Abstract
Background
Bleaching can enhance the appearance of stained teeth while maintaining their structural integrity. The antioxidant properties of green tea and pine bark extracts related to the effect on dental hard tissues have not been updated.
Objectives
This in vitro study aims to evaluate the influence of herbal antioxidants on the push-out bond strength of resin-based composite materials to dentin following intracoronal bleaching.
Materials and Methods
In this in vitro study, 60 human mandibular premolar teeth were randomly divided into six groups as follows (n = 10): GI (control), no bleaching; GII, bleaching with 35% hydrogen peroxide (HP) and sodium perborate; GI, bleaching with 35% HP and sodium perborate bleached and restored after 14 days; GIV, bleaching with 35% HP and sodium perborate followed by 10% sodium ascorbate; GV, bleaching with 35% HP and sodium perborate followed by green tea. GVI, bleaching with 35% HP and sodium perborate, followed by pine bark. The specimens were bonded to composite in all groups. The push-out bond strength of specimens was measured in megapascals (MPa). Data were entered into an Excel sheet and analyzed using one-way analysis of variance (ANOVA) (p = 0.05).
Results
The unbleached teeth (GI) demonstrated the highest shear bond strength, followed by the bleached teeth treated with a 10% pine bark extract (GVI) antioxidant. The peak stress values were significantly higher in GI (control) and GVI, followed by other groups such as GIV, GIII, GV, and GII.
Conclusion
All the antioxidants used in this study were equally effective in neutralizing the adverse effects of HP on the bonding ability of immediately restored teeth. Group VI displayed the peak load, followed by Group IV and Group V, and successfully restored the weak bond strength of bleached dentin; this can be inferred from the current in vitro study.
Introduction
Demand for esthetics in dental restorations has augmented dentistry. 1 Patients who desire a happy smile no longer accept discolored teeth that are not aesthetic. Since the cosmetic elegance of anterior teeth is crucial in contemporary dentistry,1, 2 discolored anterior teeth are frequently seen as an aesthetic denigration. 3 Numerous studies have shown that the color of the anterior teeth determines patient satisfaction.3–5 A complex combination of factors causes tooth stains. Extrinsic, intrinsic, or a combination of the two are the several categories for discolorations.6, 7 Tooth bleaching has seen increased demand because of its coherence, safety, cost efficiency, and minimally invasive method of treating such discolorations.
Along with bleaching treatments, combination therapy is believed to be effective. 8 Composite resin or resin-modified glass ionomer cement (RMGIC) can be employed to successfully restore the defects brought on by damage to the enamel integrity. 9 Bleaching can restore the aesthetics of stained teeth while preserving the tooth’s structural integrity. 10
A minimum 3-week delay must elapse before the post-bleaching composite restoration, as bleached with hydrogen peroxide, the calcium and phosphate levels of the enamel and dentin significantly decrease, and the most superficial enamel crystallites undergo morphological changes. 11 Numerous studies have found that adhesive restorations like composite resin are prevented from polymerizing because bleached teeth still contain residual peroxide and/or oxygen radicals.12–14 A primary concern following bleaching is a reduced bond strength between composite resin and enamel, particularly when bonding is done quickly after bleaching. 15 Whitening agents often reduce the composite’s micro tensile bond strength (TBS) to the enamel when bonding occurs immediately after bleaching.12, 13, 15, 16
According to various studies, antioxidants are safe to use in bleaching and can increase bond strength immediately without significantly altering it. The bond strength of a composite resin to bleached enamel has been successfully increased by adding both organic and synthetic antioxidants.17–20 Delaying the bonding time is time-consuming to strengthen the binding between resin and bleached enamel.
Various antioxidants have been utilized, including sodium ascorbate (SA), herbal extracts like green tea extract, pine bark extract, and vitamin C. These antioxidants decrease the redox potential and effectively absorb free oxygen radicals.14, 16 Numerous studies have demonstrated that herbal antioxidants improve the binding strength of composite to bleached enamel. However, a few researchers have reported varied efficacy values at various application times.17–20 Research has primarily focused on enamel, and there are not many studies on dentin. To overcome this lack of literature, the antioxidant properties of green tea and pine bark extracts related to their effect on the dental hard tissues, that is, dentin, were investigated in this study. Therefore, this study aims to investigate the antioxidant characteristics of various herbal antioxidants, such as green tea, pine bark, and SA, and their impact on dental hard tissues. The secondary aim is to evaluate the push-out bond strength of resin-based composite to dentin after the intracoronal bleaching procedure.
Materials and Methods
The institutional ethical committee approved the in vitro study protocol (No. JSSDCH IEC: 44/2022). A total of 60 extracted single-rooted mandibular premolar samples were collected and cleaned using an ultrasonic scaler and were later preserved in an antiseptic medium (deionized water with 0.1% thymol) until their subsequent use (Figure 1). Selection criteria included teeth free from caries or fractures. Endodontic therapy involved 3 × 3 mm access openings, gutta-percha obturation, and sealing with zinc phosphate cement.
