Abstract
The underlying mechanism of fibrosis in classical Hodgkin lymphoma (CHL) remains uncertain. This study aimed to investigate the association of fibrosis in the lymph nodes of patients with CHL through histological examination of the expression of cytokines associated with fibrosis and mast cell proliferation. Additionally, we sought to determine the degree of mast cell infiltration in a nodular sclerosis subtype of CHL (NSCHL) compared with that in non-NSCHL. We analyzed lymph nodes from 22 patients with CHL, of which eight were of the NSCHL and 14 of the non-NSCHL subtype, using immunohistochemical staining of forkhead box P3 (FOXP3), transforming growth factor (TGF)-β, interleukin (IL)-3, IL-13, and stem cell factor (SCF). Mast cells were positive for TGF-β and IL-13, and FOXP3-positive cells were negative for TGF-β. Only the expression of IL-13 in Hodgkin and Reed–Sternberg (HRS) cells was significantly more frequently observed in NSCHL than that in non-NSCHL (P = 0.0028) and was associated with a higher rate of fibrosis (P = 0.0097). The number of mast cells was significantly higher in NSCHL than that in non-NSCHL (P = 0.0001). A significantly positive correlation was observed between the rate of fibrosis and the number of mast cells (correlation coefficient, 0.8524; 95% CI, 0.6725–0.9372) (P <0.0001). The number of mast cells was significantly higher in the group with IL-13-positive HRS cells than that in the group with IL-13-negative HRS cells (P = 0.0157). Based on these findings, we hypothesize that IL-13 production by HRS cells may lead to fibrosis, and furthermore, promote mast cell proliferation and infiltration. This in turn might further produce the fibrotic cytokines IL-13 and TGF-β, resulting in fibrosis typical of NSCHL.
Introduction
Classical Hodgkin lymphoma (CHL) is histologically characterized by the presence of neoplastic cells of large mononuclear and multinuclear cells (Hodgkin and Reed–Sternberg [HRS] cells) as well as extensive inflammatory cells in the background. On the basis of the characteristics of reactive infiltrates and morphology of large mononuclear and multinuclear tumor cells, CHL can be classified into four morphological subtypes: nodular sclerosis subtype of classical Hodgkin lymphoma (NSCHL); mixed cellularity CHL (MCCHL); lymphocyte-rich CHL (LRCHL); and lymphocyte-depleted CHL (LDCHL). 1 NSCHL and MCCHL account for nearly 80% of all CHL cases. The most striking characteristic of NSCHL is the presence of extracellular matrix deposits, which are collagen-rich fibrotic bands surrounding nodules where inflammatory and neoplastic cells mainly reside.
Many studies have documented that CHL is associated with dysregulated cytokine production by reactive lymphocytes and HRS cells. 2 The cytokines not only contribute in determining the histological subtypes of CHL but also systematically account for some for its clinical features. 3 Transforming growth factor (TGF)-β, a cytokine growth factor that induces fibroblast proliferation and fibrosis, has been reported to play an important role in CHL-associated fibrosis. 4 HRS cells have been shown to produce TGF-β mRNA and secrete TGF-β protein in the cell line L-428. 4 TGF-β was predominantly detected in HRS cells obtained from NSCHL using both immunohistochemistry and in situ hybridization. 5 Interleukin (IL)-13 is also known to be associated with fibrosis. 6 Three possible mechanisms have been proposed to explain the role of IL-13 in NSCHL fibrosis: (1) IL-13 stimulates the production of TGF-β by macrophages and TGF-β acts on the collagen-producing fibroblasts; (2) IL-13 directly activates fibroblasts, inducing fibrosis; and (3) IL-13 upregulates arginase activity in macrophages, increasing L-ornithine synthesis from L-arginine. L-proline and polyamines, which are formed from L-ornithine, promote fibroblast proliferation and fibrosis.7,8 In addition to Th2 lymphocytes, HRS cells also produce IL-13. Higher levels of IL-13 are produced by HRS cells in NSCHL than in non-NSCHL. 9
Mast cells are associated with fibrosis in some diseases.10,11 Fibrosis in IgG4-related diseases was thought to be caused by TGF-β-producing regulatory T (Treg) cells. However, a recent study showed that TGF-β-producing mast cells, and not Treg cells, are the main participants in fibrosis. 12 Mast cell proliferation is induced by IL-3, IL-13, and stem cell factor (SCF).13–16 We investigated the association of fibrosis in lymph nodes (LNs) of patients with CHL through histological examination of the expression of cytokines associated with fibrosis and mast cell proliferation (TGF-β, IL-3, IL-13, and SCF) and by determining the degree of mast cell infiltration in NSCHL compared with that in non-NSCHL (MCCHL or LRCHL).
