Abstract
Background
Rheumatoid arthritis (RA) affects nearly 4% of the world’s population, along with an increasing rate of new cases.
Objectives
The current work was focused on investigating the anti-arthritic efficacy of the Curcuma xanthorrhiza rhizome extract (CXRE) against the complete Freund’s adjuvant (CFA)-triggered RA in rats.
Materials and Methods
The RA was triggered in the experimental rats and then administered with CXRE, respectively, for 14 days. The body weight and paw edema of the rats were measured precisely. The levels of hematological biomarkers were measured using an automated hematological analyzer. The kidney injury biomarkers level was assessed using assay kits. The status of total protein and oxidative stress biomarkers was measured using kits. Histopathological studies were done on the hind limb tissues.
Results
The treatment with the CXRE improved body weight and diminished paw edema in the CFA-triggered RA rats. The C-reactive protein and rheumatoid factor levels were appreciably attenuated by the CXRE treatment. A substantial reduction in renal markers was observed in the CXRE-treated RA rats. The CXRE increased the red blood cells and hemoglobin while reducing the white blood cell and erythrocyte sedimentation rate status in the RA rats. The antioxidants catalase, glutathione peroxidase, and superoxide dismutase were boosted, and lipid peroxidation status was depleted by the CXRE in the RA rats. The CXRE also ameliorated the CFA-induced histological alterations in the CFA-induced RA rats.
Conclusion
In conclusion, the current results demonstrated that the CXRE appreciably attenuated the CFA-induced RA progression in the experimental rats via its potential antioxidant and salutary properties. Therefore, it could be utilized as a promising anti-arthritic agent to treat RA.
Introduction
Rheumatoid arthritis (RA) is an autoimmune disorder that causes persistent inflammation in the synovium. Little diarthrodial joints connected to major joints are particularly vulnerable to the effects of RA. 1 All of these minor progressions eventually lead to the clinical manifestation of ailment in the form of discomfort, edema, inflexibility in the knee and ankle joints, and difficulties in moving due to altered bone structure at later disease phases. RA has a 2–4% worldwide prevalence. The prevalence and racial distribution of this illness are continually evolving. This condition disproportionately affects females as opposed to males. 2 However, the precise cause of RA that participates in its rapid development remains unknown.
The complete Freund’s adjuvant (CFA) is an extensively employed technique to induce autoimmune disorders like RA in experimental animal models. Hyperimmunization and elevated levels of circulating antigen-specific antibodies following subcutaneous injection of CFA cause joint disability and inflammation. 3 Arthritis is induced by CFA in rats in two stages: first, periarticular inflammation develops, and then, like arthritis, bone involvement occurs. Pathological changes caused by arthritis induction include synovitis inflammation, an elevation in inflammatory cell infiltration, a spike in inflammatory mediators, cartilage degradation, and joint deformity. 4
In most cases, non-steroidal anti-inflammatory drugs (NSAIDs) can provide only symptomatic relief for those living with RA, a chronic, debilitating condition. The usage of these drugs is often related to evident harmful effects such as ulceration, hypertension, and stroke. Consequently, safer and alternative natural therapy options represent an unmet therapeutic requirement these days. Plant-based treatments are becoming highly famous worldwide, even though contemporary medicine has made huge advances in terms of both science and technology. 5
In Southeast Asian countries, particularly Indonesia, the herb Curcuma xanthorrhiza Roxb. is extensively employed in conventional medicine and as a dietary complement. C. xanthorrhiza belongs to the Zingiberaceae family and is commonly referred to as Java turmeric or Temulawak. Furthermore, C. xanthorrhiza has been highlighted to have extensive pharmacological activities like anti-diabetic, anti-cancer, anti-inflammatory, anti-microbial, and antioxidant effects. Additionally, it was also highlighted that C. xanthorrhiza exhibits skincare and hepatoprotective activities.6–8 Apart from these biological properties, the salutary effects of C. xanthorrhiza extract against RA have not been scientifically studied yet. Thus, the current work was planned to examine the anti-arthritic property of the C. xanthorrhiza rhizome extract (CXRE) against the CFA-triggered RA in rats.
