Abstract
Background
Alzheimer’s disease (AD) is a complex neurological disorder characterized by neuronal loss and progressive cognitive decline. The disease progression is influenced by both environmental and genetic factors. Recent research highlights the significant role of vitamin D in the pathological progression of AD.
Objectives
This review aims to comprehensively analyze the interplay between vitamin D and Alzheimer’s disease, focusing on its molecular mechanisms, epidemiological evidence, and therapeutic implications.
Methodology
An extensive literature review was conducted to explore the molecular pathways by which vitamin D affects key pathological processes in AD, including amyloid-beta deposition, oxidative stress, neuronal inflammation, and tau phosphorylation. Epidemiological studies linking vitamin D status with AD prevalence and clinical outcomes were also analyzed, along with data from research trials investigating the efficacy of vitamin D supplementation in AD prevention and management.
Results
Vitamin D deficiency is associated with increased cognitive decline and heightened risk of developing AD. The review highlights the therapeutic potential of vitamin D supplementation in mitigating AD progression through its effects on pathological processes. However, challenges and controversies remain regarding the efficacy, optimal dosing regimens, and therapeutic strategies of vitamin D intake.
Conclusion
This review underscores the role of vitamin D as a modifiable factor and therapeutic target in Alzheimer’s disease. Further research is required to establish definitive dosing regimens and strategies to optimize the use of vitamin D in preventing and managing this debilitating neurological disorder.
Introduction
The condition known as Alzheimer’s is a neurological illness that exacerbates with time. This disease typically begins with subtle memory loss and progresses to confusion, disorientation, and difficulty in speaking, reasoning, and everyday tasks. Individuals may experience personality changes, inability to recognize their loved ones, and agitation as the disease progresses. 1 Alzheimer’s illness is the biggest cause of death among people aged 65 and older in the USA. Over 70% are at least 75 years old. Alzheimer’s disease (AD) is estimated to affect 70% of the 55 million people worldwide with dementia. Other risk factors include inheritance, ancestral history, alongside particular factors like poor exercise and diet, and medical conditions such as cardiovascular disease and diabetes. 2 Alzheimer’s is recognized by abnormal brain tangled and plaque-like deposits. Cell death and brain tissue loss can be caused by deposits that interfere with communication between nerve cells. There is no single test for Alzheimer’s. A thorough medical history, physical examination, neurological and cognitive tests, and sometimes brain scans can be used to rule out other possible causes of symptoms. 3 AD is currently incurable. The treatment aims to alleviate symptoms and slow disease progression. Cholinesterase inhibitors and memantine may be prescribed to alleviate cognitive symptoms and improve, as does quality of life. Non-drug interventions, like cognitive stimulation, social interaction, and physical exercises, are also vital to treatment. AD can be both psychologically and physically discerning for both patients and caregivers. 4 Caregivers play an important role in assisting, coordinating daily activities, and ensuring safety. Ongoing research aims to better understand the underlying mechanisms of AD, develop better diagnostic tools, identify new treatment targets, and, ultimately, discover a cure or effective disease-modifying therapies. Prevention strategies, such as lifestyle interventions, aim to reduce risk. 5