Materials and methods
Case samples
We examined 22 specimens obtained from the LNs of previously untreated patients (18 men, 4 women) with CHL, who were diagnosed between 2003 and 2013 at Osaka Medical College Hospital. CHL diagnosis was based on the World Health Organization classification system. 1
Histopathology and immunohistochemical staining
LNs were obtained from each patient by biopsy, fixed in 10% buffered formalin, embedded in paraffin, cut into 4-μm-thick sections, and stained with hematoxylin and eosin (H&E). Immunohistochemical staining was performed using EnVision HRP (DAKO, Glostrup, Denmark) for TGF-β, IL-3, IL-13, and SCF. We also performed double staining for the pairs forkhead box P3 (FOXP3)/CD25, and FOXP3/TGF-β using both EnVision HRP and EnVision G/2 system/AP (DAKO) (Table 1). Toluidine blue stain was used to detect mast cells. Serial sections were prepared for toluidine blue stain and IL-13 immunostain with EnVision HRP or TGF-β using the EnVision G/2 system/AP. The extent of fibrosis was analyzed in LNs stained with Masson’s trichrome stain. The whole area and fibrotic area of each LN were measured by objective quantitative analysis using the WinROOF image processing software program (MITANI Corporation, Tokyo, Japan). The number of mast cells in the entire lesion of each LN was counted under a microscope using a grid inserted into the ocular piece of the microscope by two pathologists and expressed per mm2. The rate of fibrosis was calculated as the ratio of the fibrotic area to whole area in each LN.
Antibodies used for immunohistochemical study.
Statistical analysis
Results for continuous variables are shown as the mean ± standard error (SE). The relationships between cytokine production in HRS cells and subtypes of CHL (NSCHL and non-NSCHL) were determined using the Pearson’s chi-square test. The relationship between cytokine production in HRS cells and the number of mast cells/mm2 and the relationship between cytokine production in HRS cells and the rate of fibrosis were determined using the t-test. The relationship between the rate of fibrosis and the number of mast cells/mm2 was analyzed using a correlation coefficient. The P values <0.05 were considered significant. Statistical analyses were performed using JMP Pro 11 (SAS Institute, Inc., Cary, NC, USA).
Results
Patient clinical characteristics
The average age of 22 patients with CHL was 54 years (age range, 15–88 years). Histologically, CHL cases were further classified as NSCHL (n = 8) and non-NSCHL (n = 14; 13 MCCHL, 1 LRCHL) (Table 2).
Clinical and pathological features of 22 cases of CHL.

Histological and immunohistochemical results
Fibrotic areas stained blue with Masson’s trichrome stain, with a range of 0–48% (mean ± SE; 11.6% ± 2.8%), and LNs from patients with NSCHL (Figure 1a) had a higher number of fibrotic areas than those from patients with non-NSCHL (Figure 1b), as shown in Table 2.

Representative histology of (a) NSCHL and (b) non-NSCHL (MCCHL) stained with Masson’s trichrome stain. (a) The fibrotic area of the lymph node of Case 5 is stained blue with Masson’s trichrome stain. (b) In the lymph node of Case 14 only the capsule is stained blue. Objective magnification, 2×.
