Abstract
Background
Flacourtia inermis Roxb. (Salicaceae) is an underutilized edible fruit known for its antioxidant-rich bioactive compounds. This study explores the antiobesity potential of F. inermis ethanolic fruit extract (FF2) in a high-fat diet (HFD) induced obesity model using Wistar albino rats.
Materials and Methods
Wistar albino rats were randomly assigned into five groups (n = 6). Group 1 served as the normal control and received a normal diet (ND), while Groups II–V were induced with obesity by being fed an HFD for 10 weeks. Group II acted as the obese control, receiving 100% HFD throughout the study. Groups III, IV, and V also received HFD throughout the study but were supplemented with Orlistat (30 mg/kg b.w), and FF2 extract at doses of 200 mg/kg, and 400 mg/kg, respectively, from week 11 until week 15. Treatment impact of FF2 on body weight, body mass index (BMI), serum lipid levels, glucose levels, liver function parameters, and histopathological evaluations of liver and adipose tissue were investigated.
Results
The study unveiled that F. inermis fruits exerted potent antiobesity effects, with FF2 treatment (at 200 mg/kg and 400 mg/kg) substantially decreasing body weight gain and BMI in comparison to the HFD group. Supplementation of FF2 alongside an HFD reduced body weight gain, glucose levels, and serum lipid profile. Both doses of FF2 counteracted the adverse effects induced by HFD, with lower triglycerides (TG), total cholesterol, and LDL cholesterol levels, and higher HDL cholesterol levels in the treated groups compared to the positive control. Additionally, FF2 extract showed dose-dependent improvements in serum lipid and glucose levels, as well as, hepatic profile, indicated by decreased AST, ALP, and ALT levels, suggesting potential benefits in managing dyslipidemia, hyperglycemia, and liver health associated with obesity. Histological examination of white adipose tissue provided further evidence of the antiobesity effect.
Conclusion
The study highlights FF2’s therapeutic potential in managing obesity and associated metabolic disorders. Further investigations into FF2’s mechanisms and clinical application are essential for its development as an antiobesity therapeutic agent in functional food formulations.
Introduction
Flacourtia inermis Roxb. (Salicaceae, also placed in Flacourtiaceae) (Figure 1), commonly known as Lovi lovi, is a tropical evergreen tree native to Moluccas, India, Malaysia, Sri Lanka, and New Britain in Papua New Guinea. The fruits of F. inermis, known as loobikka or lavulolika in Kerala, India, is a red, pulpy berry with an acidic taste and, is used in local culinary practices such as pickling, confectionery preparation, and wine production. Studies have reported that the fruit juice of F. inermis contains caffeoylquinic acid derivatives, phenolic glucosides, quinic acid, and malic acid, and has demonstrated antioxidant activities. 1 Additionally, the polyphenols from the fruits have been found to possess α-glucosidase and α-amylase inhibitory activities. Furthermore, the fruit has been investigated for its antibacterial, antifungal, and antiprotozoal activities. 1 In our previous studies, Bency et al. reported the pancreatic lipase (PL) inhibitory activity of the ethanolic fruit extract of F. inermis. 2
Flacourtia inermis Tree and Fruits.

Obesity is a global concern associated with various comorbidities such as atherosclerosis, diabetes, hypertension, and cancer. Epidemiological studies have established a strong link between high saturated fat intake and obesity.3–5 PL plays a crucial role in lipid absorption, and inhibiting it has been shown to reduce fat absorption, offering potential benefits for obesity management. The long-term use of Orlistat, the only approved PL inhibitor, has been associated with significant adverse effects, necessitating the search for safer and more efficient inhibitors for obesity management. Natural compounds have shown promise as lipase inhibitors, providing a basis for the development of novel and safer antiobesity agents.6–8
In this study, we aim to investigate the efficacy of FI as a natural PL inhibitor and its broader implications for antiobesity therapy. The study employed an HFD-induced obesity model in Wistar albino rats to explore the potential antiobesity activity of the ethanolic fruit extract of F. inermis. The rationale for this investigation is supported by the existing literature on the adverse effects of long-term use of Orlistat and the promising role of natural compounds as lipase inhibitors for the development of novel and safer antiobesity agents. 9 Additionally, the multifactorial nature of obesity, including its impact on various aspects of health and the contribution of socioeconomic inequalities, will be addressed in the study to provide a comprehensive understanding of the public health issue.
