Abstract
The aim of this study is to investigate the efficacy of interleukin-6 (IL-6) inhibitor antibodies as a therapeutic agent to combat severe COVID-19. Given the constant evolution of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and its variants, the study aims to assess the potential of these inhibitors in adapting to new challenges and improving treatment outcomes. A comprehensive review of relevant literature was conducted using electronic databases such as Web of Science, PubMed, EMBASE, Scopus, and Google Scholar. The study analyzed the role of IL-6 inhibitors in combating SARS-CoV-2, focusing on their therapeutic potential in treating severe COVID-19 cases, particularly in controlling the overshooting immune response associated with cytokine storms. IL-6 inhibitors proved to be versatile and effective in attenuating severe immune responses, especially cytokine storms, which are common in advanced COVID-19 cases. The literature emphasizes their ability to regulate the immune system, reduce disease severity, and improve patient outcomes. Their efficacy in controlling the immune response and reducing disease severity makes IL-6 inhibitor antibodies a promising therapeutic strategy for severe SARS-CoV-2 and its new variants. Further research is needed to optimize their use and adaptability in evolving clinical scenarios.
Keywords
Introduction
Interleukin-6 (IL-6) is a crucial cytokine that substantially impacts the control of immunological and inflammatory responses. It is generated by diverse immune system constituents, such as T cells, B cells, macrophages, and fibroblasts. Depending on the context and the specific signaling pathways triggered, IL-6 can either promote or attenuate inflammatory responses. 1 Its versatile nature, termed pleiotropy, allows IL-6 to regulate aspects of neurological and hematological system development, acute phase responses, inflammation, and immune system functions.2, 3 In the case of viral infections, signals from infected cells are critical for mobilizing and activating the necessary innate or adaptive immune cells to fight the virus. The role of IL-6 has attracted considerable attention in the wake of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic. Research has shown that severe COVID-19 patients commonly have increased levels of IL-6, and these raised levels are frequently associated with more unfavorable clinical results. 4 The immunologic profile of COVID-19-positive critically ill patients suggests hyperactivation of the humoral immune system, particularly IL-6, as a key mediator of shock, multiorgan failure, and respiration. A cytokine storm, characterized by excessive inflammation due to the overproduction of IL-6 and other pro-inflammatory cytokines, is possible. 5 The exact processes leading to the surge in IL-6 levels and the subsequent cytokine storm in COVID-19 continue to be the subject of ongoing research. Preliminary studies suggest that irregularities in immune system behavior may be implicated in the progression of COVID-19. These irregularities might include a decrease in white blood cell counts, an increase in neutrophil levels, imbalances in monocytes and macrophages, as well as subdued or postponed type I interferon responses. The spike in IL-6 contributes to an overproduction of proinflammatory signaling molecules, including IL-6 itself, ultimately culminating in a cytokine storm.6, 7
The phrase “cytokine storm” describes the reaction of the immune system to a viral infection, which leads to the release of numerous inflammatory substances. This hyperactive immune response can damage organs and tissues and potentially lead to significant respiratory problems, multiple organ failure, and, in extreme cases, death. The main cytokines involved in this process include IL-6, IL-1, and tumor necrosis factor-alpha (TNF-α). Alternatively, therapeutic strategies could focus on modulating the role of IL-6 in the immune response (Figure 1).
