Abstract
Background
Pediatric wellbeing and health outcomes are among the leading indicators of World Health Organization (WHO). Growing infections and risk of antibiotic resistance have appeared to be one of the main cause if mortality and morbidity in the pediatric population. Cephalosporins, due to their better comparative efficacy and safety, are often first line therapy approach among the pediatric population.
Objectives
This review is aiming to elaborate the use of cephalosporins in pediatric patients within the Kingdom of Saudi Arabia (KSA). In addition, this review shall also sum up the clinical outcomes after use among the pediatric patients.
Results
This review highlights the use of clinical efficacy and use of cephalosporins among pediatric population in variety of infections. It was observed that among the included studies the cephalosporins were found effective in combating infections caused by gram-negative bacteria (Escherichia coli, methicillin-susceptible Staphylococcus aureus [MSSA], and non-typeable Haemophilus influenzae [ntHi]), while recognizing their ineffectiveness against methicillin-resistant S. aureus (MRSA) and enterococci. In Saudi health care setting use of Cephalosporins is frequent and this recommendation is based on their broad-spectrum efficacy and clinical safety profile among variety of pediatric infections in different clinical settings.
Conclusion
This review emphasizes the need to ensure the supervised use of cephalosporins in Saudi health care setting. Implementation of the national clinical guidelines and their synchronization with the global standards shall be a way to ensure long term availability of cephalosporins as 1st line regimen. Promoting rational prescribing methods can reduce the risks of resistance and maintain efficacy in pediatric care.
Introduction
Cephalosporins, a significant class of antibiotics (ATB), are widely used in clinical settings to combat various infectious agents. In addition to their notable antibacterial effectiveness, these compounds exhibit an excellent safety profile, with minimal severe side effects and contraindications. Cephalosporins remain essential in treating serious conditions such as septicemia, meningitis, surgical prophylaxis, and pyelonephritis. However, their efficacy in managing common respiratory tract infections (RTIs) has been questioned due to the emergence of β-lactam resistance in pneumococci (particularly Streptococcus pneumoniae) and their role in the rise of extended-spectrum beta-lactamase (ESBL)-producing Enterobacteriaceae. 1
Significant interindividual variation exists in the pharmacokinetics of cephalosporins among pediatric patients. Alterations in body composition, size, and the developmental maturation of liver and kidney functions contribute to the differences in how these drugs are absorbed, distributed, metabolized, and excreted (ADME) between children and adults. This variation is particularly pronounced in neonates, who experience rapid postnatal changes that affect pharmacokinetics. Renal maturation plays a key role in this variability, with postnatal and gestational age at birth as major covariates. Specific pharmacokinetic parameters have been developed for neonates in the context of cephalosporin use. 2 For children beyond the neonatal period, pharmacokinetics becomes more similar to that of adults when adjusted for body surface area (BSA) or weight, allowing dosage adjustments based on these factors. 3
The primary goal of this comprehensive review is to assess the proper use of cephalosporins in neonatal and pediatric settings in the Kingdom of Saudi Arabia (KSA), as documented in studies published since 2010. As no previous systematic review has focused on cephalosporin use in the pediatric population, this study seeks to address that gap. A secondary objective is to investigate cephalosporin resistance in this population.
Materials and Methods
Search Strategy
Data sources and strategies from 2010 were sought in electronic databases, namely, MEDLINE PubMed, Scopus, Embase, Google Scholar, and Ovid, to assess the data. The search terms encompassed variations such as “cephalosporins” AND “KSA” or “Saudi Arabia” or “Kingdom of Saudi Arabia” AND “pediatric” or “children” or “infants” or “neonates” to identify studies that documented the dispensing of cephalosporins in medical centers for human use.
Study Inclusion and Exclusion Criteria
Included in the review were randomized control trials and various cephalosporin interventions examining the effectiveness of addressing microbial infections in pediatric patients younger than 18 years. Additionally, experimental single-case studies, original studies published in scientific journals, and articles in the English language were considered. Excluded from the study were in vitro studies, expert opinions, study protocols, editorials, review articles, conference abstracts, and studies involving animals. Patients who are older than 18 years are excluded from this study.
Population and Study Outcomes
The prime focus of this scoping review will be on the use of cephalosporins among pediatric, children, or neonate populations. Over time, there has been strict monitoring of the use of ATB among the pediatric, children, or neonate population; therefore, this article will look at the rationale for use, therapeutic outcomes, and recovery from infection among Saudi pediatric patients.
Data Extraction
Two reviewers independently reviewed the titles and abstracts of the records found through the electronic search. Then, they independently evaluated the entire text of all probably qualifying articles. The findings obtained for each stage were compared following the screening of the first recordings. Disagreements among reviewers were addressed through conversation.
Data on the research’s characteristics (author, study design, study population, gender, surveillance area, pathological condition, cephalosporins used, primary findings, and conclusions) were retrieved. Isolated organisms, resistant cephalosporins, resistance reasons, and conclusions were also reported.
