Abstract
Purpose
Forskolin is primarily found in the roots of the Coleus forskohlii plant, historically employed in Southeast Asian and Indian Ayurvedic medicine. Mast cells play a crucial role in fibrosis progression, yet their activation and inhibition in animal models are understudied; thus, we explored forskolin’s impact on kidney fibrosis.
Materials and Methods
Forskolin was evaluated in a mouse model for stem cell factor-induced histamine release, and plasma histamine levels were measured using the enzyme-linked immunosorbent assay. Kidney fibrosis was developed by unilateral ureteral obstruction (UUO). Renal function was assessed by spectrophotometric measurements of serum blood urea nitrogen (BUN) and creatinine. The gene expression of collagen, transforming growth factor-beta (TGF-β), α-smooth muscle actin (α-SMA), interleukin-1 beta (IL-1β), and mast cell protease-5 (MCPT-5) in the kidney was analyzed by reverse transcription polymerase chain reaction (RT-PCR). Histopathological changes in the renal tissues were examined by hematoxylin and eosin (H&E) and Masson’s trichome stain.
Results
Our results showed that 3 mg/kg forskolin inhibited SCF-induced plasma histamine release in a mouse model. In the 7-day UUO model, forskolin significantly showed inhibition of serum creatinine and blood urea nitrogen compared with the disease group. Forskolin significantly inhibited elevated expression of collagen, TGF-β, α-SMA, IL-1β, and MCPT-5 in the kidneys. Histopathological observation of H&E and Masson trichome-stained kidney forskolin demonstrated a reduction in inflammatory cells, pelvic and tubular dilation, and fibrosis.
Conclusion
Forskolin showed an anti-fibrotic effect in UUO-induced renal fibrotic mice. Most significantly, forskolin administration showed a decrease in the expression of the mast cell protease MCPT5 in the kidneys. These results imply that forskolin, through modifying SCF activity, may be a viable potential treatment for the attenuation of tubule-interstitial fibrosis.
Introduction
Chronic kidney disease (CKD) is one of the leading causes of death worldwide. Renal fibrosis is crucial for its development, regardless of the cause of CKD. Fibrosis is commonly known as scarring of tissue resulting from organ injury, including CKD, where progressive fibrosis is linked with deteriorating renal activities.1, 2 Renal interstitial fibrosis and inflammation are extensively studied using an experimental paradigm known as unilateral ureteral obstruction (UUO). Numerous variables that contribute to fibrosis and damage in UUO have also been linked to fibrotic kidney diseases in humans. Fibrosis characteristics comprise the presence of tissue proteases such as matrix metalloproteinases (MMPs), 3 elevated expression of transforming growth factor-beta (TGF-β), 4 and high expression of α-smooth muscle actin (α-SMA), which is the myofibroblast marker. Additionally, increased interstitial leukocyte recruitment with an elevation in CD4+ T cells, 5 mast cells, and macrophages 6 is accompanied by progressive renal damage in UUO.
Forskolin, a naturally derived diterpenoid observed in the root of Coleus forskohlii, has long been utilized in Southeast Asian and Indian traditional medicine. 7 Forskolin, with potential cAMP pathway interactions, has important therapeutic advantages due to its interaction with several metabolic disorders, malignancies, and other conditions. Forskolin’s effects have reportedly been explored in a variety of clinical trials, including cystic fibrosis, metabolic disorders, obesity, glaucoma, asthma, and chronic obstructive pulmonary disease (COPD). 8
Mast cells have been linked to a variety renal disorders through an examination of how they infiltrate the disease and how their accumulation corresponds to the severity of the condition and decline in renal function. These diseases include diabetic nephropathy (DN), chronic inflammation, and fibrosis. 9 Mast cells are essential for regulatory T cell activity and tolerance, but as an inflammatory mediator, they are more likely to affect acute kidney injury. Mast cells have a significant role in the synthesis of cytokines and chemokines, which are necessary for leukocyte adhesion and recruitment, as well as TNF-α. 10 Mast cells highly express Fc epsilon receptor 1 (FcεRI), the receptor for immunoglobulin E (IgE), and c-Kit, a type III receptor tyrosine kinase and activation of both receptor results in degranulation of mediators.11, 12 A multipotent growth factor that plays an important role in the formation of mast cells is the c-Kit ligand stem cell factor (SCF). SCF causes immature mast cells to proliferate and differentiate from CD34+ progenitors, as well as chemotaxis and mast cell existence. 13 SCF activation increases the inflammatory response by synergistically elevating the degranulation and synthesis of cytokines. 14 Mice with genetic deletions unique to mast cells have recently made it easier to study the roles of mast cells in vivo. Mast cells played an important role in developing hydronephrotic kidneys in WT mice with augmented collagen deposition, amplified expression of TGF-β, α-SMA, and MMP12, and enhanced leukocyte recruitment. However, mast cell-deficient KitW-sh/W-sh mice did not demonstrate fibrosis injury in the UUO model. 15 This study indicates that mast cells play a very important role in the development of renal fibrosis.
