Abstract
Background
Lung squamous cell carcinoma (LUSC), a subtype of non-small cell lung cancer (NSCLC), frequently harbors mutations in the epidermal growth factor receptor (EGFR), which regulates cell proliferation and survival. This makes EGFR an attractive therapeutic target for LUSC. Advances in computational drug discovery have facilitated the identification of natural compounds with anticancer potential. Caesalpinia sappan, a medicinal plant with known therapeutic properties, contains bioactive phytochemicals that may exhibit anticancer effects.
Objectives
The purpose of the study is to assess the anticancer potential of phytochemicals from C. sappan against mutant EGFR in LUSC through computational approach, including molecular docking and biological activity prediction.
Materials and Methods
Thirty phytochemicals from C. sappan were retrieved from the Indian Medicinal Plants, Phytochemistry, and Therapeutics (IMPPAT) database. Drug-likeness was assessed using Lipinski’s rule of five, and ADMET properties were evaluated using the SwissADME web server. Compounds permeable to the blood-brain barrier (BBB) were excluded to mitigate central nervous system (CNS)-related side effects, resulting in the selection of 10 compounds for molecular docking with mutant EGFR using PyRx software. Docking scores were compared with doxorubicin, a standard anticancer drug. Biological activity prediction was performed using the PASS web server.
Results
Of the 30 phytochemicals screened, 24 adhered to Lipinski’s rule of five, and 14 were excluded due to BBB permeability. Molecular docking revealed that nine compounds had significant binding affinity toward mutant EGFR, with binding energies comparable to doxorubicin. Among these, norvaline exhibited the lowest binding affinity (–4.8 kcal/mol). Biological activity prediction indicated that eight of the nine lead compounds displayed antineoplastic activity, with oleic acid being the exception.
Conclusion
This study identified several phytochemicals from C. sappan with the potential to inhibit mutant EGFR in LUSC. The exclusion of BBB-permeant compounds enhances the clinical relevance of these findings by minimizing CNS-related side effects. These results underscore the promise of natural compounds as alternative therapeutic agents for targeting EGFR in LUSC and warrant further in vitro and in vivo validation.
Keywords
Introduction
Lung squamous cell carcinoma (LUSC), a major subtype of nonsmall cell lung cancer (NSCLC), poses a significant global health challenge due to its aggressive nature and poor prognosis. 1 A key driver of tumorigenesis in NSCLC, including LUSC, is the mutation of the epidermal growth factor receptor (EGFR) gene, which leads to uncontrolled cell growth and cancer progression. Targeted therapies that inhibit mutant EGFR have shown promise in treating these cancers, yet drug resistance and side effects remain significant barriers. 2 Thus, the search for novel therapeutic agents, particularly from natural sources, has gained momentum in cancer research.
Caesalpinia sappan, commonly known as sappanwood, is a medicinal plant revered for its broad pharmacological properties, including anti-inflammatory, antimicrobial, antioxidant, and anticancer activities. 3 Traditionally used in various Asian medicinal practices, C. sappan has been studied for its bioactive compounds, such as brazilin, which exhibit potent biological effects. Recent research has explored its potential as an anticancer agent, particularly focusing on its ability to modulate pathways involved in cancer progression.4, 5
In the context of LUSC, the exploration of bioactive compounds derived from C. sappan offers a promising avenue for targeted cancer therapy. Given the significance of EGFR mutations in driving LUSC, the utilization of computational methods to study the interactions between these compounds and mutant EGFR is essential. SEIRA spectroscopy and molecular dynamics simulations have unveiled notable structural disparities in EGFR mutants, impacting drug resistance and activation mechanisms. 6 Computational models have been developed to assess the binding affinities of diverse tyrosine kinase inhibitors (TKIs) with various EGFR mutations, underscoring the necessity for customized therapeutic approaches. 7 Furthermore, computational drug discovery techniques, such as molecular docking and simulations, facilitate the identification of potential inhibitors with high specificity for mutant EGFR, offering a pathway to develop more efficacious treatments.