Results
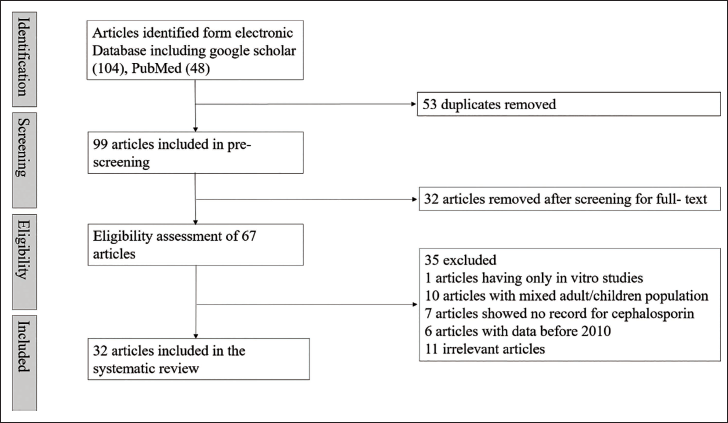
From the database searches (Figure 1), a total of 152 articles were initially identified, and after removing duplicates, 88 articles remained. Upon a review of titles and abstracts, 67 articles were chosen for a full-text review. After carefully examining the full text, 32 articles met the eligibility criteria. The exclusion of most articles was attributed to a lack of clarity regarding the study population, including issues such as mixed children and adult patients, and some of the studies only presented information on antibiotic usage without furnishing records specifically detailing the use of cephalosporins in pediatric populations. The summary of the studies included is expressed in Table 1.
PRISMA Flowchart Presenting the Process of Selecting Studies Focused on the Utilization of Cephalosporins in Pediatric Patients.
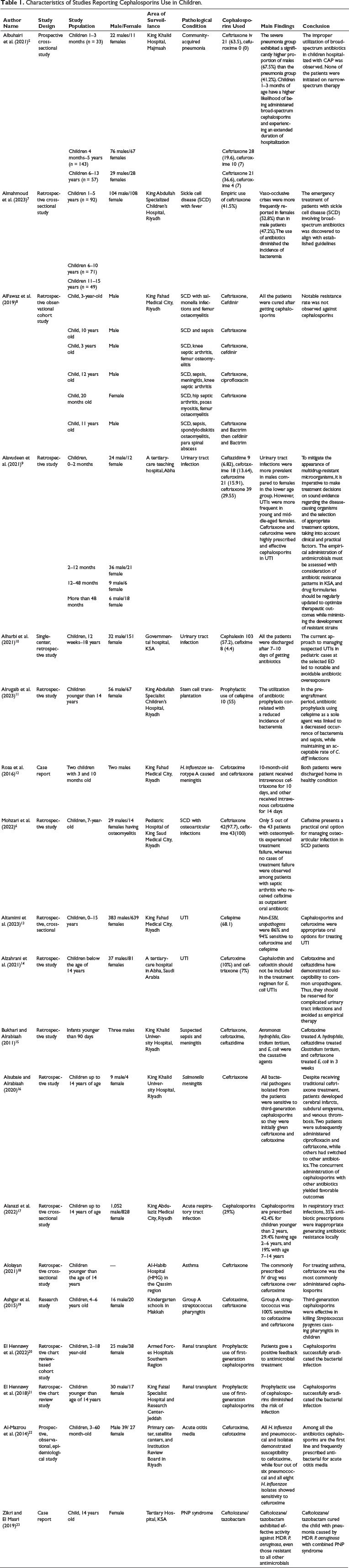
Characteristics of Studies Reporting Cephalosporins Use in Children.