Furthermore, it is essential to note that our previous studies reported the PL inhibitory activity of FF2. 2 This aligns with the extensive use of PL inhibitory activity for evaluating the potential efficacy of natural products as antiobesity agents. Therefore, building upon this previous research, the current study aims to explore deeper into the antiobesity potential of FF2 through in vivo experimentation.
Materials and Methods
Chemicals and Reagents
The study involved using adult Wistar strain albino rats obtained from Kerala Veterinary and Animal Science University, Mannuthy, Thrissur. The rats were housed in controlled conditions at Al Shifa College of Pharmacy, Kizhattoor, Perinthalmanna, Kerala, India. The rats were kept in cages with specific environmental parameters, including a temperature of 23°C ± 2°C, a light-dark cycle of 12 hours, and a relative humidity of 60% ± 5%. Each group consisted of six animals, with the normal control receiving standard commercial rat feed pellets and water, while the experimental groups were fed an HFD diet. The experimental procedures followed internationally recognized principles for laboratory animal use and were approved by the Institutional Animal Ethics Committee of Al Shifa College of Pharmacy (Approval no. IAEC 073/21). Orlistat was obtained from Sigma Aldrich (St Louis, MO, USA), and all other chemicals and solvents used were of analytical grade and purchased from SD Fine Chemicals (Mumbai, Maharashtra, India).
Collection and Authentication of Plant Materials
Fresh fruits of F. inermis were gathered from the Puliyanam area of Ernakulum district, Kerala, India, following the rainy season. The authentication of the plant specimen was conducted by the Pharmacognosy Division of the Center for Medicinal Plants Research (CMPR) at Arya Vaidya Sala, Kottakkal, Kerala, with the reference number: CMPR/AIF/PHG/317.
Preparation of F. inermis Extract
Fresh fruits of F. inermis were collected, dried, powdered, and sequentially extracted with hexane, chloroform, ethyl acetate, ethanol, and water using a Soxhlet extractor for 24 hours. The extraction process was repeated in triplicate to ensure thorough extraction. The resulting extracts were filtered, combined, and concentrated using a rotatory vacuum evaporator (Roteva, India).
In our previous study, we conducted an in vitro assessment of PL inhibition using various extracts of F. inermis fruit. Among these extracts, the ethanol extract [FF2] displayed significant inhibition of PL activity, as evidenced by its IC50 value. Based on these findings, FF2 was selected for further investigation utilizing an in vivo model in the current study.
Acute Oral Toxicity Studies (OECD 2002)
The acute oral toxicity test was conducted following OECD Guideline No. 423 using female Wistar albino rats. The study adhered to standard conditions, including a controlled temperature of 21°C, a 12-hour light/dark cycle, and access to a standard pellet diet and water. The Acute Toxic Class Method was employed, which involves a stepwise approach with three rats per step. Each rat was uniquely marked for identification. The dose level to be used as the starting dose is selected from one of four fixed levels, 5, 50, 300, and 2000 mg/kg body weight. The study commenced with the highest dose of 2000 mg/kg to evaluate the effects of the herbal extracts. 10
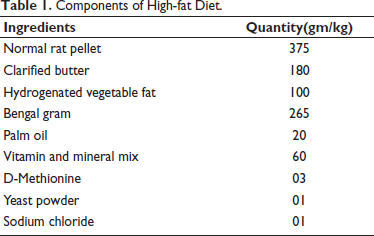
Preparation of High-fat Diet (HFD)
The HFD was prepared following a previously reported formula by Arika et al. (2019) 11 with slight modifications (Table 1). The diet was formulated by supplementing commercial rat pellets with a mixture of animal and vegetable fat, along with other essential nutrients. 11
Components of High-fat Diet.