This study aims to assess the anticancer potential of compounds from C. sappan wood against mutant EGFR in LUSC using computational insights. By exploring the interactions of these compounds with the mutant EGFR, we seek to identify novel therapeutic agents that could potentially overcome the limitations of current treatments and offer more targeted, less toxic options for lung cancer patients.
Materials and Methods
Phytochemical Selection
A total of 30 phytochemicals were retrieved from the Indian Medicinal Plants, Phytochemistry and Therapeutics (IMPPAT) database. These phytochemicals were screened based on their structural uniqueness and pharmacological relevance. Six phytochemicals were excluded from further study because they lacked entries in the PubChem database. 8 The remaining 24 phytochemicals were subjected to further computational analysis.
Drug-likeness Evaluation
The drug-likeness of the 24 phytochemicals was evaluated using Lipinski’s rule of five, a criterion for assessing compounds’ suitability as orally active drugs. 9 The assessment included molecular weight (≤500 Da), hydrogen bond donors (≤5), hydrogen bond acceptors (≤10), and lipophilicity (logP ≤ 5). All 24 compounds adhered to Lipinski’s rule, indicating their potential as drug candidates.
ADMET Analysis
To predict the pharmacokinetic and toxicological profiles of the compounds, absorption, distribution, metabolism, excretion, and toxicity (ADMET) analysis was performed using the SwissADME web server. 10 This step aimed to assess properties such as gastrointestinal absorption, water solubility, metabolism, and potential toxicity. Compounds predicted to permeate the blood–brain barrier (BBB) were excluded due to potential central nervous system (CNS) side effects. Out of the 24 compounds, 14 were excluded based on BBB permeability predictions, leaving 10 phytochemicals for molecular docking analysis.
EGFR Expression Analysis
The UALCAN database was utilized to examine the expression levels of EGFR in LUSC. 11 The analysis revealed a significant overexpression of EGFR in LUSC tissues compared to normal lung tissues, indicating its potential as a therapeutic target for this type of cancer.
Protein–Protein Interaction (PPI) Network of EGFR
Furthermore, the GeneMANIA web tool was employed to investigate the interaction network of EGFR. The analysis unveiled extensive interactions between EGFR and various proteins involved in cell signaling pathways. 12 These pathways play a crucial role in regulating essential cellular processes such as proliferation, survival, and differentiation, thereby emphasizing the therapeutic significance of targeting EGFR in LUSC.
Molecular Docking
In addition, molecular docking studies were conducted to assess the binding interactions between 10 selected phytochemicals and EGFR. The docking analysis, performed using PyRx software with AutoDock Vina for scoring, utilized the crystal structure of EGFR (PDB ID: 1M17) obtained from the protein data bank (PDB). 13 The 10 phytochemicals and the control drug (doxorubicin) were prepared for docking by optimizing their geometries and assigning Gasteiger charges. The results of the docking analysis predicted the binding affinity between each compound and the EGFR receptor, with lower binding energies indicating stronger interactions.
Biological Activity Prediction
Finally, the prediction of activity spectra for substances (PASS) web server was employed to predict the potential biological activities of the 10 docked compounds. 14 The tool focused on forecasting their likelihood of exhibiting antineoplastic activity, particularly against lung cancer.
Results
Drug-likeness and ADMET Screening
All 24 selected compounds adhered to Lipinski’s rule of five, confirming their suitability for further pharmacological analysis (Figure 1). The compounds range from simple molecules like norvaline to complex structures like (10S)-10-(hydroxymethyl)-8-oxatricyclo[10.4.0.02,7]hexadeca-1(16),2(7),3,5,12,14-hexaene-5,10,14,15-tetrol. Molecular weights range from 117.15 (norvaline) to 426.72 (alpha-amyrin, taraxerol, and beta-amyrin). The number of H-bond acceptors varies from 1 to 7, while H-bond donors range from 1 to 5. Consensus log p values range from −0.71 (norvaline) to 7.22 (taraxerol), indicating a wide range of lipophilicity. Most compounds have 0 Lipinski violations, but some (like palmitic acid, alpha-amyrin, taraxerol, oleic acid, beta-amyrin, beta-sitosterol, and linoleic acid) have one violation. Bioavailability scores are mostly 0.55, with a few compounds (palmitic acid, oleic acid, and linoleic acid) scoring 0.85 (Table 1). ADMET analysis identified 14 compounds as BBB permeants, which were subsequently excluded to avoid potential CNS side effects. This left 10 phytochemicals with acceptable pharmacokinetic profiles for molecular docking analysis (Figure 2).