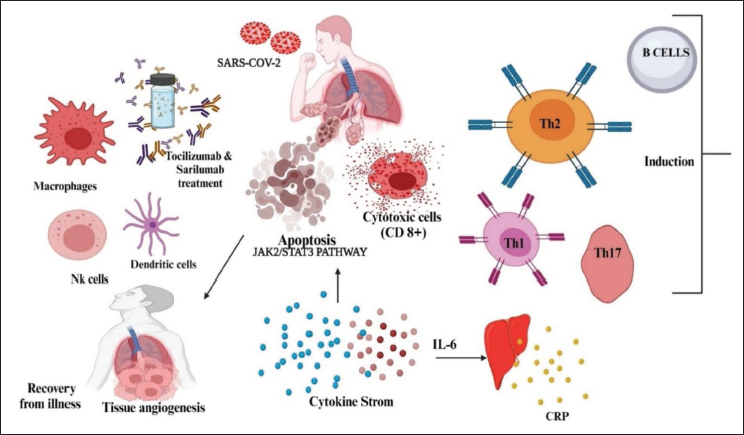
IL-6 inhibitors are a category of drugs that specifically target the function of the cytokine IL-6, which plays an important role in the treatment of inflammatory and autoimmune diseases. They offer therapeutic benefits in conditions such as rheumatoid arthritis, systemic juvenile idiopathic arthritis, and giant cell arteritis by reducing symptoms and improving quality of life. These inhibitors are used in the treatment of cytokine release syndrome, a severe reaction to cancer therapies, and have promise for use in the treatment of severe COVID-19 patients and other inflammatory diseases. IL-6 inhibitors are effective in relieving symptoms, preventing organ damage, and improving patient well-being by reducing inflammation.10–12 Table 1 illustrates the randomized clinical trial of selected IL-6 inhibitors.
Interleukin (IL)-6 Inhibitor and Selected Randomized Clinical Trials.

In the treatment of severe COVID-19 cases and to attenuate the intensity of cytokine storms, drugs targeting IL-6, such as tocilizumab and sarilumab, have been used to regulate the immune response. Tocilizumab and sarilumab are both recombinant humanized monoclonal antibodies that inhibit the IL-6R, thereby attenuating the IL-6-mediated systemic and local effects observed with COVID-19. 19 Tocilizumab and sarilumab are generally reserved for patients with severe inflammation and have shown promising results in reducing the need for mechanical ventilation and in some cases improving treatment outcomes. 20 More study is required to completely comprehend the complex function of IL-6 in COVID-19 and to maximize the use of IL-6 inhibitors as targeted therapeutics. Overall, IL-6 is an important therapeutic target in the treatment of SARS-CoV-2 infection, with potential pathways for reducing the severity and improving the prognosis of COVID-19 patients.
Overview of IL-6 and Its Role in the Immune Response
IL-6 is a central cytokine in the immune system that plays a multifaceted role in the regulation of immune reactions. It can stimulate immune activity during infection or injury but also acts to regulate this response to prevent excessive inflammation. This dual role makes IL-6 an important factor in balancing the body’s defense mechanisms. It is synthesized by several cell types including macrophages, T cells, B cells, and fibroblasts, and has a variety of functions in the immune system. IL-6 controls the immediate response phase and influences the production of immediate response proteins, such as C-reactive protein (CRP), in the liver. These proteins are involved in the early phases of the immune response. Furthermore, this cytokine plays a crucial role in hematopoiesis. Recognized as a proinflammatory cytokine, IL-6 is critical for mobilizing and activating immune cells in response to infection or tissue damage. The extensive effects of this extend to multiple immune system cells. IL-6 is vital in the adaptive immune response, as it enhances antibody production and the growth of effector T cells, growth of effector T cells. 21 The receptor system for IL-6 consists of two key elements: the IL-6R, which binds to the ligand, and the gp130 component, which is required for signal transduction. This dual-component structure is crucial for the initiation of the cell response to IL-6. After binding to its receptor, IL-6 triggers various intracellular signaling pathways, with the JAK/STAT system playing a particularly important role. This activation leads to the triggering of various transcription factors that control gene expression related to inflammation, immune activation, and tissue repair, as discussed by Uciechowski and Dempke. 22
