonal antibody from Santa Cruz Biotechnology (Santa Cruz, CA, USA; clone: 10c2), while CD146 expression (MCAM) was assessed using a mouse monoclonal antibody from Menarini Diagnostics S.A. (Greece; clone: N1238) by immunohistochemistry. Positive control included samples of normal breast from the main tissue, where nestin and CD146 were consistently expressed in myoepithelial cells and endothelial cells. Negative control was prepared by substituting the primary antibody over phosphate-buffered saline (PBS).
Immunohistochemical analysis
Histopathological evaluation was performed independently and blindly by two pathologists who are considered experts in the field of breast disease. To assess CD146 and nestin expression in the tissue specimens, normal tissue cells were used from the myoepithelium and endothelial cells of normal vessels as internal positive controls (Figure 1). Moreover, as positive controls, melanoma specimens that strongly expressed nestin and CD146 were used. Negative controls were established by omitting both the primary antibodies. The evaluation of the staining was performed using the semi-quantitative method. More specifically, immunohistochemistry was applied in a three-step method (avidin–biotin–peroxidase) for determining nestin and CD146 expression in paraffin tissue–embedded sections of 141 breast carcinomas. Each sample was considered positive for expression of nestin in cells showing staining when >5% of the cells were bonding and positive for CD146 expression when >10% cells were bonding. The staining of the specimens was scored as follows—0: no staining of the cells, 1: mild intense, 2: intermediate intense, and 3: very intense. Staining intensity 2 or 3 is defined as “increased staining intensity.” Moreover, ⩾50% of the stained cells are evaluated as “Overexpression of nestin and CD146.” Ki-67 staining assessment (nuclear reactivity) was carried out after counting the proportion of positive tumor cell nuclei in the most active (positive) area. The upper quartile (22.5%) was chosen as a cutoff value according to Gilliland et al. 24 who defined tumors with a high proportion of proliferating cells to >20%.

Intense and diffuse cytoplasmic expression of (a) nestin and (b) CD146 in the neoplastic cells of a triple-negative breast carcinoma (×200). HER2 breast carcinoma with (c) a weak to moderate expression of nestin and (d) a weak staining for CD146. (c and d) Note the intense staining of the endothelial cells (×200). A weak (e) nestin and (f) CD146 expression in a luminal B breast carcinoma. (e and f) There is an intense staining of the vessels (×200). (g) A weak nestin expression and (h) a moderate staining for CD146 in a luminal A breast carcinoma. (g and h) The endothelial cells of the vessels are strongly positive (×200). (i) Nestin and (j) CD146 are strongly expressed in the myoepithelial and endothelial cells of the adjacent to tumor, benign breast tissue (×200).
Statistical analysis
The current statistical analysis was performed with SPSS IBM statistics software (version 21.0). Correlations between nestin, CD146, and the clinicopathological characteristics were performed using t-test and χ2 test for quantitative and qualitative parameters, respectively. Α two-sided χ2 test and Fischer’s exact test were used for the assessment of the significance. The distribution of the age was controlled using histogram, Q-Q plots, Kolmogorov–Smirnov, and Shapiro–Wilk tests. The time of relapse was defined as the time in months after surgery until the occurrence of the first recurrence manifesting either with a local or with a distant relapse. Kaplan–Meier curves were analyzed for the assessment of the cumulative survival probabilities. Log-rank test was carried out to prove the significance of the relapse rates. Multivariate analysis was performed using logistic regression and the Cox proportional hazards model. The 95% was defined as the confidence interval. A P value of <0.05 was considered as significant.
Results
Descriptive characteristics of the study population
According to the criteria described above, a total of 141 patients with invasive breast cancer underwent oncological surgical resection at the 2nd Department of Propedeutic Surgery, Laiko General Hospital, National and Kapodistrian University of Athens. Mean body mass index (BMI) of the study population was 25.95 kg/m2. Other epidemiological characteristics of the patients are presented in Table 1. In Table 2, the vast majority of the patients with positive nestin and/or CD146 expression are demonstrated regarding the triple-negative cancer group compared to other patients with other molecular subtypes. Concerning nestin, a total of 20/42 patients expressed nestin in the triple-negative group but only 2/33 patients in the HER2-positive group, 3/33 patients in the luminal B group, and 1/33 patients in the luminal A group. Comparable results were demonstrated for CD146 expression in 31/42 patients of the triple-negative group, 10/33 patients of the HER2-positive group, 6/33 patients of the luminal B group, and 3/33 patients of the luminal A group. The exact TNM stage was available in 137 patients. Among them, 59 patients have been observed with lymph node metastasis while a total of 78 patients had no lymph node metastasis. Regarding the oncologic therapy, 57.4% of the patients (81/141) received radiation therapy while 73.7% of the patients (104/141) received chemotherapy. Concerning disease relapse after primary treatment, 26.2% of the patients (37/141) recurred either locally or by presenting metachronous distant metastasis.
Descriptive characteristics of the study population.