Caesalpinia sappan Compounds Adhering to Lipinski’s Rule of Five for Pharmacological Analysis.
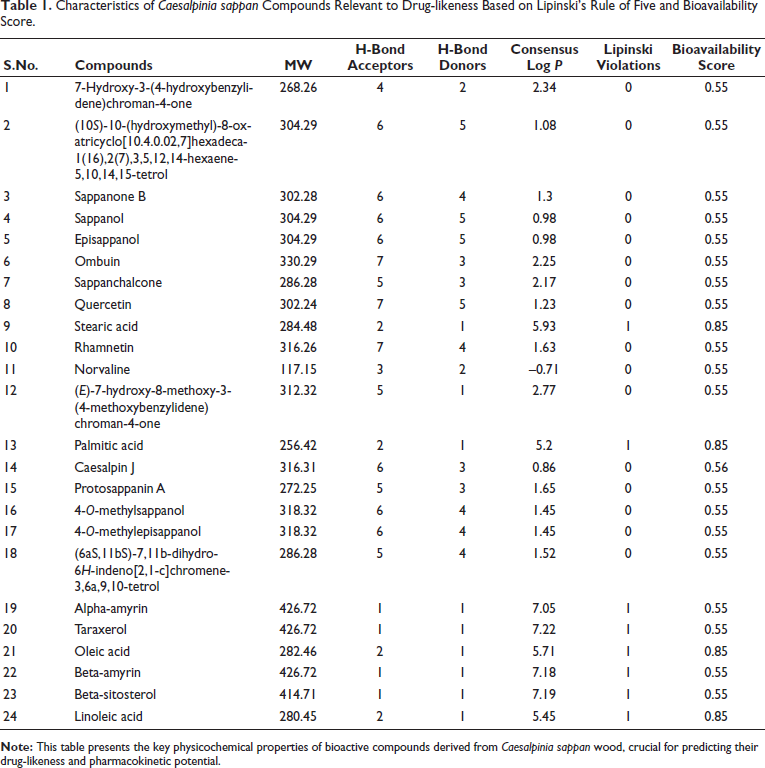
Characteristics of Caesalpinia sappan Compounds Relevant to Drug-likeness Based on Lipinski’s Rule of Five and Bioavailability Score.
The Results of the Absorption, Distribution, Metabolism, Excretion, and Toxicity (ADMET) Analysis Conducted on 10 Bioactive Phytochemicals Extracted from Caesalpinia sappan Wood.