In this study, we sought to confirm that forskolin is impeding the SCF pathway. Furthermore, we investigated whether forskolin could affect the pathogenesis of fibrosis in UUO mice by analysing blood urea nitrogen (BUN) and creatinine serum levels as well as collagen, TGF-β, α-SMA, interleukin-1 beta (IL-1b), and Mast cell protease-5 (MCPT-5) gene expression in kidney tissue by RT-PCR. MCPT5, which is known as Cma1, encodes a mast cell protease (MCP) that is expressed in tissue mast cells. Histopathological analysis was carried out using haematoxylin and eosin (H&E) and Masson’s trichrome-stained kidney sections. Pyridone class pirfenidone was used as the standard.
Materials and Methods
Reagents
Kits for analyzing the concentrations of BUN and creatinine were purchased from Siemens India (Siemens Limited). An enzyme-linked immunosorbent assay (ELISA) kit used to determine the concentration of histamine was purchased from Oxford Biomedical Research, USA. Mouse recombinant SCF protein was obtained from R&D Systems (USA). For gene expression analysis, the RNeasy Mini Kit (Qiagen), SuperScript™ VILO™ cDNA Synthesis Kit, and TaqMan™ Fast Advanced Master Mix (Thermo Fisher Scientific, USA) were used. TaqMan Primers TGF-β (Mm01178820_m1), Col-1(Mm00801666_g1), α-SMA (Mm00725412_s1), IL-1β (Mm00434228_m1), MCPT-5 (Mm01323894_m1), were purchased Thermo Fisher Scientific, USA.
Animals
Female BALB/c mice (8–9 weeks old) and C57BL/6 male mice (body weight 22–26 g) were obtained from an in-house animal facility at Aurigene Pharmaceutical Services Ltd. All animals were maintained in a temperature (22 ± 3°C) and humidity (50 ± 20% RH) controlled vivarium under a 12-h light/dark cycle provided with food and water ad libitum.
Compliance with Ethical Standards
All animal protocols were carried out in compliance with institutional policies, and the Institutional Animal Ethics Committee. Animals were treated humanely to alleviate suffering. The animal ethical protocol approval numbers are APSL/IAEC/142-01/10-2021 and APSL/IAEC/148-02/06-2022.
SCF-Induced Histamine Release Model
The model was adapted as described by Hu et al. 16 , Briefly, histamine release in the blood was measured to quantify the degree of activation after administration of recombinant SCF to mice to promote mast cell activation. Female BALB/c mice received a single intraperitoneal injection of 1 and 3 mg/kg forskolin. Intravenous injection of SCF was given an hour after the initial dose. Anti-c-kit antibody (ACK-2) was used as the standard. In total, 15 minutes after the SCF injection, the mice were euthanized, and blood was collected by heart puncture for histamine measurement. Plasma histamine levels were estimated using the Histamine EIA ELISA Kit (EA31) from Oxford Biomedical Research.
UUO-Induced Kidney Fibrosis in Mice
UUO surgery was conducted as per Atkinson et al. 17 procedure. Briefly, animals were administered preoperative analgesia subcutaneously (buprenorphine 0.05 mg/kg and carprofen 5 mg/kg), and the same was used post-surgery for three days. The anesthesia used at 2–5% isoflurane and a flow rate of 1 L/min. After placing the animal on its right side on a heated 37°C surgery table, the animal’s left flank was shaved and disinfected with povidone-iodine. On the left flank, 5 mm of the left ribs were cut vertically downward with a scalpel. The fat was cleared, and the ureter was identified, ligated with a double ligature of a 6-0 nylon suture, and placed back. The sham animals underwent a similar surgical procedure without ligation. A continuous suture made of Ethicon 5-0 was used to repair the muscle wall and the skin wound was closed using reflex skin closure clips. After recovery in a warm recovery box, the animals were returned to their home cages. Animals from Group 1 (sham control) and Group 2 were administered with vehicle, Group 3 with pirfenidone 100 mg/kg p.o., and Groups 4 and 5 with forskolin 1 and 3 mg/kg i.p. for 7 days from the day of surgery. On the day of termination, blood was collected by retro-orbital plexus under anaesthesia for BUN and creatinine. After euthanasia, kidney tissue was collected for gene expression analysis for TGF-β, α-SMA, Collagen, IL-1β, MCPT-5, and histopathology analysis for fibrosis by H&E and Masson trichrome staining. Plasma creatinine and BUN levels were estimated using the Dade Behring Dimension from Siemens.