Nestin and CD146 expression according to the molecular subtype.

CD146: cluster of differentiation 146; HER2: human epidermal growth factor receptor 2.
Nestin and CD146 are highly expressed in the cytoplasm of the tumor cells
Immunohistochemical examination showed that nestin and CD146 were located in the cytoplasm of the tumor cells (Figures 2 and 3), while the myoepithelial cells displayed strong nestin and CD146 expression as positive controls. There was no nestin or CD146 expression in the negative controls. Nestin was expressed in 18.4% of the patients (26/141) and CD146 in 35.4% of the patients (50/141) as illustrated in Table 2.

(a) Intense cytoplasmic expression of nestin in tumor cells of patients with triple-negative molecular subtype. (b) Weak nestin expression in tumor cells of patients with HER2+ subtype. Imaging of lobules, myoepithelium, and vessels. (c) Nestin expression in tumor cells of patients with luminal B subtype. (d) Least intense nestin expression in tumor cells of patients with luminal A subtype.

(a) Intense CD146 cytoplasmic expression in tumor cells of patients with triple-negative molecular subtype. (b) CD146 expression in tumor cells of patients with HER2+ subtype. (c) CD146 expression in tumor cells of patients with luminal B subtype. (d) Least intense CD146 expression in tumor cells of patients with luminal A subtype.
Nestin is correlated with low differentiated tumors, triple-negative cancers, and increased expression of the cell proliferation marker Ki-67
Nestin was significantly higher expressed in younger patients (approximately 54 vs 64 years, p = 0.029; Table 3). No correlation was observed with the tumor size or the presence of lymph node metastasis at the time of the operation. On the contrary, an association (Table 4) was revealed between nestin expression and the triple-negative subtype (p < 0.0001), lower tumor differentiation (p = 0.021), and the higher expression of the cell proliferation marker Ki-67 (p = 0.007). The nestin-positive group of patients had received chemotherapy in a significantly higher rate (p = 0.039). No significant correlation was observed between nestin expression and treatment with radiation therapy.
Correlation of nestin and CD146 with age.

Correlation of nestin and CD146 with clinicopathological characteristics.

Nestin expression is related to a significant earlier disease recurrence
Among the 37 patients who presented with postoperative disease recurrence, 29.7% of the patients (11/37) expressed nestin, while 70.3% of the patients (26/37) showed negative nestin expression. These 11 patients recurred interestingly with a distant metastasis (the opposite breast, skeletal, lung, or liver metastasis). Interestingly, 81.8% of the patients (9/11) who recurred exhibited the triple-negative subtype, while 18.1% of the patients (2/11) exhibited the luminal B subtype. The Kaplan–Meier curve (Chart 1) showed that 45.1% of the patients expressing nestin, recurred 30 months after the operation while only a 18.9% of the nestin-negative group relapsed at the same time. The nestin-positive group relapsed with a significantly higher rate 30 months after the first surgical treatment (log-rank test, p = 0.022). Cox regression analysis (Table 5), however, showed that nestin is not an independent prognostic factor for the time of disease recurrence in breast cancer.