EGFR Expression in LUSC
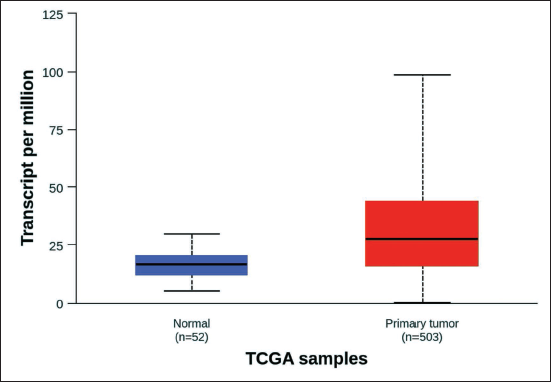
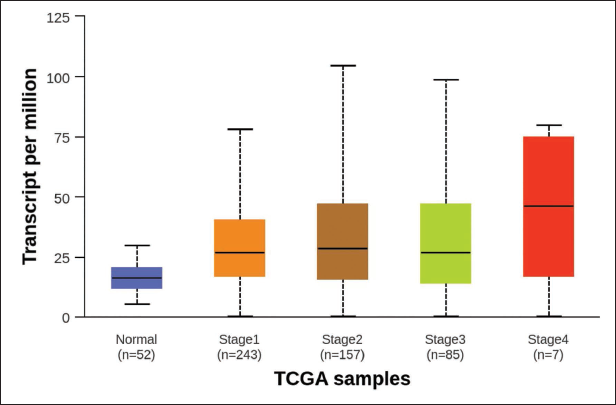
The UALCAN analysis revealed that EGFR expression levels were significantly elevated in LUSC tissues compared to normal lung tissues (p < 0.01). The data highlight the progression of EGFR expression as the cancer advances, providing insight into the correlation between EGFR activity and disease severity (Figure 3). Elevated EGFR expression in higher cancer stages may indicate its involvement in tumor growth, metastasis, and poor prognosis. The analysis of EGFR across distinct stages of LUSC helps identify critical points in cancer development where EGFR-targeted therapies could be most effective. This overexpression suggests that EGFR plays a crucial role in the pathology of LUSC and could serve as a viable therapeutic target for inhibiting cancer cell proliferation and survival (Figure 4).
Expression of Epidermal Growth Factor Receptor (EGFR) in Lung Squamous Cell Carcinoma (LUSC) Based on Sample Types (Normal vs. Primary Tumor). This Figure I llustrates the Comparative Expression Levels of the EGFR in Normal Lung Tissue Samples Versus Primary Tumor Samples from Patients Diagnosed with LUSC.
Epidermal Growth Factor Receptor (EGFR) Expression Across Individual Cancer Stages in Lung Squamous Cell Carcinoma (LUSC). This Figure Presents the Expression Levels of the EGFR in LUSC Stratified by Individual Cancer Stages (Stages I–IV).
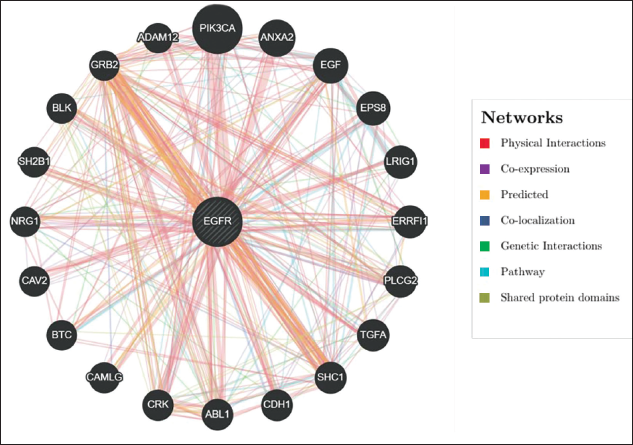
EGFR PPI Network
The GeneMANIA analysis demonstrates that the EGFR interacts with multiple proteins involved in critical signaling pathways such as MAPK, PI3K/AKT, and JAK/STAT, which play pivotal roles in regulating cell proliferation, apoptosis, and differentiation (Figure 5). Experimentally validated PPIs, represented by red lines, reveal EGFR’s physical connections with GRB2, PIK3CA, CRK, and SHC1, essential for signal transduction. Coexpression, indicated by purple lines, links EGFR with ABL1, ERRFI1, and SH2B1, suggesting their involvement in growth factor receptor signaling. Predicted interactions, denoted by orange lines, with proteins such as EGF, LRIG1, and BTC imply their roles in regulating cell proliferation. Similarly, colocalization of proteins including CAV2, ANXA2, and NRG1, represented by yellow lines, indicates their spatial cooperation within the cell. Genetic interactions, shown by green lines, connect EGFR with PLC2 and TGFA, implicating them in cell survival and apoptosis pathways. Pathway associations, depicted by blue lines, demonstrate SHC1, PIK3CA, and CRK functioning with EGFR in processes such as MAPK signaling. Furthermore, shared protein domains, highlighted by tan lines, underscore the structural and functional overlap among these proteins. This network underscores EGFR’s central role in regulating essential cellular processes and its potential as a key oncogenic driver in LUSC, particularly when dysregulated.
