Abstract
Background
The rising global trend of traditional medicine and complementary and alternative medicine use necessitates a structured approach to ensure patient safety and efficacy. Due to its unique theoretical framework, directly applying modern pharmacovigilance principles to Ayurveda may be insufficient.
Purpose
This study aims to highlight the need for a collaborative approach that integrates traditional Ayurvedic knowledge with modern safety and efficacy evaluation methods. It critiques the limitations of current pharmacovigilance practices, especially in the context of Ayurveda, and proposes improvements to better capture the complexities of traditional medicine.
Methodology
An analysis of the “AyushSuraksha” program, a key initiative for the pharmacovigilance of Ayurveda, Siddha, Unani, and homoeopathy (ASU&H) medicines in India, was conducted. The program’s focus areas, including monitoring medications, specific materials, and procedure-based therapies, were reviewed in light of their efficacy and safety reporting mechanisms. Additionally, the applicability of current pharmacovigilance methodologies, particularly in the context of personalized Ayurveda treatments, was evaluated through case studies and literature reviews.
Results
The “AyushSuraksha” program, while focused on medication and therapy monitoring, overlooks the safety of food items used in Ayurveda. Current pharmacovigilance systems, designed for standardized medications, may be inadequate for Ayurveda, as demonstrated by positive outcomes in autism treatment despite the presence of microbiologically harmful products. Furthermore, existing pharmacovigilance proformas lack a distinction between physicians’ and end-users perspectives, limiting the reporting scope.
Conclusion
A transition to a holistic, evidence-based pharmacovigilance approach that incorporates Ayurvedic assessments and nonlinear herbal interactions is crucial. Separate proformas for physicians and end-users, along with disease-specific questions, are recommended to improve reporting accuracy. Expanding the scope of pharmacovigilance beyond standardized medication monitoring is essential for the evolution and refinement of Ayurvedic science.
Introduction
Over the past two decades, complementary and alternative medicine (CAM) usage has significantly increased. A systemic review and update by Relton et al. have reported its prevalence among general populations globally varying from 9.8% to 76%. 1 In India, among the most frequently used forms of CAM, Ayurveda remains the most popular alternative medicine system being utilized. A study examining the reliance of the elderly population on CAM revealed a wide prevalence of Ayurveda being the most preferred (64.7%) practice, closely followed by homeopathy (62.4%). 2 Against this backdrop of widespread usage, it is crucial to consider the safety of these practices, ensuring that they do not pose any health risks and that their benefits are thoroughly validated.
In earlier times, community-based interventions, whether in terms of medicine or food, were rare and often individual-focused, typically administered within the vicinity of a Vaidya. However, contemporary healthcare has seen a significant shift toward a vast array of community-based medical interventions. This broader expansion means that even trivial health issues may be magnified and escalated into serious health hazards. The primary goal of pharmacovigilance is to monitor and report adverse effects, regardless of whether the medicine is from Ayurveda or allopathy. Given the increasing challenges and concerns surrounding Ayurveda, it is essential to integrate pharmacovigilance into this context to ensure safety and address potential health risks effectively.
Pharmacovigilance is defined as “the science and activities relating to the detection, assessment, understanding, and prevention of adverse effects or any other drug-related problem.” 3 As of the present moment, a universally accepted and operational definition of pharmacovigilance within the realm of Ayurveda remains elusive. A pervasive global concern exists regarding the safety and efficacy of traditional medicine (TM) and the utilization of procedure-based therapies. This heightened apprehension has contributed to widespread reservations among individuals considering the use of complementary and alternative medicine (CAM). 4 In the current landscape, the initiatives undertaken by the Government of India, notably “AyushSuraksha,” are greeted with enthusiasm as they address the prevalent concerns in the field. 5 However, it is important to note that these efforts are still in their early stages of development, reflecting a nascent phase in their implementation. Merely replicating the pharmacovigilance principles and practices from modern science can lead to epistemological compromises, ultimately culminating in the misapplication or malpractice of the science. The present moment calls for evaluating our existing practices to enhance the safety and effectiveness of Ayurvedic implementation. This revision is imperative to transition toward a more evidence-based approach, ensuring the evolution and refinement of Ayurvedic science for optimal outcomes. Raising the concerns regarding the pharmacovigilance of Ayurvedic medicines used in pediatric practice, the article discusses potentially implementable documentation protocols for the clinical safety of such medications.
