Abstract
Diabetes is known to cause an array of central and peripheral neurological conditions. Among the neuropathic complications associated with diabetes are distal symmetrical polyneuropathy, amyotrophy, polyradiculopathy, autonomic neuropathy, and cranial mononeuropathies. Notably, brachial plexopathy has not been documented as a complication of diabetes. A 50-year-old man with a history of diabetes mellitus presented with severe pain and progressive weakness in his right upper limb. Laboratory investigations, immunopathological analyses, and cerebrospinal fluid examination yielded normal results. The electrodiagnosis and imaging studies were consistent with bilateral brachial plexus neuritis. This unique presentation does not align with the rare diabetic polyradiculopathy of the upper limbs commonly linked to diabetic amyotrophy. The association between diabetes mellitus and brachial plexopathy is uncommon, indicating a possible occurrence of a rare subtype within diabetic neuropathies.
Keywords
Introduction
Neuropathy emerges as a common complication of diabetes mellitus (DM), affecting around 50% of individuals with diabetes throughout their condition. 1 A broad categorization of diabetic neuropathy includes symmetric polyneuropathies, focal neuropathies, and multifocal neuropathies. This classification covers a range of conditions such as sensorimotor polyneuropathy, autonomic neuropathy, cranial neuropathy, mononeuropathies impacting the trunk and limbs, mononeuropathy multiplex, and diabetic lumbosacral radiculoplexus neuropathies. 2 Brachial plexus involvement as a complication of DM is an uncommon occurrence that has been seldom reported. Within this context, we present a case of a 50-year-old male patient who suffered from bilateral brachial plexopathy in the setting of DM.
Case Presentation
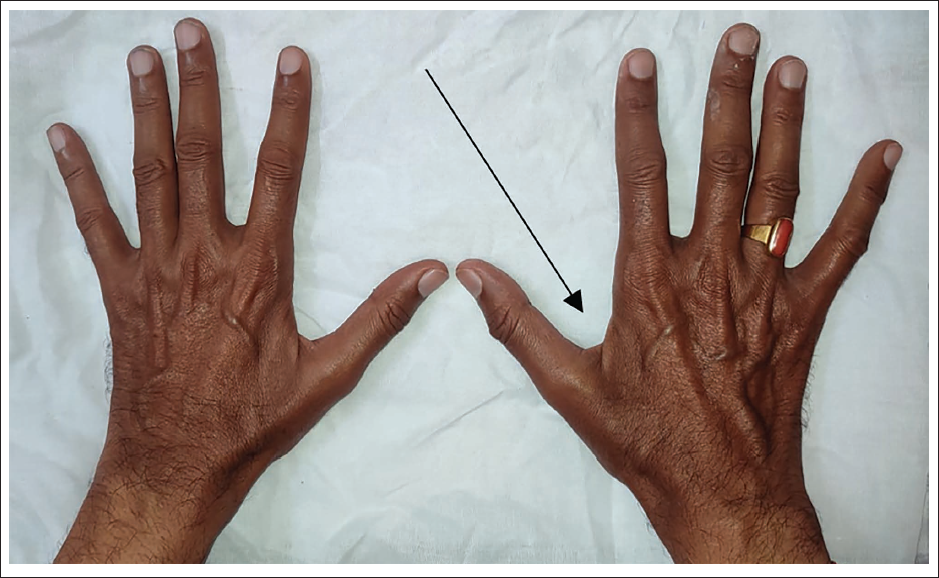
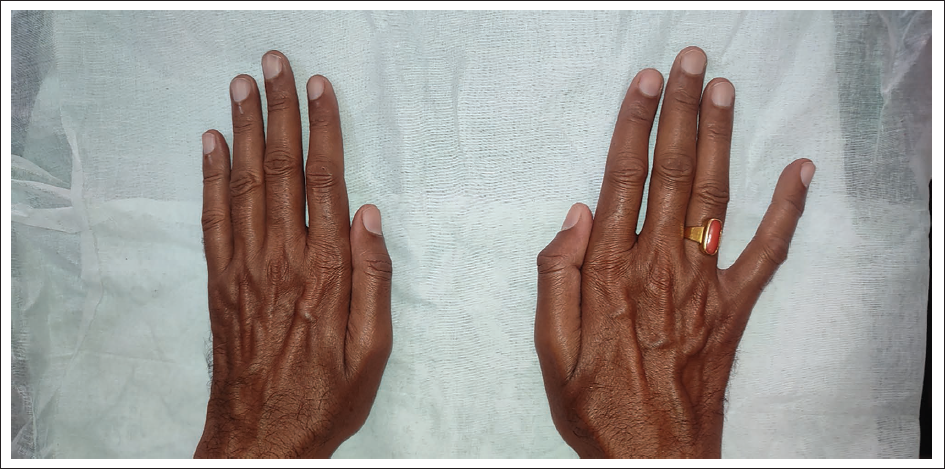
A 50-year-old man presented to Vivekananda General Hospital in Hubballi, Karnataka, India, with extreme pain and weakness in his upper limbs over the past year. He had a history of insulin-dependent DM for 20 years. The patient reported that the pain was sudden in onset and interfered with his sleep for the past year. He had been managing the pain with amitriptyline and gabapentin tablets prescribed by a local physician. The patient reported progressive weakness in his right hand and a decline in his dexterity especially while attempting to grasp and lift a teacup using his right hand. The extent of weakness he experienced in his right hand led him to rely on assistance from his left hand. Upon presentation, atrophy of the first dorsal interosseous muscle between the thumb and index finger of the right hand was noted (Figure 1). A positive Wartenberg’s sign was observed in the right hand (Figure 2).
