Abstract
Background
Di-(2-ethylhexyl) phthalate (DEHP), a plasticizer compound affecting female reproduction, leads to scenarios, such as polycystic ovarian syndrome (PCOS) and infertility through oxidative stress (OS) mechanisms. Ascorbic acid (AA) is one of the antioxidants in infertility issues.
Objectives
The present study investigates the ameliorative effect of AA on DEHP-induced ovarian toxicity in pubertal female Wistar rats.
Materials and Methods
Thirty female Wistar rats of four weeks of age were stratified into five groups. Group I was treated with corn oil (Vehicle), groups II and III with low and high dose DEHP, and groups IV and V with low and high dose DEHP+AA were administered for 30 days.
Results
Increased body weight gain was noted in DEHP groups. Estradiol hormone was considerably reduced, whereas progesterone levels were increased in both low- and high-dose DEHP-treated groups. DEHP+AA groups have shown significant (p < 0.005) protection of these hormone levels as equal to the control group. The high-dose DEHP group shows an increased, ovarian estrogen receptor (ER) alpha, ER-beta, and progesterone receptor gene expression, and DEHP+AA groups have significantly (p < 0.005) showed expression similar to the control. OS was noted with decreased superoxide dismutase and increased malondialdehyde expression in Group III (GR III) compared to control, whereas the DEHP+AA treated group significantly protected OS by restoring the expression levels. DEHP-treated groups show elevated levels of both Bcl-2 and BAX which is specific to apoptotic expression and restored by AA treatment (p < 0.005).
Conclusion
Evidence suggests that AA may protect against DEHP-induced ovarian toxicity by decreasing OS levels, improving folliculogenesis, and restoring the hormonal with receptor level alterations.
Keywords
Introduction
Phthalate esters are a type of chemical that can interfere with our hormones on excessive exposure and have been demonstrated to trigger reproductive toxicity in animals, fetal death, cancer, deformities, and liver and kidney damage in humans. 1 Di-(2-ethylhexyl) phthalate (DEHP) is one such phthalate, present in daily consumables and polyvinyl chloride for increasing flexibility. DEHP bonds to the plastics non-covalently hence it leaches out to the environment making it a pollutant causing endocrine disruption. 2 In humans, DEHP exposure is through ingestion, inhalation, and dermal contact. 3 Thus, DEHP disturbs the body’s physiological mechanism resulting in toxicity. 4 Phthalates can disrupt normal reproductive function in women’s ovaries at critical developmental phases.5, 6 Phthalates are known to have an impact on folliculogenesis and steroidogenesis altering ovarian function. 5 DEHP, an estrogen agonist, and likely androgen antagonist, changes gene expression, many of which are needed for offspring maturation. 7 The consequences of DEHP on rat ovaries decreased the maturation rate, ovarian size, and the number of antral follicles. 8 DEHP reduced antral follicle size and more apoptotic cells in ovaries. 9 The experiment showed that DEHP-cultured ovaries dramatically increased oxidative stress-related gene expression. 10
Vitamin C or ascorbic acid (AA) is a water-soluble vitamin with antioxidant characteristics. AA is an important antioxidant that plays numerous roles in reproductive processes, such as follicle development, hormone synthesis, tissue remodeling, follicle repair, and protection from oxidative impairment. 11 The antioxidant activity of AA was shown to reduce the damage caused by DEHP exposure in male rats.12, 13 AA decreases folliculogenesis in neonatal mouse ovaries through mono-(2-ethylhexyl) phthalate induction through decreasing ovarian oxidative stress.8, 14 Mice treated with mono-(2-ethyl-5-hexyl) phthalate (MEHP) had more primordial follicles and fewer immature follicles than control rats. AA reduces oxidative stress and protects against MEHP-induced ovarian dysfunction. 8 Furthermore, antioxidants AA reduce ovarian reactive oxygen species (ROS) levels and induce oxidative stress, apoptosis of granulosa cells, disruption of oocyte communication, and oocyte nourishment, which degrades the oocyte. 15 Estradiol is critical for the folliculogenesis process as well as for the preservation of the female character of the ovarian somatic cell. 16 MEHP increases the percentage of primary and secondary follicles and decreases the number of juvenile follicles in the ovaries, speeding up primordial follicle acquisition in mice. MEHP also increased malondialdehyde (MDA), glutathione, and superoxide dismutase (SOD) levels, causing ovarian oxidative stress. AA and DEHP treatment groups returned the oxidative stress and reduced follicular composition. MEHP-treated ovaries had higher MDA levels and reversed with AA+MEHP treatment confirming the ameliorative effect of AA in vitro in neonatal mice ovaries. 14 Few published data are available on how the antioxidant vitamins protect DEHP toxicity in male rats. Though data on AA protecting the DEHP-induced female reproductive toxicity in vitro is available the evidence in animals is vague concerning the female reproductive system. This study aims to investigate the influence of AA on DEHP-induced ovarian gene alterations in pubertal female Wistar rats and evaluate its potential as a protective intervention against the reproductive toxicity of DEHP.