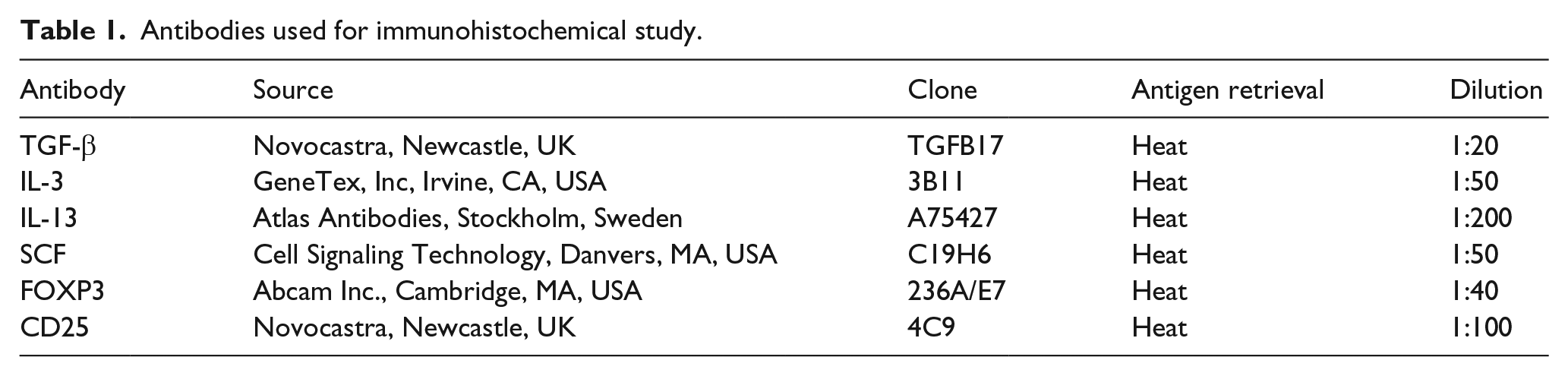
Of the 22 patients with CHL, seven (32%; 1 NSCHL, 6 non-NSCHL) were TGF-β positive (Figure 2a) and 15 (68%; 7 NSCHL, 8 non-NSCHL) were TGF-β negative in HRS cells. Ten patients (45%; 7 NSCHL, 3 non-NSCHL) were IL-13 positive (Figure 2b) and 12 patients (55%; 1 NSCHL, 11 non-NSCHL) were IL-13 negative in HRS cells. Ten patients (45%; 4 NSCHL, 6 non-NSCHL) were SCF positive (Figure 2c) and 12 patients (55%; 4 NSCHL, 8 non-NSCHL) were SCF negative. No patient had IL-3-positive HRS cells (Table 2).
Histological analysis of HRS cells in representative LN specimens collected from patients with CHL reveals (a) TGF-β positivity, (b) IL-13 positivity, and (c) SCF positivity (objective magnification, 40×).