Materials and Methods
Plant Material
The C. xanthorrhiza rhizomes were collected from Bogor, West Java province, Indonesia, during the month of February 2023 with the help of traditional experts residing around that location.
Extract Preparation
The rhizomes (1 kg) were rinsed, dehydrated in the shade, ground into a fine powder, and then heat macerated with 2 L of deionized water at 60°C for 35 min. After that, the suspension was cooled to 37°C and filtered using Whatman No. 1 filter paper. The suspension was condensed using a rotary evaporator at 45°C, and the final extract was stored at 4°C, till further use.
Experimental Rats
The work was executed at the 6-week age. Wistar rats weighing around 150–200 g were purchased from the Biocon animal house in Bangalore, India. All rats were caged in a clean polypropylene confine and sustained in a well-ventilated cabin at 24 ± 1°C with a 12 h light/dark sequence. The rats were given access to the standard rat-pelleted diet and clean drinking water ad libitum. All rats were acclimated at least 1 week before starting the experiments. The present animal study was approved by the animal ethics committee at KMCH College of Pharmacy, Coimbatore (approval number: KMCRET/ReRe/I.P/64/2022).
Preparation of Extract and Standard Drug
The CXRE was dissolved in 1% sodium carboxymethyl cellulose (Na CMC) and treated through an oral gavage route to the rats at the concentrations of 250 and 500 mg/kg/day. The control rats received the only vehicle, 1% w/v Na CMC. Leflunomide (10 mg/kg) was dissolved in 1% w/v Na CMC and given for 14 days to rats as a positive control drug.
Experimental Groups and Treatment Protocols
The experimental rats were arbitrarily distributed into five groups, with six rats in each (n = 6). The group I rats were served as a control group and treated only with Na CMC. The rats from group II were RA-triggered rats by injecting 0.1 mL of CFA through a subcutaneous route in the plantar surface of the hind limb. The group III rats were treated with the standard drug leflunomide (10 mg/kg) for 42 days. The rats from group IV and group V were RA-induced as described in group II and then administered with 250 and 500 mg/kg of CXRE, respectively, for 42 days. After that, the body weight of all rats was measured.
Determination of Body Weight and Paw Edema
The body weight of each rat was detected precisely before the initiation of RA and then after every 3 days until the completion of experiments. The paw thickness of the experimental rats was measured with the help of a plethysmometer, which is proportional to the degree of inflammation in the joints. Paw edema was detected in each rat by the blinded method. The arthritis score was determined before and after the treatments. The score was evaluated on a scale of 0–4: 0—no swelling or edema, 1—very mild, 2—mild, 3—moderate, and 4—severe edema.
Analysis of Hematological Parameters
After the completion of the treatments, all the experimental rats were anesthetized using pentobarbital sodium. The blood was gathered from the experimental rats via retro-orbital procedure. The gathered blood was subjected to an examination of the hematological biomarkers such as red blood cells (RBCs), white blood cells (WBCs), and hemoglobin (Hb) using the automated hematological analyzer (Beckman Coulter, USA).
Quantification of Renal Function Biomarker Levels
The changes in the kidney injury biomarker levels, that is, urea, uric acid, and creatinine, in the experimental rats were investigated using the kits (Thermo Fisher Scientific, USA).
Assay of C-reactive Protein (CRP), Rheumatoid Factor (RF), and Total Protein in the Experimental Rats
The corresponding assay kits were employed to detect the CRP, total protein, and RF levels in the experimental animals. All the assays were performed in triplicates using the described guidelines by the kit’s manufacturer (Abcam, USA).
Measurement of Oxidative Stress Marker Levels
The status of superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), and lipid peroxidation (LPO) in both control and experimental animals was investigated using the kits following the protocols recommended by the manufacturer (Elabscience, USA).