Induction of Obesity
Thirty Wistar albino rats were utilized in this study to investigate the induction of obesity. Among these, 24 rats were subjected to an HFD for 10 weeks to induce obesity. The rats were provided with water ad libitum throughout the study period. An additional six rats were fed a standard rat pellet diet and served as the control group. After the 10-week period, randomization was performed, and 24 rats from the HFD group, weighing approximately 225–250 g, were selected for further antiobesity studies. These rats were considered obese-prone based on their weight gain and metabolic changes induced by the HFD. The control group consisted of six rats fed an ND to compare the effects of obesity induction.
Animal Study Design
The study involved five groups of rats: (n = 6)
Group I (Normal Control): Received a regular diet throughout 15 weeks of study. Group II (HFD): Received an HFD throughout 15 weeks of study. Group III (Standard): Fed with the HFD for 10 weeks, followed by Orlistat administration at 30 mg/kg body weight along with the HFD for the next 5 weeks. Group IV (FF200): Fed with the HFD for 10 weeks, followed by administration of the low dose of ethanolic fruit extract of F. inermis at 200 mg/kg body weight along with the HFD for the next 5 weeks. Group V (FF400): Fed with the HFD for 10 weeks, followed by administration of a high dose of ethanolic fruit extract of F. inermis at 400 mg/kg body weight along with the HFD for the next 5 weeks.
Throughout the 15-week study, animals had ad libitum access to water and were subjected to specific dietary interventions. Orlistat, FF200, and FF400 were orally administered once daily. Body weights were measured weekly using a digital balance, and daily feed intake was documented. At the end of the study, the animals were euthanized following an overnight fast using carbon dioxide. Blood samples were obtained via cardiac puncture, centrifuged to isolate serum, and stored at 20°C. The liver and white adipose tissue were dissected, washed with a cold sodium chloride solution, and prepared for subsequent biochemical and histopathological analyses.
Determination of Body Weight
The body weight of each rat was assessed weekly using a standard weighing machine throughout the 15-week study period to monitor changes over time. Net weight gain was calculated as the difference between the final and initial weights. 12
Determination of Feed Intake
Feed intake was measured daily for each cage, and the average daily intake was computed by quantifying the remaining feed using an electronic balance. 13
Determination of Body Mass Index (BMI)
BMI was calculated at the 11th and 15th weeks of the study. This involved measuring the body length from the nose to the anus in centimeters and calculating the BMI using the formula: BMI = (Body mass in grams)/(Body length in cm2). 14
Measurement of Serum Glucose and Lipid Profile
Upon completion of the study, blood samples were collected through cardiac puncture. Subsequent serum analysis was performed to evaluate glucose and lipid metabolism parameters. The levels of total cholesterol, triglycerides (TG), high-density lipoprotein cholesterol (HDLC), and glucose in the serum were assessed utilizing commercial kits and analyzed using a FUJI DRICHEM NX500 analyzer (Fujifilm, Tokyo, Japan). Furthermore, VLDL levels were determined by applying the Friedewald formula.13, 15
Measurement of Serum Liver Parameters
Additionally, serum liver function tests were performed to assess key parameters such as aspartate aminotransferase (AST), alanine aminotransferase (ALT), and alkaline phosphatase (ALP). These tests were conducted using a combination of established methods and commercially available biochemical kits, alongside an autoanalyzer (FUJI DRICHEM NX500). 16
Histopathological Analysis
Histopathological analysis was performed on preserved adipose tissue and liver samples. The samples were sliced into 5 µm sections, stained with hematoxylin and eosin, and viewed under a light microscope at 40× magnification. 16
Masson’s Trichrome Staining
In the study, Masson’s trichrome staining was performed on paraffin-embedded liver sections to visualize collagen and detect liver fibrosis, with subsequent examination under an optical microscope. The analysis focused on the high-dose FF400 and standard Orlistat treatment groups, chosen based on observed histopathological improvements and potential therapeutic effects on liver morphology and adipose tissue fat accumulation, as previously discussed. This staining technique aimed to evaluate liver fibrosis, providing valuable insights into the impact of the antiobesity drug and plant extract on liver health and fibrotic changes. 17
Statistical Analysis
Statistical analysis was performed using GraphPad Prism version 9.5.10 software. The in vivo data were presented as mean ± SEM and analyzed using one-way ANOVA followed by Tukey’s multiple range test, with a significance level of p < .05 considered statistically significant.
