Abstract
Background
Lifitegrast, a lymphocyte function-associated antigen-1 antagonist, was found to be useful in the treatment of psoriasis.
Objectives
The study aimed to evaluate semisolid formulations of lifitegrast for their physicochemical and efficacy performance.
Materials and Methods
Emulgel and ointment formulations were prepared with permissible excipients that help in increasing permeability and retention in the skin. The rheological characteristics, release rate, in vitro permeation, and biological activity in the imiquimod-induced psoriasis model in C57 mice were studied for these formulations.
Results
The formulations (emulgel and ointment) were found to be homogeneous and stable, with characteristic shear-thinning and viscoelastic behavior suitable for topical use. The gel formulation had the highest mean release rate, while in vitro permeation studies showed that the emulgel had superior drug permeation and retention. These formulations of lifitegrast topically (3%, 5%, and 7%) were applied twice a day on the psoriatic skin of C57 mice for 6 days, and Psoriasis Area and Severity Index scores were measured. The formulations reduced psoriatic inflammation, and the emulgel formulations were found to be significantly more efficacious than the ointment formulations.
Conclusion
Lifitegrast formulations showed substantial improvement in psoriasis, owing to the optimum permeation and retention in psoriatic skin.
Introduction
Penetration and retention of drugs in the skin are important and challenging, and dermatological formulation techniques are used to achieve optimum skin permeation and retention for anti-psoriatic agents. 1 The formulation approaches are useful to increase the penetration of poorly soluble drugs in the skin, with a minimum requirement of excipients for formulation. The optimum particle size and the use of suitable copolymers, surfactants, and permeation enhancers are some of the factors responsible for the success of dermatological formulations.2, 3 Increased permeation and increased residence time in the skin can be achieved using these techniques, which are useful for the treatment of psoriasis. 4 Psoriasis is a disease where increased immunological reactions coupled with multifactorial skin inflammation cause infiltration of immune cells, leading to erythematous, raised, scaly skin lesions, epidermal acanthosis, parakeratosis, and hyperkeratosis. 5 Topical delivery of agents is always preferred for the treatment of mild-to-moderate psoriasis because of low systemic side effects and increased patient compliance. 6
Lifitegrast is a novel lymphocyte function-associated antigen-1 (LFA-1) antagonist, which is designed to mimic the binding epitope of intercellular adhesion molecule-1 (ICAM-1) and it is used for treating dry eye disease. 7 LFA-1 is involved in the activation and recruitment of T-cells, inflammation, and plaque formation, which is the major pathogenetic cause of psoriasis. Earlier, we established that lifitegrast, a small molecule that blocks the LFA-1 and ICAM-1 interaction, exhibits anti-psoriatic efficacy by reduction of T-cell mediated immunity. 8 However, lifitegrast topical formulation for the use of dry eye treatment has a limited efficacy potential in the treatment of psoriasis because the active ingredient used in the ophthalmic formulation is a water-soluble salt of lifitegrast. 9 Studies indicate that the outermost layer of skin, the stratum corneum, can easily allow the lipophilic compounds to penetrate. 10 On the other hand, the partitioning into the epidermis and the dermis is favorable for hydrophilic agents due to the more aqueous nature of these matrices. 11 Hence, the development of a suitable topical formulation of lifitegrast that would penetrate faster and reside more in the skin across the dermis and epidermis would be needed for the treatment of psoriasis. 3
This study aimed to prepare a formulation of lifitegrast that would ensure the penetration as well as retention of lifitegrast in the skin and investigate its potential for the treatment of psoriasis in an imiquimod (IMQ)-induced psoriasis model in mice.
Materials and Methods
Lifitegrast was procured from Cayman Chemical (USA). Polyethylene glycol 400 (PEG400), propylene glycol, carbopol, and other reagents were procured from MilliporeSigma (USA). All the reagents were of analytical or high-performance liquid chromatography (HPLC) grade of high purity.