Community-acquired Pneumonia
Saudi Pediatric Infectious Diseases Society (SPIDS) suggests amoxicillin as the primary treatment for immunized patients diagnosed with community-acquired bacterial pneumonia (CAP). For infants and children who have not been appropriately immunized, specifically with Haemophilus influenzae type b (Hib) vaccine (three doses), SPIDS approves amoxicillin–clavulanate or cefuroxime, 30 mg/kg/day, as an alternative. If a child with CAP is admitted to a hospital, SPIDS advises using penicillin G or ampicillin. Additionally, for inappropriately immunized children who have not completed the Hib vaccine series, amoxicillin–clavulanate or cefuroxime, 150 mg/kg/day, should be considered an alternative. In cases of hospitalized infants and children with complicated pneumonia, SPIDS recommends contemplating a third-generation parenteral cephalosporin, that is, ceftriaxone (75 mg/kg/day) or cefotaxime. 4 A study in King Khalid Hospital in Majmaah assessed the prescription pattern of ATB in children aged 30 days to 13 years suffering from community-acquired pneumonia. As anticipated and aligning with established guidelines, the youngest participants (below 90 days) and those having severe pneumonia notably received cephalosporins, which has broad-spectrum coverage, and experienced prolonged hospital stays. Participants were categorized into three distinct age groups: Group A (30–90 days), group B (4–60 months), and group C (6–13 years). Over the study period, 233 children were hospitalized with CAP, comprising 127 males (54.5%) and 106 females (45.5%). The age of the children upon hospital admission ranged from 30 days to 13 years. Approximately 23.6% of the patients had asthma. Ninety-one children (39%) among 233 were divided into two categories: pneumonia and severe pneumonia. In the severe pneumonia group, male patients were significantly higher (67.5%) than the pneumonia group (41.2%). Notably, ampicillin and ceftriaxone were given more frequently in group A: 12.1% and 63.5%, respectively, than in group B: 0.7% and 19.6%, respectively, and group C: 0% and 36.8%, respectively. Conversely, using piperacillin–tazobactam and cefuroxime was less common, and the differences among the three groups were not statistically significant (1.4–3.5% and 0–7%, respectively). Children with severe pneumonia were more suitable for receiving ceftriaxone (55%) than the pneumonia group (23.5%). Like the comparison between age groups, cefuroxime and piperacillin–tazobactam were administered less frequently, with no remarkable differences between these two groups. In summary, 30% of the patients received ceftriaxone, and cefuroxime was administered to only 6% of the participants. 5
Sickle Cell Disease (SCD)
A study on pediatrics having SCD evaluated the use of cefixime for treating osteoarticular infection. Among 260 patients, 43 with osteomyelitis (OM) and 8 with septic arthritis (SA) were selected. Among OM patients, males constituted 67.4%, and the median age was 7 years. The median time for intravenous ATB and hospital stays was 10 and 11 days, respectively. For SA and OM, the median time of antimicrobial administration was 25 and 37 days, respectively. The success percentage was 88% for OM cases and 100% for SA patients. 25% of SA patients experienced reinfection, while readmission occurred in 39.5% of OM cases. In summary, the study’s results suggest that cefixime serves as an oral antimicrobial alternative for managing osteoarticular infections in children with SCD. 6 A retrospective cross-sectional investigation evaluated the prevailing emergency practices for managing patients with SCD in tertiary hospitals. 52.8% of the children exhibited documented fever upon presentation to the emergency department (ED). Nevertheless, blood cultures were obtained from 82% of these patients. Almost 41.5% of patients with fever were subsequently admitted and injected with ceftriaxone for empirical therapy. 7 The study focused on SCD patients who had salmonella invasions and got treatment at King Fahad Medical City from 2012 to 2018. The researchers followed the six patients who got ceftriaxone (I.V.) either by itself or with other drugs to fight the infection. The cephalosporins worked well against the bacteria. 8
Urinary Tract Infection
SPIDS recommends cephalexin (50 mg/kg/day in four doses) and cefprozil, 30 mg/kg/day in two doses, or cefuroxime (30 mg/kg/day in two doses) as first-line oral ATB for empirical therapy of community-acquired urinary tract infections (UTIs) in children (3 months–14 years) for a time period of 3–7 days. Recommended intravenous cephalosporins are ceftriaxone with dose 50–75 mg/kg, every 24 h and cefotaxime with a dose of 150 mg/kg/day, divided every 8 h. 24 A study investigated the emergence and antimicrobial susceptibility patterns of uropathogenic bacteria in children with UTIs at a tertiary-care center in KFMC between 2019 and 2020. It included 1,022 patients and indicated that Escherichia coli was the major causative factor for UTIs in pediatric patients with ages below 15 years. Non-ESBL isolates were susceptible to ATB such as cefepime (94%) and ceftazidime (93%). ESBL isolates demonstrated 100% susceptibility to ertapenem and meropenem, followed by cefoxitin (90%). Conversely, ESBL isolates showed limited susceptibility to ceftriaxone, cefotaxime, cefuroxime, and cephalothin, (0% for all), and only 1% for cefepime and ceftazidime. Overall, both ESBL and non-ESBL strains exhibited greater susceptibility to cefoxitin. Furthermore, the study disclosed that the frequency of E. coli infection was notably greater among females than males. In contrast, Klebsiella, the second most frequent pathogen, and Pseudomonas infections were significantly more common in males than females. 