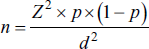Abstract
Background
Drug–drug interactions (DDIs) are a significant concern among cardiac inpatients, potentially impacting therapeutic outcomes and patient safety. Early identification and management of potential DDIs are crucial in preventing adverse events, especially in complex treatment regimens.
Objectives
This study aimed to evaluate the prevalence, severity, and clinical relevance of potential DDIs among cardiac inpatients at a tertiary care hospital.
Materials and Methods
The study was a cross-sectional, observational study design that was conducted over 6 months among cardiac inpatients receiving multiple medications to analyze the incidence of DDIs in patients treated in the cardiology department. Patient data, including demographic details, diagnosis, and prescribed medications, were collected and analyzed. Potential DDIs were identified using standard drug interaction databases, and their severity was classified into minor, moderate, and major categories.
Results
The occurrence of DDIs was observed to be higher in male patients compared to female patients and was more prevalent in individuals aged 51–70 years. Minor to moderate DDIs were more frequently detected than severe interactions. Additionally, pharmacodynamic interactions were more common than pharmacokinetic interactions.
Conclusion
Potential DDIs are common among cardiac inpatients, emphasizing the need for vigilant monitoring. Involving clinical pharmacists and individualized medication management, especially for elderly patients, can minimize risks, ensuring better patient safety, effective treatment outcomes, and optimized pharmacodynamic interaction management.
Introduction
Cardiovascular diseases (CVDs) account for 17.9 million deaths per year, making them the leading cause of mortality worldwide. Cardiac and vascular diseases, collectively referred to as CVDs, include acute coronary syndrome, valvular heart disease, cerebrovascular disease, and rheumatic heart disease. Over four out of five deaths from CVD are caused by heart attacks and strokes, and one-third of these deaths occur in individuals under the age of 70. According to statistics, India has recorded the highest number of deaths from CVDs globally. The number of deaths has increased significantly from 2.26 million in 1990 to 4.77 million in 2020. 1
CVDs are a group of interrelated conditions where complications from one disease may lead to the development or worsening of another. Consequently, cardiovascular patients are more likely to suffer from multiple medical conditions and to be prescribed multiple medications, exposing them to a variety of medication-related challenges, such as drug interactions. 2 A drug–drug interaction (DDI) refers to the pharmacokinetic (PK) or pharmacodynamic (PD) effects that medications exert on one another, which may result in desirable effects, reduced efficacy, or increased toxicity. 3
Current research indicates that polypharmacy is most prevalent among the older population. 4 However, individuals under the age of 70 account for two-thirds of all polypharmacy cases. 4 This trend is particularly evident among cardiovascular patients due to their increased multimorbid susceptibility and the complexity of their conditions, often necessitating the use of multiple drugs. 5 As a result, polypharmacy affects not only the elderly but the entire cardiovascular patient population. 5 CVDs are among the most common conditions associated with high rates of polypharmacy prescriptions. 6
Several factors have been identified as contributing to polypharmacy in CVD patients, including the development of complications, the presence of multiple comorbidities, advanced disease stages, and increasing age. 7 In cardiac patients, polypharmacy increases the risk of DDIs, leading to adverse outcomes such as acute kidney injury (AKI), diverse side effects, and metabolic disturbances. The combination of diuretics, angiotensin-converting enzyme (ACE) inhibitors/angiotensin receptor blockers (ARBs), and non-steroidal anti-inflammatory drugs (NSAIDs)—often termed the “triple whammy”—is particularly known to impair renal function and precipitate AKI. Polypharmacy also raises the risk of falls, hospitalization, reduced functional status, poor medication compliance, inappropriate prescriptions, morbidity, and mortality. Additionally, it increases the chances of both DDIs and adverse drug reactions.8, 9 Analyzing the prevalence of DDIs in hospitalized patients is essential for improving clinical outcomes, minimizing preventable harm, optimizing therapeutic regimens, and enhancing the overall quality and safety of healthcare delivery. Therefore, the present study aims to investigate the prevalence, types, and clinical significance of potential drug-drug interactions (pDDIs) in patients with CVDs, where complex therapeutic regimens frequently involve polypharmacy.