Atrophy of the First Dorsal Interosseous Muscle Between the Thumb and Index Finger of the Right Hand.
Wartenberg’s Sign in the Right Hand. The Fifth Digit Remains Abducted When Attempting to Adduct the Fingers.
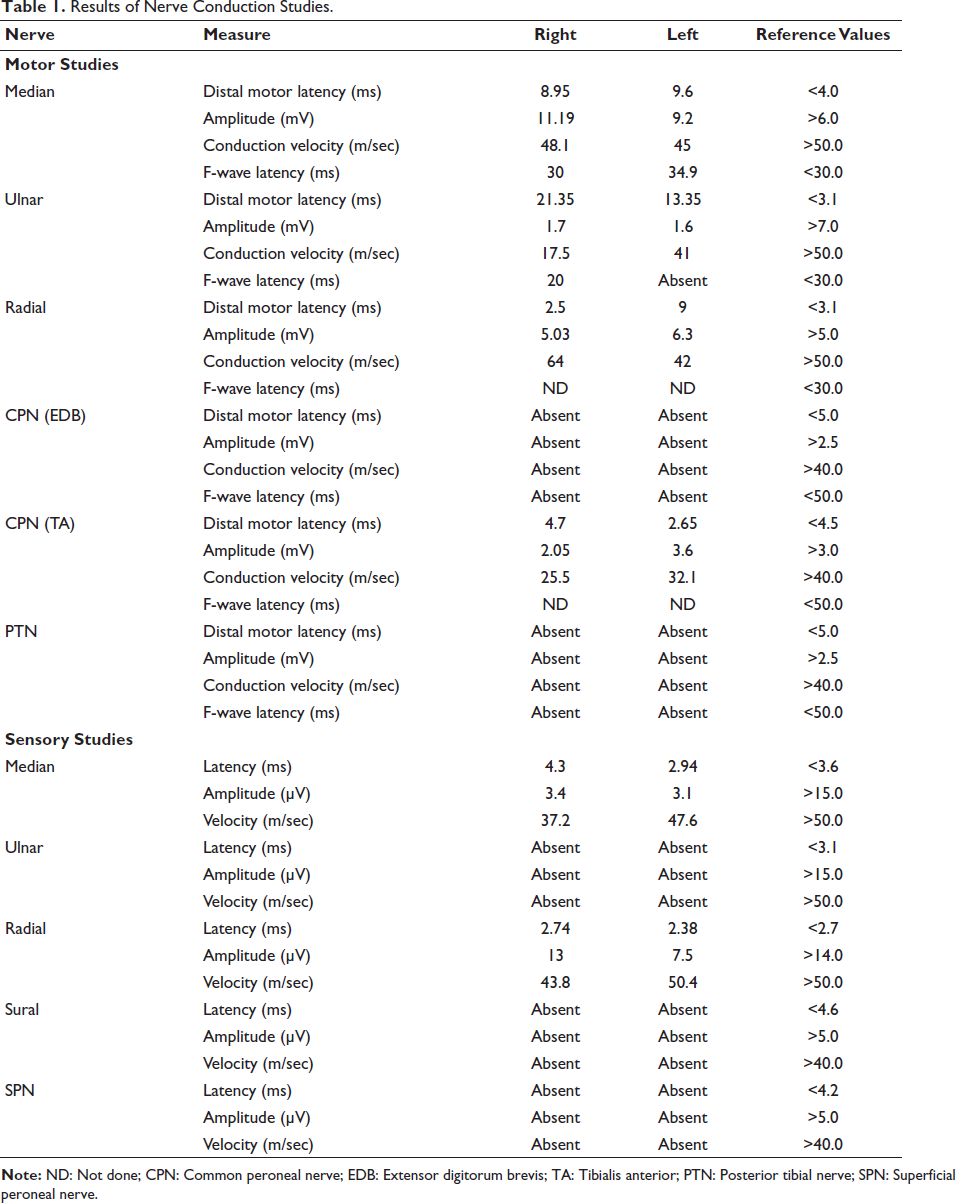
The patient’s hematological report showed no abnormalities. His random blood glucose levels were measured at 171 mg/dL, and his HbA1c level at that time was 8.0%. He tested negative for HIV and Hepatitis B and C antigens. The nerve conduction studies (NCS) were conducted 3 days after the initial presentation (Table 1). Examination of the right ulnar nerve revealed a prolonged distal motor latency (DML) of 21.35 ms, with nerve conduction velocity slowed to 17.5 m/s. The compound muscle action potential (CMAP) amplitude was recorded at 1.7 mV, and the F wave latency was measured at 20 ms. CMAP amplitude was absent in both the right common peroneal nerve (CPN) and the right posterior tibial nerve (PTN). Furthermore, there were absent sensory nerve action potentials (SNAPs) in the right ulnar, superficial peroneal nerve (SPN), and sural nerves. The right median nerve displayed a reduced SNAP amplitude. NCS of the left upper and lower limbs showed that left ulnar DML was prolonged, measuring at 13.35 ms, accompanied by a diminished CMAP amplitude. Additionally, there was an observed prolongation of the F wave latency of the left median nerve. Reduced nerve conduction velocity was evident across all nerves in the left upper limb. Within the left lower limb, both the CPN and PTN, CMAP were absent. SNAP was absent in the left ulnar nerve and the left lower limb, while the left median SNAP exhibited reduced amplitude and prolonged latency. There was an asymmetric involvement of multiple nerves in all four limbs. A provisional diagnosis of mononeuropathy multiplex was made.
Results of Nerve Conduction Studies.
Further, immunopathological tests were conducted to rule out the possibility of an autoimmune disorder. The outcomes of all these tests turned out to be negative. The lumbar puncture performed on the patient yielded normal findings, with no discernible elevation in cerebrospinal fluid (CSF) protein levels. The magnetic resonance imaging (MRI) utilizing short tau inversion recovery (STIR) sequence of the whole spine and brachial plexus revealed mild thickening of the bilateral brachial plexus. The NCS was consistent with the demyelination of nerves, especially in the right upper extremity. Unfortunately, the patient refused to have a nerve biopsy.
