Abstract
Background
Sodium butyrate (SB) used to increase oxidative stress was reduced and the glutathione redox pathway was boosted. It inhibits proliferation, induction of differentiation, and induction or repression of gene expression and it has also anti-inflammatory action. This is endorsed for suppression of nephrotic symptoms.
Objectives
The present study was carried out to evaluate the nephroprotective activity of SB on gentamicin-induced nephrotoxicity in rats.
Materials and Methods
Present research work was designed to estimate pharmacological potential of SB in gentamicin-induced nephrotic disorder. The pharmacological prospective was estimated in gentamicin-induced rats through the oral administration of SB formulation 200 mg/kg and 300 mg/kg BW.
Results
The study observed that the SB significantly protects the kidneys from gentamicin-induced nephrotic disorders. Gentamicin 100 mg/kg for 7 days was used for induction of nephrotoxicity in Wistar rats by intraperitoneal administration. The administration of SB was observed to decrease the occurrence of glomerular congestion, vascular congestion, epithelial desquamation, inflammatory cell build-up, and kidney cell necrosis induced by gentamicin. The dose of SB maintained the gentamicin-induced increased creatinine (0.62±0.05), serum urea (18.10±0.79), uric acid (2.10±0.28), and blood urea nitrogen (BUN) (17.56±0.55) levels. This fact is further sustained by the histopathological examinations.
Conclusion
Sodium butyrate formulation with antinephrotic activity in maintaining uremic solutes such as urea, uric acid, creatinine, BUN, ketone, urobilinogen, glucose, specific gravity, and urine pH in Wistar rats with gentamicin-induced glomerulonephritis (GN). The SB was administered to the animals at doses of 200 and 300 mg/kg body weight. Pretreatment with the drug showed promising results in maintaining elevated parameters in nephritic rats within a short duration.
Introduction
Nephrotoxicity is a class of disorders that damage the parts of the kidney and create a severe illness that requires speedy treatment.1, 2 The term nephrotic disorder (ND) covers a collection of clinical findings associated with massive loss of protein in urine via the kidney and also characterized by the occurrence of protein excretion (proteinuria ≥ 3.5 g per 24 h), edema, hyperlipidemia (≥220 mg/dL), lipiduria, and hypoalbuminemia (<2.5 g/dL).3–5 The signs of edema occur due to localized fluid increase in interstitial tissues which attributed to mechanisms involving sodium retention in the face, legs, and stomach. 6 NDs, also known as nephrotic syndrome, refer to a group of conditions that affect the kidneys, leading to increased presence of protein in urine, reduced protein levels in the blood, elevated cholesterol levels, and swelling in various parts of the body, particularly the legs and around the eyes. 7 These disorders primarily result from injury to the bundles of tiny blood vessels in the kidneys responsible for filtering waste and surplus water from the bloodstream to produce urine. 8 Sodium butyrate used to increase oxidative stress was reduced, and the glutathione redox pathway was boosted. It inhibits proliferation, induction of differentiation, and induction or repression of gene expression and it also has anti-inflammatory action. Sodium butyrate protects the kidneys from gentamicin-induced NDs. 9
Prevalence and Incidence of Nephrotoxicity
Nephrotoxicity, which damages the kidneys, significantly contributes to the global kidney disease burden. It is responsible for 14%–26% of acute kidney injury (AKI) cases, especially in hospitalized patients and those in intensive care units (ICUs), with incidence rates reaching up to 60%. High-risk patients can experience contrast-induced nephropathy at rates up to 50%, while chronic exposure to heavy metals and nephrotoxic herbal remedies increases prevalence, particularly in developing countries. Worldwide, nephrotoxicity is a leading cause of AKI, affecting 13.3 million people annually and often leading to chronic kidney disease (CKD). CKD impacts about 10% of the global population and can advance to end-stage renal disease, requiring expensive treatments like dialysis. Mitigation strategies include raising awareness, enforcing stricter regulations, investing in research for safer alternatives, and minimizing environmental pollutants.
Prevalence
Drug-induced nephrotoxicity: Medications are a major cause of nephrotoxicity, responsible for about 20% of AKI cases in hospitals. Common nephrotoxic drugs include nonsteroidal anti-inflammatory drugs (NSAIDs), certain antibiotics (such as aminoglycosides and vancomycin), and chemotherapy agents like cisplatin.