Histopathological Analysis
After the scarification, the hind limb tissues were gathered from the experimental animals. The collected tissues were processed with 10% formalin and then dehydrated with graded ethanol. The nitric acid (10%) was employed to decalcify the tissues and then embedded in paraffin. The tissues were then cut into slices using rotary microtome at 5 µm diameter. The sliced tissues were then stained with hematoxylin and eosin, and finally, the slides were investigated under an optical microscope.
Statistical Analysis
The results from each assay were examined using SPSS software. The values are given as a Mean ± SD of triplicate measurements. The one-way analysis of variance (ANOVA) and Duncan’s multiple range test (DMRT) were employed to measure the alterations between the groups with p < 0.05 as significant.
Results
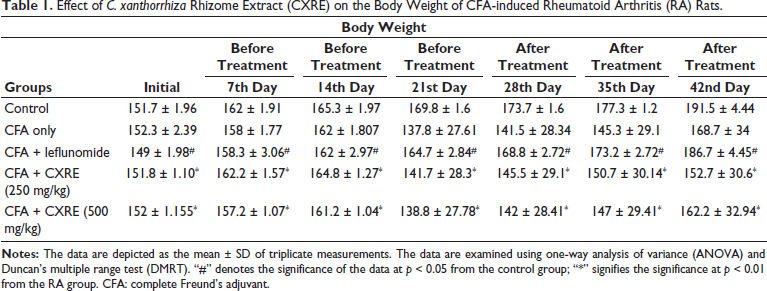
Effect of CXRE on the Body Weight of CFA-induced RA Rats
Table 1 shows the influence of CXRE on the body weight of the experimental rats. The RA rats exhibited a substantial decrease in body weight when compared to the control. A decrease in body weight may resemble the onset of the disease. However, the 250 and 500 mg/kg of CXRE treatment effectively increased the body weight of the CFA-induced RA rats, which indicates the salutary properties of the CXRE. The standard drug leflunomide also potentially increases the body weight of RA rats, which supports the therapeutic activity of CXRE.
Effect of C. xanthorrhiza Rhizome Extract (CXRE) on the Body Weight of CFA-induced Rheumatoid Arthritis (RA) Rats.
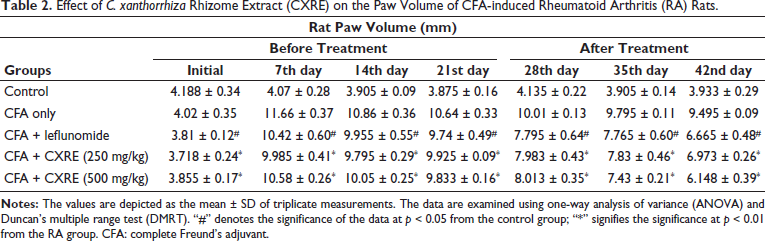
Effect of CXRE on the Paw Volume of RA Rats
The impact of CXRE on the paw edema of the experimental animals was determined, and the outcomes are revealed in Table 2. A considerable increase in the hind paw volume was noted in the RA rats, which indicates the increased level of inflammatory response in the joints. Interestingly, the 250 and 500 mg/kg of CXRE appreciably reduced the hind paw volume in the RA rats. These findings denote the anti-inflammatory properties of the CXRE. The activity of the CXRE was supported by the results of the standard drug leflunomide, which also substantially reduced paw edema in RA rats.
Effect of C. xanthorrhiza Rhizome Extract (CXRE) on the Paw Volume of CFA-induced Rheumatoid Arthritis (RA) Rats.