A diagnosis of bilateral diabetic brachial plexopathy was established based on the patient’s medical history and evaluation results. Treatment with corticosteroids was contraindicated due to a history of diabetes. Thus, the following treatment regimen was initiated: Cyclophosphamide injections administered in 500 mL of normal saline over 6–8 hours for 2 days per month, for 3 months. Concurrently, injection of MESNA 100 mg was administered intravenously at the 0th, 4th, and 8th hour of cyclophosphamide infusion days. Intravenous Ondansetron and Pantoprazole were prescribed to manage potential drug-induced nausea and vomiting. The patient received guidance to ensure ample fluid intake and regular urination on the days of infusion. After 3 months of treatment, the patient achieved recovery, communicated the absence of pain, and that the weakness declined.
Discussion
The correlation between DM and neuropathy has been acknowledged for over a century, and it did not take long to recognize the existence of different subtypes. As early as 1893, Leyden introduced the first classification encompassing hyperesthetic (painful), paralytic (motor), and ataxic forms of diabetic neuropathy. 3 The term “diabetic neuropathy” encompasses a multitude of neuropathy variations, some closely tied to elevated blood sugar levels and consequent metabolic and ischemic alterations, while others arise from compressive causes. Additionally, there are variations associated with inflammatory and immune processes. Based on the pathophysiology, diabetic neuropathies are classified as Metabolic-microvascular-hypoxic (e.g., Diabetic polyneuropathy, Diabetic autonomic neuropathy), Inflammatory immune (e.g., chronic inflammatory demyelinating polyradiculoneuropathy [CIDP] in DM, cranial neuropathies), compression and repetitive injury, complications of diabetes and treatment-related neuropathies. 4 Instances of brachial plexopathy within the context of DM have been rarely documented. 5 A particular report detailed the occurrence of brachial plexopathy coinciding with the correction of hyperglycaemias along with acidosis during an episode of diabetic ketoacidosis. 6 Similarly, one case documented the occurrence of diabetic brachial plexopathy as the initial manifestation in a patient newly diagnosed with DM. Another case highlighted brachial plexopathy in a patient with effectively controlled type 2 diabetes.5, 7 Notably, in our case, the patient experienced brachial plexopathy during a phase of prominent hyperglycaemias and inadequately managed DM.
