Abstract
Background
Cancer has become the second leading cause of death among the Indian population.
Objectives
The purpose of the research was to elucidate and compare the suppressive impact on cell proliferation of metformin, teneligliptin, and canagliflozin on MCF-7 breast cancer cell lines.
Materials and Methods
The MTT assay was employed to assess the anti-proliferative impact of the anti-diabetic drugs metformin, teneligliptin, and canagliflozin on MCF-7 breast cancer cell lines. After the MCF-7 cells achieved total growth, they were transferred to 24 well plates. Various quantities of metformin, teneligliptin, and canagliflozin were then administered to the appropriate wells. The maximum concentration was set at 1,000 µg/mL, but eight other concentrations were also set, with the lowest dosage being 7.8 µg/mL.
Results
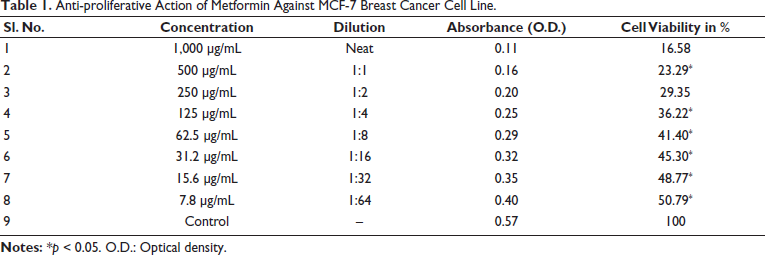
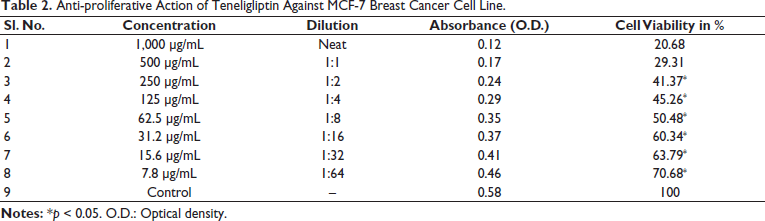
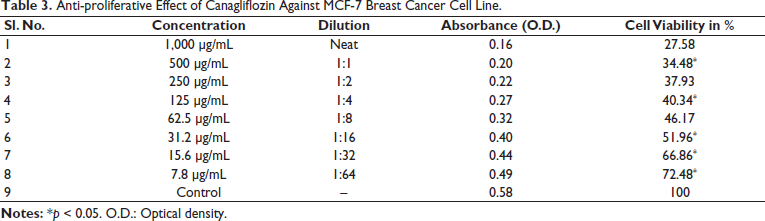
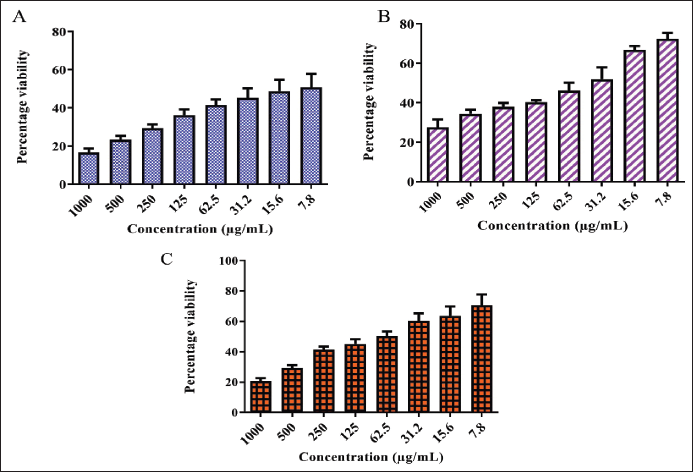
Through MTT assay, the cell viability of metformin was found to be 50.79, 48.77, 45.30, 41.40, 36.22, 29.35, 23.29, and 16.58% respectively. At concentrations of 7.8, 15.6, 31.2, 62.5, 125, 250, 500, and 1,000 µg/mL, the cell viability of teneligliptin was found to be 70.68, 63.79, 60.34, 50.48, 45.26, 41.37, 29.31, and 20.68%, respectively, and the cell viability of canagliflozin was found to be 72.28, 66.86, 51.96, 46.17, 40.34, 37.93, 34.38, and 27.58%, respectively. The inhibitory concentration 50 (IC50) values for metformin, canagliflozin, and teneligliptin were 7.8, 31.2, and 62.5 µg/mL, respectively.