Materials and Methods
A hospital-based cross-sectional study was conducted to evaluate the pDDIs in cardiac inpatients for 6 months, that is, August 2022 to January 2023, from the Department of Cardiology at a tertiary care hospital. A convenience sampling technique was employed to select participants. Of the 163 patients approached for the study, 150 met the inclusion criteria and were included in the final analysis. Thirteen patients refused to participate, resulting in a final sample size of 150. There was no follow-up phase, as this was a cross-sectional study, and all data were collected at a single point during each patient’s hospital visit. The inclusion criteria for the study were patients of both genders aged over 18 years with cardiovascular illness, admitted for more than 24 h, and receiving one or more medications. Patients who were unwilling to give consent or had an inpatient admission of less than 24 h were excluded from the study. The sample size formula employed for the present study is as follows:
The ethical clearance was obtained from the Ethics Committee of the institution for the approval of the study (JSS/MC/PG/91/2022-23). The 163 patients approached for the study and the 13 who refused to participate are essential for assessing the response rate and potential selection bias. The data were collected from medical charts, prescription records, and electronic health records of inpatients admitted to the cardiology department. Medication prescribed was noted from the inpatients’ records, and the potential DDI was assessed using online software (
Workflow Chart.

Statistical Analysis
Data were analyzed using Statistical Package for the Social Sciences (SPSS) version 23, with a significance level set at p < .05. Missing data were handled using listwise deletion, where only complete cases were included in the analysis. No sensitivity analyses were conducted as the study primarily focused on descriptive and inferential statistics to assess the prevalence and severity of DDIs. To account for potential confounding factors, multivariable logistic regression analysis was conducted, adjusting for key confounders including age, gender, number of medications, and comorbidities, as these factors are known to influence the risk of DDIs in cardiovascular patients. Continuous variables (e.g., age, hospital stay) were presented as means (standard deviation (SD)) or medians (interquartile range (IQR)), depending on distribution, while categorical variables (e.g., gender, DDI severity) were expressed as frequencies and percentages. The categories were defined as 29–40 years, 41–50 years, 51–60 years, 61–70 years, 71–80 years, and 81–90 years. This grouping was chosen to capture the typical age distribution of cardiac inpatients and to assess age-related variations in the prevalence of DDIs. No subgroup analyses or interaction tests were conducted due to the sample size and scope of the study. Similarly, sensitivity analyses were not performed as the study primarily focused on descriptive and inferential statistics to assess the prevalence and severity of DDIs
Results
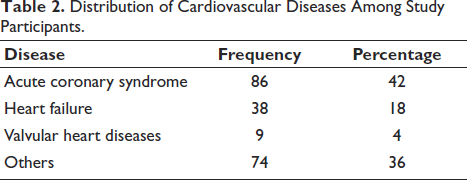
The study was conducted in 150 patients with CVD. Data obtained show that the proportion of male patients, at 80.7%, was considerably higher than that of female patients, at 19.3%. The age group distribution of the study population shows that the largest portions are middle-aged to older adults, specifically those in the 51–70 years range (24.7% each for 51–60 and 61–70 years), as shown in Table 1. Acute coronary syndrome was the most common condition, accounting for 42% of cases, followed by heart failure at 18% and valvular heart disease at 4%, as shown in Table 2.
Gender and Age Group Distribution of Study Participants.

Distribution of Cardiovascular Diseases Among Study Participants.
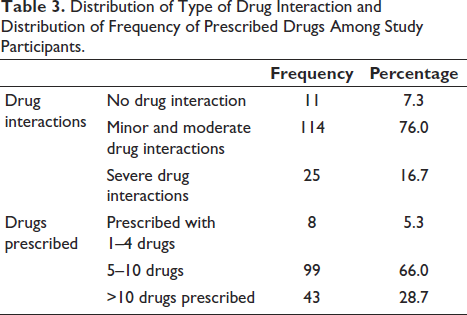
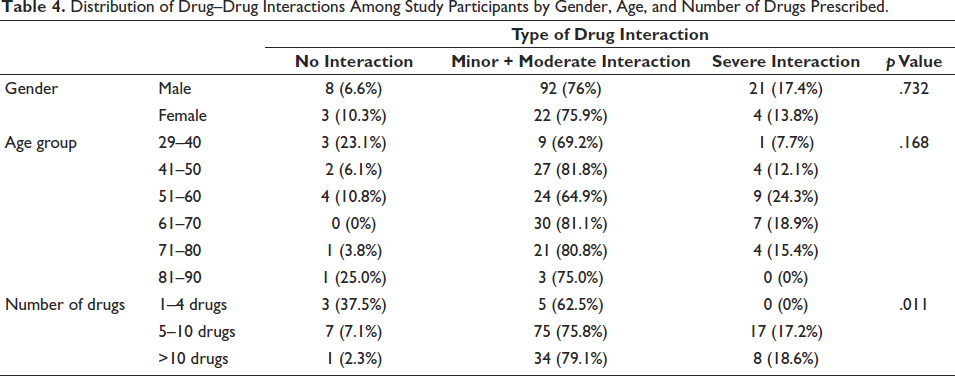
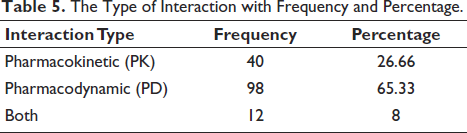
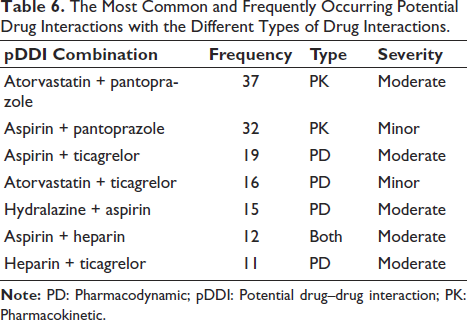