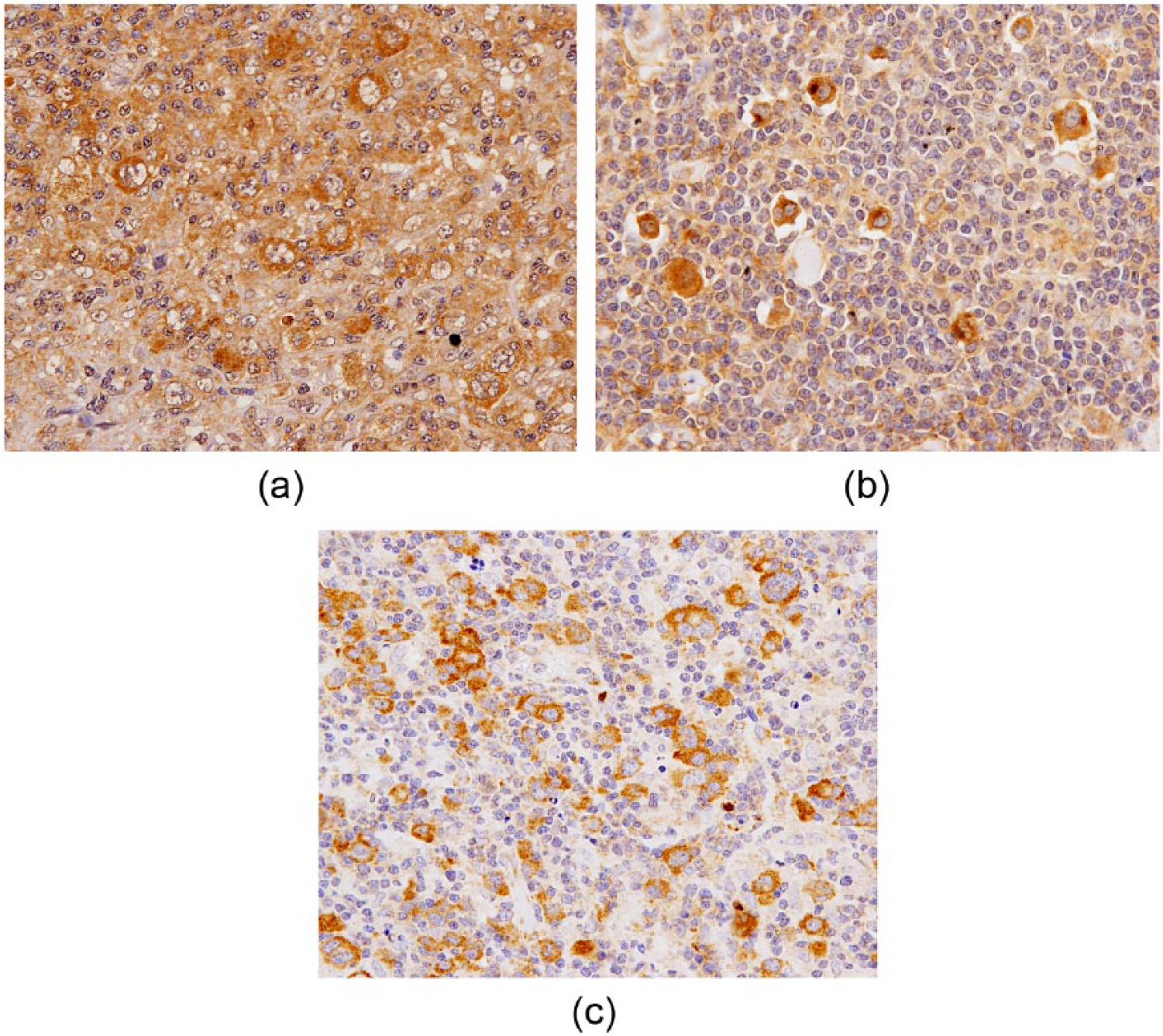
A variable mixture of non-neoplastic inflammatory cells was observed in the background. Some mast cells were scattered in the fibrotic areas within the LNs stained with toluidine blue and eosin (Figure 3a). The number of mast cells were in the range of 0.02–3.60/mm2 (mean ± SE, 0.77 ± 0.21/mm2) and LNs from patients with NSCHL had a higher number of mast cells than those from patients with non-NSCHL (Table 2). Many FOXP3-positive cells were observed in all cases, and a majority of these cells were also positive for CD25 (Figure 3b). In contrast, TGF-β-positive cells (Figure 3c) were small in number and more sparsely distributed than FOXP3/CD25 double-positive cells. Moreover, FOXP3/TGF-β double staining revealed that TGF-β-positive cells comprised a different population than FOXP3-positive cells, as no FOXP3/TGF-β double-positive cells were observed (Figure 3d). In serial sections, mast cells were positive for TGF-β and IL-13 (Figures 3e, f).
