Abstract
Introduction
Shivering is a common complication during neuraxial anesthesia, leading to metabolic and cardiovascular challenges. Existing treatments vary in effectiveness, and this study compares intravenous tramadol and nalbuphine, both administered with midazolam, for shivering control.
Objectives
To evaluate and compare the efficacy of IV tramadol and IV nalbuphine, alongside midazolam, in controlling post-spinal anesthesia shivering.
Materials and Methods
A prospective, randomized comparative clinical study involving 100 patients undergoing various surgical procedures under spinal anesthesia. Patients were divided into two groups to receive either IV Tramadol or IV Nalbuphine with midazolam. Parameters like shivering severity, time to cessation, recurrence, and side effects were recorded and analyzed.
Results
Both tramadol and nalbuphine effectively controlled shivering with comparable response times and side effect profiles. There was no significant difference in the incidence of nausea, vomiting, and sedation between the groups. Hemodynamic stability was maintained throughout the study.
Conclusion
IV tramadol and IV nalbuphine, in conjunction with midazolam, are effective in controlling post-spinal anesthesia shivering, with similar efficacy and safety profiles. They provide valuable options for clinicians in managing this common anesthetic complication. Further research is encouraged for more refined application in diverse patient populations.
Keywords
Introduction
Shivering is a reflex activity of the skeletal muscles that occurs in response to cold, especially during neuraxial anesthesia, to regulate body temperature. 1 The occurrence of shivering in patients receiving anesthesia varies between 40% and 50%, presenting difficulties beyond patient discomfort, such as metabolic and cardiovascular repercussions.1, 2 Dysfunction of the autonomic control of body temperature, along with circumstances such as the cold surgical setting, leads to a decrease in the body’s core temperature, which triggers shivering.2, 3 Although not life-threatening, shivering during anesthesia can result in negative consequences such as increased oxygen consumption, increased pressure in the eyes and brain, intensified wound pain, slowed healing, and longer stays in the postanesthesia care unit. 3
Currently, nonpharmacological therapies involve the use of external heating technologies. However, there has been significant research on pharmacological approaches that specifically target core thermoregulatory control pathways.4, 5 Among the drugs investigated, tramadol and nalbuphine have shown anti-shivering effects.6, 7 Tramadol, a man-made opioid, has been studied for its ability to effectively reduce shivering after spinal anesthesia. Research has shown that its efficacy varies depending on the dosage.8–11 Nalbuphine, a medication that activates and blocks opioid receptors, has demonstrated potential in reducing postanesthetic shivering.12–16
Several comparative studies have examined the efficacy of these medications in conjunction with others. According to Onyekwulu et al., a dosage of 0.5 mg/kg tramadol was found to be more effective at managing shivering than a lower dosage. 17 Similarly, Mittal and Gupta conducted a comparison between dexmedetomidine and tramadol and reported that dexmedetomidine resulted in faster cessation of shivering. 18 Kyokong et al. established the superiority of tramadol plus nalbuphine compared to ondansetron and placebo.19, 20 Wang et al. and Tamdee et al. provided additional evidence supporting the effectiveness of nalbuphine and tramadol for managing shivering.20, 21
In addition to tramadol and nalbuphine, studies on various substances have shown different levels of efficacy.8, 5, 14–16 Butorphanol, an alternative opioid analgesic, was shown to be marginally more effective than tramadol in managing shivering.22–24 Fern and Misiran discovered that dexmedetomidine had greater effectiveness, albeit it was accompanied by an increase in problems. 25 According to Reddy et al., tramadol was associated with a shorter reaction time, a greater rate of response, and an elevated occurrence of adverse effects in comparison to clonidine. 26 Ketamine and dexmedetomidine, as proven by Akram et al. and Magella et al., respectively, have also been shown to be effective at managing shivering.25, 27–29 However, it is important to note that they have varying rates of response and potential consequences.
Our research aimed to thoroughly assess the effectiveness of intravenous tramadol and intravenous nalbuphine, both of which are administered with midazolam, for treating shivering after spinal anesthesia. Recognizing the complex difficulties associated with shivering throughout the perioperative period, our main objective is to provide detailed insights to assist in therapeutic decision-making. In this study, we will conduct a double-blinded randomized controlled trial with the following objectives: first, to assign patients to two study groups (Group T and Group N) and administer intravenous tramadol (0.5 mg/kg) to Group T and intravenous nalbuphine (0.1 mg/kg) to Group N. Second, we will thoroughly assess and compare the time needed for shivering to stop, examine the rates of shivering recurrence, and evaluate the occurrence of nausea, vomiting, and sedation in both groups. Our study aimed to provide significant evidence on the comparative efficacy and safety profiles of tramadol and nalbuphine when taken along with midazolam by thoroughly examining these characteristics. In essence, our research aimed to provide physicians with factual and data-driven knowledge, enabling them to make the best decisions and improve the quality of treatment throughout the perioperative period.