The data reveal that the vast majority of patients are experiencing some level of drug interactions, with 76% having minor to moderate interactions and 16.7% having severe interactions. Only a small fraction of the population (7.3%) has no drug interactions. The data show that the majority of patients in the study are prescribed 5–10 drugs (66%), followed by more than 10 drugs (28.7%), and a small fraction of patients (5.3%) are with few medications (1–4 drugs), as shown in Table 3. For both males and females, approximately 76% have minor to moderate drug interactions. This has a p value of .732, indicating no statistically significant difference between males and females regarding drug interaction types. The highest percentage of no-drug interactions is observed in the 29–40 years (23.1%) and 81–90 years (25.0%) age groups. The 41–50 years (81.8%), 61–70 years (81.1%), and 71–80 years (80.8%) age groups have the highest percentages of minor to moderate drug interactions. The highest percentage of severe drug interactions is in the 51–60 years age group (24.3%). There is no statistically significant difference in the drug interaction types between different age groups, where the p value is .168, and between genders, where the p value is .732. There is an important relationship between the number of drugs the patient is prescribed and the types of drug interactions, as supported by the statistically significant p value of .011 obtained. The more drugs a patient receives, the more severe the interactions are, as shown in Table 4. PD interactions were the most common (65.33%), followed by PK interactions (26.66%), while a smaller proportion (8%) experienced both, as highlighted in Table 5. The most frequently observed pDDI was between atorvastatin and pantoprazole (37 cases), classified as PK with moderate severity, followed by aspirin and pantoprazole (32 cases), which was PK with minor severity. PD interactions were common, with aspirin and ticagrelor (19 cases) and hydralazine with aspirin (15 cases) showing moderate severity, and a combination of both PK and PD interactions was observed in aspirin and heparin (12 cases), as shown in Table 6.
Distribution of Type of Drug Interaction and Distribution of Frequency of Prescribed Drugs Among Study Participants.
Distribution of Drug–Drug Interactions Among Study Participants by Gender, Age, and Number of Drugs Prescribed.
The Type of Interaction with Frequency and Percentage.
The Most Common and Frequently Occurring Potential Drug Interactions with the Different Types of Drug Interactions.
Discussion
The study was a cross-sectional, observational study that mainly focused on the cardiology population aged above 18 years. The study population included 150 cardiovascular inpatients. The objective of the study was to identify the frequency of pDDIs in cardiac patients who had been admitted for more than 24 h. The study was mainly concentrated on cardiac patients, as the drug interaction is comparatively higher due to many comorbidities and polypharmacy. This discussion highlights recent research findings on the prevalence and mechanisms of DDIs among cardiovascular patients, focusing on gender, age distribution, and the types and severity of interactions.
In a study conducted by Mosca et al., the research indicates that male patients constitute a larger proportion of the cardiovascular population experiencing DDIs compared to female patients. This is consistent with epidemiological data showing a higher prevalence of CVD among males (64%), potentially due to differences in lifestyle, genetic predisposition, and hormonal factors. Similarly, in our study, out of 150 inpatients, 80.7% were male patients, followed by 19.3% female patients. 10
In our study, the data analysis on age group distribution of the study population shows that the largest portions are middle-aged to older adults, specifically those in the 51–70 years range (24.7% each for 51–60 and 61–70 years). This indication corresponds to the study conducted by Hajjar et al., demonstrating the highest incidence of morbidity and polypharmacy in patients aged between 61 and 70 years. This age group is particularly susceptible to DDIs due to age-related physiological changes. 11
The frequency of drug interactions obtained from the data analyzed showed the highest prevalence of PD interaction (65.21%), followed by PK interaction (30.43%), and both (2.17%). This is cognizant of the study done by Varsha Shetty et al., which says that most drug interactions involve PD (63.65%), followed by PK (36.35%), in the 663 patients taken into consideration with CVDs. 12
In a study conducted by Straubhaar et al., out of these 739 potential DDIs, 190 (25.7%) were severe drug interactions, followed by moderate and minor, accounting for 549 (74.3%) of drug interactions. 13 Hence, this study concluded that the majority of drug interactions were moderate and minor. Similarly, in our study, the interpretation of the dataset indicated that the majority of drug interactions were moderate and minor (76%), followed by severe (16.7%) drug interactions.