Kaplan–Meier curve shows the 5-year time of disease recurrence in months between the nestin-positive and nestin-negative group (log-rank test, p = 0.022). The vast majority of patients expressing nestin presented disease relapse 30 months after surgery.
Multivariate analysis using Cox regression in order to determine which parameter predicts disease recurrence.
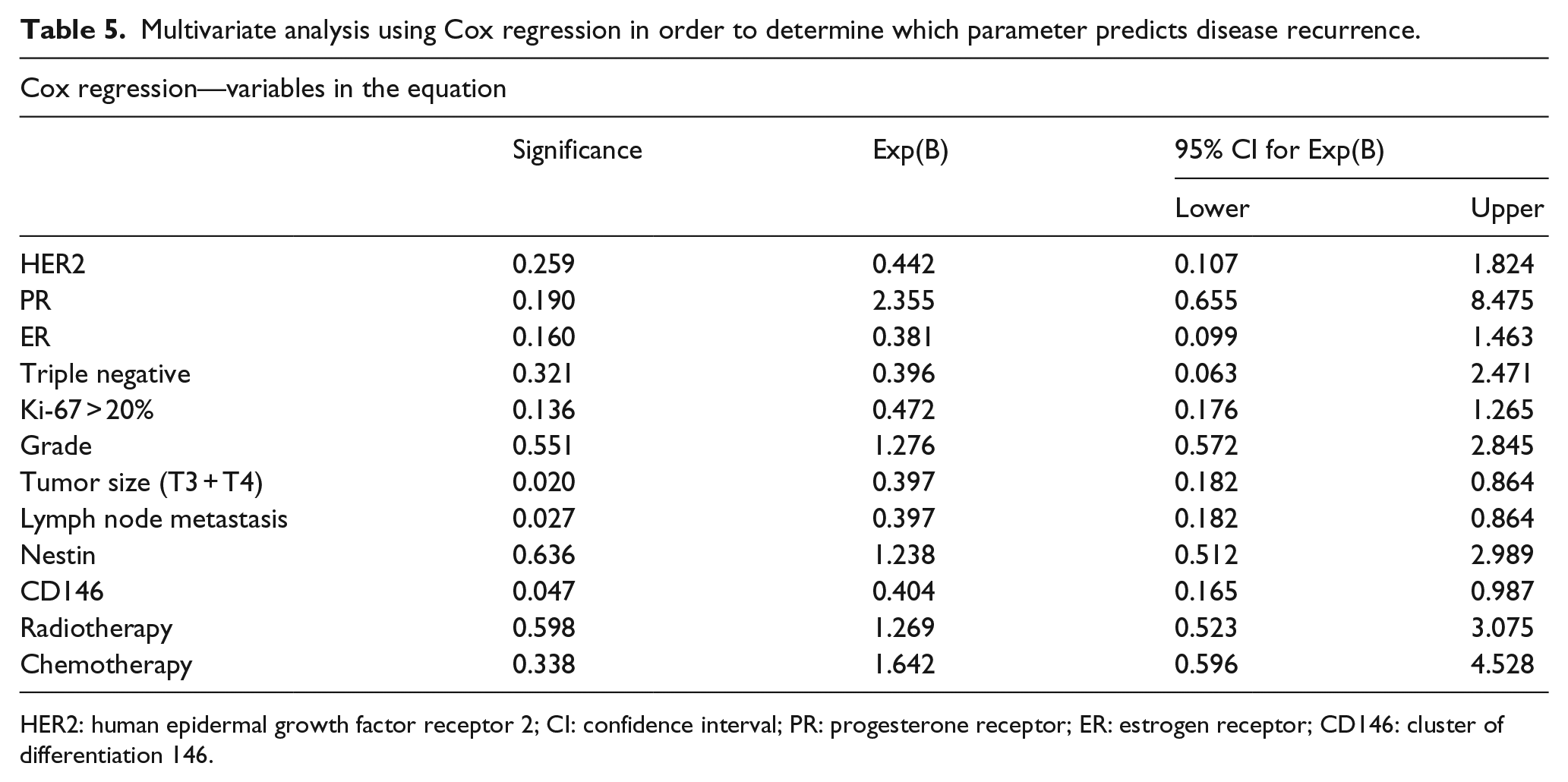
HER2: human epidermal growth factor receptor 2; CI: confidence interval; PR: progesterone receptor; ER: estrogen receptor; CD146: cluster of differentiation 146.
CD146 is correlated with low differentiated tumors, triple-negative cancers, and increased expression of the Ki-67 cell proliferation marker, but no correlation was observed regarding tumor size and lymph node metastasis
CD146 expression revealed no significant correlation with age, tumor size, and the presence of lymph node metastasis. However, CD146 expression was associated (Table 3) with triple-negative breast cancers (p < 0.0001), low tumor differentiation (p = 0.008), and higher expression of the cell proliferation marker Ki-67 (p < 0.0001). Patients expressing CD146 had not received chemotherapy or radiation therapy with a significantly higher rate.
CD146 is an independent prognostic factor of earlier time of relapse
Among 21 patients who recurred and expressed CD146, 13 exhibited the triple-negative phenotype. A total of 20 patients recurred with a distant metastasis (the opposite breast, skeletal, liver, lungs, and brain) and 1 with lymph node metastasis. Kaplan–Meier analysis revealed (Chart 2) that almost 40% of the patients who recurred and expressed CD146 relapsed 30 months after surgery. On the contrary, only 17.4% of the patients, who did not express CD146, presented occurrence of relapse. This observation was statistically significant (log rank, p = 0.003). In order to evaluate which factor could independently predict recurrence, multivariate analysis using Cox regression was performed, that determined that CD146 (p = 0.047), tumor stage (p = 0.020), and the presence of lymph node metastasis (0.027) could independently predict recurrence as illustrated in Table 5.