Key Characteristics: Unveiling the Essentials
Ayurveda prioritizes monitoring diseases before considering medicinal interventions, ensuring a thorough examination of scientifically sound treatment choices. 6 The “AyushSuraksha” program under the Ministry of AYUSH focuses on pharmacovigilance of medicines but it is imperative to extend this oversight to include food items as well, following the noteworthy example set by the Food and Drug Administration (FDA).7, 8 Many diseases stem from dietary and lifestyle factors, so broadening vigilance in “AyushSuraksha” supports comprehensive health. Ayurveda views food as “the ultimate medicine,” highlighting its role in health and disease prevention. 9 Emphasizing dietary practices in pharmacovigilance aligns with Ayurveda’s holistic well-being approach, enhancing the understanding of diet’s impact on health.
Integrating Ayurvedic Epistemology: A Call for a Unique Reporting System
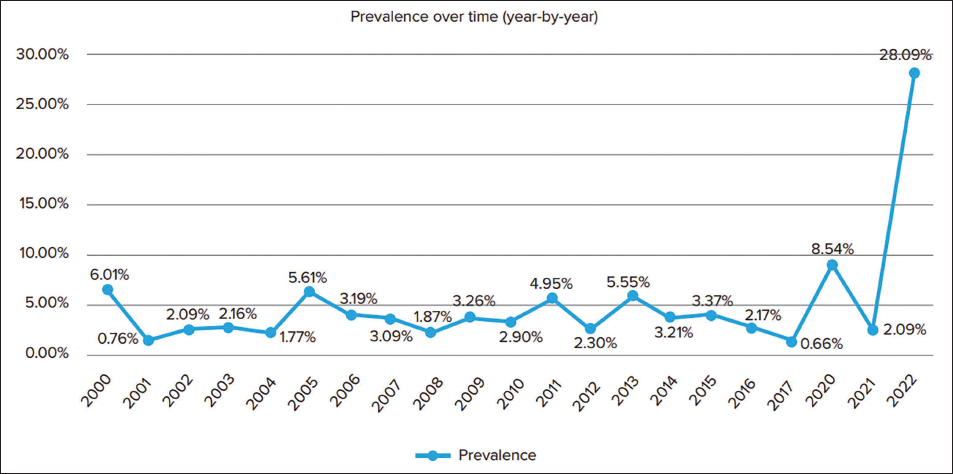
A recent meta-analysis conducted by the World Health Organization (WHO) revealed a prevalence of 5.8% of adverse drug reactions (ADRs) categorized under preventable medication errors. Based on data from the WHO-Uppsala Monitoring System, these findings encompass herbal medicine (Figure 1), including Ayurveda. 10 However, it is important to note that this reporting primarily reflects conventional analysis rather than Ayurvedic principles. Alarmingly, since 2021, there has been a significant rise in the reporting rate of preventable medication errors (Figure 2). 10 This trend poses a substantial risk to the credibility and integrity of Ayurvedic medicine.
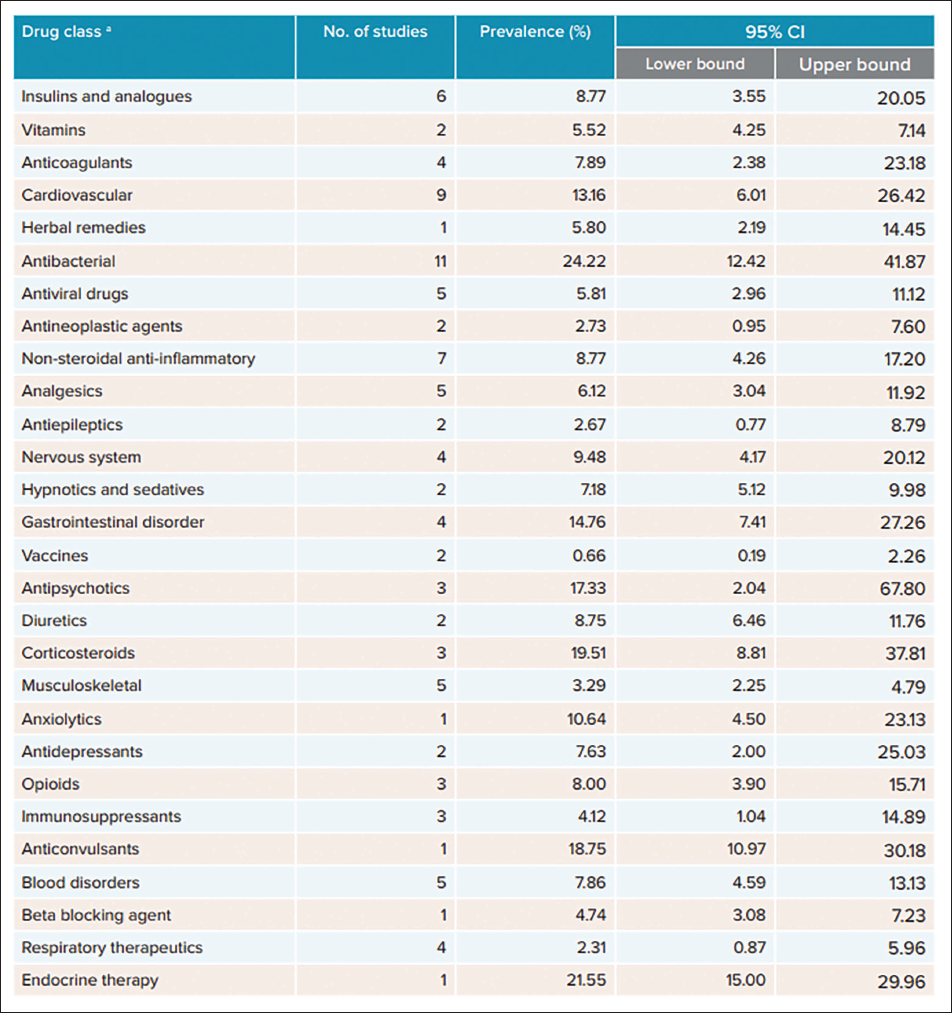
The terms “Samanam,” “Kopanam,” and “Swasthahitam” are conventional classifications found in classical Ayurvedic literature. 12
Substances classified as “Kopana dravyas,” typically associated with the potential for adverse reactions, paradoxically find therapeutic application in numerous cases like their utility for the procedures of vamana, virechana, and so on. As per the default nature of “Samanam,” primarily employed for the management of various ailments, it is theoretically expected that these substances should not induce ADRs. 13
