Abstract
Background
Statin-associated muscle symptoms (SAMS), the commonest statin-induced adverse effects, could hinder patient adherence and possibly lead to discontinuation of therapy, which then increases patients’ risks for developing cardiovascular events. Although statins are the most frequently prescribed lipid-lowering medication, the occurrence and severity of SAMS among the Malaysian population are less known.
Objectives
To examine the likelihood and types of muscle symptoms attributable to statins, and the risk factors for developing the symptoms among statins users.
Materials and Methods
This was a cross-sectional study conducted with convenience sampling of patients with type 2 diabetes mellitus undergoing phlebotomy at an endocrinology clinic of a secondary hospital in a suburban city. Demographic and clinical data for the evidence of SAMS were retrieved from the hospital information system. A total of 214 patients were screened, and 50 subjects were recruited for interviews based on the study’s inclusion and exclusion criteria. Muscle symptoms that were evident through clinician notes, and self-reported signs and symptoms by patients during the interview were assessed to determine whether they were SAMS or not. This was done using a pre-structured published questionnaire via patient interview, and the likelihood was then determined using the SAMS-clinical index (SAMS-CI) tool.
Results
In this cohort, the probability of having had a statin-associated muscle adverse event was 0.48, i.e., 48% “probable” and “possible” likelihood of SAMS having occurred for a patient who consumed a statin. The mean SAMS-CI score was 6.32 ± 0.470 (±7.4%) [CI: 5.85–6.79], which fell under an “unlikely” category. The most frequent type of muscle symptom was myalgia, which was experienced as muscle aches, stiffness, cramps, symptoms worsening after exercise, muscle tenderness, and trouble moving knees or arm joints. These symptoms were proximal and symmetrical in nature and commonly involved large muscle groups such as the thighs, buttocks, calves, and back muscles. An elevation in creatinine kinase was not commonly observed.
Conclusion
A minority (6%) of the patients that we sampled experienced SAMS, which were probably attributable to statins. Nevertheless, caution must be exercised for female patients with a familial history of heart disease being prescribed with the lipophilic statins, atorvastatin, and simvastatin, and those who report aches in the large muscle groups.
Keywords
Introduction
Statins are the first-line treatment for dyslipidemia, which is a condition in which there are elevations of low-density lipoprotein cholesterol (LDL-C) and triglyceride levels while the high-density lipoprotein cholesterol (HDL-C) level is reduced. Statins are currently the most frequently prescribed lipid-lowering medication in clinical practice in Malaysia. The most prescribed statins include simvastatin, atorvastatin, and rosuvastatin, with varying doses for each type. Statins’ lipid-lowering effects are attributed to their actions on 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase, the rate-limiting enzyme in hepatic cholesterol synthesis, which makes it an excellent target for statins to exert their lipid-lowering effects on patients with dyslipidemia. 1 Statins have been proven to reduce LDL-C levels by 20%–50% and triglyceride levels by 10%–20%, and increase HDL-C levels by 5%–10%. 2 It has also been proven that statins can significantly reduce the risk of cardiovascular diseases, such as myocardial infarction and stroke. 3
Statins are generally safe and well-tolerated, and the cardiovascular benefits have been proven to outweigh the risks of adverse effects.4, 5 However, the musculoskeletal adverse effects, which are known as statin-associated muscle symptoms (SAMS), have at times been severe enough to cause life-threatening conditions, for instance, with rhabdomyolysis. These adverse effects, when intolerable, can lead to non-adherence and even discontinuation of statin therapy by the patients. 6 A previous study reported that the discontinuation rate of statins due to muscle symptoms was high (up to 75%) within the first year of therapy. 7