Preparation of Gel
Carbopol 974P was dissolved in hot water. Separately, lifitegrast was dissolved in PEG400 and propylene glycol and mixed with the solution of carbopol. The neutralizing agent triethanolamine and isopropyl myristate were added, and the gel was allowed to be set, which was filled in aluminum tubes.
Preparation of Ointment
White petrolatum was heated until a clear solution was formed. Lifitegrast was dissolved in transcutol and mineral oil, and it was added to the hot solution of petrolatum with continuous stirring. To it, butylated hydroxyanisole and isopropyl myristate were added, and the mixture was allowed to cool to form an ointment, which was filled in aluminum tubes. The ingredients of the gel and ointment formulations are shown in Tables 1 and 2, respectively.
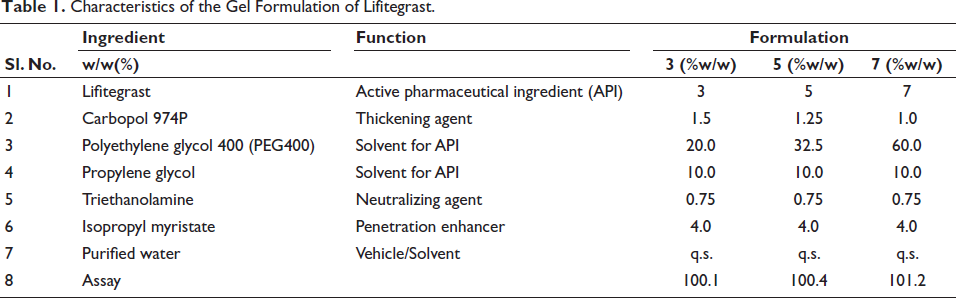
Characteristics of the Gel Formulation of Lifitegrast.
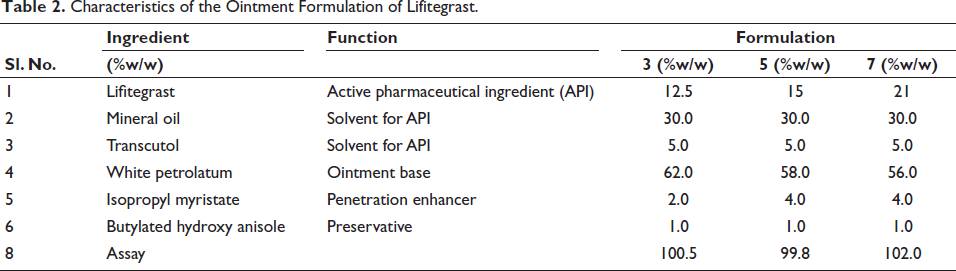
Characteristics of the Ointment Formulation of Lifitegrast.
Evaluation of Physicochemical Aspects of the Formulations
The compatibility of the active pharmaceutical ingredient (API) with excipients was evaluated with Fourier transform infrared spectroscopy (FT-IR) spectroscopy (FTIR-8400-S, Shimadzu, Japan). The spectra of the API lifitegrast and the final formulations were compared to ascertain the common peaks. All the formulations were also tested visually for color, appearance, homogeneity, and phase integrity. The pH of the formulations was tested using a digital pH meter (Lab India, Thane, India), which was calibrated using standard buffers at pH 4.7 and 10.
The rheological behavior of different formulations was evaluated using a stress-controlled rheometer (MCR-302, Anton Paar USA Inc., Ashland, VA, USA). The formulation was mounted in a Peltier hood to maintain the formulation temperature at 32°C and prevent evaporation. Approximately 0.5 g of each sample was placed on the lower plate with a preset gap of 100 µm with the upper plate. Steady-state flow tests (0.1–100 s–¹) were conducted to characterize flow properties and obtain viscosity values at different shear rates. 12
The in vitro release test was conducted using vertical diffusion cells with a 5 mL volume and a recirculating water bath at 32 ± 0.5°C. Each cell had a donor and receptor chamber separated by a synthetic membrane. Approximately 300 mg of the drug product was applied to the membrane in the donor chamber. At the same time, the receptor compartments were filled with 10 mM phosphate-buffered saline (PBS, pH 7.4) and continuously stirred with a magnetic stirrer. Samples were taken from the receptor compartment at specific time points up to 6 h to determine the drug release rate, which was analyzed using HPLC.