Environmental and occupational exposure: Nephrotoxicity is also caused by exposure to heavy metals (such as lead and cadmium), industrial chemicals, and environmental toxins. In areas with high levels of environmental pollution, the prevalence of nephrotoxicity can be considerable.
Incidence
Acute kidney injury: AKI, frequently caused by nephrotoxic agents, has an incidence rate that varies by population and setting. Hospitalized patients experience higher rates, with studies indicating incidences between 5% and 30%. In critically ill patients, the incidence can exceed 50%.
Chronic kidney disease: Sustained or repeated exposure to nephrotoxic agents can lead to CKD. The global prevalence of CKD is estimated at around 13.4%, with nephrotoxic agents playing a significant role in its development. The incidence of CKD varies worldwide, with higher rates in areas with prevalent nephrotoxic exposures. 10
Contribution to Kidney Disease Burden
Hospitalization and Healthcare Costs
Nephrotoxicity-related AKI and CKD: These conditions result in higher hospitalization rates and extended hospital stays. Managing nephrotoxicity demands significant healthcare resources, such as dialysis, monitoring, and treating complications.
Financial burden: The costs include direct medical expenses and indirect costs from loss of productivity and long-term care needs.
Morbidity: Patients with kidney disease caused by nephrotoxicity often suffer from a reduced quality of life due to chronic pain, fatigue, and decreased physical function. Morbidity related to nephrotoxicity also includes a higher risk of cardiovascular diseases, infections, and other complications due to impaired kidney function.
Mortality: Nephrotoxicity greatly increases mortality risk. AKI, especially in critically ill patients, has a high mortality rate, ranging from 20% to 50%. The progression of CKD, often worsened by nephrotoxic exposure, is linked to higher mortality due to cardiovascular events and complications from end-stage renal disease (ESRD).
Public health and global impact: The increasing prevalence of diabetes and hypertension, both of which are risk factors for nephrotoxicity, heightens the global burden of kidney disease. The growing aging population also contributes to this burden, as older adults are more vulnerable to drug-induced kidney damage. In low- and middle-income countries, the impact of nephrotoxicity is often greater due to limited access to safe medications, inadequate healthcare infrastructure, and higher exposure to environmental toxins.
Children
Minimal change disease (MCD): Most common, typically ages 2–6.
Focal segmental glomerulosclerosis (FSGS): Less common, seen in older children/adolescents.
Congenital nephrotic syndrome: Rare, presents in the first few months of life.
Adults
FSGS: Common, more prevalent among African Americans, can occur at any age.
Membranous nephropathy (MN): Common in white populations, typically middle-aged to older adults.
Diabetic nephropathy: Common in middle-aged/older adults with diabetes.
Amyloidosis: More common in individuals over 50.
Common Etiologies
Primary Causes
MCD: Often idiopathic, sometimes linked to infections or allergies.
FSGS: Often idiopathic, can be secondary to obesity, hypertension, and HIV.
MN: Often idiopathic, can be linked to autoimmune diseases, infections, and malignancies.
Secondary Causes
Diabetic nephropathy: Due to prolonged hyperglycemia in diabetes.
Amyloidosis: From systemic amyloid deposits, often related to multiple myeloma or chronic inflammation.
Lupus nephritis: Immune complex deposition in systemic lupus erythematosus.
Infections: Hepatitis B/C, HIV, and malaria.
Medications/toxins: NSAIDs, penicillamine, and gold therapy.
Potential Therapeutic Strategies for Nephrotic Disorders
Medications
Corticosteroids: First-line treatment for conditions like MCD.
Immunosuppressants: Drugs like cyclophosphamide, cyclosporine, and tacrolimus for steroid-resistant cases.
Angiotensin-converting enzyme (ACE) inhibitors: Used to reduce proteinuria and protect kidney function.
Lifestyle and Supportive Care
Dietary modifications: Low-sodium and low-protein diets.
Diuretics: To manage edema.
Statins: To control hyperlipidemia.
Targeted Therapies
Biologic agents: Such as rituximab for refractory cases.