SAMS can be explained as muscle symptoms attributed to statin therapy that may be accompanied by biochemical changes, such as elevations of creatine kinase (CK). SAMS cover a wide range of clinical presentations. According to the American College of Cardiology/American Heart Association (ACC/AHA), the spectrum of SAMS ranges from myalgia, myositis, and rhabdomyolysis in order of increasing severity. 4 Myalgia is defined as muscle ache or muscle weakness without CK elevation; myositis refers to muscle symptoms with an increase in CK levels. In contrast, rhabdomyolysis refers to muscle symptoms with significant CK elevation, typically 10 times the upper limits of normal readings. 8 Besides, SAMS may be asymptomatic, with only CK elevation evident. SAMS were recorded to typically occur within 4–6 weeks after statin initiation, after a statin dose increase, or due to interaction between statins and other drugs. The determination of SAMS is still not clearly elucidated as there is no gold standard method of measuring muscle symptoms accurately. Nevertheless, the likelihood of muscle symptoms attributable to statins can be assessed by the SAMS Clinical Index (SAMS-CI), as proposed by the National Lipid Association (NLA) Task Force on Statin Safety. 10 The SAMS-CI does not give a definitive association, instead, it categorizes the association into: “unlikely,” “possibly,” and “probably.” This categorization is done based on a scoring system that takes into account the regional distribution or muscle symptom patterns, the onset, and the patient’s response upon discontinuation and statin rechallenge. Although it has yet to be tested on a larger population, SAMS-CI has been proven to demonstrate inter-rater reliability and validity in a relatively large population. 11 Additionally, the comprehensiveness, brief nature, standardized questions, easily comprehensible language, and easy interpretation of scores make it a very promising tool for diagnosing SAMS. It was agreed that SAMS-CI was suitable for use in SAMS assessment by physicians and non-physicians, which can be convenient in clinical practice. 9
Despite statins being the commonest lipid-lowering drug prescribed in Malaysia, the extent to which the adverse effects impacted the population is less known. According to previous studies, the most commonly reported muscle symptoms related to statin use were muscle pain, cramps, and weakness. 9 Therefore, in this study, we aimed to determine the prevalence of SAMS and the pattern of occurrence, and to investigate the correlation between patient demographic and clinical variables with SAMS.
Materials and Methods
Ethics Approval
Ethics approval was obtained from the Research Ethics Committee of MARA University of Technology with the approval number REC/04/2022. This study was conducted in accordance with the Helsinki Declaration and the International Conference of Harmonization of Technical Requirements for Registration of Pharmaceutical Human Use (ICH) Guideline for Good Clinical Practice.
Study Design and Sampling
This was a cross-sectional study of patients with type 2 diabetes mellitus undergoing phlebotomy and sampled conveniently at the endocrinology clinic of a suburban hospital. With a 95% confidence interval and a 5% margin of error, the sample size was calculated using the Raosoft sample size calculator based on population size of 824,168, which was the number of adults aged 18 and over in the state of Selangor in 2023, and a prevalence of 13.5% statin users according to the data reported by the National Health Morbidity Survey in 2019. The minimum sample size required to give the study sufficient power was 48.
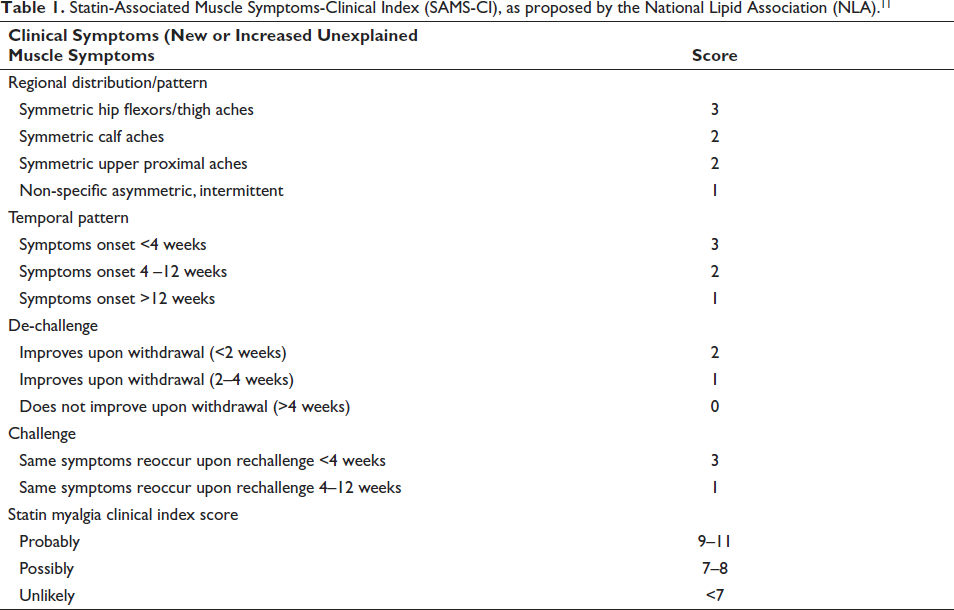
The list of patients coming for phlebotomy appointments to the clinic from January to June 2023 was used as a starting point to screen for potential study participants. Patient interviews were conducted during their phlebotomy appointment for their convenience. During the appointment, patients were briefed face-to-face about the study; a participant information sheet was handed out, and written consent was obtained before the interview. SAMS were assessed using a pre-structured published questionnaire, and the likelihood was determined using SAMS-CI following the interview (Table 1).