Physiology
Physiology of Central Neuraxial Blockade
Central neuraxial blockade results in the inhibition of autonomic, sensory, and motor functions. There is a distinct difference in the level of anesthesia required for autonomic fibers compared to sensory or motor fibers, with weaker solutions being sufficient for autonomic fibers at the higher end.2, 30 The autonomic sympathetic blockade normally extends two to four segments above the sensory level, but the motor blockade usually affects one to four segments below. Stout’s principles, which include concentration, quick fixation, injection speed and volume, spinal fluid pressure, and solution-specific gravity, have a significant impact on the propagation of anesthesia. Control and spread pertain to factors such as medication dosage, injection location, volume, pace, and barbotage.25, 31 Following injection, the concentration of anesthetic drugs in the subarachnoid space decreases as a result of dilution, mixing in cerebrospinal fluid, diffusion to neural tissues, uptake by neural tissues, and absorption by blood vessels. 32 The distribution of the subarachnoid space is regulated by several factors, including age, height, position, spinal cord disorders, injection location, needle orientation, Baricity, and dosage. Sympathetic blockade has a crucial role in determining physiological reactions to subarachnoid anesthesia, leading to both immediate and delayed consequences. 33
Physiology of Shivering
Thermoregulation in homeothermic animals encompasses the process of perceiving temperature changes, regulating temperature changes in the central nervous system, and producing appropriate responses. 30 Afferent thermal sensing is largely facilitated by delta fibers, which transmit signals from cold receptors, and C fibers, which transmit signals from warm receptors. The preoptic area of the anterior hypothalamus, which regulates autonomic thermoregulation, consolidates thermal information. 23 Effector responses included changes in behavior, vasomotor reactions, and shivering. Shivering, a repetitive contraction of muscles, is triggered by cooling of the preoptic area. 34 Thermoregulatory disruptions arise during general anesthesia, affecting the thresholds for warm and cold responses. 31
Afferent Thermal Sensing
Thermal regulation encompasses complex mechanisms for detecting temperature changes, coordinating reactions in the central nervous system, and executing appropriate actions. 35 Cold sensors, primarily sent by delta fibers, have the highest response to temperatures between 25°C and 30°C. 36 On the other hand, warmth receptors, transmitted by C fibers, have the most activity when exposed to temperatures between 45°C and 50°C. Central cold thermoreception is less important than peripheral cold sensory reception. It becomes activated when the core temperature approaches its minimum. 37 Thermal information that is increasing in temperature travels through the spinothalamic tracts located in the front part of the spinal cord. 30 It is important to note that no tract is essential for the transmission of thermal information. The nucleus raphe magnus and subcoeruleus region in the brainstem serve as crucial intermediary centers for relaying heat information from the skin to the hypothalamus. The hypothalamus, brain, spinal cord, and deep tissues collectively contribute approximately 20% of the temperature input to the central regulating system. 23
Central Regulation
The preoptic region of the anterior hypothalamus dominates autonomic thermoregulation in mammals. It houses both temperature-sensitive and temperature-insensitive neurons. Heat-sensitive neurons, which are four times more abundant than cold-sensitive neurons, increase discharge rates in response to local heat, activating heat loss mechanisms. Warm-sensitive neurons integrate local and thermal information and compare it with synaptic afferents arriving from ascending pathways. 37 The hypothalamus, the primary thermoregulatory control center, modulates thermoregulatory responses, integrating information from various sources. The intensity versus core temperature relationship defines the gain in thermoregulatory responses. 38 Warm-sensitive neurons in the preoptic anterior hypothalamus not only sense core temperature but also compare local information with thermal and nonthermal synaptic afferents. Classic neuronal models distinguish integrative and effector neurons; however, some anterior hypothalamic neurons function both as sensors and integrators. 39 The gain of a thermoregulatory response varies, and the complexity of a system is influenced by interactions between responses and time-dependent effectors. 26
Efferent Responses
Effector mechanisms respond to integrated thermal signals, ensuring optimal thermoregulation. Altered behavior, a potent mechanism, involves increased motor activity or seeking warmer surroundings. 30 Vasomotor responses include vasoconstriction and piloerection in response to cold and vasodilation and sweating in response to heat. Shivering and an increased metabolic rate are critical responses to cold. The activation of thermoregulatory effector responses occurs at specific temperatures, with behavior modification often overpowering autonomic mechanisms. 34 Cutaneous vasoconstriction, a consistent autonomic response, regulates heat loss without the major involvement of sweating or shivering. 38 Thermoregulatory vasoconstriction reduces cutaneous heat loss, preserving metabolic heat within the core. Shivering, the last line of defense, is activated when behavioral compensation and vasoconstriction are insufficient to maintain core temperature. 23
Thermoregulation
In homeothermic species, thermoregulation is a complex interplay of afferent thermal sensing, central regulation, and efferent responses orchestrated to maintain internal body temperature within a narrow range for optimal physiological function. Three primary components govern thermoregulatory responses: afferent thermal sensing, central regulation, and efferent responses. 23
During General Anesthesia
Major disturbances in thermoregulation are observed during and after general anesthesia. The induction of general anesthesia impairs the function of neurons in the preoptic nuclei and hypothalamus, reducing the temperature at which hypothermia usually occurs. Behavioral adaptations become irrelevant in unconscious and often paralyzed patients, as most general anesthetics markedly inhibit autonomic thermoregulatory control. Anesthesia-induced thermoregulation slightly elevates the warm response and markedly reduces the cold response threshold. When the inter-threshold range is increased, the gain and maximum intensity of some responses remain normal, while others are reduced by general anesthesia. 34
During Regional Anesthesia
Epidural and spinal anesthesia, encompassing regional anesthesia, also impact thermoregulation, albeit to a lesser extent than general anesthesia. Both the vasoconstriction and shivering thresholds decrease by 0.6°C, but this reduction is less pronounced than that measured above the upper level of the block. The gain of the shivering response is reduced, and the maximum intensity is diminished during regional anesthesia. Shivering above the block compensates for the inability of muscles below the block to engage in shivering. The core temperature decreases initially due to core-to-peripheral heat distribution, but prolonged epidural anesthesia results in less core hypothermia than does general anesthesia, attributed to vasoconstriction compensating for heat loss. 23
Understanding these variations in thermoregulation during different types of anesthesia is critical for perioperative management and patient well-being.