Sodium butyrate has shown potential protective effects against nephrotoxicity by reducing inflammation and oxidative stress in the kidneys. This could make it a promising therapeutic strategy for preventing or mitigating kidney damage caused by toxic substances. 11
Material and Methods
Collection of sodium butyrate: Sodium butyrate (SB) salt was collected from CDH chemicals Pvt Ltd, Daryaganj, New Delhi, and prepared a formulation SB-300 mg tablet.
Experimental rats: Wistar male rats weighing between 160 and 185 g were utilized for the research after prior approval of IAEC, the IAEC-approved protocol No. HIPER/IAEC/41/13/23. All the animals were maintained as per Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA) guidelines. 11 The rats were fed with a regular pellet diet and unrestricted access to fresh water. All animals underwent a 1-week acclimatization period in laboratory conditions in accordance with CPCSEA guidelines before experimentation. 12
Experimental design: Wistar experimental rats remained allocated into five groups consisting of six animals. Group I assisted in place of the normal group, whereas Group II gave gentamicin at a dosage of 100 mg/kg/day for a duration of 7 days. Group III was treated through sodium butyrate tablet at a dose of 200 mg/kg b.w. Group IV was treated with sodium butyrate tablet 300 mg/kg/day. And Group V was cured through standard treatment drug, cystone 500 mg/kg b.w. daily continued for 35 days. 13
Assessment of renal function through serum: Blood urea nitrogen, urea, uric acid, and creatinine levels alongside total bilirubin and total protein in blood were analyzed by autoanalyzer.
14
BUN (blood urea nitrogen) mg/dL Creatinine mg/dL Urea mg/dL Uric acid mg/dL Potassium mmol/L Calcium mmol/L Magnesium mmol/L Sodium mmol/L
15
Urine profile: Some parameters in urine sample were analyzed by a commercial test kit.
Ketone mg/dL Blood Ery/µL Urobilinogen mg/dL Leukocyte Leu/µL Bilirubin mg/dL Nitrite Nit/µL Glucose mg/dL SG Protein mg/dL pH
16
Histological analysis: Kidney tissues were obtained on the last day after the scarification of rats and stored now 10% formalin buffer solution and tissues of the rat’s section were examined under the light microscope. 17