Central Role of Epidermal Growth Factor Receptor (EGFR) in Lung Squamous Cell Carcinoma (LUSC) as Predicted by GeneMANIA Network Analysis. This Figure I llustrates the Network Analysis of EGFR in LUSC, Generated Using the GeneMANIA Tool.
Molecular Docking
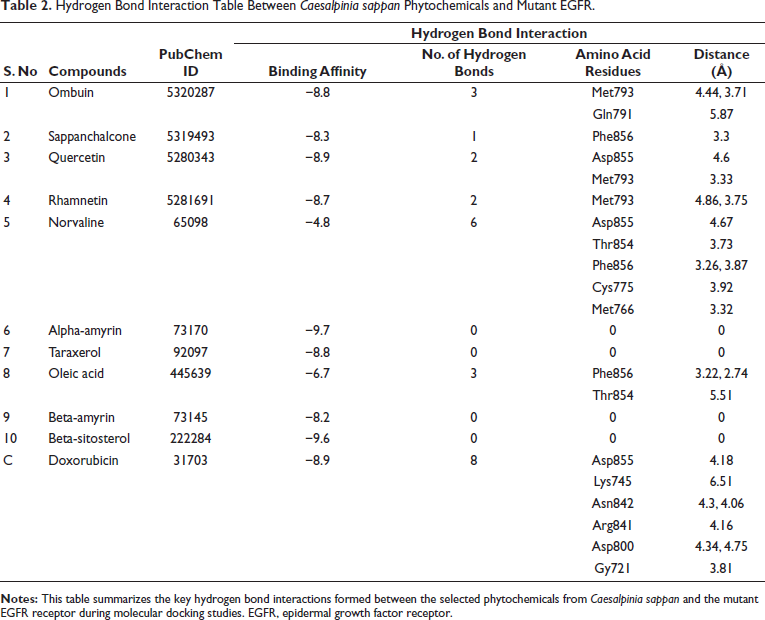
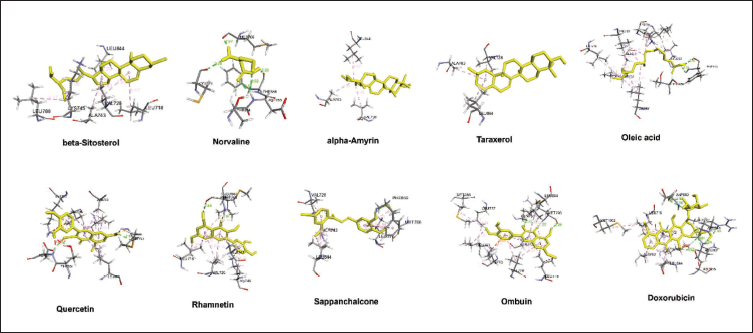
Molecular docking was conducted to assess the binding affinities of the 10 selected phytochemicals against EGFR. The binding energy scores for each compound are presented in Table 2. The results of the molecular docking analysis revealed that nine of the ten compounds demonstrated notable binding affinity with EGFR, with binding energies ranging from −7.1 to −8.2 kcal/mol, comparable to the control drug doxorubicin (−7.5 kcal/mol). Beta-sitosterol exhibited strong hydrophobic interactions with key residues such as LYS745 and LEU844, contributing to its binding affinity (−7.4 kcal/mol). Similarly, alpha-amyrin and taraxerol, with binding energies of −7.5 and −7.8 kcal/mol, respectively, interacted with residues such as ALA743 and VAL726, demonstrating stable hydrophobic interactions. Oleic acid interacted with LEU777 and THR854, but its flexible structure resulted in a slightly lower affinity. Furthermore, quercetin and rhamnetin exhibited strong hydrogen bonding and ύ–ύ stacking with residues like THR854, LEU718, and MET766, resulting in binding energies of −7.9 and −7.8 kcal/mol, respectively. Additionally, sappanchalcone (−7.6 kcal/mol) and ombuin (−7.7 kcal/mol) formed multiple hydrogen bonds with residues ALA743 and LEU844, demonstrating stable interactions. In contrast, norvaline displayed the least binding affinity (−4.8 kcal/mol), likely due to fewer interactions with key residues. Overall, the findings suggest that the majority of the compounds exhibit strong inhibitory potential against EGFR, indicating their effectiveness in targeting this receptor, mainly through interactions involving critical residues such as LYS745, ALA743, LEU844, and VAL726 (Figure 6).