Materials and Methods
Animals
Thirty Female Wistar rats of post natal day (PND) 30 were acquired through the Central Animal House Facility, Manipal Academy of Higher Education, Manipal for the study purpose. Animals were kept following the regulations and practices of the Committee for Control and Supervision on Experiments on Animals (CPCSEA) with the permission of the Institutional Animal Ethics Committee (IAEC NO: IAEC/KMC/84/2022). The rat was kept in cages with 12-h light/dark cycles at 21°C–23°C. Female Wistar rats of PND 30, were kept under observation with appropriate body weight measurement. On PND 35, the rats were evenly assigned into below mentioned groups. The rats were kept in cages made of polypropylene and fed with a standard amount of food pellets and water was kept at ad libitum.
Reagents and Chemicals
Chemicals were purchased from Sigma-Aldrich, Dioctyl phthalate (D201154), L-Ascorbic acid/Vitamin C (A92902), and Corn oil (C8267) were used. Reagents brought from Takara RNA iso plus (CAT# 9108), Prime Script RT reagent kit (CAT# RR037A), and TB green premix Ex TaqTM II (CAT# RR820A) were used for gene analysis. Furthermore, the remaining substances needed for the processes were bought from MP Bio and SRL.
Experimental Design
The DEHP treatment was given for 30 days. The dosage quantity was modified in conjunction with the animal’s body mass, and the new solutions were made on consecutive days. AA was administered through water consumption since it is a water-soluble vitamin. PND-35 rats were oral gavage to treatment according to the appropriate group protocol. Group I was the vehicle group of controls, Group II 10 mg/kg of body weight of DEHP (low dose), Group III 100 mg/kg of body weight of DEHP (high dose), Group IV 10 mg/kg of body weight of DEHP with 100 mg/kg of body weight of AA, and Group V 100 mg/kg of body weight of DEHP with 100 mg/kg of body weight of AA. At PND 50–65 days the Estrous cycle was evaluated. After 30 days of treatment the animals in diestrus were carried for blood collection and further sacrificed for ovarian tissue collection. Sacrifice was done after Pentobarbitunol––IP and decapitation. Blood was drawn and the separated serum was kept at −20°C until hormone estimation. Ovarian tissue was promptly removed, and the adjoining fat tissues were excised and stored in a nucleic acid sample protector for gene expression studies.
Ovarian Hormone Analysis
Serum samples stored were used for Estradiol (E2) and Progesterone (P4) analysis. The test was performed using the ELISA technique. The estimation of E2 and P4 is done using the commercially available Rat ELISA kit from MY BIOSource. E2 kit (Cat No. MBS2607338) and Progesterone (Cat. No: MBS3809064) were performed according to the manufacturer’s instructions. The sensitivity of the assay was 5 pg/mL for E2 with ≤8% intra-assay precision. Sensitivity for P4 was 10 ng/mL with 15% in the intra- and inter-assay CV.