IL-6 has a function in stimulating the proliferation and development of B cells, as well as facilitating the production of antibodies, as described by Maeda et al. 23 Furthermore, IL-6 plays a role in regulating T cell functions and facilitates the transformation of immature CD4+ T cells into Th17 cells, which are recognized for their ability to generate IL-17, a crucial component in protecting against extracellular pathogens. IL-6 can also prevent the growth of regulatory T cells (Tregs), which maintain the tolerance level of the immune system and prevent overwhelming immune responses, as mentioned by Li et al. 24 and Tanaka et al. 1 Beyond its typical functions, IL-6 has links to several pathological conditions. Rheumatoid arthritis, systemic lupus erythematosus and chronic inflammation are autoimmune diseases associated with excessive or unbalanced production of IL-6. It also supports tumor cell survival, proliferation, and angiogenesis. 25 Targeting IL-6 has become a treatment approach for various disorders due to its varied functions. IL-6 inhibitors work by blocking the IL-6R, thereby interrupting the JAK/STAT signaling pathway. This interruption reduces the production of proinflammatory cytokines, which play a central role in inflammatory and autoimmune processes. By suppressing the production of cytokines and the recruitment of immune cells, these inhibitors help to regulate the excessive immune response that occurs in diseases such as rheumatoid arthritis, cytokine release syndrome, and other inflammatory diseases. This targeted mechanism not only relieves inflammation but also protects tissue from damage caused by excessive immune activation. Monoclonal antibodies specifically targeting IL-6 or its receptor have been developed and approved for the treatment of diseases such as rheumatoid arthritis, juvenile idiopathic arthritis, and cytokine release syndrome associated with certain cancer immunotherapies. By influencing inflammation, activating immune cells, and supporting tissue repair, IL-6 plays a central role in the immune response. Dysregulation of IL-6 molecule can result in numerous health challenges, underscoring its significance in both research and therapeutic avenues.
IL-6 and Antigen-presenting Cells (APCs): A Classical Network of Immune Response
An essential element of the immune system that plays a role in both initiating and controlling immune responses is the APCs. They consist of B cells, macrophages, and dendritic cells (DCs). To trigger an immune response, APCs collect antigens, refine them, and pass them on to T cells and other immune cells.26, 27
Macrophages
Macrophages are crucial components in the human immune system, fulfilling multiple roles, such as ingesting and breaking down microorganisms, removing cellular debris and dead cells, and activating additional cells contributing to immune functions. Table 2 illustrates the specific activities of macrophages during SARS-CoV-2 infections. In response to various triggers like infections, tissue injuries, or the existence of other inflammatory agents, macrophages can generate IL-6. When macrophages release IL-6, it can increase inflammation by attracting and activating various immune cells such as neutrophils and T cells.38–40 Macrophages play a key role in regulating both normal and disease-related activities, and they monitor both innate and adaptive immune responses. SARS-CoV-2 manipulates monocytes and macrophages to suppress the host immune response, facilitating the progression of COVID-19. Systemic effects of the virus include acute respiratory distress syndrome (ARDS), widespread intravascular coagulation, edema, and pneumonia.41, 42
The Effector Functions of Macrophages in Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection.
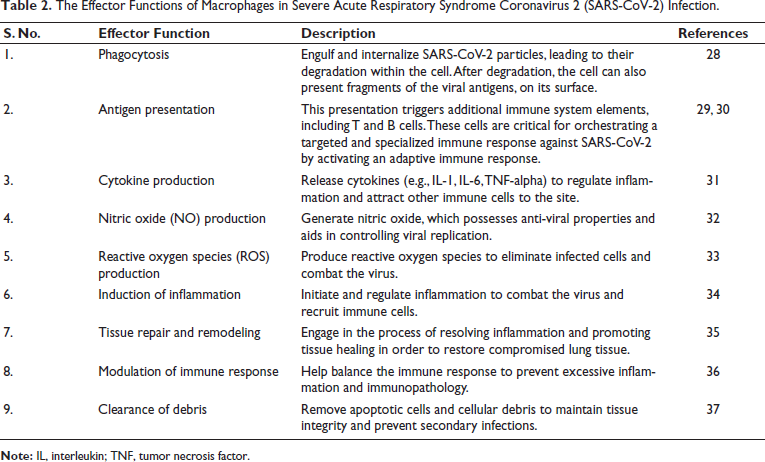
Macrophages, which serve as the first line of defense in our respiratory system, are equipped with detectors known as pattern recognition receptors (PRRs), including toll-like receptors (TLRs), which identify viral elements. When these macrophages encounter SARS-CoV-2, they can be activated. In severe cases of COVID-19, the immune response can become overly aggressive, leading to a condition known as cytokine release syndrome or cytokine storm, as described by Knoll et al. 43 This condition is characterized by a significant increase in proinflammatory cytokines, particularly IL-6. Macrophages and other immune cells can exacerbate this cytokine storm by producing high levels of IL-6. Consequently, IL-6 measurements are an indicator of the severity of COVID-19. As Vainer et al. found, higher levels of IL-6 in the blood correlate with poorer treatment outcomes and higher mortality rates. 44 Monitoring IL-6 levels is crucial for physicians to make informed treatment decisions, such as administering anti-inflammatory drugs like tocilizumab, which directly targets IL-6 signaling pathways.