Statin-Associated Muscle Symptoms-Clinical Index (SAMS-CI), as proposed by the National Lipid Association (NLA). 11
Inclusion and Exclusion Criteria
All the patients from the endocrine clinics aged between 18 and 75 years who were diagnosed with dyslipidemia, and who were on statins for at least 6 weeks were included. However, patients who had muscular pain prior to the initiation of a statin, who had creatinine levels more than 2 mg/dl (normal range 0.59–1.35 mg/dl) or alanine transaminase levels of more than two times the upper limit of regular readings (normal range 0–33 IU/L), those with inherited metabolic disorders or with pre-existing muscle or joint diseases were excluded to minimize potential confounding factors which may impede the estimation of true SAMS. Patients diagnosed with terminal illnesses were also excluded.
Sociodemographic and Clinical Data Collection
Following the approval granted by the REC, demographic and clinical data of patients registered to the endocrine clinics were retrieved from the electronic hospital information systems. Patients were anonymized, and their demographic data, such as registration number, gender, age, as well as clinical data, including the number of concomitant medications, co-morbidities, and family history of chronic diseases, were collected from the hospital’s electronic systems. Patients’ records were screened based on the inclusion and exclusion criteria and the eligible patients were shortlisted and were invited to participate in the interview.
Patient Interview
The interview was conducted in either English or Malay, according to the participant’s preferred language. The pre-structured questionnaire included information on the onset, nature, and the degree to which the muscle symptoms affected patients’ lives. Patients with exposure to statins, who developed SAMS, and who had shown progressive muscle symptoms were identified via entries by the nurses and clinicians within the progress notes. Clinical indicators included fatigue, muscle pain, muscle soreness, muscle stiffness, muscle tenderness, muscle cramps, and joint pain. In any event, where rhabdomyolysis was suspected, the clinician in charge was informed. This is because it may have been necessary to discontinue the medications, and the CK levels were then measured.
The likelihood of the patient’s muscle symptoms being attributed to statins was analyzed and categorized under the “unlikely,” “possibly,” and “probably” categories according to the scoring system of SAMS-CI (Table 1). A score of less than 7 was categorized as “unlikely”, 7–8 was categorized as “possibly” and a score of 9–11 as “probably.” The SAMS-CI categorization was also used to assess the correlation between sociodemographic data and clinical characteristics of participants with the occurrence of muscle symptoms associated with statin therapy.
Data Analysis
The Statistical Package for the Social Science (SPSS) software version 28.0 was used to perform the statistical analyses. Descriptive statistics were used to describe the distribution of demographic and clinical profiles of respondents as well as the types of muscle symptoms experienced. A normality test was conducted using Kolmogorov Smirnov. Inferential statistical analyses, i.e., Mann–Whitney U test and Kruskal–Wallis tests, were used to investigate the occurrences and the likelihood of SAMS adjusted for sociodemographic variables and clinical characteristics of the participants. A p < .05 was set to indicate a significant association for all analyses.
Results
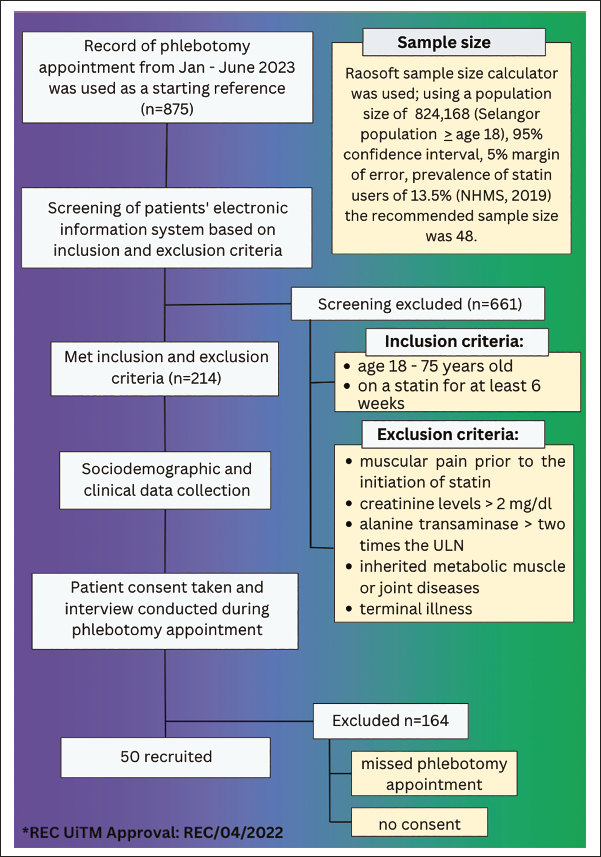
Of the 875 patients listed for phlebotomy appointments during the period, 214 patients from the endocrine clinics who met the inclusion criteria were shortlisted for an interview. From this number, 50 patients who presented on the phlebotomy dates and who consented to be included were selected to participate in this study (Figure 1). The procedures followed the COVID-19 Standard Operating Procedure (SOP).