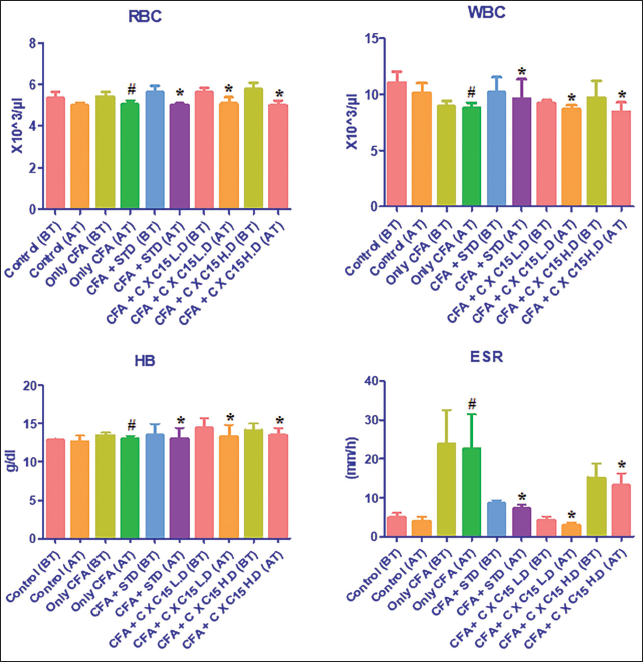
Effect of CXRE on the Hematological Parameters in the CFA-induced RA Rats
The status of hematological parameters such as RBC, WBC, Hb, and erythrocyte sedimentation rate (ESR) in the experimental rats was assessed, and the findings are demonstrated in Figure 1. The outcomes exhibited a remarkable diminution in the RBC and Hb, while the elevation in the levels of WBC and ESR was observed in the RA rats. Whereas the 250 and 500 mg/kg of CXRE treatment remarkably increased the RBC and Hb while decreasing the WBC and ESR levels in the CFA-induced RA rats. The leflunomide also effectively modulated the changes in hematological parameters in the RA rats, which reinforced the results of CXRE treatment.
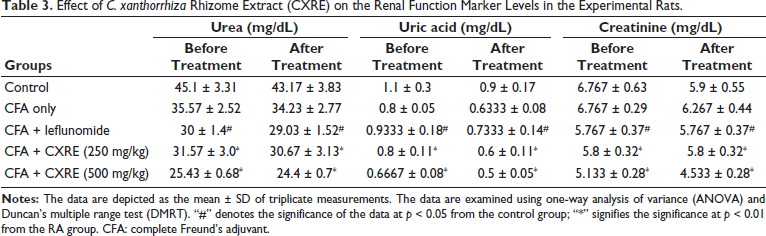
Effect of CXRE on the Renal Function Marker Levels
Table 3 demonstrates the status of kidney injury biomarkers, including urea, uric acid, and creatinine, in the control and CXRE-treated RA rats. The considerably increased status of these markers was observed in the RA rats. Contrarily, the 250 and 500 mg/kg of the CXRE treatment substantially decreased the urea, uric acid, and creatinine in the RA rats. The leflunomide treatment also diminished these markers. The activity of CXRE was supported by the results of standard drug leflunomide treatment.
Effect of C. xanthorrhiza Rhizome Extract (CXRE) on the Renal Function Marker Levels in the Experimental Rats.
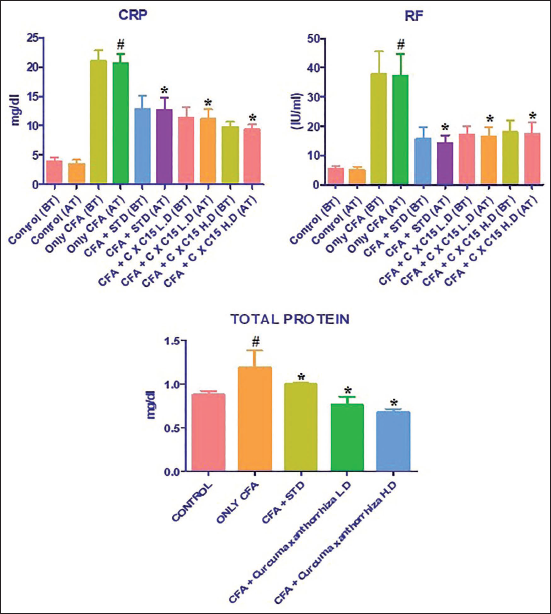
Effect of CXRE on the Total Protein, CRP, and RF Levels in the CFA-induced Rats
The CRP, RF, and total protein levels were assessed, and the findings are shown in Figure 2. As indicated in Figure 2, substantially increased levels of total protein, CRP, and RF were noted in the CFA-induced RA rats. Fascinatingly, considerable diminution in the status of total protein, CRP, and RF was noted in the 250 and 500 mg/kg of CXRE-administered RA rats. The levels of CRP, RF, and total protein were also decreased by the standard drug leflunomide treatment, which supports the activity of CXRE treatment.