Considering the study conducted by Assefa et al., the most common CVDs identified were hypertension (46.6%), congestive heart failure (14.5%), and stroke (13.3%). The most frequent comorbid conditions were endocrine disorders (24.7%), followed by respiratory problems (10.6%) and chronic kidney diseases (9%). This study develops a foundational base to the observation of our study, which shows 42% of patients suffered from acute coronary syndrome, 18% from heart failure, followed by 4% from valvular heart diseases, and others (36%), including hypertension, diabetes mellitus (DM), and chronic obstructive pulmonary disease (COPD). 14
In a study conducted by Shah et al., the mean number of drugs per prescription was 7.31 ± 4.39. In our study, the mean number of drugs per prescription was 7.5 ± 5.3. Therefore, it corresponds to the other studies that highlight the fact that with the increase in the number of drugs prescribed, the frequency and prevalence of polypharmacy also rise, which results in drug interactions. 15
The most common drug interactions in our analysis were found to be aspirin + ticagrelor (19), aspirin + pantoprazole (32), and atorvastatin + pantoprazole (37). Subsequently, a similar study conducted by Ismail et al. demonstrated the most common pDDIs to be heparin + aspirin (39 cases), digoxin + spironolactone (35 cases), and spironolactone + ramipril (23 cases). 16 These findings indicated the incidences of drug interactions in cardiovascular prescriptions. The administration of variable prescription medications, the severity of CVDs, length of stay, and the existence of comorbidities all influence the incidence and severity of these drug interactions.
Limitations of the Study
Variability in drug regimens due to differing cardiovascular conditions results in heterogeneity among patients, affecting the generalizability of findings. Differences in drug dosing and adjustments based on clinical indications may alter the severity of DDIs. Additionally, the interaction checker used (
Generalizability of the Study
The findings of this cross-sectional study, conducted at a single tertiary care hospital among cardiac inpatients, may have limited generalizability to broader populations. Variability in drug regimens due to differing cardiovascular conditions and heterogeneity among patients may affect how applicable these results are to other settings or patient groups. The use of a convenience sampling technique and exclusion of patients with hospital stays less than 24 h further restricts representativeness. Additionally, the drug interaction database used (
Conclusion
The present study examines the prevalence, type, and severity of DDIs among 150 cardiovascular inpatients using a cross-sectional design. Patient data were collected from medical records, focusing on demographics, prescribed medications, and potential DDIs, identified using online software. PD and PK interactions were categorized based on severity, and statistical analysis determined their frequency across age groups and cardiovascular conditions. The results revealed a higher number of male patients, with the 51–70 age group being most affected. PD interactions were more frequent than PK interactions, with common drug combinations including atorvastatin + pantoprazole (37), aspirin + pantoprazole (32), and aspirin + ticagrelor (19). Moderate interactions were predominant, underscoring the need for careful medication management. These findings highlight the influence of demographics on DDIs and emphasize the importance of understanding PD interactions to minimize risks. Future research should explore long-term effects and mitigation strategies.
Footnotes
Abbreviations
ACE: Angiotensin-converting enzyme; AKI: Acute kidney injury; ARBs: Angiotensin receptor blockers; COPD: Chronic obstructive pulmonary disease; CVDs: Cardiovascular diseases; DDIs: Drug–drug interactions; DM: Diabetes mellitus; FDA: Food and Drug Administration; IQR: Interquartile range; MS Excel: Microsoft Excel; NSAIDs: Non-steroidal anti-inflammatory drugs; PD: Pharmacodynamic; pDDIs: Potential drug–drug interactions; PK: Pharmacokinetic; SD: Standard deviation; SPSS: Statistical Package for the Social Sciences.
Acknowledgments
The authors would like to thank JSS Medical College and Hospital and JSS Academy of Higher Education and Research for all the support.
Authors’ Contributions
SP, PVH, MM, RR: Conceptualization, methodology, visualization, investigation, original draft—writing. SBC: Statistical analysis. VGD: Writing—review and editing, supervision.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was conducted after approval from the Institutional Ethics Committee with Ref No.: JSS/MC/PG/91/2022-23.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
The study participant (patient) was informed and written informed consent for their willingness to publish the data was obtained.