This is attributed to the fact that a “Samana Oushadha” does not generate any new dosha dushti or kopana according to its definition and merely does the pacification. 14 The concept of three Doshas, namely, Vata, Pitta, and Kapha lays the foundation for all aspects of Ayurveda 15 and thus a Samana dravya does not cause any impairment to the balance of these three doshas.
Even for a knowledgeable physician, achieving absolute correctness can be challenging, as clinical decision-making involves analyzing numerous minute variables. The ancient texts, or Acharyas, specifically emphasize the importance of a comprehensive approach. According to their teachings, a practitioner who diligently considers and analyses at least 10 factors in a clinical setting significantly reduces the likelihood of making mistakes in their judgment. 15 This underscores the significance of thorough and multifaceted assessments in ensuring accurate clinical decisions.
In contemporary times, there is a tendency to equate pharmacovigilance in Ayurvedic drugs with that of herbal pharmacovigilance. However, it is crucial to recognize their inherent differences. Merging them could potentially compromise the entire epistemology of Ayurveda. Furthermore, this approach may lead to an undue focus on the toxic components of Ayurvedic herbal medicines rather than appreciating their therapeutic virtues. Maintaining a nuanced and context-specific understanding is essential to ensure the preservation of Ayurveda’s comprehensive and holistic approach to healthcare. Herbs, diseases, and the human body indeed constitute complex and dynamic systems characterized by nonlinear interactions. The conventional linear approaches commonly employed in modern pharmacovigilance may not be entirely suitable for these intricate systems. Recognizing the nonlinear nature of these interactions is essential for developing pharmacovigilance methodologies that better align with the complexities inherent in herbal medicines, diseases, and the human body within the framework of Ayurveda. Characterization of a drug should extend beyond the individual components to encompass the entire plant or substance. Relying solely on marker compounds can indeed be misleading, as the synergistic interactions among various constituents within the total plant contribute to its therapeutic effects. A comprehensive characterization that considers the entirety of the plant’s composition and properties is vital for a more accurate understanding of its impact on the human body within the context of Ayurvedic principles.