Postanesthetic Shivering
Postanesthetic shivering is a common physiological response following surgical procedures and anesthesia. It also results in patient discomfort, increased pain, and potential complications. It can increase intraocular and intracranial pressure, affecting specific surgical scenarios. 34 Shivering leads to a significant increase in oxygen consumption (VO2), with potential implications for high-risk patients and adverse myocardial outcomes. 30 Preventive measures include passive insulators, active warming systems, and pharmacotherapy, aiming to reduce core hypothermia and enhance patient comfort and safety. 23 The exact etiology of this disease is multifactorial, and the contributing factors include perioperative hypothermia, postoperative pain, and residual concentrations of anesthetics. 38 Shivering may occur as a manifestation of sympathetic overactivity, spinal reflex hyperactivity, or a response to altered central thermoregulatory control. The discomfort experienced by patients is often compounded by increased pain due to muscular contractions, tension on suture lines, and potential adverse effects on surgical sites. 34
Several hypotheses have been proposed to explain the origins of postanesthetic shivering, with perioperative hypothermia and pain being the most substantiated. It is crucial to differentiate between a decrease in core temperature and a reduction in overall heat content, as hypothermia, rather than decreased heat content, facilitates the emergence of shivering. Awareness of core hypothermia may be impaired by the effects of regional anesthesia, complicating the assessment of shivering triggers.31, 36
Measures for Combat Shivering
Efforts to prevent postanesthetic shivering are essential for patient comfort, safety, and overall perioperative outcomes. Various measures can be employed to reduce core hypothermia and, consequently, the incidence of shivering. Passive insulators, such as cotton blankets, surgical drapes, disposable plastic drapes, and plastic bags, play a crucial role in reducing heat loss to the environment. The extent of heat conservation is directly proportional to the body surface area covered. While a single layer of each material reduces heat loss by approximately 30%, adding extra layers does not proportionally increase the benefit. Passive insulation strategies are effective but may have limitations in prolonged or extensive surgeries. 30
Active warming systems, including convection warming and radiant heat systems, are instrumental in preventing perioperative shivering. Convection warming involves forcing warm air through a quilt-like porous blanket, directly warming the skin and creating a warm air envelope around the body. Radiant heat systems, such as infrared light or thermal ceiling lights, can also effectively warm the body. These measures significantly contribute to heat conservation and a reduction in shivering. 38
Various drugs with anti-shivering properties have been identified, spanning biogenic monoamines, cholinomimetics, cations, endogenous peptides, and N-methyl-D-aspartate (NMDA) receptor antagonists. These drugs modulate central thermoregulatory control mechanisms, although their primary functions are diverse. Pharmacotherapy represents a promising avenue for preventing postanesthetic shivering, but further research is needed to understand its specific roles and mechanisms. 35
In addition to being a source of discomfort, postanesthetic shivering poses potential complications for patients, especially high-risk individuals, by impacting oxygen consumption and myocardial outcomes. Preventive measures, including passive insulators, active warming systems, and pharmacotherapy, play critical roles in maintaining core temperature and enhancing patient comfort and safety during the perioperative period. Further investigation into the specific mechanisms of pharmacotherapeutic agents is warranted for a comprehensive understanding of their potential in preventing postanesthetic shivering. 40
Pharmacology
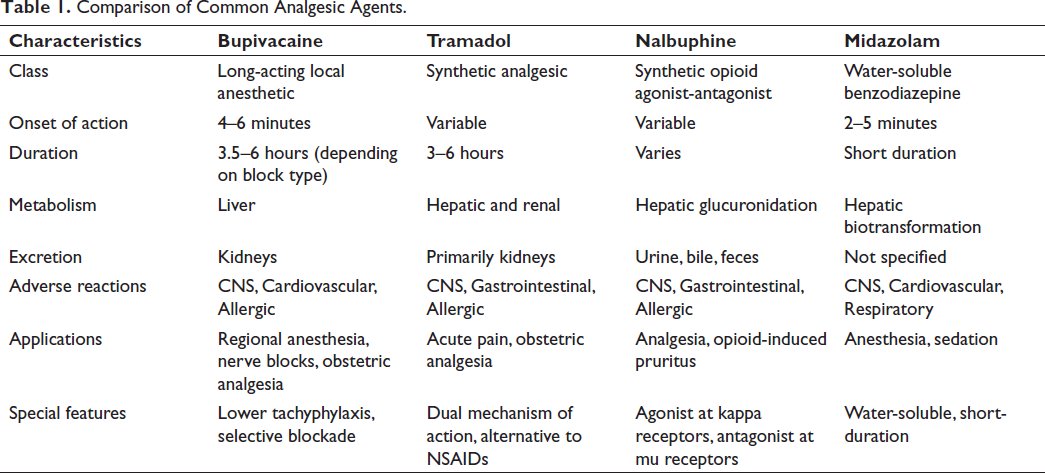
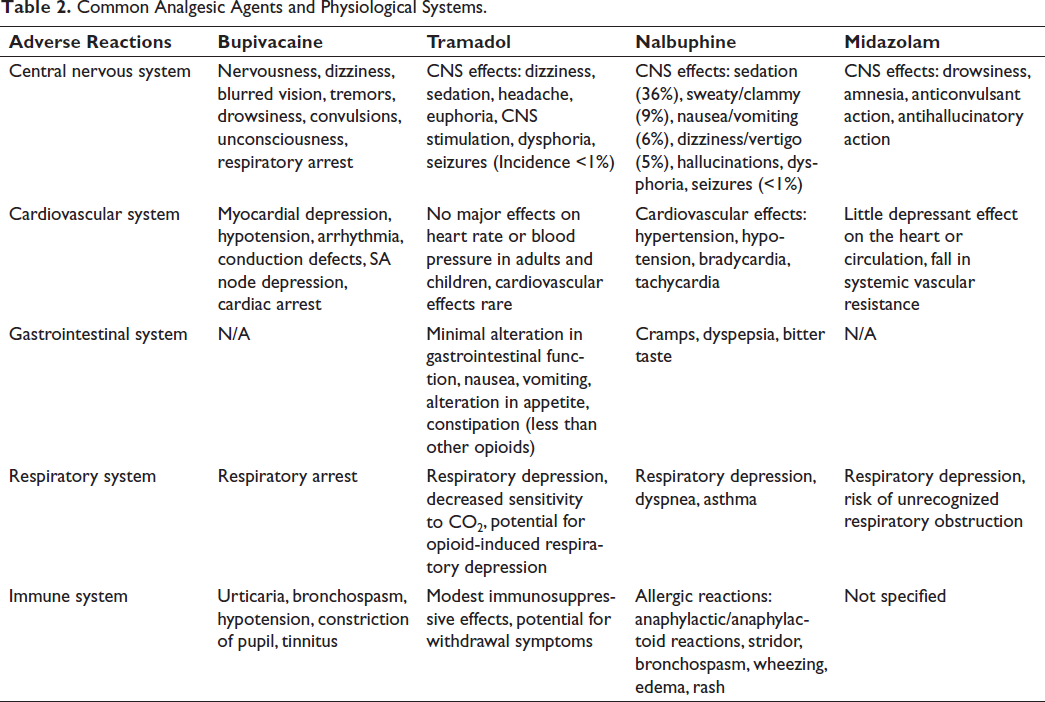