AD has an immense effect on relatives, caregivers, and the community as a whole, in addition to the individuals who have been diagnosed. It poses substantial economic and healthcare burdens and requires a comprehensive approach involving medical, social, and community support systems. Vitamin D was discovered as a vitamin in the early 1900s but is now understood to be a prohormone. Vitamin D has the unique property of being able to be produced by the body using sunlight. Vitamin D, also known as calciferol, belongs to the fat-soluble seco-sterol family. Vitamin D and vitamin D3 are the two most common types. Vitamin D2 (ergo-calciferol) is majorly produced by people and incorporated into food. Vitamin D3 (cholecalciferol) is formed in human skin from 7-dehydrocholesterol and can also be obtained through diet by eating animal-derived foods. 6 It is a lipid-soluble nutrient and is well-known for its vital role in regulating the immune system and bone health-like bodily functions. Vitamin D, perhaps best known for its association with calcium absorption, is important in maintaining optimal bone density and strength. 7 It helps in aiding the absorption of phosphorous and calcium, which are necessary minerals for bone mineral deposition. As a result, lack of vitamin D causes rickets in children and osteomalacia in adults, characterized by weak bones and an increased risk of fractures. Beyond its skeletal benefits, it has a significant impact on the immune system. It helps to regulate immune responses, affecting both innate and adaptive immunity. 8
According to studies, adequate vitamin D levels help to decrease the risk of autoimmune diseases, infectious illnesses, and certain cancers by modulating inflammatory processes and strengthening the body’s defense mechanisms. Also, evidence suggests that vitamin D influences various physiological processes, including muscle function, cardiovascular health, and mental well-being. It is linked to increased muscle strength and performance, potentially lowering the risk of falls and fractures, especially among old people. Moreover, lack of vitamin D is linked to an elevated risk of cardiovascular disease, depression, and cognitive decline, but more exploration is required to understand these associations clearly.9, 10
Pathological Progression of AD
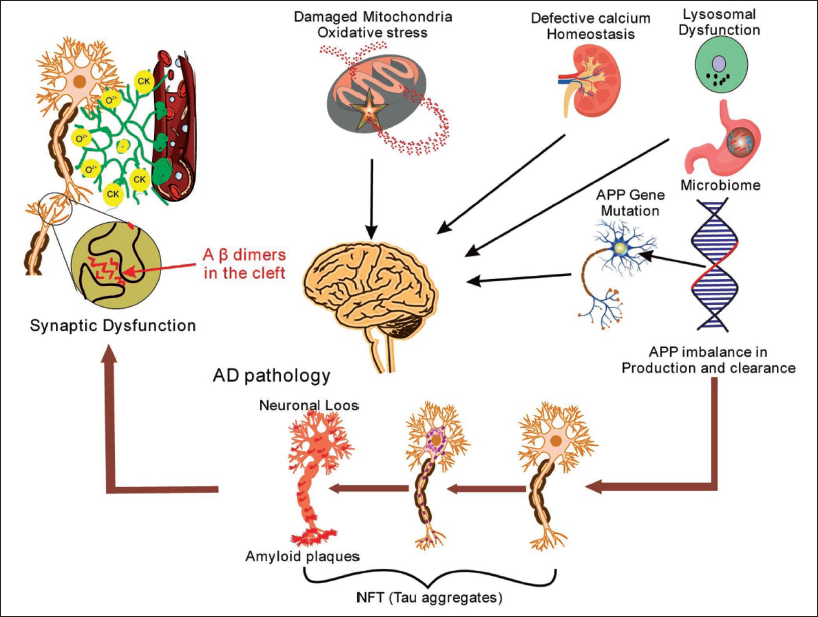
Alzheimer’s illness is an intricate neuro-degenerative illness marked by progressive cognitive impairment and memory loss. While the exact reason for Alzheimer’s remains unknown, investigators have identified several key factors that are linked to its pathogenesis. One of the hallmarks of this disease is the deposition of unnatural protein deposits in the brain. Tau tangles and amyloid-beta (Aβ) plaques are two of the most common protein deposits seen in the brains of Alzheimer’s patients. 11 Aβ plaques are formed when misfolded Aβ proteins accumulate, whereas tau tangles are caused by abnormal tau protein aggregation within neurons (Figure 1). 12 These protein clumps cause abnormal cell division, neuronal damage, and cellular death. Genetics also plays a vital part in the occurrence of AD.
Early-onset familial variants of the illness have been linked to gene mutations such as presenilin 1 (PSEN1), presenilin 2 (PSEN2), and amyloid precursor protein (APP). Increased production is the result of these alterations. 13
In addition to genetic factors, environmental and lifestyle influences also impact Alzheimer’s risk. Chronic inflammation, vascular disease, oxidative stress, and impaired glucose metabolism have all been implicated in disease progression. These factors can exacerbate protein aggregation, promote neuronal injury, and contribute to the breakdown of brain networks involved in memory and cognition. 14– 16 In addition, disruptions in neurotransmitter systems, particularly acetylcholine and glutamate, are observed in Alzheimer’s. Impairment in learning and memory are the causes of acetylcholine deficiency, while excessive glutamate signaling can lead to excitation toxicity and neuronal death.17–19 AD has a complex etiology that involves multiple causes, including genetic, environmental, and lifestyle factors. To create therapies and interventions that effectively delay or stop the progression of the disease, it is imperative to comprehend these mechanisms.
Overall Pathogenesis of Alzheimer’s Disease (AD).