Process of Selecting Study Participants.
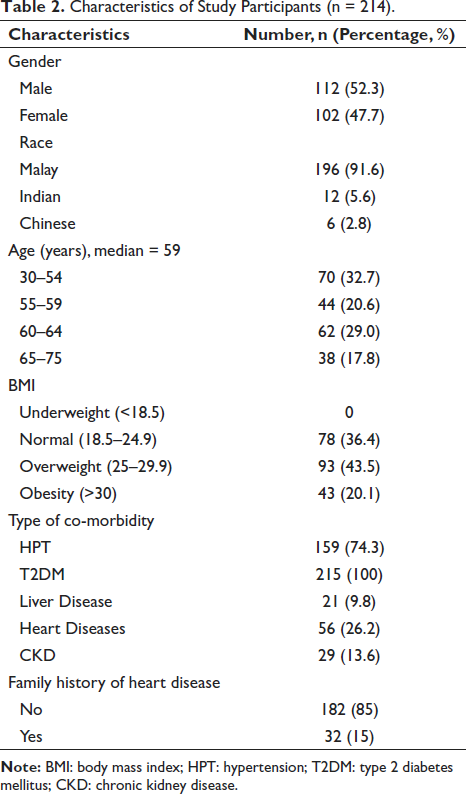
The 214 patients’ demographic information and medical history are summarized in Table 2. Subjects’ median age was 59. They comprised 112 males (52.3%) and 102 females (47.7%). A majority of the study participants were Malay (91.6%), followed by Indians (5.6%) and Chinese (2.8%). For the body mass index (BMI), there were no underweight patients. A majority of the participants had overweight BMI (43.5%), while the remainder were normal (36.4%), and obese (20.1%). All the subjects were diagnosed with type 2 diabetes mellitus. Most of them had hypertension (74.3%), and a proportion had heart diseases (26.2%), chronic kidney disease (13.6%), and liver diseases (9.8%). A family history of heart disease was reported among 15% of them.
Characteristics of Study Participants (n = 214).
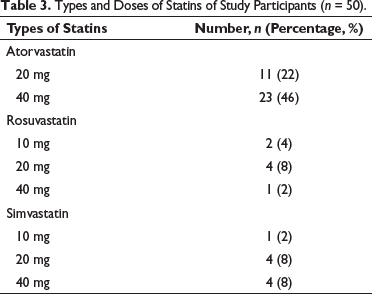
In the endocrinology clinics, there were only three types of statins prescribed to the patients (atorvastatin, rosuvastatin, simvastatin) with varying strengths. The most commonly prescribed statin was atorvastatin. Of the 50 subjects who were present for their interviews, many of them received atorvastatin 40 mg (46%), followed by a decreased strength of atorvastatin 20 mg (22%). The type that was least prescribed was rosuvastatin 40 mg (2%) and simvastatin 10 mg (2%), as shown in Table 3.
Types and Doses of Statins of Study Participants (n = 50).