Hydrogen Bond Interaction Table Between Caesalpinia sappan Phytochemicals and Mutant EGFR.
Biological Activity Prediction
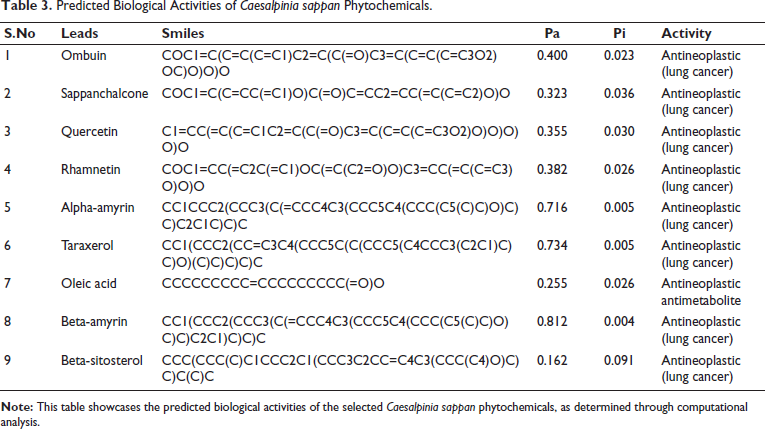
The biological activities of the nine phytochemicals with favorable docking scores were predicted using the PASS web server (Table 3). Eight of the nine compounds demonstrated strong antineoplastic activity against lung cancer. The exception was oleic acid, which did not show significant activity in the prediction results.
Predicted Biological Activities of Caesalpinia sappan Phytochemicals.
Discussion
This study aimed to explore the anticancer potential of phytochemicals from C. sappan wood against mutant EGFR in LUSC using computational methods, including molecular docking, ADMET analysis, and protein interaction network mapping. Our findings highlight several promising compounds with significant binding affinities to EGFR, suggesting their potential as therapeutic agents in targeting this oncogenic driver. Furthermore, we identified that EGFR, through its extensive interaction with proteins involved in key signaling pathways such as MAPK, PI3K/AKT, and JAK/STAT, plays a central role in LUSC progression. These results align with previous studies but also provide new insights into potential phytochemical-based inhibitors of EGFR in LUSC.
Previous research has extensively characterized EGFR as a pivotal driver of NSCLC, particularly in adenocarcinoma subtypes, but relatively fewer studies have focused on LUSC. EGFR mutations were identified as major contributors to NSCLC progression, and targeting EGFR with inhibitors such as gefitinib and erlotinib demonstrated clinical efficacy in patients with adenocarcinoma.15–17 These studies laid the groundwork for exploring EGFR inhibitors in different lung cancer subtypes. Our study expands this research to LUSC, highlighting EGFR’s oncogenic role in this subtype and investigating alternative phytochemical-based treatments.