HPLC was employed to perform an assay of lifitegrast in the topical formulations. The drug was extracted from the formulation by dissolving it in diluent media containing 40% methanol, 40% acetonitrile, and 20% water. The formulation in diluent solution was collected and further diluted to achieve a 300-ppm concentration of lifitegrast. The solution was analyzed using an HPLC system (Shimadzu LC-20AD) after being filtered through a 0.45 µm membrane filter. A gradient program was used with pH 5.5 buffer and acetonitrile and buffer in the ratio of 9:1. The sample was injected into Hypersil ODS (250 × 4.6, 3µ) column at a flow rate of 1 mL/min and column temperature at 35°C with a detection wavelength of 260 nm. The assay was quantified as a percentage of the labeled amount.
Cryopreserved, dermatomed human skin was obtained and prepared for the experiment by cutting into sections for mounting on vertical Franz diffusion cells with a diffusion area of 0.64 cm2. The receptor chamber was filled with 10 mM PBS (pH 7.4), and the temperature was controlled at approximately 32°C. The dermatomed skin thickness ranged from 400 to 500 µm. The barrier integrity was assessed by measuring electrical resistance at 100 Hz and 100 mV before applying the formulations. Acceptable skin specimens underwent further permeation studies with a finite dose of 10 mg/cm2 formulation added to the donor compartment. After 24 h, the excess formulation was removed by cotton swabbing and tape stripping, followed by overnight stirring in PBS for drug extraction. The estimation of the drug was done using the HPLC method mentioned above.
Evaluation of Biological Characteristics of the Formulations
C57 male mice were maintained under a controlled 12-h light/dark cycle, with a temperature of 25–28°C and air humidity of 50–70%. The Institutional Animal Ethics Committee (IAEC) of the institute approved the experimental protocol, which was constituted as per the directions of the Committee for Control and Supervision of Experiments on Animals (CCSEA), with protocol no. DMIHER/IAEC/22-23/33. For the psoriasis study, mice were anesthetized with ketamine and xylazine, and an area of approximately 4 cm2 on their backs was shaved. After 24 h, the mice were divided into five groups: control (petrolatum application), IMQ (62.5 mg of 5% IMQ cream daily), IMQ + lifitegrast gel formulation (62.5 mg of 5% IMQ cream daily, and lifitegrast gel 3%,5%, or 7% w/w, twice a day), IMQ + lifitegrast ointment formulation (62.5 mg of 5% IMQ cream daily and lifitegrast ointment 3%, 5%, or 7% w/w, twice a day), IMQ + 0.05% clobetasol (Clop, Zydus) 100 mg once a day. The volume of lifitegrast formulations was 50 mg at one application. This treatment continued for 6 days.
The psoriasis severity was monitored using the Psoriasis Area and Severity Index (PASI), evaluating erythema, scaling, and thickness on a scale from 0 to 4. Mice were restrained for 15 min after lifitegrast application to ensure proper absorption, with a 2-h gap between IMQ and lifitegrast applications. A cumulative score (erythema plus scaling plus thickening) served as an objective evaluator of the severity of the induced psoriasis (scale 0–12).
Statistical Analyses
The data were analyzed with GraphPad Prism 10 and are presented as Mean ± SEM. One-way analysis of variance with Bonferroni or Newman–Keuls correction was used for multiple comparisons. Probability values <0.05 were considered significant for a 95% confidence interval.
Results
Three gels and three ointments of lifitegrast were prepared as mentioned in Tables 1 and 2. The formulations were found to be non-transparent, and the gels were smooth, homogenous and translucent. All the formulations had a pH in the range of 7.0–7.3. The formulations were evaluated under a microscope and found to be free of crystal formation.