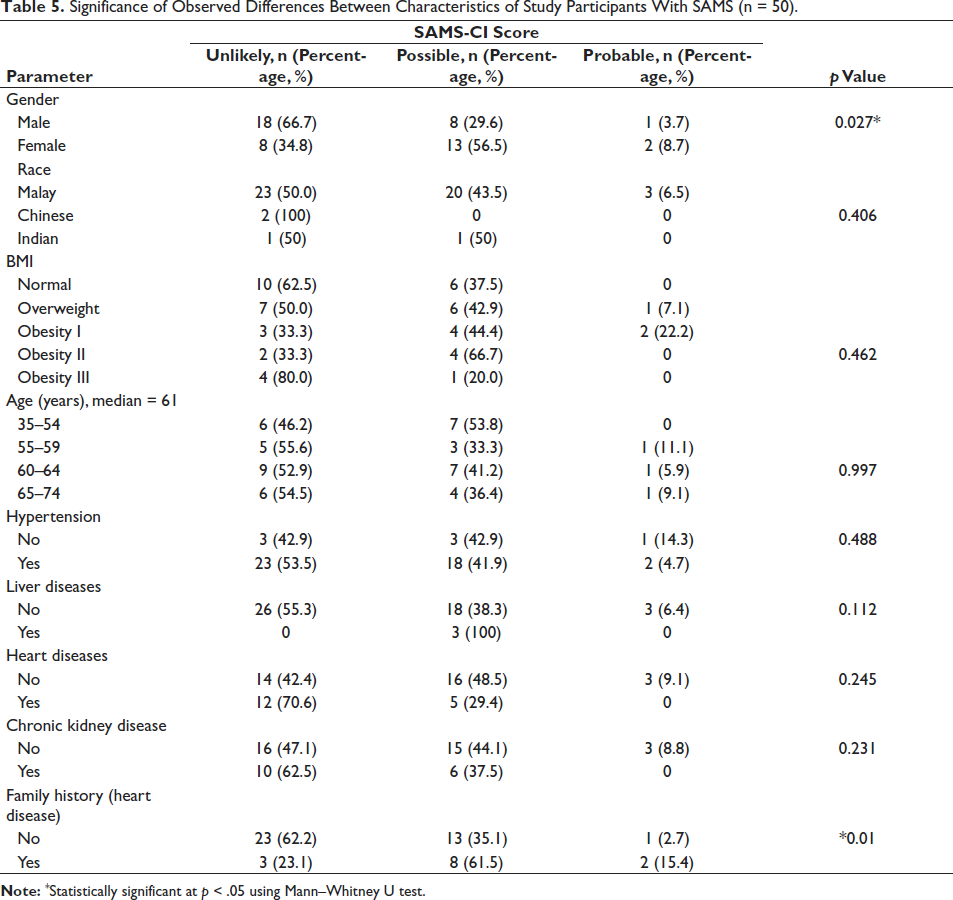
For the SAMS-CI scoring system, of the 50 participants, 26 patients were “unlikely” to have experienced SAMS (52%), 21 “possibly” experienced SAMS (42%), and three cases were considered probable (6%), as shown in Table 4. The mean SAMS-CI score was 6.32 ± 0.470 (±7.4%) [CI: 5.85–6.79], which fell under an “unlikely” category. A significant difference was found between gender with SAMS (p = .027) as well as family history of heart disease and SAMS (p = .01) (Table 5).
The Likelihood of SAMS Among Participants.

Significance of Observed Differences Between Characteristics of Study Participants With SAMS (n = 50).
Further analysis using the Kruskal–Wallis and Mann–Whitney U tests found that the SAMS-CI score differed significantly across the types of statins. Atorvastatin was found to be significantly associated with the SAMS-CI score (p = .001). The same scenario was observed with simvastatin (p = .003), but not with rosuvastatin (p = .053) (Table 6).
Significance of Observed Differences Between Types of Statins With SAMS (n = 50).
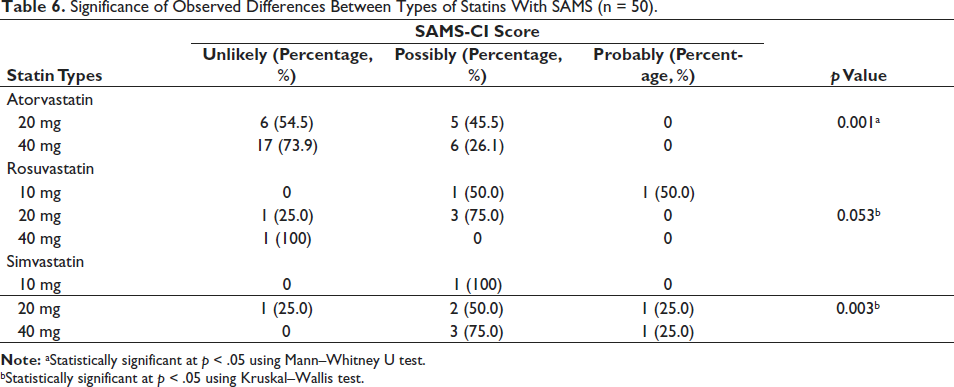
bStatistically significant at p < .05 using Kruskal–Wallis test.