Ovarian Gene Expression Analysis
Takara RNA iso plus was used to isolate total RNA by following the manufacturer’s guidelines. Agilent BioTek Epoch microplate spectrophotometer was used to quantify the extracted RNA. Prime Script RT reagent kit from Takara was used to produce complementary DNA (cDNA) from total ovarian RNA. This produced cDNA with its forward and reverse primer (Table 1) along with TB green premix Ex TaqTM II was incorporated into for qRT PCR estimation process. A Real-Time PCR machine (Applied Biosystems-Quant studio 5) was used to conduct the reactions in triplicate under predetermined PCR reaction parameters. The NCBI BLAST program was utilized in the primer creation process. Using the 2−∇∇CT approach, the comparative threshold cycle (CT) was measured and estimated for comparative expression. Each fact point was normalized to the housekeeping gene, beta-actin, to calculate the normalized subjective value for each gene.
Gene Expression Primer Sequence.

Statistical Analysis
Using GraphPad Prism 8.0, all data were analyzed, and represented as Mean ± SEM. Two-way analysis of variance (ANOVA) was applied to enumerate the variations in body weight gain between groups on Days 1, 7, 14, 21, and 28. One-way ANOVA was employed to assess the alterations among groups, following the Tukey test for multiple comparisons. The level of significance was set to 0.05.
Results
Effect of AA on DEHP Induced Ovarian Toxicity Concerning Body Weight
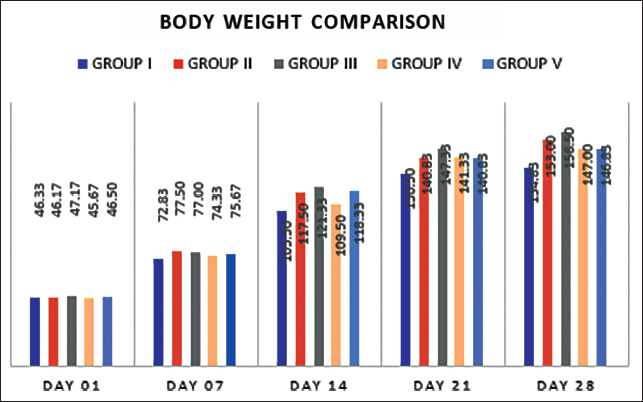
Our findings show that low and high doses of DEHP groups have shown an incline in body weight in contrast to the control population (Figure 1). We also compared the body mass index of rats treated with AA, which showed to have a low body mass index compared to the low dosage and high-level DEHP. The body weight was significantly lowered in AA-treated groups (GR III vs GR V) compared to the DEHP groups with a (p = 0.05).