13 A retrospective study involved 132 children with bacterial UTIs in the inpatient department of a tertiary-care hospital in Abha, Saudi Arabia. Throughout the study period, Klebsiella pneumoniae and E. coli, along with 15 other bacterial strains, were identified. Fever was identified as a prominent symptom (84.09%), with seizures documented as a major concomitant problem in 9% of UTI cases. Among pediatric cases, E. coli accounted for 31.82% of the most reported uropathogens, followed by 25% for K. pneumoniae. E. coli exhibited a susceptibility of 92.86% to ceftriaxone and ceftazidime, 78.57% to cefepime, and 71.43% to cefoxitin and cefotaxime. The sensitivity of K. pneumonia was 54.44% to cefotaxime, while Pseudomonas aeruginosa was 100% susceptible to cefepime and ceftazidime. Cephalosporins with ceftriaxone (29.55%), and cefuroxime (15.91%) were identified as the most frequently prescribed drug category across various drug classes in this patient population. 9 A retrospective investigation of patient records was conducted on individuals treated in the pediatric department with a diagnosis of UTI in a large tertiary-care hospital in Al Baha, Saudi Arabia, from 2017 to 2018. The study encompassed children of both genders below 14 years of age. Results indicated that among 118 urinary samples, E. coli emerged as the primary causative agent for both hospital- and community-acquired bacterial infections. The infection rate was lower in boys, 31.36%, compared to girls, 68.64%. Predominant isolates included coagulase-negative Staphylococci (3.39%), methicillin-resistant Staphylococcus epidermidis (4.24%), Enterococcus faecalis (7.63%), K. pneumoniae (9.32%), ESBL-producing E. coli (11.86%), and E. coli (44.07%). The susceptibility profile of E. coli indicated sensitivity to cefotaxime, ceftazidime (100%), ceftriaxone (75%), cefepime (71.4%), and cefuroxime (53.33%). However, resistance was reported with cefoxitin (76.92%) and cephalothin (92.30%) in treated individuals. Notably, ESBL-positive E. coli displayed resistance to cephalothin, cefoxitin, cefepime ceftazidime, cefuroxime, and ceftriaxone. In the case of K. pneumoniae, high sensitivity was observed to cefepime (100%) and ceftriaxone (66.67%), while it exhibited high resistance against cefoxitin, cefuroxime, and cephalothin (100%). E. faecalis demonstrated high sensitivity to cephalothin (100%). The study revealed that 19% nitrofurantoin was the most frequently used antibiotic, followed by cefuroxime (10%) and ceftriaxone (7%). 14 The researchers looked at the children who were 12 months–18 years old and had UTIs when they left the ED from January to August 2022. Cephalexin was the antibiotic that most of the patients got, with 57.2% of them receiving it. 10
Meningitis
A retrospective study on 304 infants younger than 3 months with suspected sepsis was examined. Among them, 20 infants were recognized with neonatal sepsis in the 30 days of their lives. Twelve blood cultures gave positive results (one group B streptococcus, one Enterobacter cloacae, one Bacillus species, one with diphtheroids, two Staphylococcus aureus, two Staphylococcus hominis, and four isolates of S. epidermidis). Only three cerebrospinal fluid (CSF) cultures were positive. Blood culture is positive with S. aureus, S. hominis and diphtheroids were treated with cefotaxime and ampicillin, while S. hominis was also treated with ceftriaxone. All the CSF cultures were treated with cephalosporins, that is, culture of E. coli was treated with ceftriaxone, C. tertium with ampicillin and ceftazidime, and Aeromonas hydrophila was exposed to cefotaxime and ampicillin/gentamicin. 15 The researchers at King Khalid University Hospital studied pediatric patients who were 14 years old or younger and had Salmonella meningitis from 1999 to 2016. They checked the neurologic problems and long-term results of all the affected children for 3 years. Out of 141 patients who had invasive Salmonella infection, 14 (10%) had meningitis. The infection started when the children were about 4.7 months old on average. The patients got cefotaxime or ceftriaxone at first. If they had any trouble or the infection came back, they got either ceftriaxone with ciprofloxacin, two patients, or meropenem, eight patients. They received IV ATB for 3–12 weeks. 16 Another study described the first two instances of H. influenzae serogroup A causing meningitis in Saudi Arabia. The babies had sepsis signs, and the blood culture and CSF samples showed the presence of the bacteria. The patient was taken to the intensive care unit (ICU) with a suspected case of meningitis and sepsis and received vancomycin and ceftriaxone treatment (ceftriaxone: 100 mg per kg per day; cefotaxime: 200 mg/kg/day, and vancomycin: 60 mg/kg/day). When the blood culture confirmed the infection, vancomycin was discontinued, and the patient was moved to the ward after 72 h in a stable state. The fever went away after 48 h of admission, and the blood culture was clear after 48 h. The patient received ceftriaxone for 10 days and went home in healthy condition. 12
Respiratory Tract Infections (RTIs)
Another scientific investigation was to assess the spread and antimicrobial-treatment patterns for RTIs and to evaluate the prevalence and types of antibiotic-prescribing errors in patients coming to the ED. Throughout the investigation, 3,185 cases of RTIs were identified, with pediatrics, less than 15 years of age, constituting 44% of the cases. 68.7% of pediatric cases (981/1,428) reported upper respiratory tract infections (URTI), while 447/1,428 reported lower respiratory tract infections (LRTI). Among URTI, the age group with the highest representation was 2–6 years, followed by 7–14 years, with the group less than 2 years old having the lowest representation.