Vitamin D Role in AD
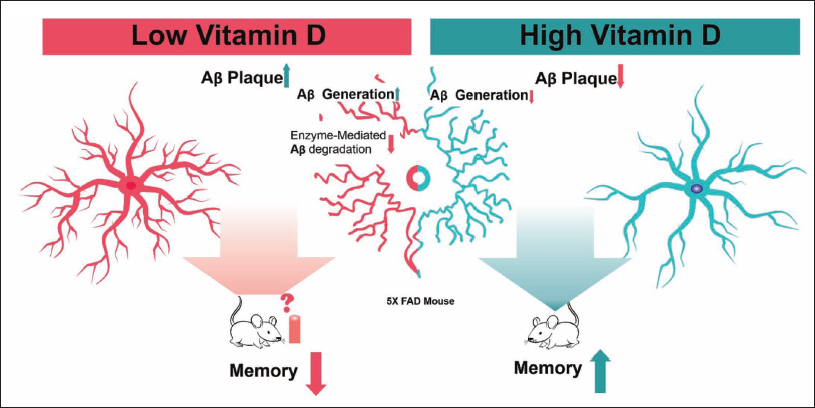
Vitamin D in AD has garnered significant attention in recent research. While the exact mechanisms are still being explored, here is a short overview: The neuroprotective effect shown by vitamin D may potentially reduce the risk of cognitive decline and AD development. It is believed to modulate various pathways involved in neuronal function and survival. 20 Vitamin D has anti-inflammatory properties that may help reduce neuroinflammation, an attribute of AD. Inflammation in the brain over time contributes to the onset of this disease, and vitamin D could help reduce this process. 21 Aβ plaques are the pathological feature of AD. Research suggests it can regulate Aβ production and clearance, potentially slowing disease progression. 22 Vitamin D receptors (VDRs) are frequently found throughout the brain, including areas controlling mood and cognition. It may influence neurotransmitter synthesis and function, potentially impacting cognitive function and lowering the risk of AD (Figure 2). 23 Vitamin D regulates calcium homeostasis, which is required for neuronal function. Calcium deregulation in the brain is linked to neurodegenerative illnesses such as Alzheimer’s. It also may help maintain optimal calcium levels, promoting neuronal health. 24 These mechanisms reveal a potential role for vitamin D in AD prevention and management.
Role of Vitamin D in Alzheimer’s Disease (AD).
Vitamin D Role in Aβ Accumulation
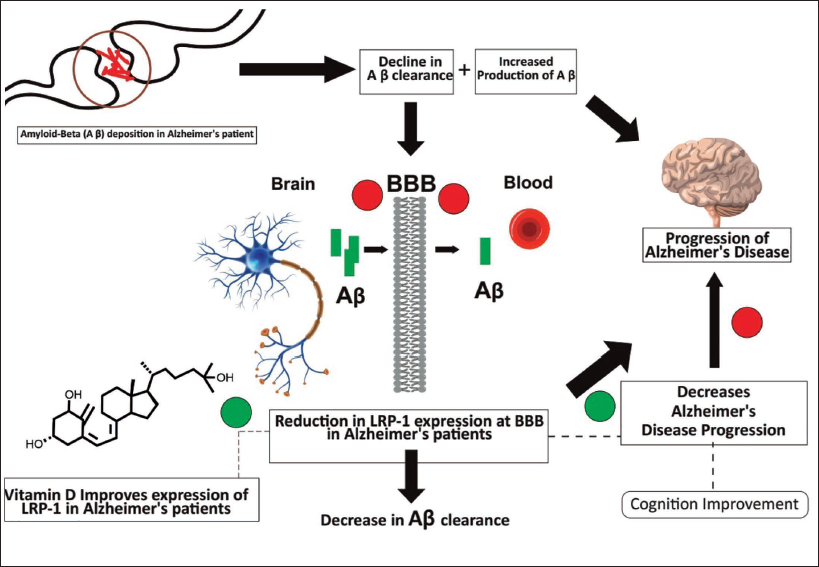
One emerging area of study is the potential impact on Aβ accumulation, a feature of Alzheimer’s pathology. Understanding the role of vitamin D in this study is critical, given the growing prevalence of AD and the lack of effective treatments. Aβ is a protein fragment accumulating abnormally in the brain, resulting in typical AD plaques. 25 These plaques impair normal neuronal function, contributing to cognitive decline and neurodegeneration. While the precise mechanisms underlying Aβ accumulation are unknown, resulting proof indicates that this vitamin may have a direct as well as indirect impact on this process. 26
First, vitamin D has been shown to impact gene expression in Aβ metabolism. According to research, VDRs exist in the brain, including regions affected by AD pathology. 27 Vitamin D can activate these receptors, regulating the gene expression in Aβ production, clearance, and degradation. For example, vitamin D has been shown to elevate the enzyme expression encompassed in Aβ degradation, promoting its clearance from the brain. 28 Also, it has antioxidant and anti-inflammation features, which may influence Aβ accumulation. Chronic inflammation and stress due to oxidation are linked to the pathogenesis of this disease and are shown to exacerbate Aβ deposition (Figure 3). 29 It is shown to reduce neuro-inflammation and oxidative damage by inhibiting the production of pro-inflammatory cytokines and reactive oxygen species. It may help to minimize Aβ accumulation in the brain by inhibiting these pathological processes. 30
Vitamin D also interacts with additional neurotrophic factors and signaling pathways involved in AD pathology. For example, Vitamin D can boost the expression of brain-derived neurotrophic factor (BDNF), a protein required for neuronal survival, synaptic plasticity, and memory function. In AD, BDNF signaling is deregulated, which is linked with synaptic dysfunction and cognitive impairment. It may improve neuronal health and resilience to Aβ toxicity by increasing BDNF expression and signaling.31, 32