Histological analysis of background inflammatory cells in representative LN specimens from CHL patients. (a) Scattered mast cells displaying metachromasy in the fibrotic area are seen with toluidine blue and eosin stain (arrows). (b) Many FOXP3- (brown nuclei) and CD25-positive (red cytoplasm) Treg cells are also observed in FOXP3/CD25 double immunohistochemical staining. (c) Scattered TGF-β-positive cells (arrow). (d) FOXP3-positive (brown nuclei) and TGF-β-positive cells (red cytoplasm) (arrow) are separate, with no double-positive cells in FOXP3/TGF-β double immunohistochemical staining. (e) With toluidine blue stain (left) and TGF-β immunostain (right) of serial sections, a mast cell (left, arrow) is positive for TGF-β (right, arrow). (f) Toluidine blue stain and immunohistochemical staining for IL-13 in serial sections. A mast cell (left, arrow) is positive for IL-13 (right, arrow) (objective magnification, 40×).
Statistical analysis
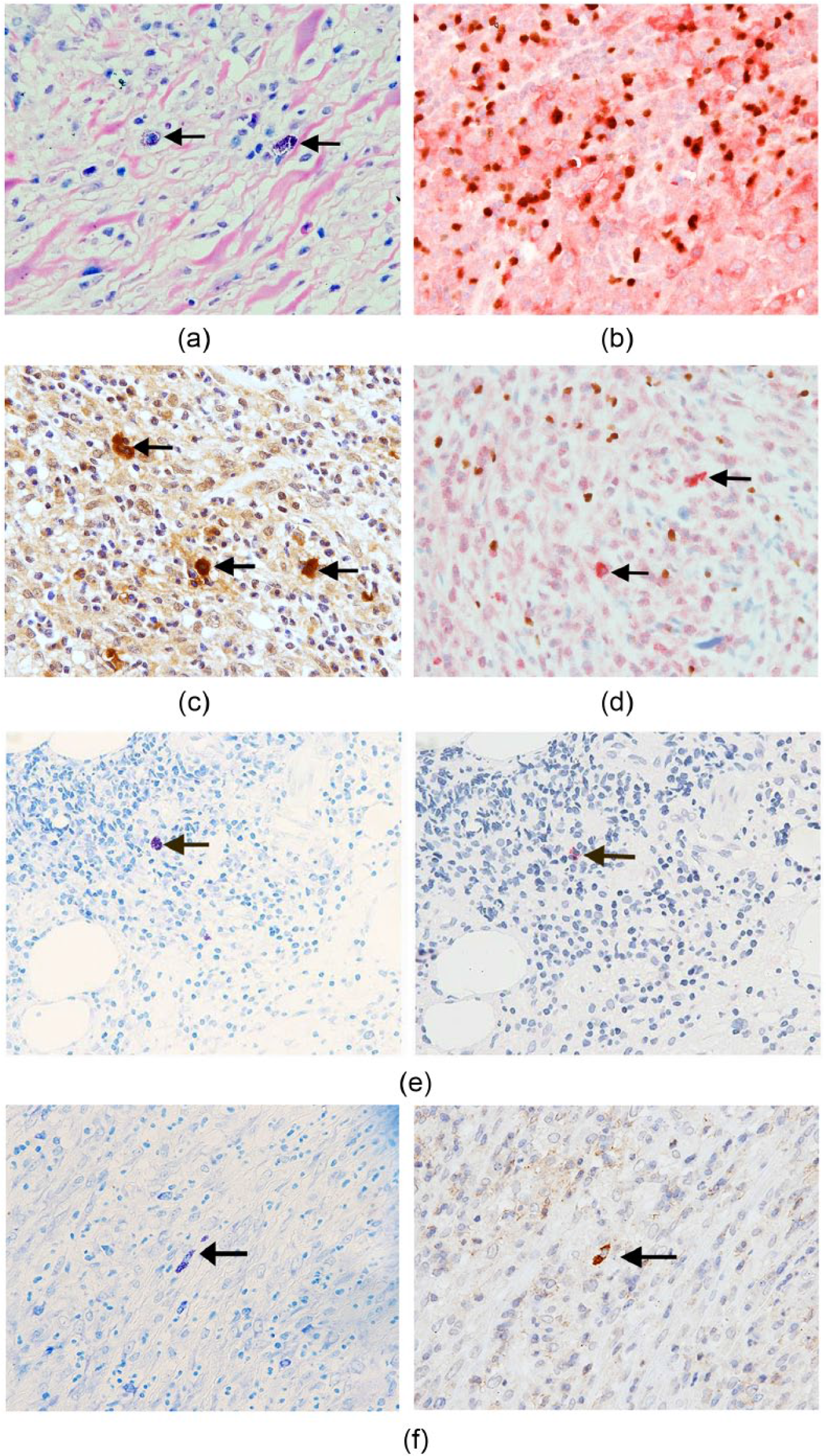
The rate of fibrosis was significantly higher in NSCHL (26.0 ± 4.1%) than that in non-NSCHL (3.3 ± 0.7%) (P <0.0001) (Figure 4a). No significant difference in TGF-β or SCF expression in HRS cells was observed between NSCHL and non-NSCHL (P = 0.1414 and P = 0.7462, respectively). The rate of fibrosis did not differ between HRS cells with or without SCF or TGF-β expression (P = 0.2110 and P = 0.1436, respectively). In contrast, IL-13-positive HRS cells were observed more frequently in NSCHL than in non-NSCHL specimens (P = 0.0028). Moreover, the group with IL-13-positive HRS cells also had a significantly higher rate of fibrosis (19.2 ± 5.1%) than that of IL-13-negative HRS cells (5.2% ±1.5%) (P = 0.0097) (Figure 4b). The number of mast cells was significantly higher in NSCHL (1.69 ± 0.37/mm2) than that in non-NSCHL (0.24 ± 0.09/mm2) (P = 0.0001) (Figure 4c). Moreover, the number of mast cells showed a significantly strong positive correlation with the rate of fibrosis (correlation coefficient, 0.8524; 95% CI, 0.6725–0.9372) (P <0.0001). There was no correlation between the number of mast cells and the expression of TGF-β or SCF in HRS cells (P = 0.3188 and P = 0.5546, respectively). In contrast, the number of mast cells was significantly higher in the group with IL-13-positive HRS cells (1.30 ± 0.37/mm2) than that in the group with IL-13-negative HRS cells (0.33 ± 0.14/mm2) (P = 0.0157) (Figure 4d).
(a) The rate of fibrosis is significantly higher in NSCHL than that in non-NSCHL (P <0.0001). (b) The rate of fibrosis is significantly higher in the group with IL-13-positive HRS cells than that in the group with IL-13-negative HRS cells (P = 0.0097). (c) The number of mast cells is significantly higher in NSCHL than that in non-NSCHL (P = 0.0001). (d) The number of mast cells is significantly higher in the group with IL-13-positive HRS cells than that in the group with IL-13-negative HRS cells (P = 0.0157).