Dendritic Cells (DCs)
DCs are characterized as specialized immune cells that are essential for recognizing and fighting external entities such as pathogens. Table 3 details their function in the immune system’s response to SARS-CoV-2. As experts in antigen handling, DCs seize and process antigens and introduce them into other components of the immune system to trigger and drive the immune response, as noted by Liu et al. Upon encountering pathogens or when exposed to inflammatory stimuli, DCs can release IL-6, an integral component of immune defense. 48
Roles of Dendritic Cells in Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection Effector Activities.
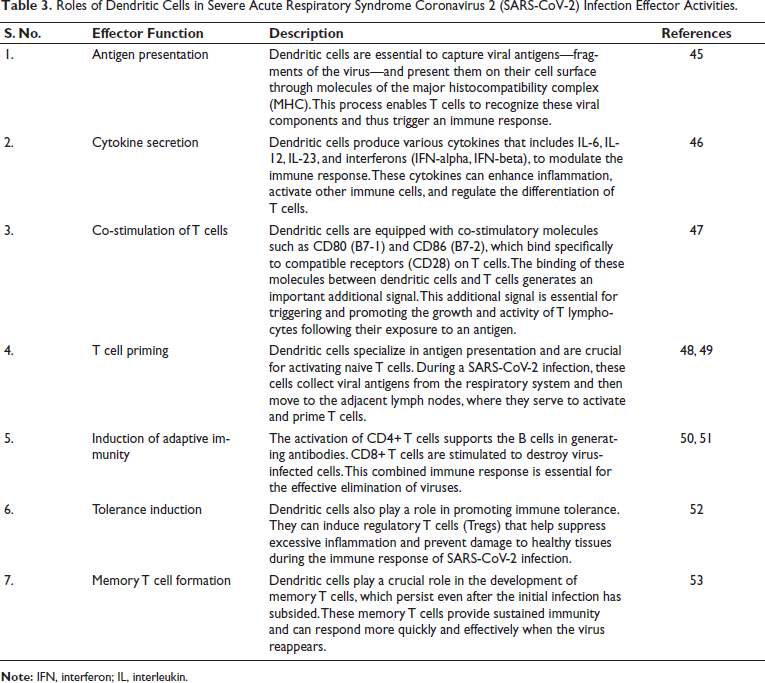
IL-6 is crucial for the mobilization and activation of additional immune cells, such as T and B cells, and for directing these cells to the required sites. In this context, as highlighted by Xu et al., the interaction between DCs and T cells can lead to the release of IL-6. 54 This cytokine, along with other cytokines, plays an important role in shaping the subsequent immune response and determines the outcome for the activated T cells. In addition, DCs undergo maturation and activation when they encounter antigens or inflammatory stimuli. During this phase, they can secrete IL-6, which acts as an autocrine or paracrine signal, supporting their own maturation process and stimulating nearby immune cells, as described by Jones et al. 55 DCs also play a role in immune regulation during SARS-CoV-2 infection. They can promote the development of Tregs, which help to suppress excessive immune responses and prevent tissue damage. This regulatory function is important for maintaining immune homeostasis and preventing immunopathology. DCs can secrete various cytokines, including IL-6, in response to SARS-CoV-2 infection. 56 IL-6, along with other cytokines, helps to orchestrate the immune response by promoting inflammation, activating immune cells, and regulating the balance between proinflammatory and anti-inflammatory processes.