The potential diagnoses to consider encompass CIDP, diabetic amyotrophy, and mononeuropathy multiplex. The presentation observed in this patient does not align with diabetic amyotrophy. In cases of diabetic amyotrophy, individuals typically aged 50 years or older manifest symmetrical, asymmetrical, or unilateral weakness alongside muscle wasting in the pelvic and femoral muscles, often accompanied by pain. It is common to observe a concomitant diabetic distal sensorimotor or sensory polyneuropathy. Additionally, upper limb involvement is infrequent in this condition. Contrarily, in our case the patient exhibited significant muscle wasting of the dorsal interossei muscle in his right hand. The dorsal interossei are innervated by the ulnar nerve. Atrophy of the first dorsal interosseous is commonly seen in severe ulnar neuropathy. 8 We also observed a positive Wartenberg sign in our patient’s right hand. Wartenberg’s sign manifests as the fifth digit remaining abducted during an attempt to adduct the fingers. This phenomenon arises from the weakness of the palmar interossei muscles. The extensor digitorum communis and extensor digiti minimi, which are innervated by the radial nerve, operate without opposition, resulting in a vector that pulls the fifth digit into abduction. This distinctive sign often becomes evident in cases of severe ulnar neuropathy. 9
CIDP usually exhibits a slow and gradual emergence of weakness that can affect both proximal and distal muscles. Those with DM are more prone to developing CIDP due to an elevated susceptibility. Currently, the widely accepted EFNS/PNS criteria (established in 2005, and revised in 2010) combine clinical and electrophysiological evidence. 10 They differentiate typical CIDP—proximal and distal weakness with sensory dysfunction in all extremities—from atypical CIDP with varied symptom patterns. Supportive criteria involve factors such as CSF protein elevation, treatment response, and abnormal sensory electrophysiology. 11 In our case, despite the electrodiagnostic studies pointing towards demyelination, the lumbar puncture conducted on the patient revealed unremarkable results, showing no noticeable increase in CSF protein levels. While biopsies can assist in diagnosing demyelinating neuropathies like CIDP, 12 unfortunately our patient refused to undergo a nerve biopsy.
Diabetic mononeuropathy multiplex is characterized by the abrupt and sometimes sequential dysfunction of peripheral nerves, occurring at irregular intervals. This condition affects two or more peripheral nerves, often in different limbs. Both peroneal and ulnar nerves were affected in our patients, mirroring the involvement seen in the majority of previously documented cases.13–15 Considering our patient’s presentation featuring multiple neuropathies, the possibility of mononeuropathy multiplex as the underlying cause cannot be entirely ruled out. It is possible that the brachial plexopathies in this patient were a result of diabetic mononeuropathy multiplex. This type of inflammatory vasculopathy tends to be acute or subacute, leading to the degeneration of nerve fibers due to ischemia, which is evident in this case. The occurrence of infarctions in various components of the brachial plexus could be responsible for the uneven upper limb weakness and sensory impairment observed. Thus, we suspect that uncontrolled DM might have contributed to the development of bilateral brachial plexopathy in our patient.16–18
Conclusion
The presented case underscores the intricate nature of neuropathic manifestations in the context of DM. The diagnostic journey illuminated the challenges of differentiating between various neuropathies that can coexist with diabetes. The patient’s symptoms, initially suggestive of mononeuropathy multiplex, later became more consistent with the diagnosis of diabetic brachial plexopathy. This change highlights how complex neuropathic conditions can be and emphasizes the importance of careful diagnostic methods to differentiate and understand overlapping neuropathic disorders.
Abbreviations
Footnotes
Acknowledgments
The authors would like to thank the invaluable contributions of Dr. Bharati Kangrali (MBBS, MD, General Medicine) in the thorough clinical assessment and clerking of the case presented in this case report.
Authors’ Contribution
All authors contributed equally to conceptualizing the study and conducting the literature review, and they collaborated in writing and revising the manuscript. All the authors have read and approved the final version of the manuscript and agreed to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
A single case report is exempt from ethical approval in our institution. Written informed consent was obtained from the patient for the publication of this report and the accompanying images.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