Results
Acute Oral Toxicity Studies FF2
In acute oral toxicity studies, Wistar albino rats were administered FF2 extract at 2000 mg/kg body weight, showing no mortality or morbidity over 14 days. Clinical parameters like body weight, food and water intake, respiration, convulsion, tremor, temperature, constipation, and skin/eye color were monitored and found within normal ranges. The absence of acute toxicity was observed. The oral LD50 of FF2 in Wistar albino rats exceeded 2000 mg/kg, indicating low acute toxicity. For in vivo antiobesity evaluation, doses of 200 mg/kg and 400 mg/kg were chosen, representing 1/10th and 1/5th of the maximum extract dose, respectively.
Determination of Feed Intake
The analysis of feed intake revealed that the HFD group exhibited the highest daily feed intake, followed by the normal control (NC) group. In contrast, the Orlistat, FF 200, and FF 400 groups demonstrated lower daily feed intake levels compared to both the NC and HFD groups.
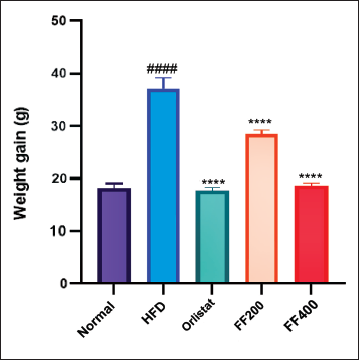
Effect of FF2 on Body Weight
Baseline body masses were similar across all study groups. The HFD group exhibited a significant increase in weight gain from the 11th to the 15th week (p < .0001). The Orlistat group displayed a significantly lower weight gain trajectory compared to the HFD group (p < .01). The FF200 and FF400 groups also demonstrated distinct patterns of weight gain, with statistically significant differences observed compared to the HFD group (p < .05 for both FF200 and FF400). These results highlight the varying effects of different interventions on weight gain in the experimental groups (Figure 2).
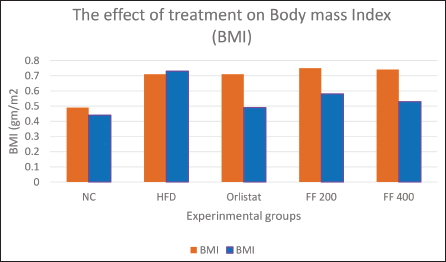
Effects of Treatment on BMI in Wistar Albino Rats
The effects of treatment on BMI in Wistar rats were assessed over the treatment period. The NC group, which received an ND, maintained a relatively stable BMI, with only slight fluctuations observed. In contrast, the HFD group exhibited a noticeable increase in BMI over the same period. The Orlistat group, receiving Orlistat in addition to the HFD diet, experienced a significant reduction in BMI, indicating a potential antiobesity effect of Orlistat in this model.
Regarding the FF2 extracts, the FF 200 group showed some changes in BMI, while the FF 400 group demonstrated a notable decrease in BMI compared to the initial measurement.
The findings indicate a potential antiobesity impact of FF 400 in HFD-induced obese Wistar rats (Figure 3).
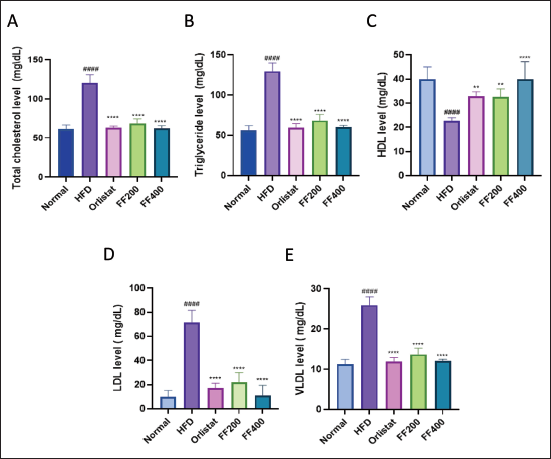
The Effect of FF2 Extracts the Serum Lipid Level of Wistar Albino Rats
The FF2 extracts potentially impacted serum lipid levels in Wistar albino rats with induced obesity. The HFD group exhibited significant alterations in total cholesterol (TC), TG, high-density lipoprotein (HDL), low-density lipoprotein (LDL), and very low-density lipoprotein (VLDL) levels compared to the baseline NC group. Orlistat positively affected the lipid profile, while the FF200 and FF400 groups also displayed dose-dependent changes in lipid levels (Figure 4).