Effect of AA on DEHP Induced Ovarian Toxicity Concerning Serum E2 and P4 Status
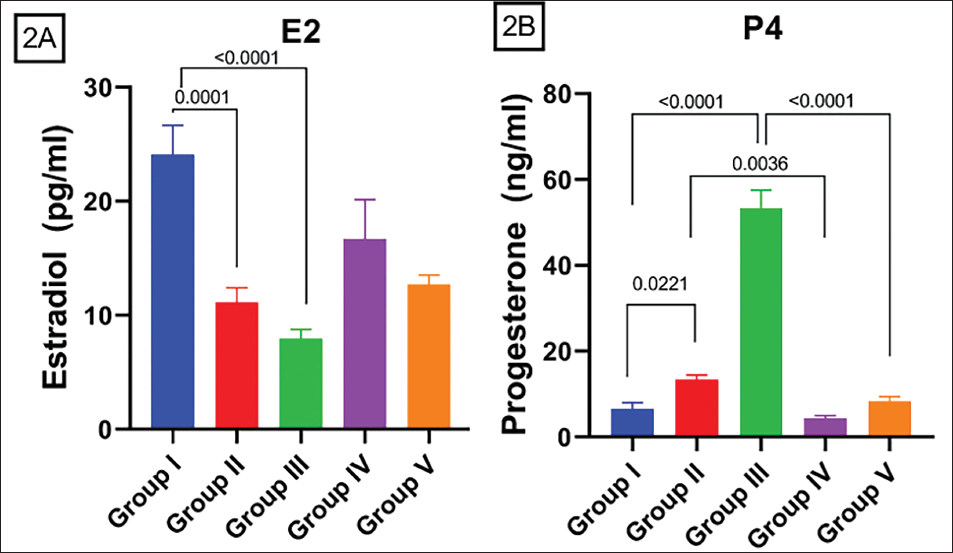
Serum estradiol level has fallen on exposure to DEHP in both groups treated solely with DEHP. E2 concentration lowered significantly compared to Control and GR II (p = 0.05) and compared to Control and GR III (p < 0.0001). This trend of diminishing the E2 hormone level is reversed in groups treated with AA. Significant decrease in hormone levels in comparison to GR II and GR IV (p = 0.0499) (Figure 2A). There is a significant elevation in P4 concentration in contrast to Control and GR II (p = 0.0221), compared to GR I and GR III (p = 0.05) (Figure 2B). There is an increase in P4 concentration with significance compared to GR II and GR IV (p = 0.05) and between GR III and GR V (p = 0.05) (Figure 2B).
Effect of AA on DEHP Induced Ovarian Toxicity Concerning Ovarian Hormonal Receptor Gene Alterations
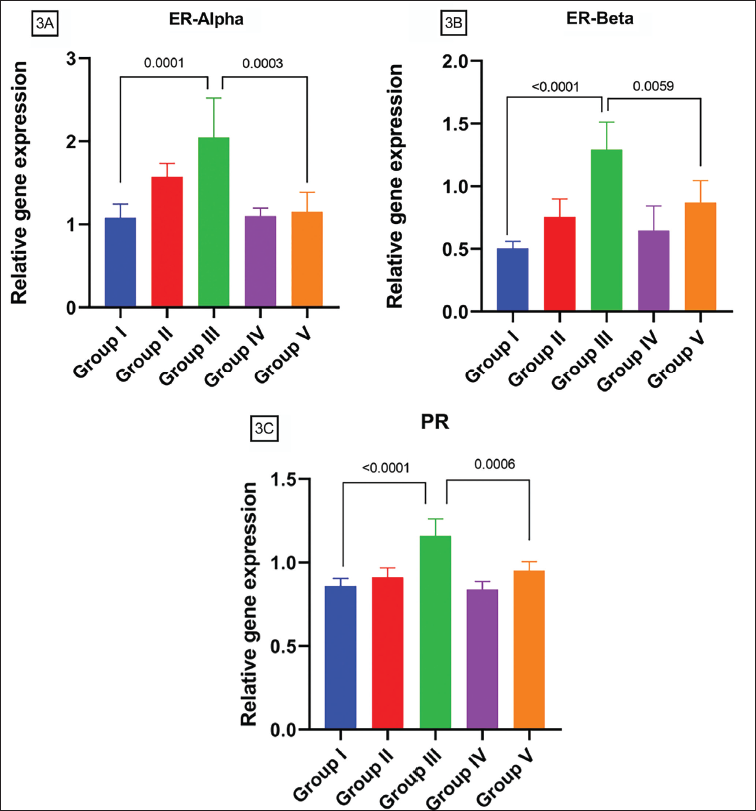
ER alpha and ER beta (Figure 3A and B) have a similar trend with exposure to DEPH. Both genes’ concentrations were raised in a dose-dependent pattern with a significance in comparison of Control and GR III (p = 0.05) but only ER beta has a significant change in GR II and GR III. AA reduces both genes’ levels of DEHP exposure significantly compared to GR III and GR V (p = 0.05) for ER alpha and ER beta. PR receptor concentration is increased in GR III compared with Control (p = 0.05) and enhanced frequency is also observed in high dose to low dose comparison (Figure 3C). AA significantly downregulated the alteration of increased PR gene levels in rats exposed to DEHP in comparison to GR III and GR V (p = 0.05).