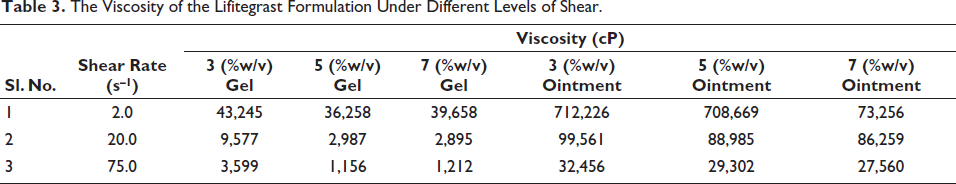
As seen in Table 3, the ointments were found to have high viscosity. Since the formulations were thinning with shears, the stability of the product was indicated, and the ease of spreadability was demonstrated since the viscosity decreased by increasing shear.
The Viscosity of the Lifitegrast Formulation Under Different Levels of Shear.
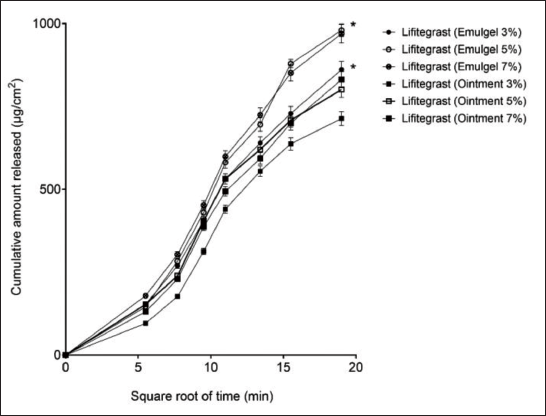
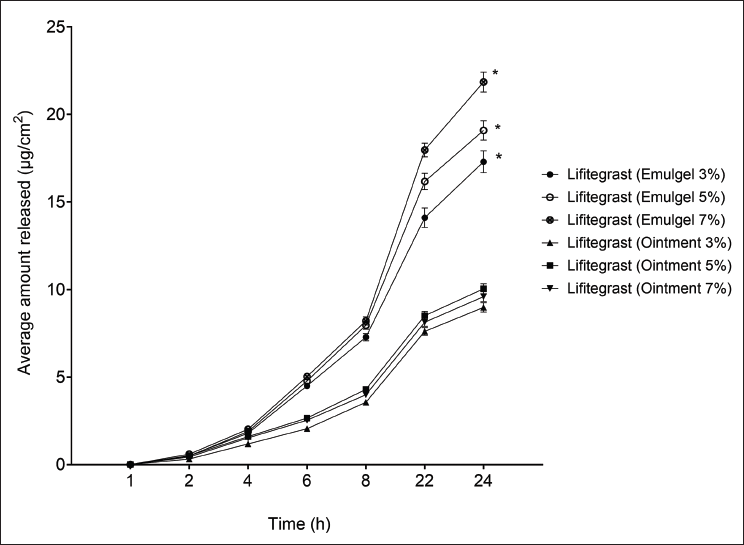
These formulations were tested for in vitro release profiles. The drug release was found to follow a linear pattern, as shown in Figure 1. The cumulative amount released at the end of 6 h for the gel was higher than the ointment at all strengths. The ointment formulations showed the release of 713.33 ± 20.08, 800.98 ± 23.35, and 831.13 ± 24.22 µg/cm2 for 3%, 5%, and 7% strengths, whereas the gel formulations showed the release of 861.28 ± 25.10, 979.99 ± 18.31, and 969.82 ± 28.27 µg/cm2, respectively. Thus, the release was found to be optimum at 5% strength, and at all strengths, the ointment formulation showed significantly lower release than the gel formulation. The in vitro drug permeation was tested through human skin, which showed that at the end of 24 h, permeation of gel at 3%, 5%, and 7% strength was 17.3 ± 0.25, 19.09 ± 0.56, and 21.85 ± 0.57 µg/cm2, and for ointment, it was significantly lower at 8.98 ± 0.26, 10.01 ± 0.26, and 9.6 ± 0.28 µg/cm2. This significant change in penetration seems to be the result of the high viscosity of the ointments, as shown in Figure 2.