Kaplan–Meier curve shows the 5-year time of disease relapse in months between the CD146-positive and CD146-negative groups (log-rank test, p = 0.003). The vast majority of patients expressing CD146 presented disease relapse 30 months after surgery.
Discussion
Nestin
In this study, it was demonstrated that nestin is expressed in breast cancer cells and myoepithelial cells. A significant higher expression was observed in the group of patients with triple-negative breast cancers. Those results are in accordance with other studies.25,26 Furthermore, the nestin-positive group of patients was 10 years significantly younger compared to the patients who did not express nestin. Another study has presented evidence through multivariate analysis that octamer-binding transcription factor 4 (Oct-4) and nestin expression were correlated with younger age at disease onset and shorter survival. 27 Interestingly, triple-negative cancers present large heterogeneity and >40% relapse in time. 28 Therefore, the ability to classify more subgroups based on molecular characteristics, from heterogeneous tumors such as triple-negative breast cancers, seems to be challenging. The question raised from these data would be whether a specific clinical profile based on nestin-positive younger patients with a greater life expectancy and exhibiting the triple-negative phenotype can be recognized and therefore plan a specific therapy.
Interestingly, nestin expression was correlated with low-differentiated tumors and increased expression of the Ki-67 cell proliferation marker. This could furthermore indicate that the molecular profile of breast malignant tumors that express nestin is aggressive and has a potential of quick disease progression. In our previous study, 29 we suggested that nestin expression in cancer is detected in cells with stem cell properties. This observation is enhanced in this study as low differentiated cells and increased mitotic activity are characteristics of stem-like cells. In vivo neural stem cells isolated from nestin knockout mice have been found to exhibit a remarkably decreased ability of self-renewal and an increase in apoptotic rate. 30 Data from studies on CSCs in breast cancer indicate that nestin-positive CSCs enhance tumorigenicity by regulating the Wntβ-catenin pathway. 26 It has been supported that tumor-initiating cells with stem-cell properties are the main cause of disease progression; invasion; resistance to radiation and chemotherapy; and therefore recurrence.31,32 It seems that nestin-positive cells in breast cancer, possibly as tumor-initiating cells, might conserve the potential for disease progression and possibly for metastasis.
In this study, no correlation was observed between nestin and lymph node metastasis at the time of diagnosis. This might suggest that the impact of nestin expression in lymph node metastasis might be low as a mechanism of disease progression. Kaplan–Meier analysis as well as the log-rank test suggested a significant recurrence rate in the nestin-positive group of patients. The vast majority of these patients (81.8% (9/11)) had triple-negative cancers and all of them have recurred with a distant metastasis. Accumulating evidence suggest that nestin is implicated in tumor neovascularization.12,33 Interestingly, Krüger et al. 34 used nestin and Ki-67 expression as an index of activated angiogenesis by identifying proliferating and immature endothelial cells in breast cancer and demonstrated that angiogenesis is increased in basal-like phenotypes compared to the ER and HER2-positive subtypes. Solid tumors seem to be more dependent on new formed capillaries considering that devascularized tumors are more unlike to promote metastasis and given that in the absence of neovascularization, 2–3 mm in size tumors do not further grow. 35 Calabrese et al. 36 reported in brain tumors the existence of a perivascular niche in which CSCs are located and interact with blood vessels. Boraas et al. 37 showed that pluripotent stem cells have a less developed cytoskeleton and expressed nestin in a lower rate in comparison to fibroblasts. During early differentiation however, they expressed similar genes which encoded cytoskeletal proteins, suggesting that the up-regulation of such genes provided the necessary cytoskeletal changes inducing further remodeling in stem cells. The above presented data suggest that nestin-positive cancer cells might provide the necessary molecular changes or might interact with stroma cells providing the necessary cytoskeleton changes to promote angiogenesis and therefore metastasis. Consequently, nestin might be a potential molecular target of anti-angiogenesis treatment in breast cancer.
CD146