Despite encouraging findings from preclinical studies and observational research connecting lower vitamin D to an elevated risk of AD, clinical investigations examining the therapeutic potential of vitamin D supplementation in AD have produced mixed results. These inconsistencies may be due to issues with study design, patient heterogeneity, and variability in vitamin D dosing and treatment duration. In addition, more study is required to know the complex interplay among vitamin D and other environmental and genetic factors that influence AD pathogenesis.33, 34
The Molecular Mechanism of Vitamin D in Alzheimer’s Disease (AD).
Vitamin D Receptor Gene Polymorphism and Possible Risk of AD
Alzheimer’s is an important community health challenge everywhere, characterized by continuous cognitive decline and neurodegeneration. While age remains the most prominent risk factor, emerging research implicates genetic factors in disease susceptibility. Among these, the role of VDR gene polymorphisms has garnered considerable attention.35, 36
Gene Polymorphism of Vitamin D Receptor
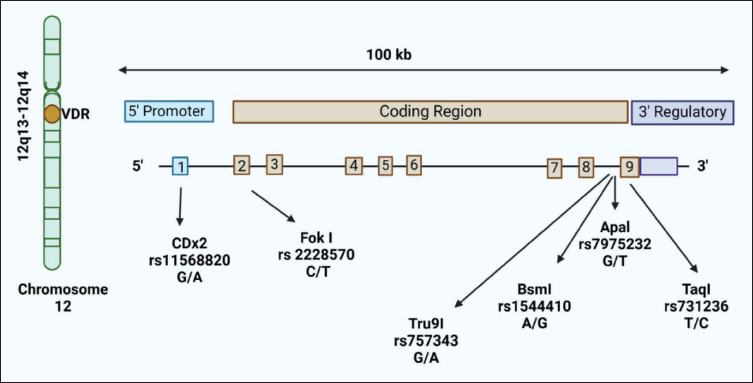
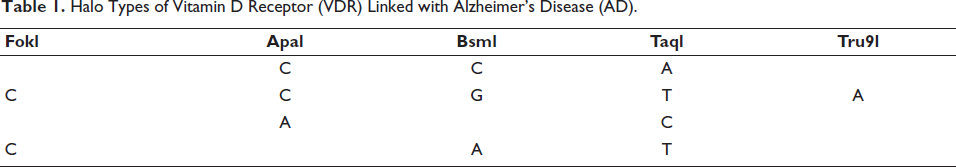
Vitamin D, frequently recognized for its part in bone strength, has garnered accelerating awareness for its potential effect on neurological disorders, including Alzheimer’s. The relationship between vitamin D and AD risk is very complex, influenced by various factors including genetics. One area of focus in understanding this relationship lies in the investigation of VDR polymorphism of genes and their molecular-level mechanisms in influencing AD risk.37–39 This gene, situated on chromosome 12q13.11, encodes the VDR, a basic hormone receptor important for mediating the biological effects of vitamin D. Polymorphisms within the VDR gene have been extensively prepared due to their potential influence on vitamin D absorption, cellular signaling, and expression of the gene (Figure 4 and Table 1).40, 41 This gene encodes a protein that functions as a receptor for calcitriol, the active form of vitamin D. Polymorphisms within this gene can bring about alterations in the makeup and function of the VDR protein, moving its capability to bind accompanying calcitriol and regulate gene expression. Several polymorphisms inside the VDR gene have been identified, with the most studied ones including FokI, BsmI, TaqI, and ApaI.42–44
Research suggests that these VDR gene polymorphisms may contribute to the pathogenesis of AD through multiple molecular mechanisms. One such mechanism involves the deregulation of calcium homeostasis. Vitamin D, through its interaction with the VDR, plays a vital role in calcium balance maintenance in the brain. Polymorphisms in this gene may disrupt this balance, leading to neuronal dysfunction and increased susceptibility to neurodegeneration, a hallmark of AD.45–47 VDR polymorphisms have been implicated in modulating inflammatory processes within the brain. Vitamin D exhibits anti-inflammatory properties and its interaction with the gene that regulates gene expression is involved in immune responses. Polymorphisms that alter gene function may impair this regulatory mechanism, promoting neuroinflammation and exacerbating neuronal damage observed in AD. Another molecular mechanism linking VDR gene polymorphisms to