Effect of AA on DEHP Induced Ovarian Toxicity Concerning Ovarian Oxidative Stress Gene Alterations
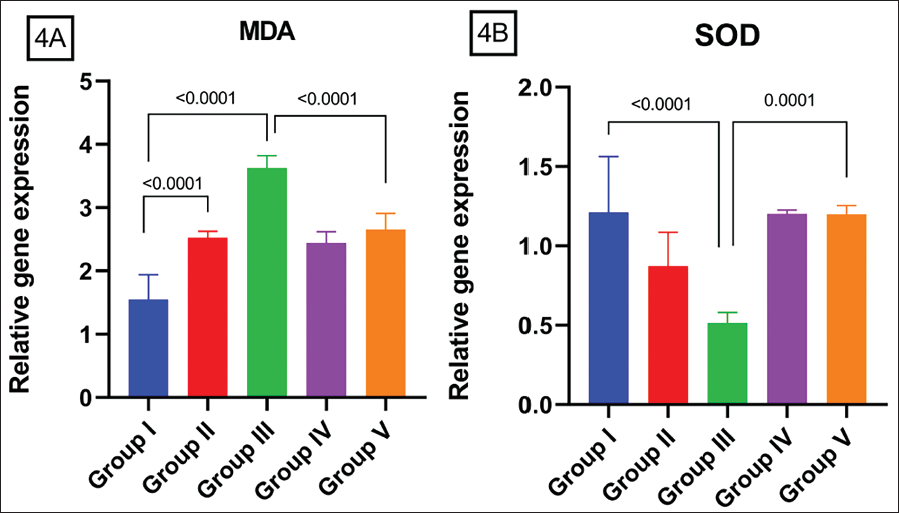
Compared with the Control group MDA is upregulated in both low and high doses of DEHP groups in a dose-dependent fashion with a significance of (p = 0.05) (Figure 4A). And AA has been shown to decline this raised MDA level significantly (p = 0.05) when compared to GR III and GR V. SOD level is downregulated on exposure to DEHP on both doses of DEHP significantly compared to GR I and GR III (Figure 4B) and GR II and GR III (p = 0.05). And this trend is reverted with AA administration showing a significant increase in SOD level (p = 0.05) in the high dose group (GR III vs GR V).
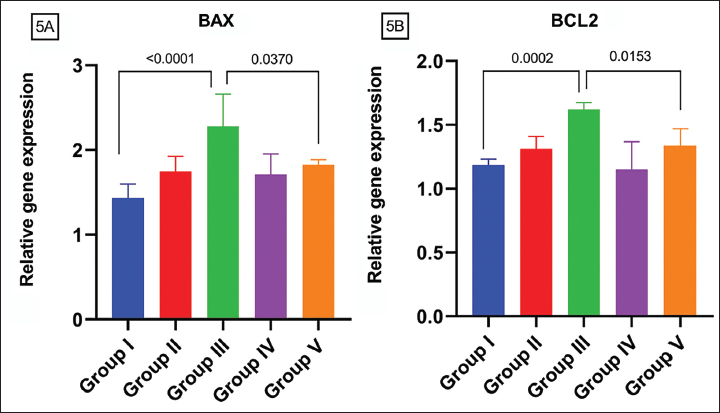
Effect of AA on DEHP Induced Ovarian Toxicity Concerning Ovarian Apoptotic Gene Alterations
BAX gene level is uplifted on exposure to DEHP exposed groups significantly (p = 0.05) in comparison to GR I and III and compared to GR II and GR III. AA declined this trend in comparison to GR III and GR V (p = 0.05) (Figure 5A). Bcl-2 gene expression has a similar trend as BAX showing an upraised tendency among both doses of DEHP, more significantly among GR I and GR III (p = 0.05) than GR II and GR III (p = 0.05). AA subsided this trend significantly compared to GR III and GR V (p = 0.05) (Figure 5B).