The Release Profile of the Emulgel and Ointment Formulations of Lifitegrast. The Data Indicate Mean ± SEM (n = 6). *0.05, When Compared to the Respective Strength Ointment Formulation.
The In Vitro Permeation of the Emulgel and Ointment Formulations of Lifitegrast. The Data Indicate Mean ± SEM (n = 6). *0.05, When Compared to the Respective Strength Ointment Formulation.
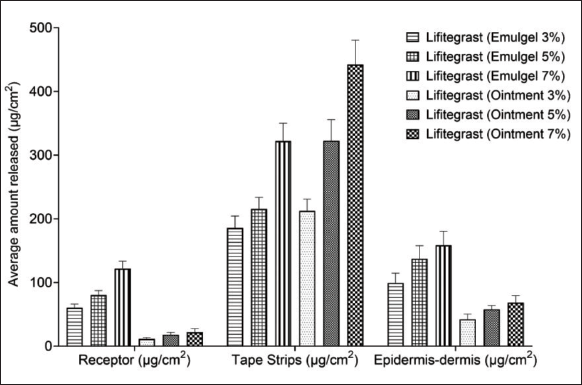
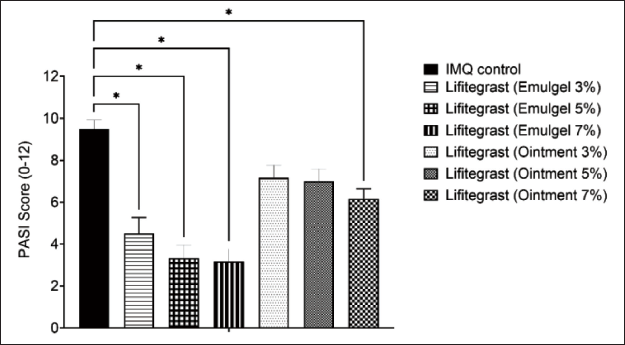
The skin retention of the formulation was tested. As observed in Figure 3, the concentration of lifitegrast stuck in the tape strips was higher in the ointment formulation. In contrast, the amount of the drug in the epidermis-dermis region was significantly higher for the gel formulation at all strengths. The topically applied lifitegrast formulation was well retained in the skin. On the other hand, the gel formulation was able to partition more drug concentration to the dermis-epidermis portion of the skin in a highly significant ratio. The pharmacological activity of the formulations was tested in the IMQ-induced psoriasis model in C57 mice. Data indicate a significant decrease in the PASI score after the treatment of all concentrations of the emulgel and after 7% ointment application, which was comparable to the effect of the standard-of-care clobetasol (Figure 4).
Comparative Profile of the Lifitegrast Formulation on Skin Permeation. Data Indicate Mean ± SEM, n = 6.
Effect of Lifitegrast Formulations on the PASI Score in C57 Mice. Data Indicate Mean ± SEM, n = 6. *<0.05, When Compared to the Imiquimod-control Group.