Discussion
Mast cells release histamine granules in immediate hypersensitivity and allergic reactions. They also play a role in various immune responses, including host defense, immune regulation, chronic inflammation, and autoimmune diseases via the production of various cytokines. 17 Moreover, mast cells have been reported to participate in the development of fibrosis.10–12 Mast cells may promote fibrosis by inducing fibroblast proliferation, migration, contraction, and collagen production through the release of a range of stored cytokines such as TGF-β from mast cells that act in a paracrine fashion to stimulate fibroblasts. 18 Recently, it was shown that fibrosis in IgG4-related diseases is induced by cytokines (e.g. TGF-β) secreted from infiltrating mast cells using dual immunofluorescence assays. 12 IL-3 and SCF have been identified as important cytokines for regulating mast cell growth and differentiation.13–15 IL-3 is produced by activated T cells, monocytes/macrophages, and stroma cells and represents a potentially important link between the immune and hematopoietic systems. 19 SCF is the ligand for the tyrosine kinase receptor c-kit, which is expressed on membrane of mast cells and is involved in cell survival and proliferation. 20 SCF is produced by stromal cells, fibroblasts, and endothelial cells. 21 Abnormal SCF expression has been reported in neoplastic cells of a restricted number of human solid tumors of the cervix, kidney, lung, and pancreas.22–25 IL-13 is also known to promote mast cell proliferation.16,26 IL-13 is produced by Th2 lymphocytes and activated mast cells and is implicated in the immunopathogenesis of various allergic diseases.16,26,27
The relationship between mast cell infiltration and the histological subtype of CHL (NSCHL or non-NSCHL) remains controversial. Keresztes et al. reported that mast cell infiltration was similar in MCCHL and NSCHL. 28 On the other hand, some authors reported that NSCHL correlated with higher numbers of tumor-infiltrating mast cells, compared with non-NSCHL.29–31 Immunodeficient NOD/SCID mice inoculated with both mast cells and human HRS cells grew tumors with a more marked fibrotic component, compared with tumors in mice inoculated with HRS cells alone. 32 Thus, there is mounting evidence that the fibrosis observed in association with mast cells in NSCHL is primarily a consequence of the mast cell infiltration, rather than the cause.