Quantitative RT-PCR
RT-PCR was performed as described by Taketomi et al. 18 The Applied Biosystems 7500 instrument was used for qRT-PCR. Total RNA was extracted from mouse kidneys using TRIzol reagent, in accordance with the manufacturer’s recommendations. High-capacity cDNA Reverse Transcription Kit was used to create first-strand cDNA. Using a real-time PCR system, quantitative PCR was carried out using a predesigned primer-probe pair (TaqMan Gene Expression Assay), and TaqMan Gene Expression Master Mix. TaqMan Gene Expression Assays for TGF-β (Mm01178820_m1), Col-1(Mm00801666_g1), α-SMA (Mm00725412_s1), IL-1β (Mm00434228_m1), and MCPT-5 (Mm01323894_m1) were used. Expression levels of the transcripts were normalized to GAPDH (Mm99999915_g1), and fold changes were calculated using the ∇∇Ct method. In the treatment groups, the percentage change in gene expression was calculated by comparing disease control gene expression. 18
Histology
On day 7, kidney tissue from each mouse was fixed in 10% (v/v) formalin solution, embedded in paraffin, and cut into 4 µm sections. These sections were then stained with Masson’s Trichome stain and H&E dye. The quantification matrix score was utilized for the quantitative analysis of tubular dilation, pelvic dilation, glomerular atrophy, and fibrosis. 19
Statistical Analysis
GraphPad Prism 9 software was used for the creation of graphs and statistical analysis. P values were determined using either a Student’s t-test or a one-way ANOVA with Dunnett’s post-test. Statistical significance was set at p < 0.05.
Results
SCF-Induced Histamine Release
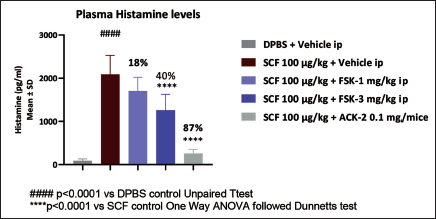
Forskolin was investigated in a therapeutically appropriate mouse pharmacodynamic model of mast cell activation to assess its effectiveness in vivo. Clinical studies have demonstrated that systemic treatment of recombinant human SCF causes functional activation of mast cells in vivo. In addition, SCF stimulates mast cell hyperplasia in non-human primates. 20 To stimulate mast cell activation in mice, we used recombinant rodent SCF. The histamine release in the blood was measured to determine the degree of activation. The study began with IP administration of forskolin to mice at 1 and 3 mg/kg. SCF (100 µg/kg) was injected intravenously an hour after the initial dose. The mice were euthanized 15 minutes after receiving SCF injection, and blood was collected for histamine measurement by heart puncture. In Figure 1, forskolin administration demonstrated a dose-dependent reduction in SCF-induced histamine release. A significant reduction in histamine release was observed at 3 mg/kg. The ACK-2, used as a standard, significantly inhibited histamine release.
Forskolin Inhibits SCF-Induced Histamine Release.
Forskolin Effect in UUO Model
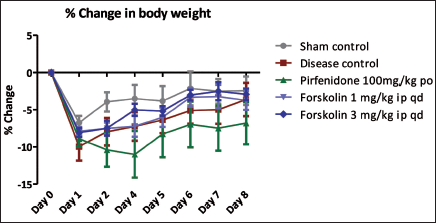
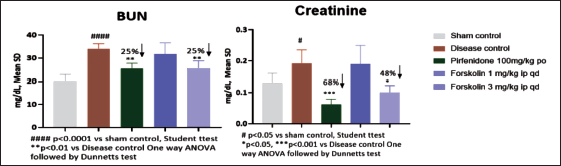
In the current study, UUO surgery inhibited body weight change from day 0 onwards. However, no significant changes were observed across the treatment groups (Figure 2). The UUO group showed a significant increase in BUN and creatinine quantities at the end of the study period. Forskolin and pirfenidone treatment caused significant inhibition of creatinine and BUN levels compared to the disease control group (Figure 3).