In our research, we focused on the four most common anesthetic agents, namely, bupivacaine, tramadol, nalbuphine, and midazolam. Table 1 shows a quick comparison of the key characteristics of bupivacaine, tramadol, nalbuphine, and midazolam, including their class, onset of action, duration, metabolism, excretion, adverse reactions, applications, and special features. Table 2 summarizes the adverse reactions of bupivacaine, tramadol, nalbuphine, and midazolam to different physiological systems.
Comparison of Common Analgesic Agents.
Common Analgesic Agents and Physiological Systems.
Bupivacaine
Bupivacaine is a long-acting local anesthetic widely employed for regional anesthesia and pain management. It exhibits an onset of action between 4 and 6 minutes, reaching maximum anesthesia within 15–20 minutes. The duration of surgery varied from 3.5 to 5 hours for peridural blocks to 5–6 hours for nerve blocks. 41 Metabolism occurs in the liver, and excretion occurs through the kidneys. 42 Notably, bupivacaine has advantages, such as decreased tachyphylaxis efficacy, selective blockade, and limited blood-brain barrier penetration.15, 22, 43, 44 Adverse reactions include CNS effects such as nervousness and dizziness; cardiovascular effects such as myocardial depression and hypotension; and allergic reactions such as urticaria.45, 46 Additionally, bupivacaine has a lower CC/CNS ratio than lignocaine.47, 48 Its applications include infiltration, nerve blocks, caudal and epidural blocks, and subarachnoid blocks, demonstrating effectiveness in various surgeries and obstetric analgesia. 49
Tramadol
Tramadol, a synthetic analgesic, acts as an opioid agonist and inhibits monoamine neurotransmitter reuptake. 40 It is available in various forms, including oral formulations, suppositories, and solutions. The mechanism of action of tramadol involves the action of an opioid agonist with multi-receptor selectivity and the inhibition of monoamine neurotransmitter uptake. 50 Its pharmacokinetics include rapid absorption after intramuscular injection, distribution in various tissues, metabolism in the liver, and excretion primarily via the kidneys. Pharmacodynamically, tramadol has minimal cardiovascular and gastrointestinal effects.50, 51 The effects on the CNS include dizziness, sedation, and headache, and these agents have antidepressant effects. 52 Tramadol is used in acute pain management, providing an alternative to other opioids, especially in patients for whom NSAIDs are contraindicated. It is also effective in providing obstetric analgesia and reducing shivering thresholds.19, 53, 54
Nalbuphine
Nalbuphine, a synthetic opioid agonist-antagonist analgesic, shares characteristics with both naloxone and oxymorphone. 11 It has a chemical structure belonging to the phenanthrene series and is water-soluble. 27 The pharmacokinetics of nalbuphine involve oral bioavailability, protein binding, and hepatic glucuronidation for metabolism. It acts as an agonist of kappa opioid receptors and an antagonist of mu-opioid receptors. 19 It is used for analgesia in various scenarios, opioid-induced pruritus, reversal of severe esophageal spasm, and its anti-shivering effects. Adverse reactions include sedation, nausea, dizziness, and allergic reactions.20, 21
Midazolam
Midazolam, a water-soluble benzodiazepine, is primarily used for anesthesia due to its short duration and potency. It possesses a unique chemical structure with an imidazole ring, making it water-soluble at lower pH. The pharmacokinetics of midazolam include rapid absorption, high protein binding, and hepatic biotransformation, leading to a metabolite devoid of pharmacological activity. Pharmacodynamically, midazolam acts on specific benzodiazepine receptors, potentiating gamma-aminobutyric acid (GABA) for CNS effects. It induces sleep, drowsiness, and amnesia. Notably, midazolam has minimal irritant effects following intravenous injection. Its applications include the induction of anesthesia, sedation, and adjunct therapy in various clinical scenarios.19–21
Materials and Methods
Study Design and Setting
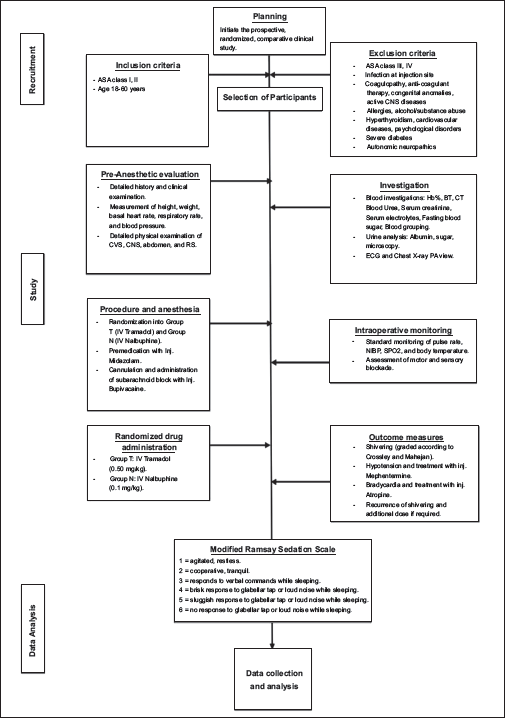
This prospective, randomized, comparative clinical study was conducted at Osmania General Hospital, Modern Government Maternity Hospital, and Government Maternity Hospital (Sultan Bazaar), Hyderabad. The research involved patients who underwent elective lower abdominal surgeries; orthopedic surgeries of the lower limbs; urological and gynecological procedures; and Cesarean section. A summary of the key steps in the procedural aspect of the study is shown in Figure 1.