Conclusion
All the three selected anti-diabetic drugs produced significant anti-proliferative activity in a dose-dependent manner. Further studies are needed to find the molecular mechanism behind it.
Introduction
Cancer has become a significant threat to human life. The word “Cancer” means Crab, and it was derived from the Greek word “Karkinos.” This condition is non-communicable, meaning it cannot be transmitted from person to person. The number of reported instances of this illness in India has quadrupled in the last 26 years. Cancer has emerged as the second most prevalent cause of mortality among the Indian people. Various studies have revealed that by the end of this decade, cancer deaths may reach 900,000. 1 There are several forms of cancers detected in various organs such as skin, blood, lungs, breast, cervix, rectum, stomach, prostate, liver, esophagus, bladder, mouth, and so on. Cancers of the breast, cervix, mouth, and lungs account for 41% of all cancer cases in India. Breast cancer is a prevalent kind of cancer among women in India, both in terms of occurrence and death rates. The study found that out of 100,000 women in India, there are 25.8 new instances of breast cancer and 12.7 deaths from the disease. 2 The term cancer shocks every individual because of the expensive treatment, side effects, and low survival rate. Most cancer drugs are cytotoxic, and they are not specifically acting toward the cancer cell line; they also affect normal healthy cells and produce harmful effects in our body.
Diabetes is a metabolic disorder; India is becoming a hub of diabetes. It is a silent killer because people who have it are often unaware that they are affected. Hyperglycemia and disruptions in the metabolism of fats, carbs, and proteins are hallmarks of the non-communicable disease known as diabetes mellitus (DM). 3 The International Diabetes Federation reports that approximately 40.9 million people suffer from diabetes in India, which is expected to grow to 60.9 million by 2025. 4
Cancer and DM are the most common health issues among several people, and various studies revealed that diabetic patients are more prone to get cancer. The relationship between these two diseases is associated with risk factors such as obesity and the lack of physical activity, aging, and diet. 5 The interrelation between diabetes and cancer was explained by a clinician 80 years ago. 6 Many epidemiological studies revealed that diabetic people have more chance (~2-fold) of getting liver, pancreas, and endometrial cancer and comparatively less chance (1.2- to 1.5-fold) of getting colorectal, breast, and bladder cancer. 7 However, no association was found between diabetes and lung cancer. 8 Recent meta-analyses revealed an increased risk of cancer in DM patients. 9
Metformin is the most common oral anti-diabetic drug that belongs to the class of biguanides. It is the most prescribed medicine, as it is safe and inexpensive. Various clinical research studies have shown that the usage of metformin reduces the risk of cancer in diabetic patients. Numerous research studies are in the pipeline to find the role of metformin in non-diabetic patients. Thus, metformin plays a significant role in cancer prevention. The use of metformin reduces the risk of cancers such as colorectal, pancreatic, stomach, liver, and esophageal. In contrast, no consistent results were found on cancers from the breast, prostate, and lung.10, 11 Previous research work on metformin shows that it is effective against colorectal, 12 prostate, 13 and ovarian cancer cell lines. 14
Teneligliptin is a newer anti-diabetic drug developed in Japan; it is a dipeptidyl peptidase-4 (DPP-4) inhibitor, 15 used for the treatment of type II-DM. The chemical structure of teneligliptin is unique, and it looks J-shaped when joined with five consecutive rings. 16 Incretins have a short half-life because of the rapid inactivation by the enzyme DPP-4. This increases the levels of incretins, glucagon-like peptide-1 (GLP-1), and gastric inhibitory polypeptide (GIP) by inhibiting the enzyme DPP-4, resulting in improving hyperglycemia by increasing serum insulin level.17, 18 It can be given alone or in combination with other oral anti-diabetic drugs or insulin. 19 Teneligliptin possesses many clinically significant properties, such as no drug interaction and a low risk of hypoglycemia.20, 21
Canagliflozin is an oral anti-diabetic drug used to reduce blood glucose through an insulin-independent approach that inhibits the sodium–glucose transporter system. The sodium–glucose transporters are divided into two types: sodium–glucose cotransporters (SGLT1 and SGLT2). 22 SGLT1 is present in the proximal tubule of nephrons, small intestine, and myocardium, whereas SGLT2 is present mainly in the brush border of proximal renal tubules. SGLT is involved in the reabsorption of glucose, water, and sodium. 23 So, by blocking SGLT, the reabsorption of glucose is inhibited, resulting in reduced blood glucose levels and glucosuria. Canagliflozin is also able to reduce body weight. 24
It is necessary to find a drug that is used to cure both cancer and DM. Previous research studies showed that some anti-diabetic drugs such as metformin and DPP-4 inhibitors produce anti-cancer activity against colon cancer. Hence, an attempt has been made to compare the anti-cancer activity of selected anti-diabetic drugs.