Statistical examination: Each set of data was presented as Mean ± SEM, and then one-way ANOVA was performed using Bonferroni’s multiple comparison test. A #p < 0.05 indicates statistical impact compared to the normal rat’s group, whereas p < 0.05 indicates statistical impact compared to the disease rat’s group. NS denotes nonsignificant comparisons among the group. 18
Results
Assessment of Plasma Profile
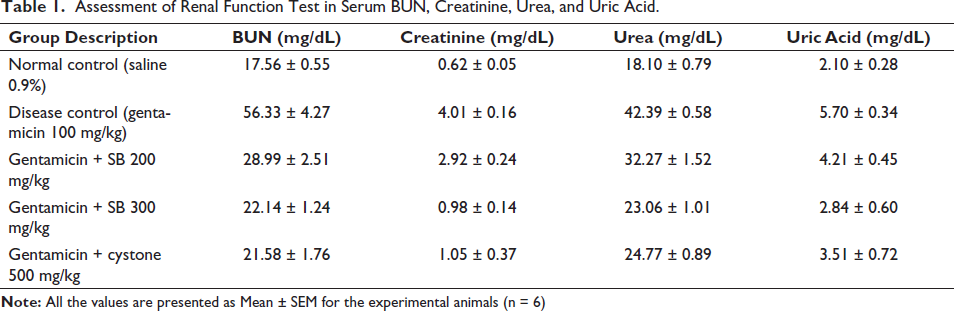
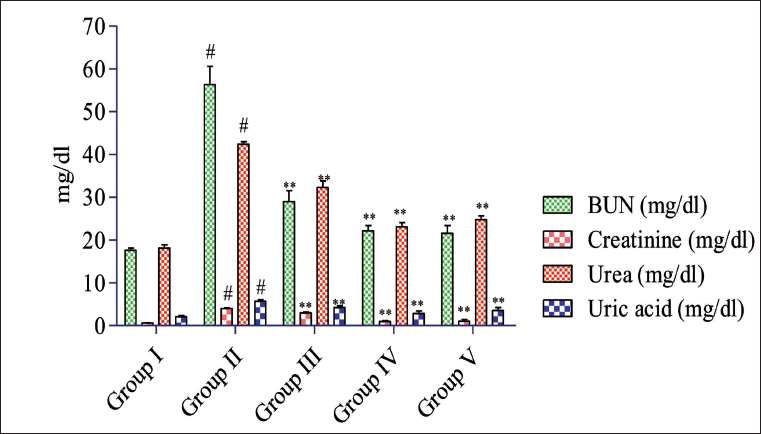
Rats treated with gentamicin exhibited a notable (#p < 0.05) rise in the overall BUN levels (56.33 ± 4.27 mg/dL), creatinine (4.01 ± 0.16 mg/dL), urea (42.39 ± 0.58 mg/dL), and the uric acid (5.70 ± 0.34 mg/dL) in the blood samples compared to normal rats group as shown in Table 1 and Figure 1. Conversely, both the standard and sodium butyrate treatment group’s impact (p < 0.05) maintained these raised levels in comparison with the gentamicin-administered rat, with values approaching those of the normal control group. Notably, the sodium butyrate 300 mg/kg treated group demonstrated further significant improvement (#p < 0.05) in maintaining total BUN (22.14 ± 1.24), creatinine (0.98 ± 0.14), urea (23.06 ± 1.01), and uric acid (2.84 ± 0.60) mg/dL, reaching levels closer to those of the normal control group.
Assessment of Renal Function Test in Serum BUN, Creatinine, Urea, and Uric Acid.
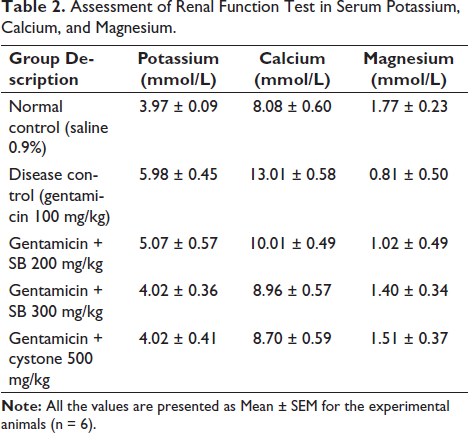
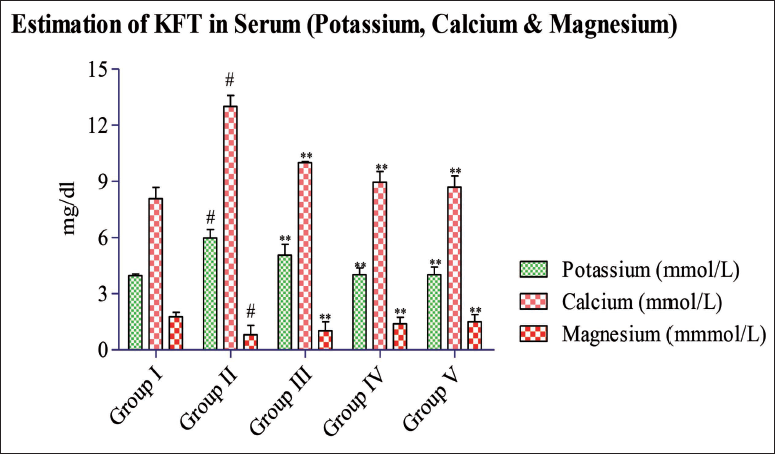
The analysis of kidney function tests (KFTs) in serum reveals a significant (#p < 0.05) rise in serum potassium (5.98 ± 0.45 mmol/L) and calcium (13.01 ± 0.58 mmol/L) levels in the disease group, while the magnesium level (0.81 ± 0.50 mmol/L) is notably decreased as shown in Table 2 and Figure 2. Conversely, treatment using formulated doses of 200 and 300 mg/kg per day, as well as the standard drug (cystone), impact (#p < 0.05) decreases the raised serum calcium and potassium values, whereas magnesium value remains depressed compared to the disease rat group. Subsequent treatment using the formulated dose of 200 and 300 mg/kg as well as cystone significantly (p < 0.05) maintains the magnesium level, with SB 200 mg/kg (5.07 ± 0.57) and SB 300 mg/kg (4.02 ± 0.36) comparable to that of the normal control group (3.97 ± 0.09) mmol/L.