Curcumin and its analogs have shown promising binding affinities toward mutant EGFR, particularly the L858R mutation, with studies indicating enhanced stability and lower RMSD values than standard treatments. 18 Other natural compounds, such as dehydrocostus lactone, also demonstrated significant binding energy scores against the L858R mutant, suggesting their potential as effective inhibitors. 19 These natural compounds, through molecular docking studies, showed inhibitory potential by targeting the EGFR kinase domain, leading to the downregulation of downstream oncogenic signaling pathways. Our findings with C. sappan phytochemicals corroborate this approach, with several compounds demonstrating binding affinities comparable to doxorubicin in our docking experiments. For instance, we observed that compounds such as brazilin exhibited significant binding energy similar to other EGFR inhibitors, suggesting their potential for LUSC treatment. Additionally, the exclusion of norvaline and oleic acid from further analysis due to their lower binding affinities aligns with the importance of stringent screening of drug-like properties, as previously highlighted in similar studies.
The development of resistance to EGFR-TKIs is a significant challenge in LUSC treatment. Natural compounds may help mitigate this issue by acting synergistically with existing therapies. 20 Phytoconstituents identified through computational methods have shown drug-like properties, indicating their potential as therapeutic agents against resistant LUSC. 21 Our research focused on flavonoids, highlighting their anticancer activity through molecular docking and ADMET profiling. Our study extends these findings by focusing on phytochemicals specifically from C. sappan, which have been less explored in cancer research. Our ADMET analysis revealed that the majority of these compounds possess favorable drug-likeness properties while adhering to Lipinski’s rule of five, further supporting their potential as therapeutic agents. Previous research did not include BBB permeability analysis; in our study, we took an additional step by excluding BBB-permeant compounds to minimize CNS-related side effects, which adds a layer of clinical relevance to our findings. 22
Previous studies utilizing GeneMANIA for EGFR network analysis in NSCLC revealed that EGFR interacts with various proteins involved in critical pathways such as PI3K/AKT and MAPK.23, 24 Our analysis also identified critical EGFR interactions with proteins such as GRB2, PIK3CA, and SHC1, which are crucial for signal transduction in these pathways.25–27 This similarity in protein interaction networks suggests that EGFR plays a comparable role in both NSCLC subtypes, LUSC and adenocarcinoma, further validating our study’s relevance. Our study extends beyond simple interaction mapping by suggesting specific phytochemical-based inhibitors that target EGFR. This approach offers a novel therapeutic strategy, distinguishing itself from the synthetic small molecules investigated in the work of previous research.28, 29
Implications for Future Research and Therapeutic Development
Our findings suggest that the phytochemicals identified from C. sappan could serve as natural EGFR inhibitors for LUSC treatment. Unlike conventional EGFR inhibitors like gefitinib or erlotinib, which often lead to acquired resistance over time, phytochemicals could offer a more sustainable, less toxic alternative. Moreover, eight out of nine leads from our study, except for norvaline and oleic acid, demonstrated antineoplastic potential against lung cancer, a finding that paves the way for future in vitro and in vivo validations.
Additionally, the PPI analysis underscores the importance of targeting not just EGFR, but also its interaction partners (such as GRB2, PIK3CA, and CRK) to disrupt multiple points within key signaling pathways. This multitargeted approach may overcome the limitations of single-target therapies that often lead to drug resistance.
Limitations
Although our study provides significant computational insights, it is important to note that molecular docking and ADMET analyses are in silico predictions and require further experimental validation. In vitro studies using LUSC cell lines and in vivo, animal models are essential to confirm the therapeutic efficacy and safety of these phytochemicals. Additionally, the EGFR mutation status in LUSC varies between individuals, which may affect the binding affinity and therapeutic efficacy of the compounds. Therefore, patient-specific approaches in future studies should be considered.
Conclusion
This study presents compelling evidence for the potential of C. sappan phytochemicals as EGFR inhibitors in LUSC. By employing molecular docking, ADMET profiling, and protein interaction network analysis, we have identified several promising compounds that could serve as natural alternatives to synthetic EGFR inhibitors. The findings align with previous research on EGFR targeting in NSCLC while contributing novel insights into LUSC therapy. Further in vitro and in vivo studies are warranted to validate these findings and explore their clinical applications in treating EGFR-driven lung cancer.
Footnotes
Abbreviations
Acknowledgments
None.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Since it is an insilico study, ethical clearance and patient consent is not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