Methodology Flow Chart.
Participants
Patients who met the inclusion criteria of ASA class I or II, aged between 18 and 60 years, and underwent the specified surgical procedures were included. The exclusion criteria included refusal to participate, ASA class III or IV, infection at the injection site, coagulopathy, anticoagulant therapy, congenital anomalies of the lower spine and meninges, active CNS diseases, allergies to specific drugs, alcohol or substance abuse, hyperthyroidism, cardiovascular diseases, psychological disorders, severe diabetes, autonomic neuropathies, and musculoskeletal disorders.
Ethical Approval and Preanaesthetic Evaluation
Ethical approval was obtained from the ethical committee of Osmania Medical College (registration number 16102001030D). Preanesthetic evaluation included a detailed history; clinical examination; and various measurements, including height, weight, basal heart rate, respiratory rate, blood pressure; and thorough examinations of the cardiovascular system, central nervous system, abdomen, and respiratory system.
Investigations
Blood parameters included Hb%, BT, CT, blood urea, serum creatinine, serum electrolytes, fasting blood sugar, and blood parameters. Urinalysis included albumin, sugar, and microscopy. Additionally, ECG and chest X-ray PA were performed.
Preliminaries and Equipment
Patients provided written informed consent. Intravenous access was established using an 18G intravenous cannula. The equipment used for the procedure included a portable tray with sterile towels, a disposable syringe, a 25-gauge Quinke’s needle, iodine, sponge-holding forceps, towels, towel clips, a drug (0.5% bupivacaine), and normal saline. Emergency resuscitation equipment and drugs were also prepared.
Procedure and Anesthesia
Patients were randomly assigned to two groups (Group T and Group N). They were premedicated with Inj. The patient was then cannulated before receiving a subarachnoid block with Inj. Bupivacaine. Motor and sensory blockade were assessed using the modified Bromage score and pinprick test, respectively. Standard monitoring was established before spinal anesthesia, including pulse rate, NIBP, SpO2, and body temperature.
Randomization and Drug Administration
Patients were randomized into Group T (received IV tramadol) or Group N (received IV nalbuphine). The attending anesthesiologist administered the study drugs without revealing the allocation.
Outcome Measures
Parameters such as shivering (graded according to Crossley and Mahajan), hypotension, bradycardia, recurrence of shivering, and sedation level (using the Modified Ramsay Sedation Scale) were recorded during the intraoperative period.
Statistical Analysis
Statistical analysis was performed using appropriate tests to compare the outcomes between Group T and Group N.
This study was conducted in adherence to ethical principles and guidelines, ensuring the safety and well-being of the participants. The efficacy of IV tramadol and IV nalbuphine in addition to midazolam for controlling shivering after spinal anesthesia was assessed in the specified patient population.
Results
A total of 100 ASA grade I or II patients, evenly distributed across sexes, aged 18–60 years, and who underwent various surgeries under spinal anesthesia were included. All patients received midazolam premedication and lactated Ringer’s preloading. No significant demographic differences were observed between the two groups.
Table 3 presents the demographic and surgical characteristics of patients in Group T (Tramadol) and Group N (Nalbuphine) undergoing spinal anesthesia. Gender distribution demonstrated a balance between males and females in both groups. The mean age in Group T was 41 years, with a standard deviation (SD) of 14, while in Group N, the mean age was 35 years with an SD of 12 (p = 0.163). Average weights were 59 kg in Group T and 57 kg in Group N, with no statistically significant difference (p = 0.209). The duration of surgery showed a mean of 72.5 minutes in Group T and 63.7 minutes in Group N, with a p value of 0.09, indicating a trend toward significance. SD for age, weight, and duration of surgery are also provided.
Demographic and Surgical Characteristics of Patients Undergoing Spinal Anesthesia with IV Tramadol or IV Nalbuphine.