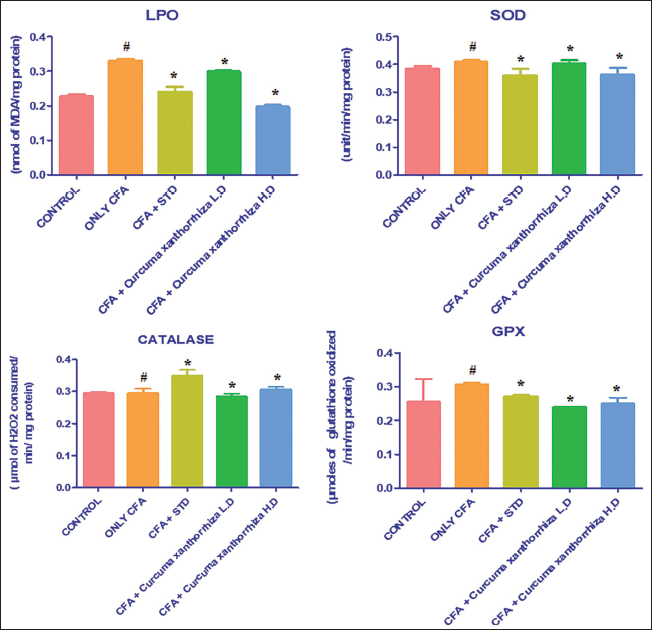
Effect of CXRE on the LPO and Antioxidant Levels in the CFA-induced RA Rats
Figure 3 exhibits the levels of LPO and SOD, CAT, and GPx in the CXRE-treated RA rats, which are assessed by assay kits. As shown in Figure 3, the considerable diminution in the antioxidants like CAT, GPx, and SOD while elevation in the LPO was observed in the CFA-induced RA rats. However, the 250 and 500 mg/kg of the CXRE effectively promoted the CAT, SOD, and GPx levels while substantially decreasing the LPO level in the CFA-induced RA rats. The leflunomide also boosted the antioxidants and decreased the LPO in the RA rats, which supports the antioxidant activity of CXRE.
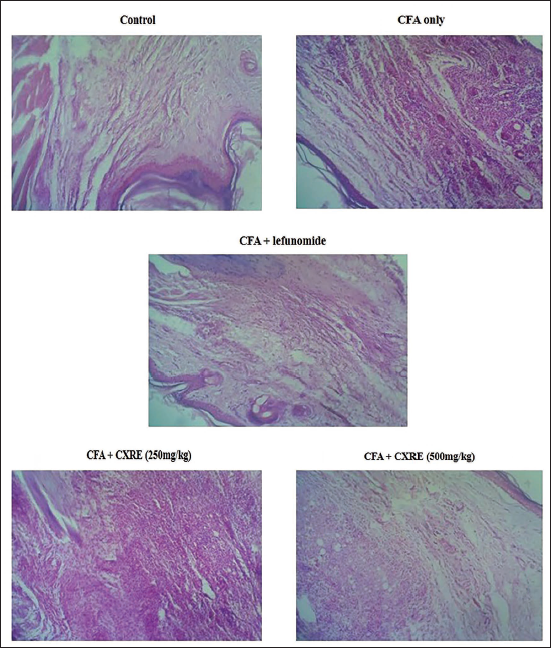
Effect of CXRE on the Histopathological Analysis of the Hind Limb of the RA Rats
The results of the histopathological investigation of the hind limbs of control and CXRE-treated RA rats are illustrated in Figure 4. The hind limb tissues of RA rats revealed clear signs of inflammation and tissue degeneration, such as synovial hyperplasia, inflammatory cell infiltrations, pannus development, and cartilage and joint destructions. Interestingly, the 250 and 500 mg/kg of CXRE administration attenuated the histological changes and reduced the cartilage, synovial, and bone joint destructions. The CXRE treatment also reduced the inflammatory cell infiltration and hyperplasia. The leflunomide also decreased the CFA-induced histological alterations in rats, which supported the activity of CXRE treatment.