Sixty Healthy Human Mandibular Premolar Teeth Samples.
Sample Size
The sample size for this study is 53. However, it has been rounded to 60 samples to account for an effect size of 0.5, a significance level of 0.05, and a power of 0.80.
Preparation of Solutions
Ten percent solutions of green tea extract, pine bark extract (Shipra Organic Ltd), and SA (OXFORD LAB FINE CHEM LLP) were prepared by dissolving 10 g of each extract in 100 mL distilled water (Figure 2).
Herbal Extract of Antioxidants which Include 10% Sodium Ascorbate Solution, 10% Green Tea Extract Solution, and 10% Pine Bark Extract Solution.

Bleaching Procedure
Teeth were divided into six groups, each receiving 0.1 mL of 35% hydrogen peroxide and sodium perborate (walking bleach) in the exposed dentin (pulp chamber), temporarily sealed with zinc phosphate cement, and incubated at 37°C with 100% humidity for five days. The solutions were prepared fresh and stored in amber-colored bottles.
Antioxidant Application
Post-bleaching, the exposed dentin surface of the pulp chamber was treated with 0.1 mL of 10% SA, green tea extract, and pine bark extract solutions, respectively, for 30 s, then rinsed with deionized water in Group IV–VI teeth.
Restorative Procedures
Access cavities were etched with 37% phosphoric acid, later adhesive bonded (Tetric N Bond), and the composite Tetric N Ceram (Ivoclar Vivadent) was cured with light emitting diode (LED) light. Samples were stored in deionized water at 37°C for 24 h, decoronated at the cementoenamel junction, and standardized discs were prepared for push-out testing. A custom metal jig and a universal testing machine with a 0.5 mm/min speed were used to measure push-out bond strength in megapascals (MPa) (Figure 3) (Table 1).
Universal Testing Machine and Samples Prepared for Push-out Test.

The Sample Procedure and Grouping.

Failure modes were evaluated with a stereomicroscope at 25× magnification, identifying cohesive, adhesive, or combined failures. All the data were entered into the Excel sheet, and statistical analysis was performed with Statistical Package for the Social Sciences (SPSS) software version 20.0 using analysis of variance (ANOVA), with significance set at p ≤ 0.05.
Results
The analysis of push-out bond strength across different study groups revealed significant variations. Group I (control) had the highest mean bond strength at 16.91 MPa with a standard deviation of 2.70, demonstrating strong bond performance. Group II, which had no bleaching or antioxidant treatment, showed the lowest mean bond strength of 0.76 MPa. With bleaching but no antioxidants and delayed composite application, Group III had a mean of 4.92 MPa. Groups IV–VI, which received different antioxidants (10% SA, green tea extract, and pine bark extract, respectively) and immediate composite application, displayed improved bond strengths with means of 10.18, 4.61, and 11.20 MPa, respectively. ANOVA results indicated statistically significant differences across the groups (p value = 0.000*), underscoring the impact of various treatments on bond strength.
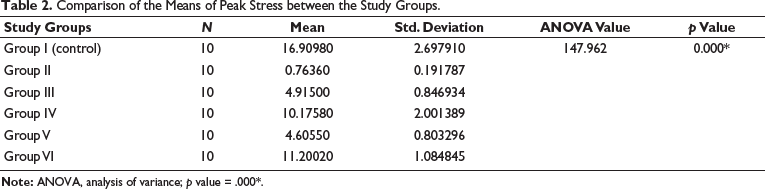
The peak stress values were observed to be significantly higher in group I (control group) (16.91 ± 2.7) and group VI (11.20 ± 1.08), followed by other groups such as group IV (10.17 ± 2.00), group III (4.91 ± 0.84) group V (4.61 ± 0.80) and group II (0.76 ± 0.19) as shown in Table 2.
Comparison of the Means of Peak Stress between the Study Groups.
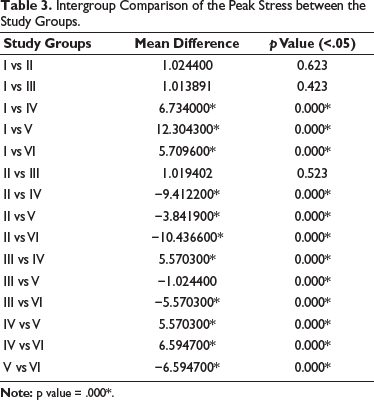
The intercomparisons between most groups show significant differences in peak stress, as indicated by the p values being less than 0.05, suggesting that the differences are statistically significant except for the groups that do not use antioxidants, as shown in Table 3.
Intergroup Comparison of the Peak Stress between the Study Groups.
Following the push-out test, the distribution of failure modes shows that 12 adhesive failures were the most common, cohesive failures were observed in 11 samples, and mixed failures were 9 out of 60 samples (Table 4).
Distribution of Failure Modes Following the Push-out Test.