The example of tomatoes is intriguing, as it has been studied for its potential benefits in cancer prevention due to compounds like lycopene. Yet, some studies also suggest associations with cancer risk.16, 17
This duality can be attributed to the intricate interplay of multiple factors, including an individual’s overall health, genetic makeup, and the complex interactions among various components within the food. It highlights the importance of considering the holistic context and the need for comprehensive research when evaluating the impact of substances on the human body, which aligns with the holistic principles of Ayurveda.
Learning from Practical Encounters
In certain clinical instances, inappropriate prescriptions have been observed to contribute to ADRs. An example of this is the prevalent occurrence of recurrent respiratory tract infections in children, which is frequently encountered in Ayurveda outpatient departments. Recurrent Upper Respiratory Tract Infection (RURTI) primarily manifests with a presentation dominated by the Kapha and Pitta elements in Ayurveda. The Pitta component in this context is highly responsive. According to Ayurvedic texts, when dealing with a Kapha–Pitta-related condition, it is essential to address the Pitta first. Administering Deepana Pachana dravyas having both appetizer and digestive components, such as Dasamoolakatutrayam Kashaya (a classical Ayurveda decoction) or Vettumaran Gulika (a classical Ayurveda tablet), may result in a slight reduction of Kapha-related symptoms like cough and rhinitis. However, there is a risk of increasing the Pitta, leading to skin eruptions, eventually termed eruptive fever or hand, foot, and mouth disease. In this scenario, it is noteworthy that hand, foot, and mouth disease (HFMD) is an infectious condition. However, the internal conditions conducive to infection are potentially triggered by the improper prescription of Ayurvedic medicine. The use of Ayurvedic remedies that focus on reducing Kapha symptoms without adequately managing the Pitta element may inadvertently create an environment within the body that facilitates the onset of HFMD.
Thirty-two children diagnosed with autism spectrum disorder (ASD) as per Diagnostic and Statistical Manual of Mental Disorders (DSM) criteria from 2007 to 2012 were selected and analyzed for six months after receiving snehapaana with tiktakam ghrita or Mahakalyanakam ghrita at Vaidyaratnam P. S. Varier Ayurveda College. The study assessed changes in hyperactivity, gastrointestinal problems, and behavioral issues using various scales, with results categorized as mild, moderate, or severe.
Gastrointestinal severity index: Mild/Moderate cases reduced (12–1, 13–10), and severe cases increased (7–21).
Visual analog scale for hyperactivity: Mild/Moderate cases decreased (10–5, 20–18), and severe cases increased (1–7).
Child behavior checklist: Mild/Moderate cases decreased (14–10, 15–9), and severe cases increased (3–11).
The treatment showed mixed effects, with improvements in some areas, but an increase in severe cases post-treatment could be attributed due to inaccurate prescription. 18
In ASD, the presence of hyperactivity often prompts physicians to consider prescribing a classical formulation of medicated ghee, Kalyanaka Ghrita, given the dominance of Vata and Pitta elements in this condition. However, if symptoms such as heightened gastrocolic reflex, constipation, and accompanying skin rashes are overlooked, the prescription of such medicated ghee may be deemed irrational. This approach could potentially lead to iatrogenic outcomes, including an initial increase in hyperactivity, subsequent fluctuations, or a lack of response to hyperactivity. Careful consideration of comprehensive symptomatology is crucial to avoid unintended and adverse effects. The level of hyperactivity tends to rise in individuals with ASD when consuming milk and dairy products as well as sweets. This suggests the presence of “Koshta gata Ama,” a condition contraindicated for the use of Ghrita Kalpana (medicated ghee).
In a recent extramural research initiative funded by the Ministry of AYUSH, Government of India, an analysis of Vilwadi Gulika and Rajanyadi Churnam, two common formulations in Ayurveda were conducted using modern scientific tools, specifically 16S rRNA sequencing (Figure 3). The findings revealed the presence of various phyla that are traditionally considered both beneficial and detrimental within the context of microbiology. This underscores a challenge in explaining the mode of action of Ayurvedic medicine through conventional scientific methods, as certain components in the medicines exhibit a complex interplay with microbial elements that may not be fully elucidated by current scientific approaches.