Discussion
RA affects nearly 4% of the population around the world. The detailed etiology of RA remains unknown, and the formation of a pannus, a hyperplastic synovial membrane, is known to be a sign of RA. Bone and nearby articular cartilage can be destroyed by the pannus, which is one of the defining features of RA, along with the synovial membrane, inflammation, autoantibody production, and joint swelling. 9 Structural problems, injuries, premature deaths, and societal costs have all been attributed to RA. Previous research has shown that middle-aged women (between the ages of 50 and 60) are adversely affected by RA. 10 Clinical research has demonstrated that RA primarily manifests itself in the joints of the feet and hands, resulting in chronic painful swelling, abnormal enlargement of the synovium, pannus development, exaggeration, and alterations in joint morphology. The precise cause of RA origin is unknown, but research suggests that inflammatory responses and immune cells contribute significantly to the progression of RA. 11
Joint pain, edema, and stiffness are common early signs of RA; later stages bring bone loss and increased disability risk along with joint stiffness and deformity of varying degrees. Paw edema is associated with increased fluid output, vascular penetrability, and cellular permeability in the inflammatory area. Paw edema analysis is a standard technique for assessing the efficiency of anti-arthritic medications. 12 Paw edema is a sensitive and rapid indicator of inflammation and can be used to evaluate the efficacy of alternative treatments. A drug or plant extract’s anti-inflammatory activity can be shown in a decrease in paw diameter because of the decreased release of inflammatory mediators. 13 Evidence from prior studies suggests that paw edema is elevated in RA. In the current study, RA rats exhibited a similar elevation in paw volume, as highlighted previously. The results of this study corroborated those of previously reported research showing that CFA administration increased rat paw volume. 14 The anti-arthritic efficacy of CXRE treatment was supported by the diminution in paw edema in the CFA-induced RA rats.
RA is associated with weight loss, and muscle atrophy syndrome called rheumatoid cachexia. Consistent with earlier studies, 15 in this work, we discovered that the body weight of the RA rats was reduced. A lack of glucose and leucine absorption in the intestine has been connected to the low body weight seen in RA conditions. 16 The fact that CXRE treatment significantly increased body weight in the RA rats suggests its therapeutic effects against the RA condition. Arthritic severity can be described with the help of the arthritic score, a measure for evaluating joint inflammation. Our current findings also demonstrated that the CXRE treatment substantially decreased the arthritic score in the RA rats, which evidences the anti-arthritic effects of the CXRE.
Changes in hematological parameters are a well-documented observation in patients with arthritic diseases. Arthritis causes erythrocyte deformability because of a decrease in RBC level, a situation that frequently occurs and results in an animatic state (a shorter lifespan for erythrocytes). 17 Hemoglobin levels drop due to the death of immature RBCs and a subsequent drop in bone marrow erythropoietin (Hb). The WBC is a key immune system component associated with the development of inflammatory reactions and associated diseases. Kumar et al. 18 have proposed that inflammatory parameters like macrophages and granulocytes are produced, and their numbers are increased due to the increased levels of WBC. In this study, we found a substantial decrease in the RBC and Hb levels, while the WBC and ESR levels were increased in the CFA-induced RA rats. The hematological parameters were significantly impacted by CXRE treatment, which increased the RBC and Hb levels while reducing the WBC and ESR levels which may be due to its anti-arthritic effects.