Discussion
DEHP is an environmental endocrine disruptor chemical causing multiple organ toxicity consequences for humans and other primates. Humans are exposed to such endocrine disruptors and their metabolites in several ways. Several studies have demonstrated that DEHP interferes with several ovarian hormone production and disrupts its functions.17, 18 DEHP exposure could cause obesity by disrupting metabolic homeostasis. This is potentially because hypothyroidism and hypothalamic suppression of leptin cause obesity. 19 The impact of DEHP on the growth status of rats can be assessed using body weight, a significant nonspecific measure that fully captures the toxicity of chemicals. 20 The current experimental study, showed DEHP treatment increased the animal’s body weight and the AA-treated groups demonstrated low body weight compared to both low- and high-dose DEHP groups restoring adiposity significantly as control-treated animals. According to Davis et al. 21 after treatment, preovulatory follicle granulosa cell estradiol synthesis was dramatically decreased by DEHP. Ovulation failed to occur when blood estradiol levels dropped because this led to an independent rise in FSH without simultaneous triggering of the LH spike. DEHP exposure to animals and subsequent F1 offspring demonstrated decreased serum estradiol levels in the group that was administered 20 g/kg/day of DEHP. 18 DEHP exposure dose-dependently elevated serum progesterone when compared to the control group.14, 18
Previous studies on DEHP and MEHP decrease the growth of antral follicles with decreased estrogen production and cell-cycle factors which are essential for proliferation and prevent apoptosis. 22 Our study also confirmed decreased serum estradiol levels in DEHP-treated groups which was in line with the previous study. DEHP exposure increased progesterone hormonal levels.18, 23 Our study also showed an increase in serum progesterone levels in DEHP-treated groups compared to the control. The level of alteration depends on the DEHP concentration, and it was in a dose-dependent manner.
An antioxidant is a molecule that is sufficiently solid to give an electron to a free radical that has caused damage in the body, this neutralizes the free radical and reduces its ability to cause harm. 24 The available evidence25, 26 suggests that free radicals play a role, at least partially, in the process behind the hormonal dysregulation that is caused by DEHP. According to Choi et al. 25 inhibition of DEHP-mediated enhanced production of free radicals was achieved by supplementation with antioxidants, such as Vitamin C and/or Vitamin E, free radicals that are accountable for the oxidation of lipids and DNA, and which may be associated with hormone instability. AA protects against these negative effects by changing these hormone levels.
Both ER-alpha and ER-beta play crucial functions in biological functions involving estrogens. 27 Issues related to estrogen signaling are associated with a variety of pathological conditions, including cancer and metabolic disorders. 27 The progesterone receptor (PR) belongs to the nuclear receptor superfamily, which regulates the expression of target genes in response to hormonal stimulation. 28 Studies also suggest that many phthalate esters operate as hormone receptor agonists and antagonists simultaneously. 29 Mice treated with DEHP had reduced cyst breakdown; ovary organ cultures demonstrated that the impairment was mediated via estrogen receptors (ERs), which were reversed. 30 In our study, the ovarian gene expression is altered after DEHP exposure. PR, ER-alpha, and ER-beta were upregulated by DEHP exposure and DEHP with AA treatment restored these levels as compared to DEHP treated groups. In experiments by Tripathi et al., 31 the results suggest that DEHP generates ROS, modulates steroidogenic genes to prevent steroid production, and triggers necrosis in rat granulosa populations of cells by activating mitochondrial apoptotic pathways. As previously published by Fu et al.15, 32 on how DEHP-induced alterations in the fecal microbe profile and their intermediates produce reproductive damage in females, they measured ovarian MDA and SOD and blood proinflammatory markers in DEHP-exposed animals and results suggested that DEHP-exposed groups had higher MDA and lower SOD than the control group. According to Wang et al., 8 findings show that DEHP suppresses follicle development through increasing ROS and lowering SOD1 expression and function. 