Assessment of Renal Function Test in Serum Potassium, Calcium, and Magnesium.
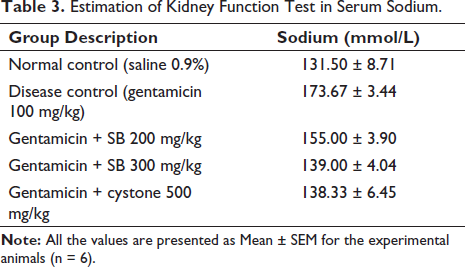
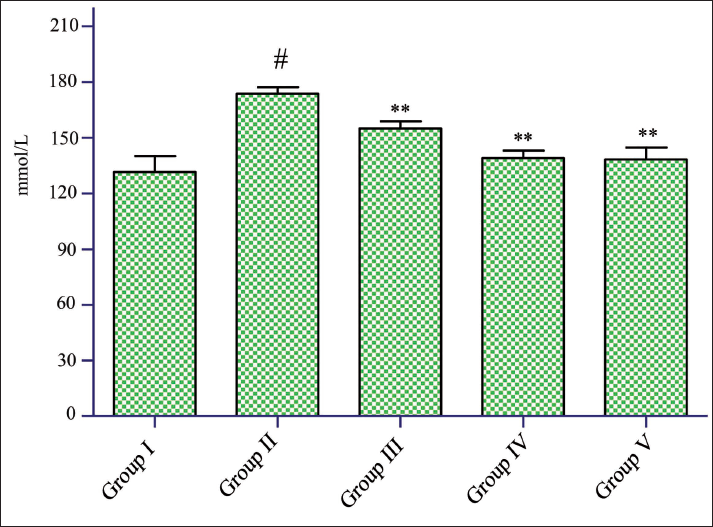
Regarding serum sodium levels, a notable rise (#p < 0.05) was noted in the disease rat group (173.67 ± 3.44 mmol/L). However, treatment with the formulated doses of 200 mg/kg (155.00 ± 3.90 mmol/L) and 300 mg/kg (139.00 ± 4.04 mmol/L), as well as standard treatment (cystone 500 mg/kg), impact (p < 0.05) conserved sodium values. By the end of the study, the standard treatment group produced impact (p < 0.05) improvement in sodium level (138.33 ± 6.45 mmol/L) data shown in Table 3 and Figure 3.
Estimation of Kidney Function Test in Serum Sodium.
Estimations of Urine Volume and Kidney Weight
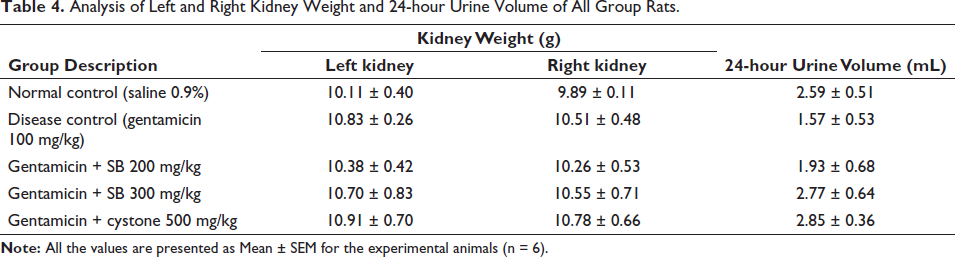
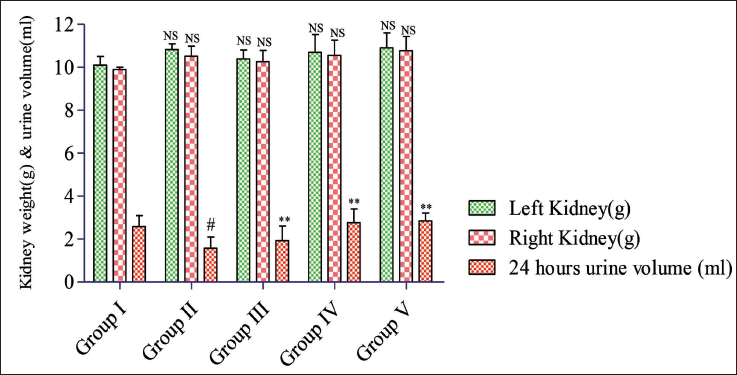
The group of rats treated with gentamicin impact (#p < 0.05) decreased 24-hour urine volume output compared to the control rat group rats and each of the treatment group rats. However, the formulation SB 300 mg/kg exhibited a significant (p < 0.05) normalized urine volume over a 24-hour period (2.77 ± 0.64 ml), similar to that of the normal rat control (2.59 ± 0.51 mL). There were nonsignificant (NS) variances observed in the weights of left and right kidneys within or between the groups as shown in Table 4 and Figure 4. The average weight of the left kidney ranged between (10.11 ± 0.40) g, and for the right kidney, it was (9.89 ± 0.11) g.