B Cells
B cells are distinct leukocytes that are crucial to the immune response. As constituents of the adaptive immune system, these components possess specialized abilities to identify infections and orchestrate a customized response. Upon encountering exogenous pathogens such as bacteria or viruses, B cells undergo activation and subsequently generate antibodies. Table 4 depicts the effector functions of B cells in SARS-CoV-2 infection. Antibodies are specialized proteins that bind to specific antigens, the unique molecular recognition features that appear on the surface of invading pathogens. Each B cell carries receptors tailored to recognize a specific antigen. In response to various triggers such as infection, tissue injury, or signals that activate the immune system, B cells can produce IL-6. IL-6 has diverse effects on different immune cells and can influence both innate and adaptive immune responses. 64
The Effector Functions of B Cells in Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection.
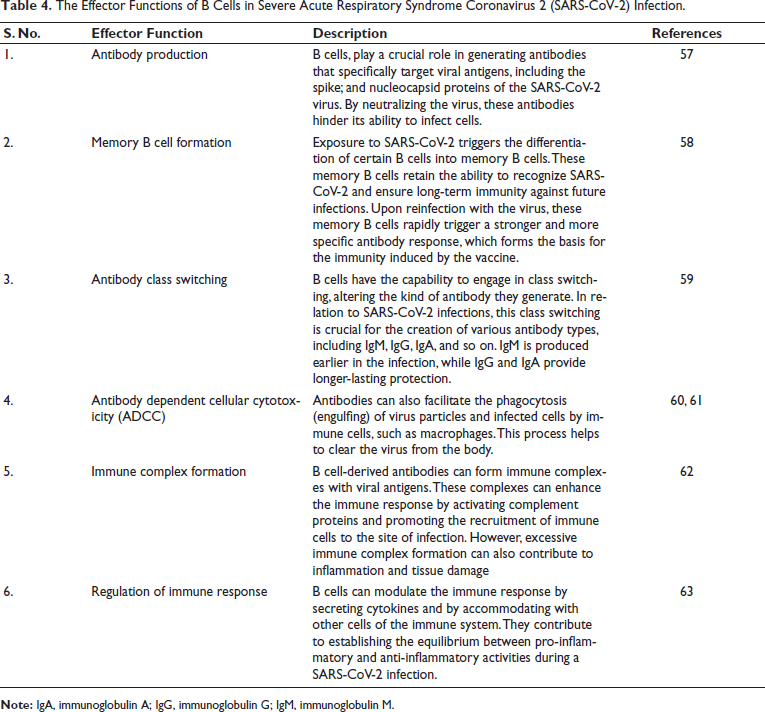
IL-6 can stimulate the growth and differentiation of B cells themselves, promoting their activation and antibody production. It can also contribute to the activation and differentiation of other immune cells, such as T cells and macrophages. Upon infection with SARS-CoV-2, B cells are activated to produce specific antibodies targeting the virus. These antibodies can bind to the spike protein of the virus, neutralizing it and preventing it from infecting host cells. B cells also contribute to the formation of long-term immune memory against SARS-CoV-2, which is important for protection against future infections. 65 IL-6 produced by various immune cells, including B cells, is mainly produced in response to SARS-CoV-2 infection. In the context of COVID-19, the disease caused by SARS-CoV-2, IL-6 levels can increase dramatically, leading to an overly aggressive inflammatory response often referred to as a cytokine storm.6, 20 While B cells contribute to fighting SARS-CoV-2 by producing specific antibodies, IL-6 plays a crucial role in controlling the inflammatory response. Uncontrolled IL-6 production can exacerbate the severity of COVID-19, which is why IL-6 inhibitors are being investigated as potential therapeutic options.