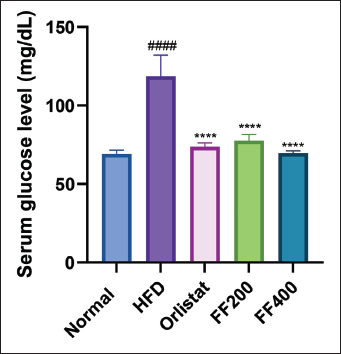
The Effect of FF2 on the Serum Glucose Level of HFD-fed Wistar Albino Rats
The effect of (FF2) on the serum glucose level of Wistar albino rats fed an HFD was investigated. The results demonstrated that the serum glucose level in the HFD group was significantly higher compared to the NC group. Treatment with FF2 at both doses (FF 200 and FF 400) decreased the serum glucose level compared to the HFD group. Notably, the serum glucose level in the Orlistat group was comparable to that of the HFD group (Figure 5).
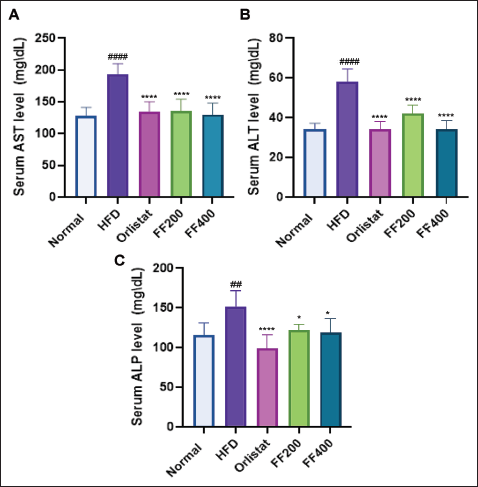
The Effect of FF2 Extracts on Serum Liver Parameters of Wistar Albino Rats
The impact of FF2 extracts on serum liver parameters in Wistar albino rats fed an HFD diet was assessed by measuring AST, ALT, and ALP levels. The HFD group exhibited elevated AST, ALT, and ALP levels, indicating potential liver damage. Orlistat treatment reduced liver enzyme levels, suggesting a protective effect against HFD-induced liver damage. FF 200 and FF 400 groups also showed improved liver health parameters, indicating potential benefits for liver function in obese rats (Figure 6).
Effects of FF2 on serum AST (A); ALT (B); ALP (C) The data are presented as mean ± SEM (n = 6), with statistical significance denoted by asterisks and hashtags. Specifically, *p < .05, **p < .01, ***p < .001, and ****p < .0001 indicate significance compared to the HFD (High-Fat Diet) group, while ####p < .0001 indicate significance compared to the NC (Normal Control) group. Statistical differences were assessed using one-way ANOVA with Tukey’s multiple comparisons test across all groups.
Histopathological Analysis
Treatment with FF2 in rats on an HFD significantly improved liver histology. Histopathological examination of the liver from rats treated with HFD revealed characteristic features of hepatic steatosis. Additionally, hepatocytes exhibit swelling, and the liver cells near the central vein appeared swollen due to fat degeneration. Compared to the HFD group, the NC group exhibited better liver histology (Figure 7). The FF200 group showed reduced steatosis and hepatocyte ballooning, indicating amelioration of liver injury. The FF400 group displayed even more substantial improvements, with minimal steatosis and near-normal hepatocyte morphology, suggesting significant protection against hepatic damage.