Discussion
This study focused on the development of a semisolid formulation of lifitegrast. Lifitegrast, a small molecule drug approved for treating dry eye disease, has the potential for managing psoriasis by inhibiting T cell-mediated inflammation. 7 Repurposing immunomodulators for topical treatment of psoriasis has significant potential in therapy, and immunomodulators, originally developed for other conditions, are being explored for their potential to treat psoriasis by modulating the immune system. 13 This approach aims to reduce inflammation and the overactive immune response that leads to psoriasis symptoms. These immunomodulators, such as tumor necrosis factor (TNF) inhibitors and interleukin-17 (IL-17) inhibitors, have shown effectiveness in treating moderate-to-severe plaque psoriasis. 6 They work by targeting specific pathways in the immune system that contribute to psoriasis, such as tumor necrosis factor-alpha (TNF-α), interleukin-12 (IL-12), interleukin-23 (IL-23), and IL-17. Studies have demonstrated the comparative effectiveness of various immunomodulators, with some showing superior results in achieving significant improvement in psoriasis symptoms. Topical formulations of immunomodulators can be more convenient and better tolerated by patients compared to systemic treatments.2, 4
Lifitegrast, primarily approved for treating dry eye disease, has shown potential for treating psoriasis and similar conditions due to its mechanism of inhibiting T cell-mediated inflammation.13, 14 Our repurposing study on lifitegrast’s effects in preclinical psoriasis models has elucidated its mechanism of action. 8 Lifitegrast is a potent inhibitor of LFA-1/ICAM-1 T cell binding, showing significant inhibition of human cutaneous T cell lymphocyte (HUT 78 T) cell adhesion and Jurkat T cell attachment. 7 Topical application of lifitegrast on IMQ-induced psoriatic skin significantly reduced the expression of IL-6, TNF-α, IL-17, and IL-23 genes associated with T cell activation. 8 These findings suggest that lifitegrast can effectively engage target receptors and reduce lymphocyte-mediated cytokine stimulation, making it a promising treatment for psoriasis.
This study focused on the composition and investigation of dermatological formulations of lifitegrast. For a suitable formulation designed for psoriasis treatment, the active ingredient must be released from the vehicle to penetrate the inflamed and thickened skin layers. 1 The correlation between the vehicle’s physicochemical and structural properties and its performance in vitro is crucial for selecting suitable topical dosage forms for therapeutic action and patient acceptance. Clinical studies show variations in potency and tolerance among different formulations, such as corticosteroid-containing aerosols versus ointments and azelaic acid gel/foam versus cream for treating rosacea and acne. 15 Despite numerous studies, inconsistencies remain in topical product development due to the complexities of semisolid products, influenced by factors like pH, viscosity, drug diffusion, and skin permeation.
This study demonstrated that semisolid dermatological formulations can be a useful therapeutic approach for the treatment of psoriasis. The in vitro and in vivo methods employed were able to distinguish between the formulations, detect the changes in dosage strength, and ensure a good correlation between the testing parameters. The gel formulations were more useful in comparison to the ointments since they were less viscous. Viscosity is a crucial parameter in the therapeutic utility of dermatological drugs since it impacts the drug’s performance, stability, patient compliance, and overall effectiveness in a range of skin ailments, 16 and highly viscous formulations may hinder the drug release in vivo. 17 The in vitro permeation test demonstrated that lifitegrast’s topical delivery was significantly higher with emulgel formulation. The ointment formulation retained more drugs on the skin surface with minimal distribution in the epidermis-dermis. Emulgel formulations demonstrated higher skin retention and permeation, attributed to the emulsion’s shear-thinning properties and the gel’s stability and penetration abilities. The rheological properties of emulgels also enhance drug release and skin penetration. 18