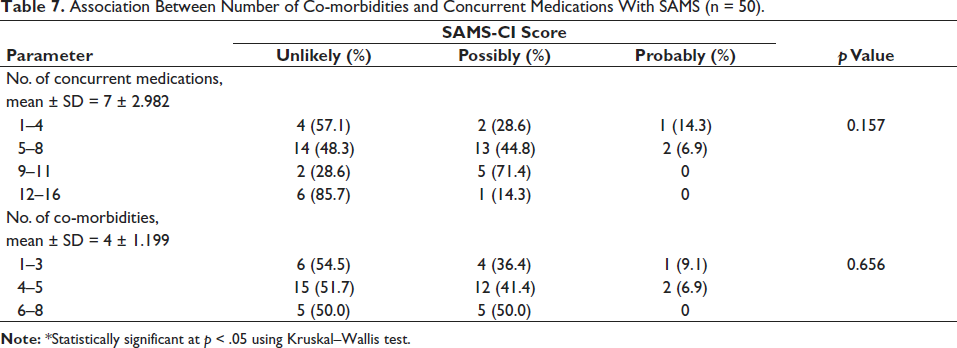
In terms of the number of concurrent medications and the number of co-morbidities among participants, there was no significant association with SAMS detected (Table 7).
Association Between Number of Co-morbidities and Concurrent Medications With SAMS (n = 50).
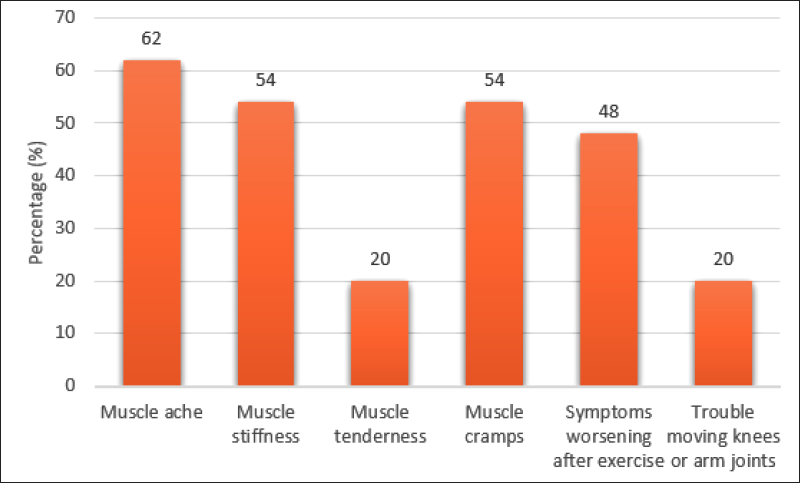
The presence of SAMS clinical indicators, which are muscle aches, muscle stiffness, muscle tenderness, muscle cramps, muscle symptoms that worsen after exercise, and trouble moving knees and arms joints, and their pattern were determined. From the patient interviews, all the participants agreed that they had experienced at least one of the muscle symptoms stated. The most frequently experienced muscle symptoms were muscle aches (62%), followed by muscle stiffness (54%) and muscle cramps (54%). In addition, 48% of the participants also experienced muscle symptoms that worsened after exercise. To a lesser extent, muscle tenderness and trouble moving knees and arm joints were independently experienced in 20% of the participants. The frequency of these muscle symptoms among participants is displayed in Figure 2.