For pediatric cases with LRTIs, the age group with the highest number was 2–6 years old, followed by those under 2 years old, and lastly, 7–14 years old. Three primary categories of ATB were prescribed: penicillin (43% for pediatrics), cephalosporins (29% for pediatrics), and macrolide (26% for pediatrics). The predominance of inappropriate antibiotic prescriptions was 53%, with a specific focus on pediatrics, accounting for 35% of cases. Among pediatric groups, the group with ages less than 2 years (11%) received 42.4% of cephalosporins, group 2–6 years (19.8%) received 29.4% of cephalosporins, and group 7–14 years (14%) got 19% of cephalosporins. Among cephalosporins, cefprozil was administered with the following percentages in pediatric groups: less than 2 years (40%), 2–6 years (30%), and 7–14 years (10%) approximately. Cefuroxime was administered to about 5% of the patients in the age group of 7–14 years. These patients were diagnosed with pneumonia and tonsillopharyngitis. 17 A retrospective study at Al-Habib Hospital in the Qassim region, from 2010 to 2019, aimed to examine the prescribing patterns of asthma therapy in children with asthma. Ceftriaxone emerged as the most frequently prescribed intravenous medication, with salbutamol following closely behind. 18 Group A Streptococcus (GAS) is one of the most encountered pathogenic bacteria. In Makah, Saudi Arabia, a study was conducted to assess the rate of streptococcal bacteria in the throat among preschool children aged 4–6 years. Samples from the throat were collected to assess the presence of Streptococcus pyogenes in healthy children. Among asymptomatic children, the predominance of GAS in throat swabs was found to be 1.5%. Notably, the highest predominance of Streptococcus pyogenes, reaching 1.6%, was detected in the age group of 5–6 years old. Importantly, Streptococcus pyogenes exhibited susceptibility to penicillins, including amoxicillin and ampicillin, and cephalosporins, such as ceftriaxone and cefotaxime. 19
Transplantation Procedures
A study was directed to examine the prophylactic utilization of antimicrobials in the pre-engraftment phase, aiming to reduce the likelihood of bacteremia, considering concerns related to antibiotic resistance, C. difficile, and the disturbance of gut microbiota. All individuals undergoing hematopoietic stem cell transplantation (HSCT), including autologous and allogeneic transplants, were administered prophylactic antimicrobials when the absolute neutrophil count fell below 1,000/µL until neutrophil engraftment. Prior to 2016, piperacillin/tazobactam was the antibiotic of choice, and subsequently, it was replaced by cefepime, according to hospital antibiogram data. Cefepime was utilized in 77% of the patients, while 21% received piperacillin/tazobactam. Two patients (1.4%) were prescribed ciprofloxacin due to allergic reactions to the antimicrobials mentioned above. In this study, the investigation revealed that the incidence of bacteremia in patients receiving prophylactic ATB was only 3.4% during the pre-engraftment phase and increased to 14% when considering the duration up to day +100 post-HSCT, compared to the literature. Sepsis and admission to the intensive care unit were observed in only 2.9% of the patients.
Collectively, employing cefepime as antibiotic prophylaxis in the pre-engraftment phase was linked with a reduced incidence of sepsis, bacteremia, and an acceptable level of C. difficile infections. 11 In the case of pediatric renal transplantation, first-generation cephalosporins were administered for 1 day for surgical site prophylaxis after renal transplantation.20, 21
Otitis Media
In a prospective epidemiological study in Riyadh from 2009 to 2011, 66 children facing new episodes of acute otitis media (AOM) were analyzed. S. pneumoniae was observed in six episodes, and nontypeable H. influenzae in eight episodes. Moreover, S. aureus, an unusual cause of AOM, was identified in 17 episodes. The pneumococcal serotypes identified were 15F (n = 1), 19F (n = 1), 23F (n = 2), and 7F (n = 2). All H. influenzae and pneumococcal strains were sensitive to cefotaxime, while 4/6 pneumococcal and 8/8 H. influenzae isolates were susceptible to cefuroxime. 22
PNP Syndrome
In the pediatric ICU, infections involving multidrug resistance (MDR) P. aeruginosa pose a significant threat, especially in immunocompromised patients. Some of the strains show resistance to all beta-lactams, including carbapenems, causing a severely restricted range of treatment options. Colistin and aminoglycosides, the available alternatives, possess suboptimal pharmacokinetic profiles and show notable toxicities. To cope with this problem, newer combinations of beta-lactam/beta-lactamase inhibitors present novel therapeutic avenues because of their favorable pharmacokinetic profiles and efficacy against MDR strains. One of them is ceftolozane/tazobactam, a cephalosporins, and beta-lactamase inhibitor that came in 2014. This drug exhibits potent activity against MDR P. aeruginosa strains, even those showing resistance to other antimicrobials. However, its current approval is limited to adult patients aged 18 years and older, and there is a scarcity of data about its pharmacokinetic parameters and clinical efficacy in pediatrics. In this scenario, a case was investigated where ceftolozane/tazobactam was successfully employed to treat pneumoniae associated with MDR P. aeruginosa in a child having PNP, purine nucleoside phosphorylase deficiency, and syndrome. 23