In the present study, mast cell infiltration and cytokine production were compared between NCHL and non-NSCHL, and mast cells were found to be positive for TGF-β. In addition, the population of mast cells was significantly higher in NSCHL than that in non-NSCHL, and the number of mast cells exhibited a positive correlation with the rate of fibrosis. However, TGF-β expression in HRS cells did not differ between NSCHL and non-NSCHL, and did not correlate with the rate of fibrosis. These findings suggest that the infiltration of TGF-β-producing mast cells may be associated with fibrosis in NSCHL rather than the production of TGF-β by HRS cells.
Moreover, IL-13-positive HRS cells were more frequently observed in NSCHL than in non-NSCHL. The group with IL-13-positive HRS cells had a significantly higher number of mast cells and rate of fibrosis than that of IL-13-negative HRS cells. Mast cells were also positive for IL-13. From these findings, IL-13 production by HRS cells appears to promote fibrosis in NSCHL via a mechanism whereby IL-13 produced by HRS cells may directly initiate fibrosis, as well as promote mast cell proliferation and infiltration. Subsequently, increased mast cells may also produce IL-13, leading to increased fibrosis. In this manner IL-13 production seems to play an important role in the increased number of mast cells and the establishment of fibrosis in CHL.
FOXP3-positive cells and IL-3 may not participate in fibrosis in CHL because FOXP3-positive cells do not produce TGF-β, and IL-3 production by HRS cells was not observed in any cases. In addition, the expression of SCF in HRS cells did not differ between NSCHL and non-NSCHL, and there was no difference in both the rate of fibrosis and the number of mast cells between HRS cells with or without SCF expression. Thus, the expression of SCF in HRS cells has no correlation with mast cell infiltration and fibrosis. However, we demonstrated aberrant expression of SCF in HRS cells for the first time. This aberrant SCF expression may be a potential therapeutic target for the future treatment of SCF-positive CHLs.
In the tumor microenvironment, the accumulation of mast cells in tumors has been well documented, and infiltrating mast cells act to regulate inflammation and immunosuppression.33,34 Mast cells promote the differentiation of T cells into FOXP3/CD25 double-positive Treg cells via the secretion of TGF-β by mast cells. 35 FOXP3/CD25 double-positive cells were recently shown to include three subpopulations: (1) effector Treg (eTreg) cells, which are terminally differentiated and highly suppressive; (2) naïve Treg cells, which differentiate into eTreg cells upon antigenic stimulation; and (3) non-Treg cells, which lack suppressive activity but secrete proinflammatory cytokines. 36 In the present study, many FOXP3/CD25 double-positive cells were observed in all LNs derived from patients with CHL, regardless of the different distribution of mast cells between NSCHL and non-NSCHL. Therefore, the FOXP3/CD25 double-positive cells observed in the present study are unlikely to be immunosuppressive eTreg cells but rather non-Treg cells.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Science Research Promotion Fund from the Promotion and Mutual Aid Corporation for Private Schools of Japan.