Analysis of Left and Right Kidney Weight and 24-hour Urine Volume of All Group Rats.
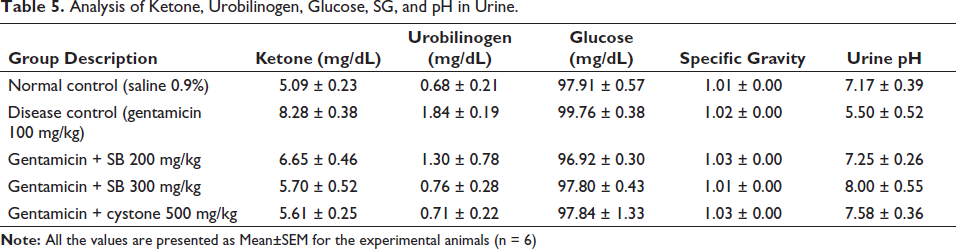
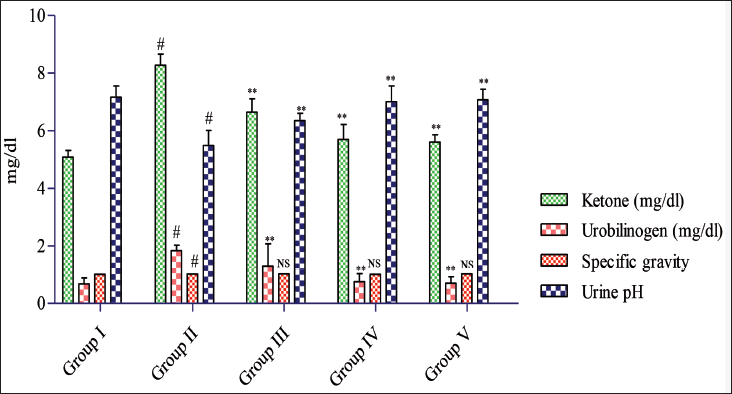
Administration of gentamicin for 100 mg/kg/day resulted in a significant rise (#p < 0.05) in the ketone (8.28 ± 0.38) mg/dL, urobilinogen (1.84 ± 0.19) mg/dL, and the decrease in urine pH (5.50 ± 0.52). Glucose (99.76 ± 0.38) mg/dL and the specific gravity (1.02 ± 0.00) showed no significant (NS) changes compared to the control rat group. However, treatment with the SB 200 mg/kg and 300 mg/kg/day proved highly effective, significantly (p < 0.05) maintaining raised levels of the ketone (5.70 ± 0.52) mg/dL, urobilinogen (0.76 ± 0.28) mg/dL, glucose (97.80 ± 0.43) mg/dL, specific gravity (1.01 ± 0.00), and the pH (8.00 ± 0.55) induced with the gentamicin. Similarly, the standard treatment rat group also impact (p < 0.05) improved these raised levels, with ketone (5.61 ± 0.25) mg/dL, urobilinogen (0.71 ± 0.22) mg/dL, glucose (97.84 ± 1.33) mg/dL, specific gravity (1.03 ± 0.00), and pH (7.58 ± 0.36) matched with the disease control group as shown in Table 5 and Figures 5 and 6. 19
Analysis of Ketone, Urobilinogen, Glucose, SG, and pH in Urine.