Materials and Methods
The MCF-7 breast cancer cell lines were obtained from the National Centre for Cell Sciences (NCCS), located in Pune. The pharmaceutical companies provided teneligliptin, canagliflozin, and metformin for the study. Fetal bovine serum (FBS) and Dulbecco’s modified Eagle medium (DMEM) were obtained from Cistron Laboratories, Chennai, India, and Hi-Media Laboratories in Mumbai, India, respectively. The chemicals used in this study, including dimethyl sulfoxide (DMSO), MTT, and trypsin, were obtained from Sisco Research Laboratory Chemicals, which is located in Mumbai, India. The remaining reagents and chemicals were supplied by Sigma–Aldrich (Mumbai).
Cell Preparation and Maintenance
The MCF-7 cell lines were cultured in minimal essential medium (MEM) supplemented with 10% FBS, penicillin (100 U/mL), and streptomycin (100 µg/mL). The cells were maintained in a controlled environment with a humidity level of 5% CO2 and a temperature of 37°C. The procurement of all other consumable materials was done through Sigma–Aldrich, Mumbai.
In Vitro Assay for Anti-proliferative Activity
The anti-proliferative effect of selected oral anti-diabetic drugs metformin, teneligliptin, and canagliflozin against MCF-7 breast cancer cell lines was estimated by the MTT assay. The MCF-7 cells, with a concentration of 1 × 10 5 per well, were cultivated in 24 well plates and subjected to incubation at a temperature of 37°C under conditions of 5% CO2. Once the required number of cells are grown, different concentrations of metformin, teneligliptin, and canagliflozin were added into the appropriate wells. The highest concentration was fixed as 1,000 µg/mL, eight different concentrations were fixed, and the lowest concentration was fixed as 7.8 µg/mL. The concentrations were obtained by performing a series of dilutions using DMSO and then incubated for a duration of 24 h. Following incubation, the test solutions containing metformin, teneligliptin, and canagliflozin were extracted and rinsed with MEM or phosphate buffer. They were then subjected to an additional 4-h incubation period. The blue formazan that was produced was dissolved in 1 mL of DMSO. The absorbance of solution was then determined at a wavelength of 570 nm using an ultraviolet (UV) spectrophotometer, with DMSO acting as the reference. The cytotoxicity profiles of compound were evaluated by measuring the cleavage of the tetrazolium salt (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide) by mitochondrial dehydrogenases in living cells. 25 The cytotoxic effects of the chosen medications were determined by applying the following formula: percentage cell viability = (Absorbance of treated cells/Absorbance of control cells) × 100. The studies were conducted three times, and the concentration needed to achieve a 50% inhibition [inhibitory concentration 50 (IC50)] was determined using graphical methods.
Statistical Analysis
The statistical and IC50 values calculations were carried out using GraphPad Prism 5 version 5.01. The anti-proliferative activity was assessed by conducting multiple t-tests. The data were displayed as Mean ± Standard Deviation (SD). The asterisk (*) indicates a significant difference when the p value is less than 0.05.