Discussion
In our exploratory study, we found that there were 6% of participants who had a “probable” likelihood of experiencing SAMS. However, the total occurrences for both “probable” and “possible” cases were 48%. The reporting of SAMS occurrence when assessed specifically using the SAMS-CI is limited in published literature. Nevertheless, the results can be compared with previous studies that reported the prevalence of SAMS. The figure reported in this study is higher than that reported by the NLA, which estimated that the prevalence of SAMS in the general population was 10%. 12 In addition, according to patient registries and observational studies, the prevalence of SAMS was reported to be between 7% and 29%.4, 13 In the Effects of Statins on Muscle Performance (STOMP) trial, which was the first randomized, double-blind, placebo-controlled study, the prevalence of SAMS was reported to be 9.4%. 14 In a Malaysian population, a pilot study with a sample size of n = 118 reported a prevalence of 18.6%. 15 A higher prevalence of SAMS (27.8%) was also observed in a lipid clinic. 16 There is a range of prevalence data published in the literature, and one of the reasons for this discrepancy could be attributed to the method of SAMS assessment, which for this study was conducted using the pre-structured SAMS-CI tool while other studies had used biomarkers such as creatinine kinase elevation alone to predict the occurrences of SAMS. It is postulated that a variety of objective assessments of muscle function, such as grip strength, and use of biochemical markers like creatinine kinase to complement the self-reported symptoms together with the application of a subjective assessment of a categorization system will yield higher accuracy of SAMS detection. It is plausible that many cases of SAMS went undetected, which underestimated the prevalence observed in this study.
There were apparent conflicting results between observational and randomized, double-blinded studies, where the prevalence for the latter was reportedly much lower. It was suggested that the incidence of muscle symptoms among patients receiving statin therapy was unlikely attributable to statins and was more due to a nocebo-like effect.17–20 Hence, the categorization of the incidences of SAMS according to the likelihood as seen in SAMS-CI may provide a more accurate representation of SAMS estimation.
There are endogenous and exogenous risk factors associated with SAMS. The endogenous risk factors include older age, female gender, underweight, Asian ethnicity, and family history of co-morbidities. Meanwhile, the exogenous factors are strenuous physical activity, diets that consist of grapefruit, pomegranate, or cranberry juice, and excessive alcohol intake or drug abuse. Additionally, co-administration of statins with CYP3A4 inhibitors can also increase the concentration of statins in the body, resulting in prolonged exposure to statins.13, 20–23
In this study, there was a significant association between gender and SAMS. This observation was supported by Skilving et al. in a randomized, placebo-controlled trial in 2002, which found that females had a higher frequency of myopathy than males. 24 This can be explained by the difference in the body fat composition between these genders, where females have a higher body fat content compared to males. This means that the volume of distribution of drugs is higher in females, especially for lipophilic statins, which leads to an increased risk for adverse effects.25, 26 Muscle symptoms are expected to be higher in lipophilic statins (atorvastatin and simvastatin) compared to in relatively hydrophilic statins (rosuvastatin) due to the better penetration into extrahepatic tissue. There was no significant association between age or BMI, with SAMS. As reported in the Prediction of Muscular Risk in Observational Conditions (PRIMO) study, there was no association between age and BMI with muscle symptoms associated with statin use. 27 This was supported by Ochs et al., who found no association between age and SAMS. 28 A systematic review and meta-analysis by Iwere et al. also found no significant association between age and SAMS. 29 In contrast, Saeed et al. reported that increased age was associated with SAMS, but gender and BMI were not. 30
In terms of co-morbidities, no significant association was found between hypertension, liver and heart diseases, as well as chronic kidney diseases with SAMS. In our study, all the study participants were diagnosed with type 2 diabetes mellitus, and without a control group, a meaningful comparison could not be done to rule out the impact of diabetes on SAMS occurrence. Hypertension was not significantly associated with SAMS. This result was supported by a meta-analysis conducted by Davis et al., who found no correlation between hypertension and SAMS. 31 However, Ochs et al. had found a significant association between hypertension and SAMS but it was further suggested that the hypertension was a result of statin cessation rather than its use being independently associated with SAMS. 28
This study also found no correlation between liver and chronic kidney diseases with SAMS. This was supported by Saeed et al., who also found no correlation between those two diseases with SAMS. 30 In contrast, Stroes et al. stated that muscle symptoms were reportedly more frequent in patients with liver diseases. 13
It was also demonstrated that there was no significant association between heart disease and SAMS. In contrast, in a meta-analysis performed by Nguyen et al., individuals with cardiovascular diseases were more likely to be associated with SAMS. 32 In addition, there was a significant association between those with a familial history of heart disease with SAMS. This was supported by Ochs et al., who stated that a family history of heart disease is the most significant predictor of SAMS. 28 This correlation can be explained by the fact that patients with a family history of heart diseases are at a higher risk of contracting a heart disease compared to any other population. Therefore, they are more likely to be advised to continue their statin treatment despite experiencing muscle symptoms, as the benefits would have outweighed the harm.