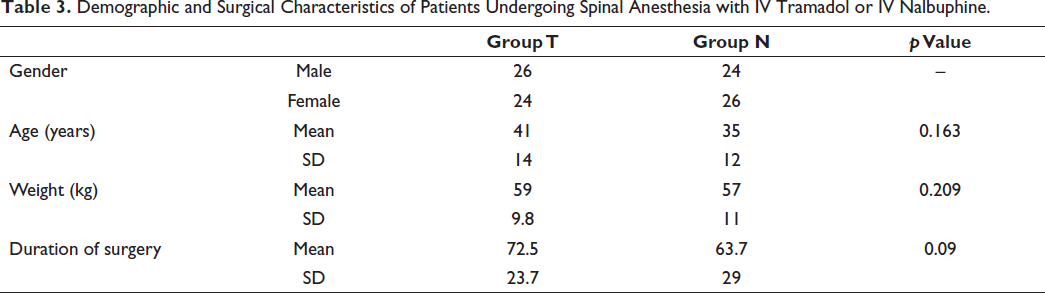
Table 4 illustrates the severity of shivering and the response to treatment in patients from Group T (Tramadol) and Group N (Nalbuphine) undergoing spinal anesthesia. The mean severity of shivering was 2.4 in Group T and 2.1 in Group N, with a p value of 0.0897, suggesting a non-significant trend. SD for shivering severity are presented, indicating variability within the groups. The mean time for the cessation of shivering after drug administration was 4.1 minutes in Group T and 4.2 minutes in Group N, with a p value of 0.66, signifying no significant difference. SD for cessation times are also provided.
Shivering Characteristics and Response to Treatment in Patients Undergoing Spinal Anesthesia with IV Tramadol or IV Nalbuphine
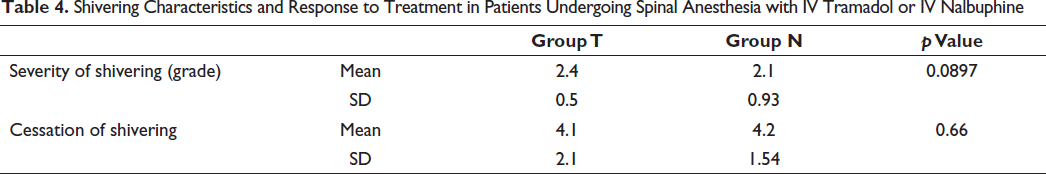
Table 5 outlines the occurrence of adverse events and recurrence rates in patients from Group T (Tramadol) and Group N (Nalbuphine) undergoing spinal anesthesia. The incidence of nausea and vomiting was present in 18 patients in Group T and 14 patients in Group N, with a p value of 0.5, indicating no significant difference. Recurrence of shivering was observed in 17 patients in Group T and 13 patients in Group N, with a p value of 0.51, signifying no statistical significance. Sedation rates were present in 15 patients in Group T and 20 patients in Group N, with a p value of 0.43, suggesting no significant difference.
Adverse Events and Recurrence Rates in Patients Undergoing Spinal Anesthesia with IV Tramadol or IV Nalbuphine.
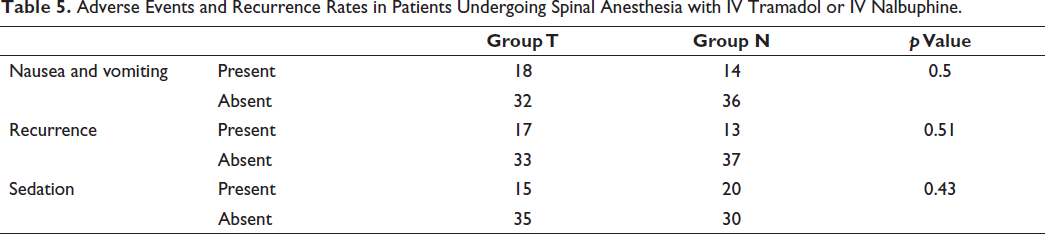
Table 6 combines the changes in heart rate and axillary temperature over time in patients from Group T (Tramadol) and Group N (Nalbuphine) undergoing spinal anesthesia. At time 0, baseline heart rates were 83.4 (6.6) in Group T and 81 (13) in Group N, with a p value of 0.24, while baseline temperatures were 36.1 (0.2) in Group T and 36.09 (0.19) in Group N, with a p value of 0.79. Subsequent time points demonstrated consistent heart rates and temperatures between the two groups, with no statistically significant differences observed at any time point (p > 0.05). The correlation analysis between heart rate and temperature over time showed a stable pattern within each group, indicating that changes in heart rate did not correspond with alterations in temperature. Both physiological parameters remained within normal ranges throughout the observation period, suggesting that neither IV Tramadol nor IV Nalbuphine had a substantial impact on the correlation between heart rate and temperature in patients undergoing spinal anesthesia.