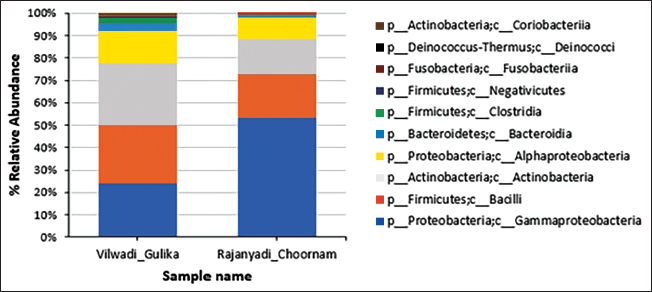
Interestingly, despite the microbiological complexity identified in Rajanyadi Churnam and Vilwadi Gulika through 16S rRNA sequencing, a significant number of children demonstrated positive responses to these Ayurvedic formulations. Particularly, improvements were noted in gut-related issues associated with dysbiosis, resulting in a reduction of symptoms related to autism. This positive clinical outcome suggests that the therapeutic effects of these Ayurvedic medicines might involve mechanisms not entirely captured by the microbiological lens, showcasing the need for a more comprehensive understanding of their mode of action.
Enhancing the Program: Specific Recommendations for Improvement
Currently, the pharmacovigilance reporting proforma comprises 12 question groups. It is proposed to distinguish between the reporting proforma for physicians and the one intended for end-users, such as patients or individuals who have used the formulations or treatments. This separation aims to capture distinct perspectives and information relevant to healthcare professionals and the individuals directly experiencing the effects of the treatments, ensuring a comprehensive and tailored approach to pharmacovigilance reporting. In the existing pharmacovigilance reporting proforma, it is notable that there is currently no question addressing the specific disease condition. Recognizing the significance of this information, it is recommended to include a dedicated question in the proforma that enquires about the patient’s underlying disease or health condition. This addition will enhance the overall understanding of the context in which the treatment or medication is being used, providing valuable insights for pharmacovigilance assessment.
When conducting an Ayurvedic assessment, it is essential to gather comprehensive information across various dimensions. Initiating with basic patient details, including name, age, gender, address, and contact information, the assessment delves into the clinical presentation. This involves evaluating the status of Agni (digestive fire), and doshas (Vata, Pitta, Kapha), and identifying specific tissues or systems affected (Dushyas). The Koshta or alimentary tract status is explored, considering the bowel movement nature and any history of abdominal discomfort. Information about the Ayurvedic drug, such as its name, perceived qualities (Gunas), and the observed beneficial or adverse effects, is then gathered. An overall assessment examines the predominant Gunas in the disease and any changes observed before and after treatment. Additional insights into the patient’s lifestyle, diet, and concomitant medications contribute to a holistic understanding. Encouraging feedback ensures a collaborative approach, fostering a detailed comprehension of the patient’s condition and treatment outcomes in Ayurvedic practice.
Conclusion
In the exploration of pharmacovigilance concerns in pediatric Ayurveda, it is recommended that actionable protocols be created for documentation. The current definition of pharmacovigilance within Ayurveda remains elusive, and global worries about TM safety persist. Despite recognizing the value of efforts such as ’AyushSuraksha,’ they are still in the process of development. Ayurveda prioritizes disease monitoring and advocates for vigilance in food-related aspects. Being a proponent of a holistic pharmacovigilance approach cautions against conventional classifications and emphasizes nonlinear herbal interactions. Practical examples highlight the importance of considering comprehensive symptomatology in Ayurvedic treatments. The authors conclude by providing concrete suggestions with specific recommendations for improving pharmacovigilance reporting and the evaluation of Ayurvedic practices.
Abbreviations
Footnotes
Acknowledgments
The authors express their sincere gratitude to the ATIDHI (Ayurveda and Therapeutic Integration for Developmental Habilitative Intervention) group and Vaidyaratnam P. S. Varier Ayurveda College for their invaluable contributions and support throughout this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
All the background studies included here were conducted with due diligence, obtaining informed consent from all participants in accordance with ethical standards. Measures were taken to safeguard participants’ privacy and rights, ensuring the confidentiality and anonymity of their data. The background studies were approved by the Institutional Ethics Committee (IEC) of Vaidyaratnam P. S. Varier Ayurveda College, India. (Proceedings No: IEC/CI/24/17).
Funding
The study received funding from the ATIDHI (Ayurveda and Therapeutic Integration for Developmental Habilitative Intervention) group, ensuring the seamless execution and accomplishment of our research objectives.