Therapeutic Antibodies in SARS COV-2 Viral Infection
Studies have investigated the use of antibodies specifically targeting IL-6 as a means of controlling severe cases of COVID-19 caused by the SARS-CoV-2 virus. IL-6, a cytokine with proinflammatory properties, plays a role in the functioning of the immune system and is thought to be associated with the increased inflammation observed in certain cases of COVID-19, as mentioned by Atal and Fatima. 66 Tocilizumab and sarilumab, originally developed for the treatment of rheumatoid arthritis, were used to treat severe cases of COVID-19 in the early stages of the pandemic. These antibodies work by suppressing the inflammatory response in COVID-19 by inhibiting the IL-6R.
Tocilizumab
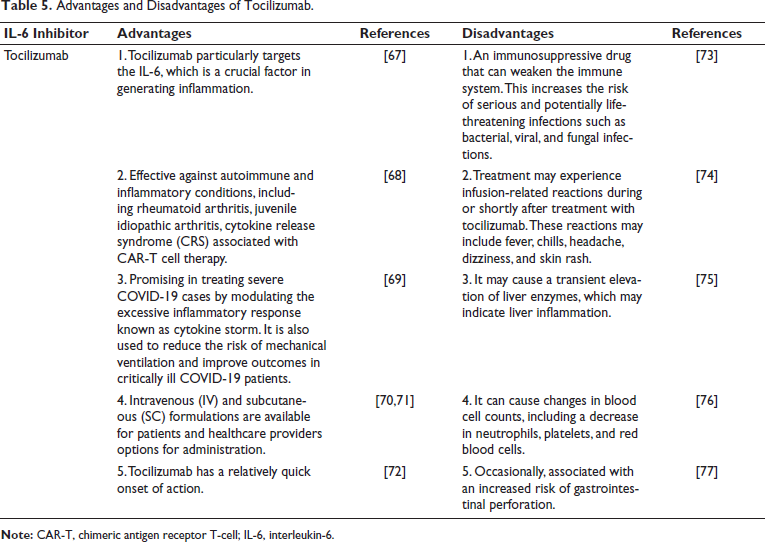
Tocilizumab is a monoclonal antibody (mAb) that suppresses the immune system and is used to treat rheumatoid arthritis. According to a new study, tocilizumab effectively reduces the inflammatory marker cytokine IL-6 and may reduce cytokine storm. The advantages and disadvantages of tocilizumab are represented in Table 5. Tocilizumab works by preventing the protein IL-6 from being activated. Tocilizumab reduces inflammation and symptoms associated with these diseases by reducing the function of IL-6, which promotes inflammation in the body. Tocilizumab has been used to treat certain SARS-CoV-2 patients, particularly those who have acquired severe respiratory problems and show signs of systemic inflammation. Tocilizumab works by inhibiting the function of IL-6, a cytokine that is often elevated in severe cases of COVID-19.78–80 Tocilizumab is used to treat COVID-19 assuming to lower IL-6 levels and modulate the immune response may help mitigate cytokine storm and reduce inflammation in the airways.
Advantages and Disadvantages of Tocilizumab.
Sarilumab
Sarilumab is a mAb used to treat certain inflammatory diseases by blocking the IL-6R. Sarilumab binds to the IL-6R, effectively interrupting the IL-6 signaling cascade, a cytokine known for its proinflammatory role and involvement in immune activities and helps to alleviate inflammation. 81 Sarilumab was originally developed for patients with moderate-to-severe rheumatoid arthritis (RA) who did not respond well to other options, such as disease-modifying anti-rheumatic drugs (DMARDs) or tumor necrosis factor (TNF) inhibitors. 82 Sarilumab has attracted considerable attention because of its prospective usefulness in treating the hyperinflammatory episodes commonly seen in patients with moderate-to-severe COVID-19 pneumonia. As a high-affinity antibody targeting the IL-6R, sarilumab is emerging as a potentially effective therapeutic strategy for these critical health scenarios. It is very important to note that; the IL-6 inhibitors target the IL-6 cytokine, which plays a role in the inflammatory response. Mutated lineages of the SARS-CoV-2 virus, such as the variants of concern, may exhibit different characteristics in terms of transmissibility, and severity of illness. The impact of IL-6 inhibitors on these mutated lineages would depend on various factors, including the specific variant’s interaction with the immune system, and the overall immune response of an individual. While IL-6 inhibitors may still be effective against these mutated lineages, their efficacy may be influenced by the specific mutations present in the variant. Ongoing research on clinical studies is being conducted to assess the effectiveness of IL-6 inhibitors against different variants of SARS-CoV-2. Regarding the mutated lineages of SARS-CoV-2, IL-6 inhibitor antibodies have primarily been used to treat severe cases of COVID-19 regardless of the specific lineage. However, it is important to note that the effectiveness of IL-6 inhibitors may vary depending on the specific mutation or variant of the virus. Certain variants of concern, such as the B.1.1.7 (alpha), B.1.351 (beta), P.1 (gamma), and B.1.617.2 (delta) variants, have been associated with increased transmissibility and, in some cases, increased disease severity. These variants have acquired multiple mutations, including those in the spike protein of the virus, which is the primary target of neutralizing antibodies like IL-6 inhibitors. 83