Our study also found a significant correlation between the types of statins used and SAMS, particularly with atorvastatin and simvastatin, which are both lipophilic statins. It was suggested that muscle symptom frequencies are high with lipophilic statin use as compared to the relatively hydrophilic statins such as rosuvastatin, due to the better penetration into extrahepatic tissues, including muscles, which then contributes to the adverse effects.33–35 In existing literature, there is a lack of direct comparison between atorvastatin, simvastatin, and rosuvastatin specifically. The result of this study was supported by Thambiah et al., who found a significant association between the types of statins being prescribed to patients in a Malaysian government health clinic and the severity of muscle symptoms. 15 However, the study involved lovastatin and simvastatin only, while in this study site, atorvastatin, simvastatin and rosuvastatin were prescribed. In the PRIMO study, there was also a significant association between types of statin used and SAMS, where SAMS prevalence was higher among atorvastatin (40–80 mg) compared to simvastatin (40–80 mg) users.
In addition, in this study, there was no significant association between the number of concurrent medications and the number of co-morbidities with SAMS, which was unexpected as the incidence of SAMS is closely related to drug-drug interactions and drug-disease interactions. According to expert opinion, the incidence of SAMS is associated with patient factors such as co-morbidities and concomitant drugs, which was not reflected in the results of this study. 36 Nevertheless, this study did not account for the effect of a specific class of medication and their doses on SAMS occurrences. Instead, this study was aimed at investigating the patterns of muscle symptoms due to statin consumption among the Malaysian population. It was noted that the clinical manifestation of SAMS is highly heterogeneous. According to the NLA, the spectrum of muscle symptoms or myalgia due to statins included muscle aches, stiffness, tenderness, cramps, and trouble moving knees and arm joints. The clinical manifestation of SAMS also included myositis, which is muscle inflammation, and rhabdomyolysis, which is a type of myonecrosis. 37 There was no incidence of myositis and rhabdomyolysis observed in this study. According to this study, the most commonly experienced muscle symptoms were muscle aches, which aligned with the findings of Rallidis et al. 38 Most patients agreed that their muscle symptoms worsened after performing an exercise. This was also demonstrated by Philips et al., where people on statins were shown to have reduced muscle strength and could not tolerate excessive physical activity. 39 Besides, it was proven that the incidences of SAMS were higher in athletes compared to the general population, which further supports the notion that statins possibly induced muscle weakness. 40 Although the majority of the participants reported experiencing muscle symptoms, it is worth mentioning that those symptoms could have arisen due to other factors, such as older age, which could have made them more prone to experiencing muscle symptoms in general.
Study Limitation and Future Research
It has long been acknowledged that it can be challenging to measure and quantify the relationship between statin use and muscle symptoms. This is due to the lack of an objective set of criteria that is able to determine the incidences of true muscle symptoms attributable to statin use alone. In the future, a larger sample size could be used to better predict SAMS. In addition, the study was conducted in one suburban town in Selangor. Future researchers could sample patients from other suburban towns to make the conclusion more representative of the Malaysian population. In addition, our study was limited in that we assumed 100% adherence to statin therapy.41, 42 Future studies could benefit from adherence assessment and pharmacist-led interventions.43, 44 Enhanced study designs could consider incorporating qualitative methods, such as in-depth interviews, to gain a deeper understanding of patients’ experiences, perceptions, and decision-making processes regarding statin use and SAMS.
Conclusion
In conclusion, a minority (6%) of the patients that we sampled experienced SAMS, which were probably attributable to statins.
SAMS, particularly those attributed to the use of lipophilic statins, i.e., atorvastatin and simvastatin, were more likely to occur in females with a familial history of heart diseases. Therefore, caution must be exercised for female patients, patients with a history of heart diseases, and those who complain of experiencing myalgia in the large muscle groups after receiving atorvastatin or simvastatin. Although the findings of this study did not find a significant association between other factors, such as co-morbidities, and concurrent medications, it is also best to study these factors in the future as there is conflicting evidence in the literature.
Footnotes
Acknowledgments
The authors thank hospital staff, their patients, and everyone who participated in the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Ethics approval was obtained from the Research Ethics Committee of MARA University of Technology with the approval number REC/04/2022.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Special Research Fund (Geran Penyelidikan Khas) 600-RMC/GPK 5/3 (107/2020).
Informed Consent
All the participants signed the informed consent form.