Analysis of Prescribing Pattern of Antibiotics in Saudi Arabia
A retrospective study of 60 days in the Jazan region was conducted to evaluate the prescribing pattern of ATB in pediatric inpatients. The number of ATB per prescription was approximately 1.45. Pneumoniae (9.7%) and fever (12.5%) were detected in the infants while other diseases made the contribution of 47.9%. Among all ATB, the cephalosporins group (ceftriaxone, cefuroxime, cephalexin) was most frequently prescribed and administered (52%). 25 A retrospective study at Children’s Hospital at KFMC examined 4,433 prescriptions that came from the pharmacy of the hospital. Among all the prescriptions, 822 contained ATB. URTI was frequently diagnosed, while patients aged less than 12 months were frequently diagnosed with pneumoniae. In general, the approximate rates of various diagnoses were tonsillitis (10.8%), pneumoniae (16.2%), otitis media (18.1%), and URTI (28.7%). For infants with age less than 12 months, cephalosporins (44.6%) were the most prescribed antibiotic. 26 Another research endeavor aimed to evaluate the frequency and factors predicting antibiotic-related prescription errors among children admitted to an emergency center within a tertiary healthcare center in the central region of the KSA. Notably, errors in dosage and duration were notably prevalent among the pediatric population. The occurrence of inappropriate antibiotic (ATB) prescriptions, encompassing one or more types of errors, stood at 46.2%, with a notable disparity between pediatrics (57.8%) and adults (38.7%), the former demonstrating a significantly higher rate. Initial bivariate analysis indicated that 77% of cephalosporins prescriptions for pediatrics exhibited a significantly elevated error rate compared to those prescribed for adults (60.9%). Concerning ATBs prescribed for pediatric patients, significant predictors of errors were identified in the context of treating UTIs and cephalosporins, as opposed to RTIs and penicillin, respectively. 27 A scientific investigation was conducted to investigate the consumption of antimicrobial medications and associated costs across various age groups in the inpatient units of hospitals in the Northwest Region (TABUK). In pediatric inpatient wards, the highest medication consumption was observed for ceftriaxone 1 g inj. (4,268), followed by Azithromycin PO 200 mg/15 mL suspension (376) and Acyclovir IV 250 mg (257). Meanwhile, in neonatal inpatient wards, the highest antibiotic consumption was ceftriaxone 1 g inj. (592), followed by gentamicin IV 20 mg (429) and vancomycin IV 500 mg (175). Consequently, the most frequently utilized antimicrobial was intravenous ceftriaxone for both neonatal and pediatric patients. 28 Similarly, the utilization of antimicrobial medications and the associated costs in ICUs at hospitals in the Northwest region (TABUK), distinguishing between pediatrics and neonates, were studied. In pediatric critical care units, the highest antimicrobial consumption was recorded for ceftriaxone 1 g inj. (592), followed by vancomycin IV 500 mg (203) and gentamicin IV 80 mg (160). The leading antibiotic consumption for neonatal critical care units was ceftriaxone 1 g inj. (214), followed by vancomycin IV 500 mg (168) and levofloxacin IV 500 mg (140). Notably, across all age groups (pediatrics and neonates), parenteral ceftriaxone emerged as the most frequently consumed drug. 29 However, concerning the utilization of antimicrobial drugs in EDs at hospitals in the North West (TABUK) region, the highest drug consumption in pediatric ED was observed for ceftriaxone 1 g (134) and Azithromycin 200 mg/15 mL (4,253). In neonatal ED, the most prominent antibiotic consumed was Azithromycin 200 mg/15 mL (83) and ceftriaxone IV 1 g (73). 30 A retrospective cohort study spanning 6 months was conducted at King Abdullah Specialized Children’s Hospital, KSA. The study’s primary objective was to delineate antibiotic consumption within the medical center. Notably, vancomycin, third-generation cephalosporins, and cefazolin emerged as the most commonly utilized antimicrobials. Furthermore, it was noticed that cefazolin and vancomycin were the ATB most frequently utilized inappropriately. The occurrence of inappropriate courses was higher in surgical admissions and in prophylactic courses. 31 The scientific professionals also monitored the prescriptions for children in the primary healthcare centers of Saudi Arabia. They noticed that cephalosporins, 12.4%, were prescribed more after penicillin, 72.5%. 32 A cross-sectional analysis, centered on scrutiny of ATB prescriptions, was carried out to evaluate the prescribing patterns of ATB in the ED of a tertiary care hospital in KSA. Among pediatric patients, cephalosporins were prescribed with a percentage of 33.3%, ranking second after penicillins, which made up 46.4% of the prescriptions (Table 2). 33
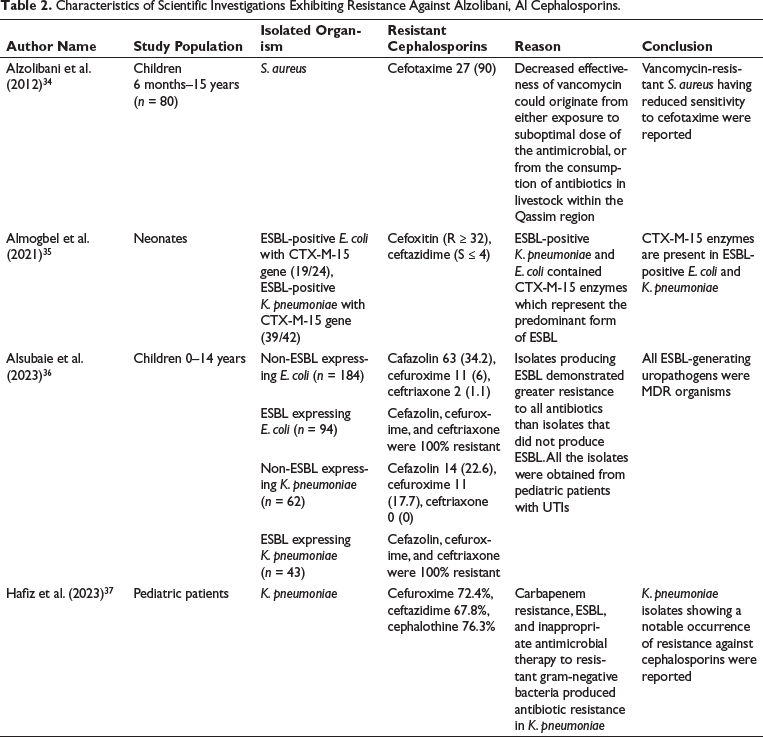
Characteristics of Scientific Investigations Exhibiting Resistance Against Alzolibani, Al Cephalosporins.