Physiological Parameters Over Time in Patients Undergoing Spinal Anesthesia with IV Tramadol or IV Nalbuphine
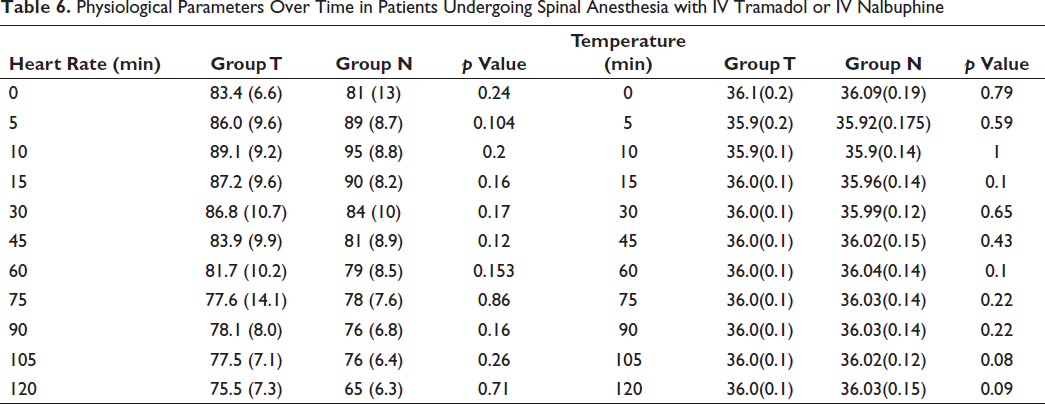
Table 7 combines the changes in both systolic and diastolic blood pressure over time in patients from Group T (Tramadol) and Group N (Nalbuphine) undergoing spinal anesthesia. At time 0, baseline systolic blood pressures were 115.7 (19.5) in Group T and 120 (22) in Group N, with a p value of 0.3. Baseline diastolic blood pressures were 80.8 (17.3) in Group T and 76 (13) in Group N, with a p value of 0.83. Subsequent time points revealed comparable blood pressures between the two groups, with no statistically significant differences observed at any time point (p > 0.05). Throughout the observation period, the trends in systolic and diastolic blood pressures were consistent, suggesting a lack of differential impact between IV tramadol and IV nalbuphine on both components of blood pressure in patients undergoing spinal anesthesia.
Blood Pressure Changes Over Time in Patients Undergoing Spinal Anesthesia with IV Tramadol or IV Nalbuphine.
Discussion
Post-spinal shivering is a common complication encountered during both general and regional anesthesia, affecting a significant proportion of patients undergoing various surgical procedures. Despite advancements in anesthesia practice, the management of shivering remains a challenge, with no established gold standard drug or definitive strategy. Shivering not only is distressing for patients but can also cause physiological stress, impacting oxygen consumption, intraocular and intracranial pressure, and routine monitoring during surgical intervention. 1
The mechanisms leading to postanesthesia shivering are multifactorial and involve factors such as redistribution of heat, perioperative heat loss, stress, direct effects of certain anesthetics, and sympathetic overactivity. Opioids, including tramadol and nalbuphine, exert their effects on various neural substrates, potentially influencing thermoregulatory responses. Tramadol, with its unique pharmacological profile, inhibits 5-HT3 reuptake and promotes 5-HT3 release, potentially through the inhibition of shivering reflex pathways. In this study, we aimed to compare the efficacy of intravenous tramadol and intravenous nalbuphine, both of which are administered in addition to midazolam, for the control of post-spinal shivering after various surgical procedures. 29 Tramadol, which acts primarily through mu-receptor mediation, was hypothesized to offer advantages over nalbuphine, a mixed mu-antagonist, and kappa-agonist. 30
Our study delves into the comparative effectiveness of intravenous tramadol (0.5 mg/kg) and intravenous nalbuphine (0.1 mg/kg), both of which are administered in addition to midazolam, for the control of post-spinal shivering. The observed response times of 4.1 minutes for tramadol and 4.2 minutes for nalbuphine are consistent with the existing body of literature, demonstrating the efficacy of both drugs in promptly mitigating shivering.
In comparison to the findings of previous studies, the response time observed with tramadol was consistent with the findings of Reddy and Chiruvella, where a higher dose (50 mg) effectively controlled shivering within 2.2 minutes during Cesarean section. 21 This faster response time can be attributed to the increased dose administered. However, our results contrast with those reported by Mostafa et al., who observed a longer response time to tramadol (6.2 minutes) when used as an adjunct to midazolam after intrathecal anesthesia. 52 The variability in response times across studies highlights the influence of factors such as dosing, patient populations, and specific procedural contexts. Similarly, our findings for nalbuphine align with the literature, supporting its effectiveness in controlling postoperative shivering. Mostafa et al noted a response time of 5.8 minutes with nalbuphine, which closely corresponds to our observation time of 4.2 minutes. This consistency across studies indicates the reliability of nalbuphine as an adjunct to midazolam for shivering control. The variability in response times observed between tramadol and nalbuphine in different studies emphasizes the complexity of drug interactiorrns and patient-specific factors. Dose-dependent responses and the nature of the surgical procedure contribute to nuanced outcomes, reinforcing the need for individualized approaches in clinical practice. While the response times were comparable between tramadol and nalbuphine in our study, the choice between these agents may be guided by additional factors, such as side effect profiles and patient characteristics. Tramadol’s unique pharmacological properties, including 5-HT3 reuptake inhibition, may present advantages in certain clinical scenarios, while the mixed agonist-antagonist properties of nalbuphine could be preferable for other agents.
Our study provides valuable insights into the ongoing discourse on shivering control, but further research is warranted to refine dosing strategies and delineate the optimal use of tramadol and nalbuphine in specific patient populations. Larger-scale trials considering diverse surgical procedures and patient demographics will contribute to a more comprehensive understanding of these issues, guiding clinicians in tailoring interventions for optimal outcomes. In conclusion, the comparative analysis underscores the efficacy of both tramadol and nalbuphine as adjuncts to midazolam for postoperative shivering control. The nuanced differences in response times highlight the need for a personalized approach that considers patient-specific factors and procedural contexts in clinical decision-making.