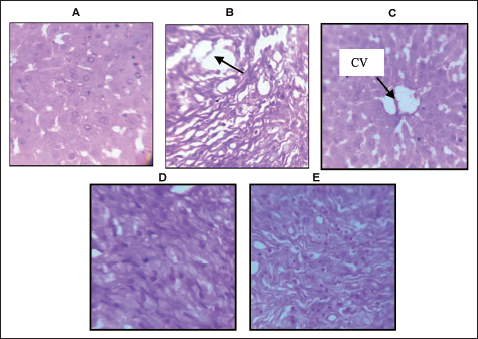
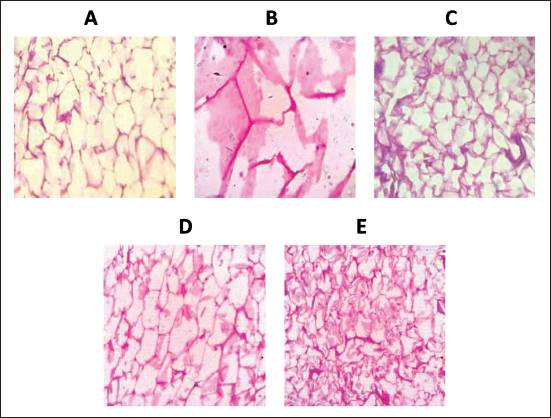
Figure 8 illustrates the adipose tissue morphology observed under H&E stain at 40× magnification. Adipose tissue from rats fed a regular diet NC exhibited typical adipocyte architecture. In comparison, adipose tissue from the HFD group displayed visibly enlarged adipocytes. On the other hand, adipose tissue from obese rats treated with Orlistat showed a reduction in fat cell accumulation. Remarkably, adipose tissue from the FF200 and FF400 treatment groups revealed significant improvements in fat accumulation, with the FF400 group demonstrating nearly normal adipocyte architecture.
(A) Adipose tissue from rats fed a normal diet, showing normal architecture of adipocytes (NC). (B) Adipose tissue from the HFD group visually appears enlarged. (C) Adipose tissue from obese rats treated with the standard drug, Orlistat, showed decreased fat cell accumulation. (D) Adipose tissue from the FF200 treatment group, demonstrated notable improvements in fat accumulation. (E) Adipose tissue from the FF400 treatment group showcasing remarkable improvements and nearly normal adipocyte architecture. (H&E Stain) 40× magnification.
Masson Trichrome Staining
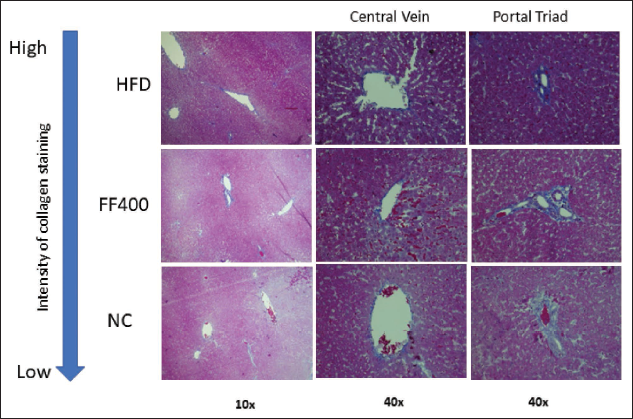
The Masson trichrome staining revealed distinct differences in collagen deposition between the HFD group and the various drug-treated groups (Orlistat and FF 400) after a 15-week treatment period (Figure 9). In the HFD group, rats exposed to the HFD displayed intense blue coloration of collagen fibers, particularly around the portal vein area. Additionally, there was a pronounced increase in collagen deposition in the areas surrounding the central vein and portal triad. In contrast, the drug-treated group (Orlistat and FF400) exhibited a prominent reduction in colorization of collagen fibers around the portal vein area compared to the HFD group.
Masson’s Trichrome Staining (Magnification 10× and 40×) Shows Collagen in Blue, Cytoplasm in Pink, and Nucleus in Black.