8 Present findings showed a similar trend where the oxidative stress-related ovarian gene SOD and MDA were altered after DEHP administration. MDA concentrations were increased due to DEHP exposure in a dose-dependent fashion relative to the control group. AA has been shown to revert this change by decreasing the MDA level more significantly in GR V compared to GR III (p < 0.05). SOD levels were reduced due to DEHP exposure and AA reverses this by increasing SOD levels in the groups treated with DEHP+AA. DEHP declined cell division, halted the cell cycle, and caused cell death in differentiated human fetal development through the apoptotic gene pathway. 7 Mitochondrial apoptotic pathway control is taken care of by certain essential proteins, such as Bcl-2 and BAX family proteins which inhibit caspases. Increased BAX leads to histological alterations, which include weakened apoptotic cell attachment to neighboring cells, genomic DNA fragmentation, ribosome release, and diminished cell survival and repair. 5 Increased concentrations of the antioxidant enzymes SOD1 and CAT and lowered levels of the pro-apoptotic proteins BAX and Bcl-2 were observed in rats exposed to DEHP10, 33. In the present study, apoptotic genes of the ovary BAX and Bcl-2 were also upregulated according to the increased dosage in the groups treated with DEHP which was in line with the previous study, and expressions were protected in the rats treated with DEHP+AA. Therefore, AA could be protective against the apoptotic expressions caused by DEHP exposure. Follicle formation in adult ovaries was inhibited in the first generation, it was disturbed in their offspring, and the primordial follicles in the neonatal ovary were hastened by DEHP exposure. 18 Kil et al.31, 34 studies on DEHP exposure in mice during prenatal and lactation, and in F1 generation demonstrate ovarian genetic abnormality and morphological alterations identical to polycystic ovarian syndrome (PCOS) patients. DEHP-treated ovaries had more oocytes in their cysts than the normal group. 10 Previous study findings confirmed the therapeutic importance of AA for the ovary by reducing MEHP-triggered oxidative stress in the ovary and follicular imbalance.14, 35 The present study complements AA as an antioxidant effect on DEHP ovarian toxicity and adds knowledge on in vivo effects.
Conclusion
According to the study, we conclude that reproductive toxicity due to DEHP exposure includes reproductive hormonal changes, bringing oxidative stress and leading to apoptotic gene activation. Since DEHP is an endocrine disruptor, enters the system through different routes and causes reproductive disorders. It is important to protect our human system from endocrine disruptors. AA could have ameliorative effects on both high- and low-dose DEHP. In this work, we could conclude that AA ameliorates oxidative stress and protects from DEHP-induced ovarian abnormalities. Study results provide valuable insights into the protective effects of AA against DEHP-induced reproductive toxicity in female rats, potentially offering a basis for future interventions and therapeutic strategies. However, more research is needed to prove that AA protects against DEHP-induced ovarian damage and to elucidate the physiological pathway underlying these alterations, PCOS: Polycystic ovarian syndrome.
Abbreviations
Footnotes
Acknowledgments
The authors gratefully acknowledge all the other faculties and technical staff of the Department of Medical Laboratory Technology, Manipal Centre for Biotherapeutic Research, Department of Pharmacology, Manipal Academy of Higher Education, Manipal, for helping us out in conducting the animal and experimental work.
Authors’ Contributions
SA was involved in experimental study, data analysis, and manuscript writing.
AZ was involved in animal experiments.
KM was involved in designing the study, manuscript correction, and data analysis.
KSRP, RU, AM, AG, and RNS were involved in designing the methodology and manuscript editing.
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Ethical Approval
Animals were kept following the regulations and practices of the Committee for Control and Supervision on Experiments on Animals (CPCSEA) with the permission of the Institutional Animal Ethics Committee (IAEC NO: IAEC/KMC/84/2022).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the Manipal Academy of Higher Education for providing the seed grant to conduct this project.
Informed Consent
Not applicable.