Results
The anti-proliferative effect of anti-diabetic drugs such as metformin, canagliflozin, and teneligliptin on the MCF-7 breast cancer cell line was estimated by the MTT assay. At concentrations of 7.8, 15.6, 31.2, 62.5, 125, 250, 500, and 1,000 µg/mL, the cell viability of metformin was found to be 50.79, 48.77, 45.30, 41.40, 36.22, 29.35, 23.29, and 16.58%, respectively, as shown in Table 1. The IC50 of metformin was at the concentration of 7.8 µg/mL. The percentage of cell viability of metformin on the MCF-7 cell line was found to be dose-dependent, and the images are given in Figure 1. At the same concentrations, the cell viability of teneligliptin was found to be 70.68, 63.79, 60.34, 50.48, 45.26, 41.37, 29.31, and 20.68%, respectively, as shown in Table 2. The IC50 of teneligliptin was 62.5 µg/mL. The percentage of cell viability of teneligliptin on the MCF-7 cell line was found to be dose-dependent, and the images are given in Figure 1. Canagliflozin at various concentrations produced cell viability of 72.28, 66.86, 51.96, 46.17, 40.34, 37.93, 34.38, and 27.58%, respectively, as shown in Table 3. The IC50 value of canagliflozin on the MCF-7 cell line was found to be 31.2 µg/mL. The dose-dependent relationship between canagliflozin and cell viability was seen in the MCF-7 cell line (Figure 1). Metformin, canagliflozin, and teneligliptin, which are all medications, have demonstrated varying degrees of anti-cancer effects at different concentrations, ranging from low to high, as compared to the control group. The IC50 values for metformin, canagliflozin, and teneligliptin were 7.8, 31.2, and 62.5 µg/mL, respectively.
Anti-proliferative Action of Metformin Against MCF-7 Breast Cancer Cell Line.
Anti-proliferative Action of Teneligliptin Against MCF-7 Breast Cancer Cell Line.
Anti-proliferative Effect of Canagliflozin Against MCF-7 Breast Cancer Cell Line.
A, B, and C Represent the Anti-proliferative Potential of Metformin, Teneligliptin, and Canagliflozin Against MCF-7 Breast Cancer Cell Lines with Inhibitory Concentration 50 (IC50) Values 7.8, 31.2, and 62.5 µg/mL.
Discussion
The anti-proliferative activity of metformin was previously reported by various researchers in colorectal cell lines, ovarian cell lines, and prostate cell lines. Glucose deprivation can enhance the anti-proliferative action in breast cancer, 26 and our results correlate with the previous results. Another drug, pramlintide, a parenteral anti-diabetic drug, inhibited the growth of HT-29 and HCT-116 cells with IC50 values of 48.67 and 9.10 µg/mL, respectively. 27 Pramlintide has anti-cancer action against colorectal cancer. Our selected anti-diabetic drugs, such as metformin, canagliflozin, and teneligliptin, have different mechanisms of action to control blood glucose. The anti-proliferative potential of these drugs may be due to the antioxidant properties of these compounds.
Conclusion
Within the scope of this research, the anti-proliferative activity of metformin, teneligliptin, and canagliflozin was assessed against MCF-7 breast cancer cell lines using a cell viability assay. A comparative analysis was conducted to evaluate their effectiveness. It was found that these drugs produce inhibitory action against cell lines with an IC50 of 7.8, 31.2, and 62.5 µg/mL. Thus, in conclusion, all three selected anti-diabetic drugs produced significant anti-proliferative activity in a dose-dependent manner. Further studies are needed to find the molecular mechanism behind it.
Footnotes
Abbreviations
DM: Diabetes mellitus; DMEM: Dulbecco’s modified Eagle medium; DMSO: Dimethyl sulfoxide; DPP-4: Dipeptidyl peptidase-4; FBS: Foetal bovine serum; GIP: Gastric inhibitory polypeptide; GLP-1: Glucagon-like peptide-1; IC50: Inhibitory concentration 50; MEM: Minimal essential medium; MTT: 3-[4,5-Dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide; NCCS: National Centre for Cell Sciences; O.D.: Optical density; SD: Standard deviation; SGLT1: Sodium–glucose cotransporter 1; SGLT2: Sodium–glucose cotransporter 2;
Acknowledgments
All the authors thank the faculties of Sri Ramachandra Medical College and Hospital, Sri Ramachandra Institute of Higher Education and Research, and Tagore Medical College and Hospital for their support and guidance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable as it is an in vitro study and does not involve the animal/human subjects.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