It is very important to know that IL-6 inhibitors target the release of IL-6, which plays a role in the inflammatory response. Mutant lineages of the SARS-CoV-2 virus, such as the variants of concern, may have different characteristics in terms of transmissibility and severity of disease. The effect of IL-6 inhibitors on these mutant lineages would depend on several factors, including the interaction of the specific variant with the immune system and the overall immune response of an individual. 84 While IL-6 inhibitors may also be effective against these mutant lines, their efficacy may be influenced by the specific mutations in the variant. Ongoing research and clinical investigations are being conducted to assess the effectiveness of IL-6 inhibitors in combating different forms of SARS-CoV-2. IL-6 inhibitor antibodies have predominantly been employed for the treatment of severe instances of COVID-19; however, their application is not restricted to a specific viral lineage. Nevertheless, it is crucial to underscore that the effectiveness of these inhibitors can fluctuate depending on the specific mutation or variation. The emergence of variants, namely, B.1.1.7 (alpha); B.1.351 (beta); P.1 (gamma); and B.1.617.2 (delta), has raised significant concerns due to their heightened transmissibility and potential to cause more severe illness under specific circumstances. 85 The observed variants have experienced numerous changes, including modifications in the spike protein, which serves as the primary target for neutralizing antibodies like IL-6 inhibitors.
Perspectives and Challenges on IL-6 Inhibitor Antibodies
IL-6 is a critical factor in the pathogenesis of various diseases, such as rheumatoid arthritis, systemic juvenile idiopathic arthritis, Castleman disease, and cytokine release syndrome (CRS).86, 87 Therapeutic antibodies, such as monoclonal antibodies and IL-6 inhibitor antibodies, are used to treat a variety of diseases, with a focus on autoimmune and inflammatory diseases. Pharmaceutical treatments capable of suppressing the action of IL-6 have been developed with the intention of mitigating inflammatory responses and regulating the activity of IL-6 in relation to these pathological situations. Although IL-6 inhibitors show promise in the therapeutic context, there are several obstacles and limitations to their use.
Management of Cytokine Release Syndrome (CRS)
Immunotherapies, such as chimeric antigen receptor T-cell (CAR-T) therapy, can induce CRS, a potentially fatal complication. IL-6 inhibitors have been used effectively to treat CRS by reducing the excessive immune response induced by CAR-T cells.88, 89
Potential in COVID-19 Treatment
The potential therapeutic application of IL-6 inhibitors in the treatment of severe instances of COVID-19 has been investigated in scientific research. The use of these inhibitors has the potential to impede the progression of respiratory problems and multiorgan failure by reducing the profound systemic inflammation usually known as a cytokine storm. However, clinical evidence for their efficacy against COVID-19 is still emerging. 90
Challenges and Future Directions
IL-6 inhibitors, like any immunomodulatory therapy, may have potential side effects and safety concerns. These may include an increased risk of infections, such as respiratory tract infections or opportunistic infections. Close monitoring of patients receiving IL-6 inhibitors is necessary to minimize adverse events. Response to IL-6 inhibitors may vary from patient to patient. Some patients may improve significantly, while others may not respond as well. Factors influencing differential response include disease heterogeneity, genetic variations, and other underlying comorbidities. Optimizing combination therapies, managing drug–drug interactions, and understanding the long-term effects of such combinations are the subject of ongoing research.