Resistance Against Cephalosporins
S. aureus obtained from children diagnosed with atopic dermatitis, aged between 6 months and 15 years, during the period spanning 2009–2010, exhibited a 90% resistance to cefotaxime. 34 A total of 48 K. pneumoniae and 24 E. coli isolates were cultured from neonates admitted to the Neonatal Intensive Care Unit (NICU). Among the E. coli isolates, the majority (19/24) were ESBL-positive, and carried the CTX-M-15 gene. Of these, 15% were coproducers of SHV-12 and CTX-M-15, and 68.4% (13/19) had TEM-1 and CTX-M-15. Considering the K. pneumoniaee organisms, 87.5% (42/48) were ESBL-positive and 92.85% (39/42) had the CTX-M-15 gene. Additionally, 97% of K. pneumoniaee had coproduced SHV-12 and CTX-M-15, and 88% had TEM-1. Moreover, 85.7% of K. pneumoniaee had coproduced TEM-1 and CTX-M-15. Pulsed-field gel electrophoresis (PFGE) grouped the 94% E. coli isolates into two genetic clusters, all identified as the ST-131 type. Contrary to that, K. pneumoniaee isolates (31/42) shared a single genotypic lineage, all categorized as the ST-14 type. The noteworthy observation is the elevated occurrence of outbreak isolates exhibiting concurrent CTX-M-15 and SHV-12 production, leading to significant resistance against oxyimino-cephalosporins. 35 Another study was carried out at a tertiary-care hospital in KSA, focusing on children aged 0–14 years diagnosed with community-acquired UTIs confirmed by culture between 2019 and 2021. The children were divided into two categories based on whether UTI was attributed to ESBL-generating bacteria. 383 children with UTI were included in the analysis. Among the 383 UTI episodes, E. coli was identified in 72.6% of cultures, and K. pneumoniaee in 27.4% of cultures. ESBL production was noted in 35.7% of isolates, with 43 (31.4%) and 94 (68.6%) belonging to K. pneumoniae and E. coli, respectively. E. coli was the predominant uropathogen in these two groups. ESBL-expressing bacteria were E. coli (33.8%) and K. pneumoniaee (41%). Multidrug resistance was present in 229 isolates (59.8%), and all ESBL-producing bacteria (137, 100%) demonstrated multidrug resistance. Regarding antimicrobial resistance (AMR) rates, ESBL-expressing organisms exhibited remarkably greater resistance than non-ESBL-expressing organisms. ESBL-expressing organisms showed 100% resistance to cefazolin, cefuroxime, and ceftriaxone compared to non-ESBL-expressing organisms, which showed 31.3%, 8.9%, and 0.8% resistance to cefazolin, cefuroxime, and ceftriaxone, respectively. 36 K. pneumoniaee stands as the second most prevalent cause of both hospital-acquired and community-acquired gram-negative bloodstream infections (BSI). A study evaluated the microbial resistance and epidemiological characteristics, as well as clinical parameters linked with K. pneumoniaee BSI in KSA. A total of 152 cases of K. pneumoniaee BSI were identified, with adults (66.4%) being more susceptible to the infection compared to pediatric patients (33.6%). K. pneumoniaee exhibited concerning levels of resistance to various ATB, including carbapenems (36.1%), ceftazidime (67.8%), amoxicillin–clavulanate and cefuroxime (72.4%), and cephalothin (76.3%). 37
Discussion
Cephalosporins were predominantly administered in both parenteral and oral forms, with a higher preference for parenteral administration. Cephalosporins have various advantages over other antimicrobials which is due to their superior efficacy, less toxicity, and lower incidence of allergic responses. Cephalosporins are the most often given ATB in the present context, particularly among the pediatric population, due to their heightened susceptibility to infections, while prolonged antibiotic use may contribute to AMR. 38 Considering the afore-mentioned remark may be the cause of the establishment of resistance. The widespread use of third-generation cephalosporins facilitates the emergence and dissemination of bacterial infections that produce ESBL. The unrestricted use of third-generation cephalosporins may elevate resistance in K. pneumoniaee and E. coli. The administration of third-generation cephalosporins may lead to adverse consequences, including pseudolithiasis and reversible cholelithiasis, in the pediatric population, particularly in cases when there is a significant decline in the child’s general development. Moreover, over time cephalosporins have served as an ideal alternative for the variety of infections among special populations. However, the growing challenge, and substantial concerns regarding the potential emergence of AMR, could provide future challenges for children or patients. 39