On the other hand, maintaining hemodynamic stability is paramount in perioperative care, and our study showed that both tramadol and nalbuphine contributed to stable hemodynamics throughout the observation period. This finding aligns with the literature, reflecting the favorable cardiovascular profiles of these opioids when used as adjuncts for shivering control.12, 17, 19, 30, 32, 53, 54 The absence of significant fluctuations in pulse rate and blood pressure in both groups suggested that these drugs can be safely integrated into anesthesia protocols without compromising cardiovascular stability. The consistency in hemodynamic stability observed in our study is particularly noteworthy when compared with findings from Magella et al., where nalbuphine offered superior stability compared to dexmedetomidine.13–16 This finding reinforces the growing body of evidence supporting the cardiovascular safety of nalbuphine, a crucial consideration in anesthesia management.
When evaluating the side effect profiles of tramadol and nalbuphine, both groups exhibited manageable and clinically acceptable outcomes. Nausea and vomiting, common side effects of opioid administration, were noted in 36% of the tramadol group and 28% of the nalbuphine group. Although the incidence was greater in the tramadol group, the difference was not statistically significant. This finding aligns with previous studies by Sharma et al. and Mittal et al., where nausea and vomiting rates associated with tramadol were within the range of 27% to 48%.13, 55, 56 Sedation, a well-recognized side effect of opioid administration, was reported in 30% of the tramadol group and 40% of the nalbuphine group. Despite being cautiously administered with midazolam, the sedation rates were not significantly greater. These rates are in line with those of Mostafa et al and Magella et al., who emphasized that the sedative effects, while present, are generally well-tolerated and do not pose undue clinical concerns. According to our findings, the rate of nausea and vomiting in the tramadol group was greater than that reported by Bansal and Jain but similar to that reported by Sharma et al.19, 55, 56 Conversely, the sedation rate in the nalbuphine group, though present, was not as pronounced as that reported by Magella et al., indicating variability in side effect profiles across different patient populations and methodologies.
Recurrence rates, an important parameter for assessing the efficacy of interventions, were 34% in the tramadol group and 26% in the nalbuphine group. These rates are consistent with the findings of Mostafa et al., who reported that both tramadol and nalbuphine may offer effective and sustained control of post-spinal shivering in the majority of cases. While our study reinforces the acceptable safety profiles of both tramadol and nalbuphine, clinicians must weigh the benefits of these drugs against potential side effects when choosing these drugs for the purpose of controlling shivering. The slight variations in side effect incidence between studies highlight the importance of individualizing treatment decisions based on patient characteristics, surgical context, and the overall clinical picture. In conclusion, our study underscores that both tramadol and nalbuphine contribute to hemodynamic stability during shivering control, with manageable side effect profiles. These findings support the safety and tolerability of these agents in clinical practice. Clinicians should consider individual patient risk factors and preferences when selecting between tramadol and nalbuphine as adjuncts to midazolam for post-spinal shivering control. Further research is warranted to explore specific patient populations and refine our understanding of the nuanced differences in side effect profiles.
This study has several limitations. Treatment response rather than the incidence of shivering was the primary focus, and the inability to compare oxygen saturation due to shivering interference was acknowledged. Randomization ensured comparable patient groups, and both tramadol and nalbuphine, which are non-controlled drugs associated with less respiratory depression, offered advantages for clinical consideration.
In conclusion, our study contributes valuable insights into the comparative efficacy of intravenous tramadol and intravenous nalbuphine, in addition to midazolam, for the control of post-spinal shivering. Both drugs demonstrated comparable effectiveness, response times, and safety profiles. These findings support the consideration of both tramadol and nalbuphine in routine anesthesia practice for shivering control. Further research, including larger-scale trials, is warranted to refine dosing strategies and explore specific patient populations, ultimately enhancing the management of postoperative shivering in clinical settings.
Conclusion
Both tramadol and nalbuphine were found to be effective at treating post-spinal anesthesia shivering, but the difference was not statistically significant. The outcomes demonstrated that both drugs offered favorable hemodynamic profiles, providing valuable options for clinicians. Notably, the incidence of side effects, such as nausea, vomiting, and dizziness, was significantly greater in the tramadol group. The observed sedation in the nalbuphine group, while statistically insignificant, may be advantageous in the intraoperative period, particularly when patient sedation is desirable. However, caution is advised when administering nalbuphine to patients with respiratory compromise or elderly patients. Importantly, neither tramadol nor nalbuphine is classified as a controlled substance, enhancing its clinical utility. The recurrence rates of shivering were comparable between the two groups, suggesting similar sustained efficacy. In conclusion, this study provides valuable insights into the choice of anti-shivering agent in the context of spinal anesthesia. The comparable effectiveness of tramadol and nalbuphine, coupled with their distinct side effect profiles, allows clinicians to tailor interventions based on individual patient characteristics and preferences. Further research is warranted to explore the broader landscape of anti-shivering drugs, optimize their use and expand the repertoire of available options for enhanced patient care.
Footnotes
Abbreviations
Author Contributions
Conceptualization: AM, DM, SP; data curation: AM, DM, SP; formal analysis: IP, VK, WHC; investigation: RSYW, LCM; methodology: ASA, KWG; project administration: IP, VK, WHC; supervision: AM, DM, SP; validation: RSYW, LCM; visualization: WHC; writing—original draft: AM, DM, SP, IP; writing—review and editing: WHC, VK, LCM, RSYW, ASA, KWG. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Ethical approval was obtained from the ethical committee of Osmania Medical College (registration number 16102001030D).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Sunway University through the research grant schemes GRTIN-RAG-DMS-11-2024 and GRTIN-IGS(02)-DBS-15-2022.
Informed Consent
All of the study participants gave their informed consent.