Discussion
The initial application of definitive treatment is hampered by the surface oxygen or peroxide residues on a resin-based restoration-like composite. This makes it difficult to treat teeth that have been bleached and prevents adhesive resin from fully polymerizing. 21 Dishman et al. 21 hypothesized that oxygen is available in significant amounts in the dentin and between the enamel prisms due to the dissociation of the bleaching agent. 21 Free radicals from the dentin and dentinal fluid can either be eliminated via the pulpal microcirculation or eventually released through surface diffusion. 22 Despite past investigations suggesting that eliminating residual peroxide breakdown product is time-dependent, there has yet to be a consensus on the required waiting period. 23 The finest microscopic analysis revealed that, when rapidly restored, the resin tags in bleached enamel were small, dense, weakly defined, structurally nonexistent, or altogether gone. 24
It is suggested that bonding be postponed for 1–3 weeks following the bleaching procedure. This, however, decreases the likelihood of a spontaneous reestablishment of other aesthetic procedures. 15 Applying antioxidants can stop the peroxide ion inclusion process. 25 However, by delaying the composite restoration for 2 weeks after the bleaching process, it would be possible to ensure that there was no loss of bond strength. 26 However, not all patients desire to wait for so long. Therefore, using antioxidants, such as herbal antioxidants, is a proven method to instantly strengthen the link between composite and bleached enamel. 27 A quick composite restoration following nonvital bleaching is made possible, according to various published literature findings, by administering 10% (SA) antioxidant on an intracoronally “bleached tooth.” 28 However, SA is mutagenic, as shown by mammalian somatic cells. According to studies, green tea catechins have far greater antioxidant activity than vitamins C and E. 29 The primary constituent of pine bark extract is oligomeric-proanthocyanidins, a polyphenolic bioflavonoid with antioxidant and free radical scavenging activities that may be found in fruits and vegetables. In addition to the aforementioned effects, they have antibacterial, antiviral, anti-inflammatory, antiallergic, anticarcinogenic, and vasodilatory properties. 30 According to in vitro research, 5% pine bark extract can effectively counteract the damaging effects of free radicals on bleached enamel. 17 Among the groups treated with pine bark (10%) and SA (10%), grape seed extract (10%), pine bark showed better binding strength, according to Subramonian et al. 6 Our study’s results are consistent with previous research that suggested antioxidant materials could successfully neutralize the adverse effects of bleaching agents on the microleakage of composite restorations. According to the study’s findings, composite restorations that were bonded to bleached samples in Group II right away significantly reduced the bond strength compared to composite restorations that were bonded to bleached samples in Group III after a 2-week waiting period. This shows that intracoronal bleaching followed by immediate bonding causes a composite restoration with insufficient adhesion. Numerous studies have demonstrated the ability of these antioxidants to strengthen the link between bleached teeth,6, 27–30 which supported the findings of our investigation. The results showed a substantial difference between the antioxidant-treated, control, and other groups.
Adhesive failures are slightly more prevalent and cohesive, and mixed failures also occur with significant frequency. The near-equal distribution among the three failure modes indicates a balanced contribution of different failure mechanisms in the tested material or interface. Understanding the prevalence and nature of these failure modes is crucial for improving the materials and methods used to enhance bonding performance and durability. Our examination under a stereomicroscope of the test samples having undergone antioxidant application immediately after bleaching and samples obtained after 2 weeks, indicated by a cohesive configuration of fracture, which shows the fracture between the resin-dentin interface was observed in fragments less than that of adhesive failure clearly showing of good bond strength. The mixed type was evident in the remaining fragments, which could be influenced by the control group, which only had direct composite restorations and experienced any bleaching effects, and the pine bark-treated specimens, which displayed the highest peak stress among the compared experimental groups.
Limitations of the Study
Antioxidants were only used for 30 s in this trial. Future research must evaluate the impact of applying antioxidants for a shorter period and at a greater concentration. Future research can concentrate on antioxidant storage techniques due to the limited shelf life of antioxidants. Since this study was in vitro, clinical trials or in vivo research are needed to determine how effectively different antioxidants restore the binding strength.
Conclusion
Delaying the composite resin bonding technique to bleached teeth by 2 weeks was essential to eliminate the bleaching chemical’s negative impact on the composite restoration’s bond strength. The push-out bond strength was greatly improved by 10% pine bark applied for 30 s. Pine bark is more effective than green tea, and SA is an antioxidant for reversing the binding strength of the resin-based composite to bleached dentin (SA). These herbal natural antioxidants have a role in enhancing dental restoration durability. By focusing on herbal antioxidants’ impact post-bleaching, the research introduces innovative, less invasive methods to improve resin-based composites’ bonding strength and longevity.
Footnotes
Abbreviations
Author Contribution
PMS, VGD: Conceptualization, methodology, investigation, writing—original draft, statistical analysis, validation, writing—review and editing; AG: Methodology, visualization, writing—original draft; ATS, SHK: Writing—review and editing, supervision.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The JSS Dental College & Hospital Institutional Ethics Committee after critically reviewing the research protocol approves the study (No. JSSDCH IEC: 44/2022).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