Effect of Forskolin on Body Weight.
Effect of Forskolin on BUN and Creatinine.
Quantitative RT-PCR
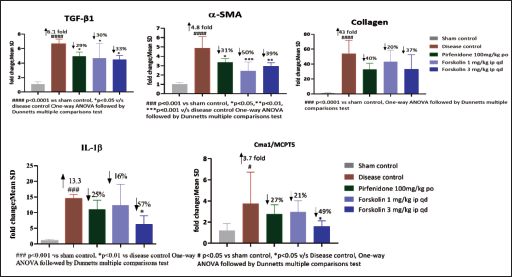
The current study investigated the effect of forskolin on fibrosis-related gene expression of TGF-β, which is believed a key mediator in the initiation and advancement of interstitial fibrosis. 21 As shown in Figure 4, the expressions of TGF-β, α-SMA, collagen, IL-1β, and MCPT5 increased in the kidney of UUO mice, however forskolin treatment inhibited this increase. These results suggest that forskolin medication can protect against the growth of fibrotic injuries in UUO kidneys and has protective benefits. The expression of MCPT5, which is a key factor in the development of fibrosis, indicates the existence of tissue mast cells. 15 In the current study, the disease control UUO group demonstrated a significant elevation in MCPT5 expression, and treatment with forskolin significantly reduced MCPT5 expression. These findings imply that forskolin may prevent mast cell proliferation through the SCF route during the pathogenesis of fibrosis.
Effect of Forskolin on TGF-β, α-SMA, Collagen, IL-1β, and MCPT5 Gene Expression.
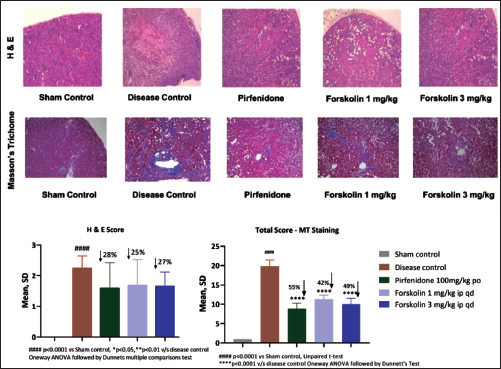
Histology
Kidney histology in the sham control group was normal. The tubular dilation, pelvis dilation, glomerular atrophy, and fibrosis were increased in the obstructed kidneys disease control group compared with the sham group. The forskolin administration showed inhibition in tubular dilation, pelvis dilation, glomerular atrophy, and fibrosis significantly compared with the vehicle group (Figure 5). The kidneys also developed collagen as a result of UUO surgery, as seen by Masson’s trichrome staining. Forskolin treatment significantly decreased the kidney’s tubulointerstitial fibrosis region. These conclusions were supported by measurement of the tubulointerstitial damage shown in the renal tissue sections stained with Masson’s trichrome and H&E (Figure 5). These findings collectively show that forskolin treatment in mice enhances renal function and ceases or delays renal injury and fibrosis.
Effect of Forskolin on H&E and Masson Trichome-Stained Kidney Histopathology.
Discussion
Clinically, UUO is associated with various diseases. According to previous studies, approximately 166 of 1 million people hospitalized in the United States had a preliminary diagnosis of UUO. 22 Intervention is required to relieve the obstruction. According to studies on the time course of experimental obstruction, canine renal function can be reversed if the obstruction is removed after four weeks. 23 However, the length of the occlusion in patients is uncertain. Even after the blockage has been removed, kidney damage may still occur. As a result, we frequently encounter patients who experience renal functional impairment as a result of blockage release. The discovery of pharmacologic medicines that could be employed to protect the renal improvement of the blocked kidney until or after relief could result from the identification of mechanisms that mediate such damage. In the present study, the effects of forskolin on kidney obstruction were evaluated for a week, and its effects on renal fibrosis development and MCP were estimated.