The effectiveness of dermatological products relies on their ability to act locally in the skin’s epidermal and dermal layers. Since there is no single method to determine the bioavailability of topical formulations, a combination of techniques is used to assess their in vitro performance before conducting costly and potentially insensitive clinical studies. In our study, emulgel or gel formulations were found to be more beneficial for psoriasis treatment due to their spreadability, elasticity, enhanced release, permeation, and skin retention. Emulsion formulations were more suitable for treating inflammatory skin conditions because of their emollient nature and better cutaneous retention compared to ointments. These findings highlight the importance of selecting the appropriate vehicle based on the site of action and intended therapeutic effect, as it plays a critical role in the performance and permeation of semisolid formulations. Carbopol 974P, a synthetic high molecular weight polymer used as a gelling agent in our formulation, was able to balance the viscosity optimally, providing a smooth and consistent texture, and formed a stable gel for all the three strengths tested. Isopropyl myristate, a fatty acid ester used as an emollient and permeation enhancer in our formulation, helped improve the absorption of lifitegrast in the dermis-epidermis layers by reducing the barrier function of the stratum corneum. 19 In the IMQ-induced psoriatic skin, there was significant parakeratosis, epidermal hyperplasia, inflammation, and hemorrhage. Clobetasol is a high-potency topical corticosteroid approved by the Food and Drug Administration (FDA) for treating moderate to severe plaque psoriasis. It works by reducing inflammation, relieving symptoms such as itching and redness, and decreasing levels of inflammatory cytokines. 20 We have used clobetasol as an effective comparator in the IMQ-induced psoriasis model in mice, and the effect of lifitegrast gel formulation was comparable to the clobetasol treatment. This study showed that dermal application of lifitegrast caused a substantial improvement in IMQ-induced psoriasis by reducing all the parameters of skin inflammation in this model. Reduction in T cell-mediated immunity is the major mechanism of lifitegrast. T cell-mediated immunity is primarily mediated through circulation and splenocytes. However, lifitegrast, primarily used for treating dry eye disease by reducing the LFA-1/ICAM-1 interaction, has shown promising potential in psoriasis therapy as well. 8 The topical solution of lifitegrast was shown to decrease the inflammatory pathways involving the LFA-1/ICAM-1 interaction, and T cell activation pathways are present in psoriatic skin inflammation. 21 Taken together, it suggests that reducing the LFA-1/ICAM-1 interaction is an established method for alleviating psoriatic inflammation. The use of lifitegrast solution approved for ophthalmic administration has been tested and found to be useful in the treatment of psoriasis. 8 The skin’s outermost layer, the stratum corneum, acts as a barrier that favors the penetration of lipophilic (fat-soluble) compounds. This makes it easier for lipophilic drugs to pass through this layer. 3 Conversely, the epidermis and dermis layers are more aqueous, making them favorable environments for hydrophilic (water-soluble) agents to partition and act effectively. 10 When it comes to lifitegrast, which is typically used as a water-soluble salt in ophthalmic formulations, the challenge would be ensuring that it can effectively penetrate through the stratum corneum to reach the deeper layers of the skin where it can exert its therapeutic effects. Since the unique characteristics of the skin layers influence the delivery and efficacy of topical treatments, we explored formulations for lifitegrast using the lipophilic form of the compound to ease its penetration and retention in the skin’s lipid barrier, which has shown significant effectiveness in the in vitro and in vivo tests. Since the skin retention of the compound is increased and the doses employed are within the safe therapeutic range of the lifitegrast, the repurposing of lifitegrast appears to be safe and effective. Further clinical studies would be required to substantiate the preclinical data that we have presented in this article.
Conclusion
The study found that semisolid formulations of lifitegrast are stable and exhibit favorable rheological behavior for dermatological topical application. The study parameters were able to differentiate changes in vehicle composition, with gel formulations showing the highest release rates. Emulgel formulations demonstrated superior skin permeation and retention as compared to the ointments. These findings underscore the importance of lifitegrast in the treatment of psoriasis, which would be validated in a suitable clinical study.
Footnotes
Abbreviations
API: Active pharmaceutical ingredient; FDA: Food and Drug Administration; FT-IR: Fourier transform infrared spectroscopy; HPLC: High-performance liquid chromatography; HUT 78 T: Human cutaneous T cell lymphocyte; ICAM-1: Intercellular adhesion molecule-1; IMQ: Imiquimod; IL-12: Interleukin-12; IL-17: Interleukin-17; IL-23: Interleukin-23; LFA-1: Lymphocyte function-associated antigen-1; PASI: Psoriasis Area and Severity Index; PBS: Phosphate-buffered saline; PEG400: Polyethylene glycol 400; TNF-α: Tumor necrosis factor-alpha.
Declaration of Conflict of Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The animal studies for the present work were approved by the Institutional Animal Ethics Committee (IAEC) of the institute constituted as per Committee for Control and Supervision of Experiments on Animals (CCSEA) directions, with protocol no. DMIHER/IAEC/22-23/33.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