CD146 is known to be highly expressed in endothelial cells, smooth muscle cells, and pericytes, while accumulating evidence proves its functional role at the endothelial junction associated with the actin cytoskeleton. 38 The current study has shown that in breast cancer CD146 was expressed in cancer cells and myoepithelial cells. An association was found between CD146 expression and triple-negative breast cancers, which has been confirmed by previous studies as well. 23 No association was observed with tumor size or the presence of lymph node metastasis. However, CD146 expression was significantly associated with low differentiated tumors as well as with increased Ki-67 expression. It has been demonstrated that CD146 expression is significantly higher in invasive components compared to in situ components in the same tissue specimens of basal-like breast cancers, suggesting that this molecular alteration is necessary for the disease progression leading to the presence of invasion. 39 Data from studies in human breast cancer cell lines show that CD146 expression is associated with an EMT phenotype. 40 These results suggest that tumor cells expressing CD146 present features of highly aggressive potency and exhibit a mesenchymal phenotype.
In this study, patients expressing CD146 had significantly higher rates of relapse. A total of two basic mechanisms related with the presence of CD146 expression have been described in tumorigenesis and tumor progression. Accumulating data from late studies suggest that CD146 acts as a co-receptor with vascular endothelial growth factor receptor 2 (VEGFR-2). Jiang et al. 41 demonstrated in vitro in melanoma and pancreatic cell lines that an interaction between CD146 and VEGFR-2 exists via CD146 enhancement of the VEGFR-2 phosphorylation which activates downstream signaling pathways and therefore, promotes endothelial cell migration and tube formation. Interestingly, in the same study it was shown in vivo in pancreatic carcinoma tumor xenografts that the anti-CD146 and anti–vascular endothelial growth factor (VEGF) combined therapy suppressed tumor growth and angiogenesis. Zeng et al. 23 showed in vitro in human breast cancer cell lines, which did not express CD146, that the mediated CD146 overexpression induced an up-regulation in the expression of the mesenchymal markers vimentin and fibronectin, while CD146 silencing induced an up-regulation of the epithelial marker E-cadherin and down-regulation of the mesenchymal markers. Those tumor cells exhibited a mesenchymal phenotype and CD146 expression induced activation of the EMT pathway. In the same study, it was shown that in vitro CD146 down-regulation inhibited cell migration and further invasion, whereas in vivo CD146 up-regulation was correlated with the presence of distant metastasis. From a clinical point of view, circulating tumor cells, which are considered as potential precursors of metastatic disease, 42 were detected largely based on the expression of CD146 in the blood of patients with metastatic breast cancer. 43
The above presented data suggest that tumor cells expressing CD146 seem to exhibit a mesenchymal phenotype and mediate disease progression through the EMT pathway and tumor angiogenesis, as CD146 seems to act as a co-receptor with VEGFR-2. The question raised is whether the conventional chemotherapy and radiation therapy given in breast cancer patients can eradicate tumor cells with mesenchymal phenotype and/or stem-cell properties. In this study, the CD146-positive group had not received significantly higher chemo- and radiotherapy, but the relapse rates were however significantly higher when CD146 was expressed. This might indicate that the tumor cells expressing CD146 were resistant to conventional therapy. According to the data presented, CD146 could be a possible target for new drug therapy since it can independently predict the relapse of malignancy. Furthermore, its expression seems to be implicated in crucial pathways such as the EMT pathway as well as in tumor angiogenesis that can potentially mediate metastasis.
Conclusion
In this study, we showed that nestin and CD146 are correlated with aggressive clinical features in breast cancer, which are harbored from cells with stem cell properties. The possible mechanisms implicate involvement of the EMT pathway and tumor neovascularization. Since it was demonstrated that when these two markers are expressed, a significantly higher disease recurrence exists, it is likely that the pathophysiological mechanisms which are implicated with these molecules might drive metastasis. Therefore, these two markers could prove to be potential targets in the field of new drug development.
Footnotes
Acknowledgements
E.C.T. and A.T. contributed equally to this work and therefore share the authorship.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