Multiple forms of renal illness have been linked to mast cells. Mast cell accumulation is positively correlated with higher kidney damage and lower immunoglobulin function. Drug-induced nephritis, polycystic kidney disease, type A nephropathy, renovascular disease, amyloidosis, allograft rejection, and diabetic nephropathy. 6 The majority of experimental investigations that have associated increased TGF-β production and interstitial fibrosis with higher mast cell counts and staining intensity have been confirmed.9, 23, 24 These studies indicate that mast cells are linked with the pathogenesis of renal fibrosis.
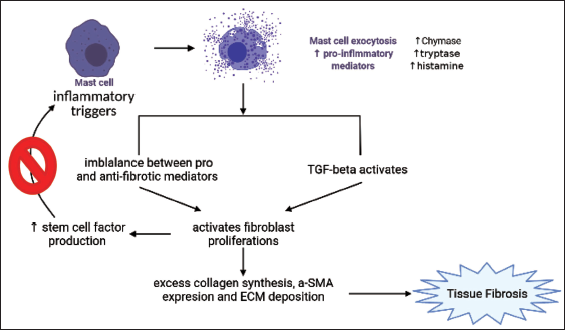
Many bioactive mediators found in mast cells, such as proteases, growth factors, leukotrienes, cytokines, and chemokines, have the potential to cause fibrosis. In a cutaneous hypersensitivity model, the ability of mast cells to enhance collagen synthesis and fibrosis was originally reported approximately two decades ago. According to this concept, mast cell stimulation encourages skin fibroblasts to produce more TGF-β, which leads to increased collagen deposition. 25 Mast cell profibrotic effects are tightly correlated with TGF-β. Mast cells produce and activate TGF-β, through chymase production. 26 Interestingly, TGF-β is also a strong mast cell chemo attractant. 27 In the SCF-induced histamine release model, forskolin inhibited SCF-induced histamine release, indicating that it interferes with the SCF pathway, which controls mast cell survival and proliferation. Furthermore, we evaluated mast cell involvement in a fibrosis model.
TGF-β and other fibrosis markers along with a mast cell marker were evaluated in the UUO fibrosis study. MCPT5 encodes a MCP that is expressed in tissue mast cells which is very important for several functions of mast cells. MCPT5 is involved in the secretion of inflammatory mediators such as chymase, tryptase and histamine from mast cells. These mediators can cause allergic reactions, inflammation and pain. 28 The MCPT5 expression in mast cells is governed by several factors, including growth factors, cytokines, and allergens. MCPT5 expression in mast cells is mainly induced by cytokines such as IL-3, TGF-β, and SCF. 29 The present study demonstrated that the disease-control obstructed kidney expressed significantly high tissue α-SMA, IL-1β, TGF-β, collagen, and more importantly MCPT5 compared to the normal unobstructed kidney group. Forskolin treatment significantly inhibited MCPT5 expression in the kidneys. This indicates that forskolin reduces mast cell activity via the SCF c-Kit axis. Figure 6 demonstrates that SCF is responsible for the activation and triggering of mast cells to initiate the pathogenesis of fibrosis. Histopathological observations revealed that forskolin treatment consistently inhibited mast cell-dependent tubular dilation, pelvic dilation, glomerular atrophy, and fibrosis in H&E and Masson’s trichrome-stained kidney sections. These data indicate that forskolin can protect obstructed kidneys in UUO by impairing the SCF-c-kit pathway for mast cell survival and activation may be a novel approach to exert its anti-fibrotic effects.
Forskolin Inhibits SCF-Induced Inflammation (created on Biorender.com).
The ability of forskolin to prevent SCF from activating mast cells and other inflammatory mediators may serve as the primary or secondary mechanism by which forskolin exerts its therapeutic effects. These findings suggest that forskolin may be a potent alternative therapy for renal fibrogenesis. To determine if forskolin’s anti-fibrotic activity is linked to the suppression of fibrosis-related receptors in the kidneys, further investigation is required. In conclusion, forskolin could be a helpful therapeutic drug for halting the growth of fibrosis, which is a feature of CKD.
The capacity of forskolin to disrupt the SCF pathway in MC survival and activation, as suggested by our results, may represent a unique strategy for forskolin to exercise its anti-fibrotic effects.
Abbreviations
Footnotes
Acknowledgments
The authors are grateful to Aurigene Pharmaceutical Services, Ltd, for approval of the publication of the manuscript. The manuscript approval number was APSL_P301_10/01/2023.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Ethical approval was obtained from Institutional Animal Ethics Committee for the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