This review illustrated that in some cases, the treatment regimen was implemented without conducting a culture sensitivity test, resulting in irrational prescriptions. Despite the financial burden associated with culture tests, physicians should strive to be more specific in their diagnoses. It is imperative to establish an antibiotic evaluating team, including a physician, pharmacist, infection control nurse, and medical microbiologist to periodically assess and monitor the proper utilization and costs of antimicrobials in the hospital. This collaborative effort aims to help clinicians in improving the clinical care of patients. With AMR emerging as a significant issue, the compilation of a hospital antimicrobial consumption policy is an urgent concern. The execution of an antibiotic order form would contribute to improving the relevancy of therapy. Although medical professionals were cognizant of the antibiotic resistance problem and its contributing factors, their practices did not present measures aimed at its reduction. The primary problem is the off-label use of ATB, characterized by many prescriptions for the same patient. The predominant use of off-label medications currently pertains to newborns. Of the most recommended antibacterial drugs in pediatrics, only six were supported by data. A specific approach is necessary for treating off-label drugs due to the increasing global risk of antibiotic resistance. 40
The recommendations given by SPIDS advocating for the use of cephalosporins as an empirical treatment for respiratory disorders and UTIs may contribute to resistance development. Moreover, in critical conditions the submission of various broad spectrum cephalosporins via intravenous route, along with the benefits there may be an immediate risk of adverse effects that are associated with the IV use of cephalosporin Therefore, the health care providers must ensure that staff is well trained not only to effectively prepare these formulations but also is capable of managing any adverse event that is associated with the use of cephalosporins.
However, it is crucial to acknowledge that the substantial prescription of cephalosporins for various other conditions is likely to exert a more significant influence on cephalosporin resistance, affecting both individuals and the community at large. Studies suggest that increased usage of cephalosporins is linked to a heightened occurrence of bacterial resistance, particularly against first-, second-, and third-generation cephalosporins. However, information regarding fourth-generation cephalosporins is currently undocumented. Our discoveries offer valuable insights into the prescription patterns of cephalosporins and suggest potential strategies for improvement in the KSA.
Future Implications
The practical implications for future healthcare policy formation concerning cephalosporin use in neonatal and pediatric settings are important. Published research emphasizes the necessity of rationalizing antibiotic usage to combat resistance, especially in vulnerable groups such as neonates and children. Policymakers should emphasize the creation of strong antibiotic stewardship programs that track and restrict cephalosporin prescriptions. This might involve implementing clinical guidelines specific to pediatric and newborn populations, ensuring that clinicians are well-versed on cephalosporin dose, duration, and indications.
Furthermore, healthcare policy should include frequent monitoring of antibiotic resistance patterns, particularly for cephalosporins, to inform real-time changes to treatment procedures. Healthcare staff in neonatal and pediatric units should receive education and training on the implications of inappropriate antibiotic use and the need to follow evidence-based procedures. Furthermore, policymakers should encourage research activities to better understand the reasons for cephalosporin resistance and create other treatment alternatives. In this setting, Saudi Arabia may lead in developing antibiotic stewardship in the Middle East, improving patient outcomes and mitigating the threat of antibiotic resistance.
Limitations
Since this study did not delve into aspects such as the total number of coadministered drugs per patient, the cost-effectiveness evaluation of cephalosporins per patient during his or her stay at the hospital, bacterial culture sensitivity tests, microbial resistance patterns along with additional costs associated with medications for treating cephalosporins-resistant infections, it is recommended that these aspects be thoroughly explored in subsequent studies.
Conclusion
Cephalosporins form a diverse category of ATB, featuring distinct pharmacokinetics and in vitro antibacterial activity that clinicians must comprehend for optimal usage. Despite their widespread application, their use contributes to the rise of ESBL-producing strains. Therefore, their deployment should be limited to scenarios requiring probabilistic extended-spectrum therapy and conditions where therapeutic alternatives are unavailable.
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank the Research Center at King Fahad Medical City, Riyadh R2, Second Health Cluster, for the valuable technical support provided for the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