Histological Studies
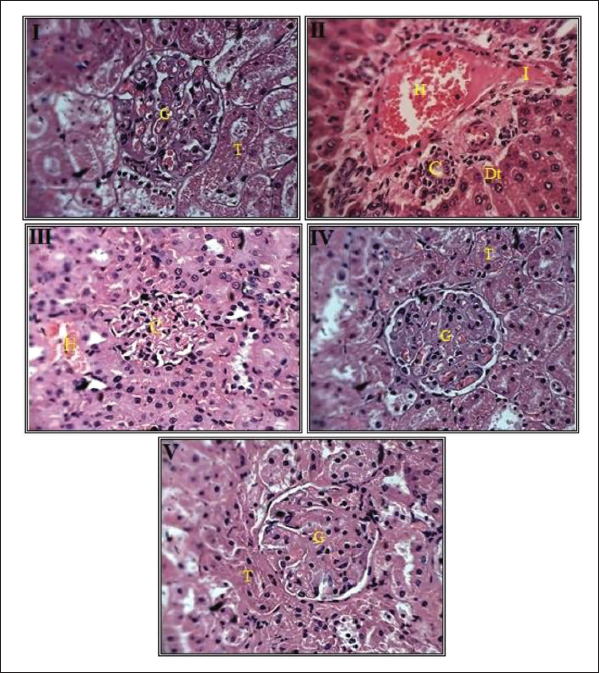
Histopathological examination of kidney tissue from all rat groups was conducted using hematoxylin and eosin staining. In Group I (control rats), kidney sections displayed usual glomeruli, Bowman’s space, and kidney tubules. In Group II (gentamicin-induced group), severe glomerular deterioration, tubular tissue necrosis, tubular cell deterioration, and dilation of Bowman’s space infiltrated by inflammatory cells were observed. Groups III and IV, treated with the SB formulation at doses as 200 and 300 mg/kg/day, consequently exhibited renal corpuscles and tubules resembling normal structures, with some tubular regeneration. Group V, treated with the standard drug cystone (500 mg/kg b.w.), also maintains normal renal corpuscles and tubules, resembling a mostly normal structure with tubular regeneration as shown in Figure 7.20, 21
G—Normal Glomerulus, T—Normal Tubule, H—Hemorrhage in Glomerulus and Renal Space, Dt—Degeneration of Epithelium and Renal Tubule, I—Inflammation, and C—Compression of Glomerulus and Renal Space.
Conclusion
The current study aimed to assess the effectiveness of a sodium butyrate formulation with antinephrotic activity in maintaining uremic solutes such as urea, uric acid, creatinine, BUN, ketone, urobilinogen, glucose, specific gravity, and urine pH in Wistar rats with gentamicin-induced glomerulonephritis (GN). The sodium butyrate was administered to the animals at doses of 200 mg/kg and 300 mg/kg body weight. Pretreatment with the drug showed promising results in maintaining elevated parameters in nephritic rats within a short duration. Serum profiling in gentamicin-induced ND in rats revealed significantly higher levels of urea (42.39 ± 0.58 mg/dL), uric acid (5.70 ± 0.34 mg/dL), creatinine (4.01 ± 0.16 mg/dL), and BUN (56.33 ± 4.27 mg/dL) in disease group matched to the normal control group. Based on calculations involving the amount of urine produced by the test animals and the variations between uremic and normal rats in 24-hour urine amount, an average of 2.77 ± 0.64 mL of urine per day was analyzed with progress in the urine color in SB 300 mg/kg group. Histopathological analysis of the kidney clearly indicated remodeling and robust glomerular capillaries. The study also suggests that sodium butyrate effectively removes urea from uremic animals, providing beneficial effects to rats with renal insufficiency. SB-200 and SB-300 demonstrated significant defense alongside gentamicin-treated ND, as proven by the body weight, biochemical, and histological parameters.
Abbreviations
Footnotes
Acknowledgments
The authors are thankful to the school of pharmacy, Sharda University, Greater Noida, UP, India for providing institutional facilities.
Conflict of Interest
The authors declare no conflicts of interest.
Ethical Approval and Informed Consent
The approval has been received from Institutional Animal Ethical Committee (IAEC-approved protocol No. HIPER/IAEC/41/13/23). All the animals were maintained as per Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA) guidelines.
Funding Sources
None