Discussion
In our ongoing pursuit to discover novel therapeutic agents from natural sources for combating obesity, we have conducted a comprehensive screening of F. inermis fruit extracts for their potential to inhibit PL, as reported in our previous study. The FF2 extract demonstrated significant PL inhibition. Additionally, our previous research reported isolating and identifying quinic acid and myricetin from these fruits. 2
The current investigation focused the antiobesity effect of FF2 in HFD-induced obese Wistar albino rats has provided valuable insights into the potential therapeutic benefits of this natural extract. The study revealed significant outcomes across various parameters, shedding light on the complex interplay between the extract and obesity-related factors.
In the evaluation of acute oral toxicity studies in Wistar albino rats administered with the FF2 extract at 2000 mg/kg body weight, no mortality or morbidity was observed over the 14-day observation period, with clinical parameters remaining within expected ranges, indicating a lack of acute toxicity. This aligns with the selection of lower doses of 200 mg/kg and 400 mg/kg, representing 1/10th and 1/5th of the maximum dose, respectively. The determination of the oral LD50 of FF2 exceeding 2000 mg/kg body weight further supports the favorable acute toxicity profile of the FF2 extract. The selection of lower doses for subsequent evaluations correlates with the absence of acute toxicity at the higher dose, providing insights into the safety profile of the FF2 extract across varying concentrations. These findings underscore the importance of dose selection in toxicity studies and contribute to the understanding of the safety margins of herbal extracts for potential therapeutic applications.18, 19
The observed reduction in feed intake in rats treated (FF2) suggests a potential role of the extract in regulating feed intake in obese rats, aligning with the concept that bioactive compounds in plant extracts can influence appetite regulation and satiety, thereby contributing to weight management. 20 Furthermore, the previous identification of quinic acid and myricetin from the same extract provides additional insights into the potential mechanisms underlying the observed effects on feed intake. Myricetin, a flavonoid, has been associated with significant antiobesity effects, potentially through its interactions with lipase enzymes responsible for fat digestion, as well as its diverse biological activities, including antioxidant and anti-inflammatory properties.21, 22 Quinic acid, on the other hand, has been chemically converted into hydroquinone, suggesting a potential role in metabolic processes related to obesity. 23
Furthermore, our previous research has elucidated the in vitro PL activity of the F. inermis Roxb. fruits ethanol extract, shedding light on the potential of bioactive compounds in plant extracts to modulate appetite regulation and satiety, consequently resulting in reduced feed intake in obese rats. 2 The observed decrease in feed intake in rats subjected to FF2 treatment signifies a regulatory effect of the extract on appetite, implying its potential role in weight management. Additionally, the investigation of the in vitro PL activity of the extract provides further insights into the potential mechanisms underlying the observed effects on feed intake. Inhibition of PL activity can impact fat digestion and absorption, potentially influencing satiety signals and overall energy balance in obese rats treated with the extract.24–28 These findings collectively support the potential antiobesity activity of FF2 and its capacity to modulate appetite regulation and satiety, thereby leading to reduced feed intake in obese rats.
In terms of body weight gain, the HFD diet group exhibited a substantial increase compared to the NC group, highlighting the obesogenic effects of the HFD. 29 However, the administration of Orlistat effectively prevented excessive weight gain, while FF200 and FF400 extracts demonstrated dose-dependent effects on weight gain, with FF200 showing more pronounced gains. This suggests that FF2 may have a modulatory effect on weight regulation, potentially through mechanisms that influence energy metabolism and fat storage.
The lipid-lowering effects of FF200 and FF400 extracts, as evidenced by improvements in TC, TG, LDL cholesterol levels, and increased HDL cholesterol levels, indicate their potential to ameliorate dyslipidemia associated with obesity. 30 These effects are crucial in reducing the risk of cardiovascular diseases and metabolic complications linked to obesity.
Furthermore, the significant reduction in serum glucose levels following treatment with FF200 and FF400 extracts suggests a potential role in improving glucose metabolism, which is often dysregulated in obesity. 1 The observed improvements in glucose metabolism are essential for managing metabolic disorders associated with obesity.