Combination therapies with IL-6 inhibitor antibodies are often used to improve the efficacy of treatment for various diseases. These inhibitors are often combined with other disease-modifying drugs or biologics to target multiple signaling pathways involved in the inflammatory process. For example, the combination of IL-6 inhibitors with corticosteroids has shown a synergistic effect in reducing mortality and shortening ICU stay in severe COVID-19 cases. However, this approach is associated with challenges, such as managing potential drug–drug interactions, tailoring treatments to the individual patient profile, and closely monitoring for overlapping side effects, including immunosuppression and an increased risk of opportunistic infections.
The immune response to SARS-CoV-2 is complex, with cytokine storms, which have a hyperinflammatory state, being a critical factor in severe cases. IL-6 inhibitors have been shown to be a targeted therapy to treat this immune dysregulation, particularly in patients with elevated IL-6 levels and signs of hyperinflammation. Early administration can prevent severe disease progression; however, not all patients benefit equally. Identifying the right candidates for this treatment remains a challenge as immune responses and disease progression vary between individuals. Accurate stratification of patients based on biomarkers is essential to ensure the effective and timely use of IL-6 inhibitors. Despite their potential, IL-6 inhibitors raise concerns about their long-term safety and efficacy. While they promise to reduce inflammation during acute disease, prolonged use could lead to adverse effects such as secondary infections or impaired tissue repair. In addition, new SARS-CoV-2 variants could compromise their efficacy, requiring constant evaluation and adaptation of these therapies. Research is increasingly focused on overcoming these challenges by exploring combination therapies, optimizing dosing regimens, and tailoring treatments to individual patient profiles through advances in personalized medicine. Innovative approaches to modulate IL-6 are also gaining attention, including small molecule inhibitors, nanomedicine delivery systems, ribonucleic acid (RNA)-based therapeutics, and gene editing technologies. These strategies aim to refine IL-6 targeting, improve the precision of drug delivery, and minimize side effects. In addition, biomarker research and genetic profiling pave the way for more personalized and effective treatments. By expanding our understanding of the role of IL-6 in inflammation and disease, these advances promise to improve COVID-19 outcomes and extend to other diseases where cytokine-driven immune responses occur.
Conclusion
The reported findings on IL-6 inhibitor antibodies in SARS-CoV-2 infections and their mutant lineages allow for important insights into the challenges, opportunities, and future prospects of using IL-6 inhibitors in treating COVID-19. IL-6 is a proinflammatory cytokine that is critical to the immune response. In severe cases of COVID-19, there is often an excessive release of IL-6, resulting in a hyperinflammatory state known as a cytokine storm. Targeting IL-6 with inhibitors has been explored as a potential therapeutic strategy. Clinical studies and practical applications reveal that IL-6 inhibitors, including tocilizumab and sarilumab, effectively reduce the severity and mortality rate of COVID-19 in certain patient groups, especially in patients with severe disease and high IL-6 levels. Although IL-6 inhibitor antibodies show promise in treating severe COVID-19 disease, particularly in patients with a hyperinflammatory response, several challenges and limitations remain to be overcome, and the impact of mutant lineages and variants on the efficacy of IL-6 inhibitors needs further investigation. Further studies and clinical trials are needed to fully assess the role of IL-6 inhibitors in treating COVID-19. This will ensure that they are used as effectively as possible in the future.
Footnotes
Abbreviations
Acknowledgments
Deanship of Scientific Research, Jazan University, Jazan, Kingdom of Saudi Arabia for the encouragement and support (Covid-19/16).
Author’s Contribution
MAB, GG, FYS, and AYS: Conceptualization and resources; SMS: Conceptualization, investigation, validation, and drafting of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This review was funded by the Deanship of Scientific Research, Jazan University, Jazan, Kingdom of Saudi Arabia, Project number Covid-19/16.