AD risk involves the regulation of Aβ metabolism. Aβ accumulation, due to abnormal processing and clearance, is the hallmark of AD pathology. Vitamin D is proved to influence the enzymatic expression involved in Aβ metabolism, such as APP processing enzymes. Polymorphisms in the gene VDR might disrupt this regulatory pathway, leading to enhanced Aβ production and deposition in the brain.48–50 Moreover, these gene polymorphisms may impact neuronal survival pathways implicated in AD pathogenesis. Vitamin D/VDR signaling has been shown to promote neuronal survival and protect against oxidative stress-induced damage. Polymorphisms that compromise VDR function may impair these protective mechanisms, rendering neurons more vulnerable to oxidative injury and apoptosis, contributing to AD progression. 51 VDR gene polymorphisms represent a potential genetic risk factor for AD, exerting their influence through diverse molecular mechanisms including deregulation of calcium homeostasis, modulation of inflammatory responses, alteration of Aβ metabolism, and impairment of neuronal survival pathways. Further elucidation in these pathways provides good directions into the progression of new and innovative therapeutic strategies targeting vitamin D/VDR signaling for the prevention and treatment of AD. 52
Structure of the vitamin D Receptor (VDR) Gene and Polymorphism Position Designed in Alzheimer’s Disease (AD).
Halo Types of Vitamin D Receptor (VDR) Linked with Alzheimer’s Disease (AD).
Future Directions
Future research on the role of vitamin D in AD should focus on clinical trials to better understand its therapeutic potential. Longitudinal studies could help clarify the relationship between vitamin D deficiency and AD progression, particularly in diverse populations. It is crucial to investigate VDR signaling pathways and their impact on neuroinflammation, synaptic function, and Aβ aggregation. Personalized approaches considering genetic variations, vitamin D status, and coexisting comorbidities may improve treatment outcomes. Further studies should also explore vitamin D’s interaction with other micronutrients and its role in brain aging to refine prevention strategies for AD.53, 54
Conclusion
Vitamin D plays a significant role in the pathophysiology of AD, influencing neuroinflammation, Aβ deposition, and cognitive decline. While evidence suggests that vitamin D deficiency may contribute to ad risk, the exact mechanisms remain complex and require further exploration. Current studies highlight the potential of vitamin D as a therapeutic strategy, but more rigorous clinical trials are needed to establish its efficacy. Understanding the interplay between vitamin D, genetic factors, and other biomarkers is essential for developing personalized interventions. Overall, vitamin D presents a promising area for prevention and treatment, warranting continued research and clinical evaluation.
Footnotes
Abbreviations
Acknowledgments
The authors extend their gratitude to the management of the School of Pharmaceutical Sciences, Sandip University, for providing the necessary facilities to conduct this work.
Authors’ Contributions
Conceptualization: N.B., M.G.B., A.P.G.; Methodology: S.M., P.K.K., N.B.; Data curation: A.D., S.N.: Formal analysis: N.B., M.G.B.; Writing – original draft: N.B., S.N., K.S.P.; Supervision: N.B., D.T.; Writing – review and editing: N.B., P.N.S.G.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