The study’s outcomes investigating the effect of FF2 extracts on serum liver parameters in Wistar albino rats fed HFD reveal promising results in alleviating liver damage associated with obesity. The elevated levels of AST, ALT, and ALP observed in the HFD group, indicative of potential liver dysfunction were notably reduced following treatment with FF2 extracts. This reduction suggests a protective effect of FF2 extracts against liver damage induced by HFD. Similarly, the FF 200 and FF 400 groups improved liver health parameters, implying the potential benefits of these treatments on liver function in obese rats. These findings underline the potential hepatoprotective effects of FF2 extracts in ameliorating liver damage associated with obesity, highlighting their therapeutic potential in preventing obesity-related liver complications such as insulin resistance and inflammation.31–34
Histopathological examination revealed that FF200 and FF400 extracts reduced fat accumulation in adipose tissue, potentially inhibiting adipogenesis and promoting lipid metabolism. This antiobesity effect on adipose tissue morphology is crucial in preventing obesity-related complications such as insulin resistance and inflammation.35–37 Additionally, the antifibrotic effects demonstrated by Orlistat and FF2 extracts in reducing collagen deposition in liver tissues suggest a protective role against liver fibrosis, a common consequence of obesity-related liver damage.
The findings from Masson’s trichrome staining confirm that HFD-induced liver fibrosis in rats. 38 In the context of HFD-fed rats treated with herbal extracts, Masson’s trichrome staining can be utilized to assess liver toxicity by evaluating changes in liver parenchyma, such as collagen deposition or fibrotic damage, which may indicate potential liver toxicity. The intense blue coloration observed in the HFD group around the portal vein area, along with increased collagen deposition around the central vein and portal triad, indicates the development of fibrosis in the liver tissues due to the HFD. This aligns with previous studies demonstrating the association between HFD diets and liver fibrosis in animal models and humans. 39
Importantly, the drug treatments demonstrated notable antifibrotic effects. The considerable reduction in collagen deposition observed in the liver tissues of the drug-treated groups, particularly around the portal vein area, indicates that these drugs effectively mitigated fibrosis induced by the HFD diet. The marked decrease in collagen deposition around the central vein and portal triad further supports the positive impact of drug treatments on liver fibrosis.40, 41 Previous studies have emphasized the importance of ensuring the safety of antiobesity drugs, particularly in the context of long-term use and potential adverse effects.42, 43 The absence of significant fibrosis in the drug treatment groups supports the safety and tolerability of these medications, providing reassurance regarding their use in the management of obesity-related conditions. The absence of significant fibrosis in the drug treatment groups, as evidenced by Masson’s trichrome staining, supports the safety and potential therapeutic benefits of FF2 extract while highlighting the detrimental effects of HFD-induced liver fibrosis.
Conclusion
In conclusion, the research on the antiobesity effects of the underutilized tropical fruit, F. inermis Roxb., has provided valuable insights into its potential therapeutic benefits in managing obesity-related complications. The FF2 extract demonstrated promising outcomes in regulating feed intake, modulating body weight gain, improving lipid profiles, enhancing glucose metabolism, and exerting hepatoprotective and antifibrotic effects. These findings highlight the diverse benefits of FI on metabolic health and its potential as a natural remedy for obesity and its associated risks. These results support further clinical exploration of F. inermis and its development as a nutraceutical product for safer and more effective obesity management strategies.
Abbreviations
Footnotes
Acknowledgments
The authors gratefully acknowledge the Pharmacognosy Division of CMPR, Arya Vaidya Sala, Kottakkal, for plant authentication, and the Biomedical Technology Wing of Sree Chitra Tirunal Institute, Thiruvananthapuram for their expertise and providing facilities for Masson trichrome staining. We also thank Al Shifa College of Pharmacy and KIMS Al Shifa Hospital, Perinthalmanna, for their support and facilities.
Ethical Approval and Informed Consent
All experimental procedures complied with internationally recognized principles for laboratory animal use, in accordance with the recommendations of the Committee for Control and Supervision of Experiments on Animals (CPCSEA). This study was approved by the Institutional Animal Ethics Committee of Al Shifa College of Pharmacy, under Approval No. IAEC 073/21.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