Several diseases are characterized by increased oxidative stress. In RA patients, the oxidative stress mechanism appears to be highly positively associated with inflammation and increased joint degradation. 19 Furthermore, oxidative stress also plays a major role in the initiation and development of the pathophysiology of RA. Gene transcription is impeded by oxidative stress. Increased levels of oxidative stress from CFA stimulate immunological cells, which then produce enzymes and cytokines that can further exacerbate RA. 20 Generally, an oxidative stress condition causes a drastic reduction in the antioxidant defensive molecules such as SOD, CAT, and GPX from their normal threshold. 21 An earlier study highlighted that the levels of antioxidant enzymes were considerably depleted in RA patients. In addition, the level of lipid peroxidation was considerably increased during the RA condition. 22 In line with this, we also discovered that the CFA challenge depleted the SOD, CAT, and GPx levels while the LPO level was augmented in the RA rats, which corroborates the earlier literature. 23 Interestingly, the current results proved that the treatment with CXRE remarkably augmented the CAT, SOD, and GPx levels while substantially decreasing the LPO level in the RA rats, which supports the hypothesis that CXRE reduces the CFA-induced RA oxidation through its antioxidant property, thereby preventing the further progression of RA condition.
It has been highlighted that RA is linked to kidney failure; however, it is not known whether this is a treatment problem brought on by the long-term treatment of anti-inflammatory drugs or an actual effect of the disease itself. 24 Renal symptoms can also be caused by membranous glomerulonephritis and proliferative mesangial glomerulonephritis. 25 Creatinine levels reflect changes in glomerular filtration rate, while uric acid and urea status may be linked to changes in tubular function. 26 In this work, our findings demonstrated the substantial upsurge in the creatinine, urea, and uric acid levels in the CFA-induced RA rats. Fascinatingly, the CXRE administration decreased the status of creatinine, uric acid, and urea in the RA rats, which shows its therapeutic potential against CFA-induced RA-associated renal toxicity.
Conclusion
In conclusion, the current results exhibited that the CXRE remarkably attenuated the CFA-induced RA progression in the experimental rats via its potential antioxidant and salutary properties. The CXRE treatment appreciably modulated the fluctuations in the hematological parameters, boosted the antioxidants, and reduced the renal injury biomarker levels in the CFA-induced RA rats, which reveals its therapeutic potential. Therefore, it could be used as a promising anti-arthritic medication for the successful treatment of RA. In addition, in-depth molecular studies are still required in the future to promote CXRE as an effective RA medication.
Footnotes
Abbreviations
CAT: Catalase; CFA: Complete Freund’s adjuvant; CRP: C-reactive protein; CXRE: Curcuma xanthorrhiza rhizome extract; ESR: Erythrocyte sedimentation rate; GPx: Glutathione peroxidase; Hb: Hemoglobin; LPO: Lipid peroxidation; Na CMC: Sodium carboxymethyl cellulose; NSAID: Non-steroidal anti-inflammatory drug; RA: Rheumatoid arthritis; RBC: Red blood cell; RF: Rheumatoid factor; SOD: Superoxide dismutase; WBC: White blood cell.
Acknowledgments
The authors are grateful to the PT. Phytochemindo Reska, Gunung Putri, Bogor, Indonesia and KMCH College of Pharmacy, Coimbatore, India, for providing facilities and their constant support during this research work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
All experimental protocols adhered to internationally accepted norms for the use of laboratory animals, in accordance with the guidelines set forth by the Committee for Control and Supervision of Experiments on Animals (CPCSEA). The present animal study was approved by the animal ethics committee at KMCH College of Pharmacy, Coimbatore (approval number: KMCRET/ReRe/I.P/64/2022).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
