Abstract
Background
Honey has been used for centuries for its medicinal benefits, where topical treatments in the folklore literature reported its beneficial role in treating different types of wounds. Despite its immense use in wound care, honey’s exact mechanism remains unclear.
Purpose
The present study aimed to explore and abridge the gap between honey and its molecular wound healing mechanism.
Methodology
An extensive literature study has been performed where these activities can be explained due to phenolic compounds, nitric oxide, non-peroxide factors, low pH, high osmolarity, and hydrogen peroxide in honey. Besides that, honey also contains carbohydrates, proteins, amino acids, lipids, minerals, and vitamins, contributing to the wound healing potential to some extent. However, these metabolites and constituents differ among different honey varieties due to the distinct nutritional requirements of different species of bees and their collection of nectar from varied sources containing different natural actives.
Results
The revival of honey-based wound care research has identified a few probable mechanistic pathways of honey primarily due to its anti-inflammatory, antioxidant, antimicrobial, and immunomodulatory potentials. It also promotes angiogenesis, facilitates reepithelialization, stimulates proliferation of extracellular matrix, reduces neutrophil formation, modulates production of TNF-α, IL-1β, and IL-6, and prevents secondary infection at the wound site.
Conclusion
The varieties of honey vary in their qualities, phenolic compounds, and safety for human use. A few of these are currently clinically employed for wound dressings. Moreover, honey can be effective for managing complicated and chronic wounds; more extensive molecular research and safety profiling would be necessary.
Keywords
Introduction
The largest organ in the body, that is, the skin, protects the body from the environment. It also works for sensory detection, fluid homeostasis, thermoregulation, immunological surveillance, and self-healing. Skin damage due to injury diminishes its protective function and, if severe damage occurs, can lead to serious impairment or even death.1, 2 Topical damage occurs primarily due to traumas leading to tears, gashes, cuts, burns, abrasions, scrapes, and bruises. These damages are potentiated by secondary underlying conditions such as diabetes mellitus, stress, uremia, fibrosis, obesity, hypertension, nutrition, and immunocompromised state.3–5 Nearly 40–60 million diabetic patients are affected with chronic diabetic foot ulcers worldwide, and 15% of the cases progress to amputation, according to the International Diabetes Foundation. Around 7.4 million people are affected by pressure and leg ulcers worldwide, most of which are venous. The projected expenditure on wound care is between $28.1 and $96.8 billion worldwide. 6 It is projected that this market will rise considerably as people age and become more susceptible to chronic wounds. 7 By 2029, the chronic wound care market is expected to grow at a 6.7% compound annual growth rate to reach a value of 19.52 billion USD. In 2021 and 2022, the market had a value of USD 11.61 and USD 12.36 billion, respectively. 8 The biological process of wound healing is intricate and dynamic, including the coordination of several cellular functions to aid in the restoration of normal function and structure to damaged skin. 9 Research of natural therapies is still in its early phases, where whole-plant extracts or other natural products contain various therapeutic metabolites, including antimicrobials, antioxidants, anti-inflammatory agents, and stimulants of reepithelialization and collagen formation, which synergize the wound healing process.10-12
Honey is one such natural product that has had an immense application in the food and beverage industry for thousands of years and has also been reported in the folklore literature. 13 It has been used as a medical agent and food supplement for thousands of years. It has also been applied to treat burns, traumatic wounds, and periodontitis. 14 15 In 2007, the Federal Drug Administration (FDA) approved MEDIHONEY®, a Manuka honey (MH)-based medication, for treating different types of wounds, which was for the first time a biologically derived substance had been authorized for wound management. 16 The historical uses of honey as a wound healer are illustrated in Table 1. In World War I, Russians and Chinese utilized honey to treat soldiers’ wounds. In the mid-20th century, the effectiveness of honey in treating septic wounds was discovered in Europe and the United States. However, once modern antibacterial medications were available in the 1920s, honey lost its necessity as a wound-healing agent. Researchers’ interest in these honey-related characteristics did not resurface until the 1980s. The increased interest in wound healing as a field of research is most likely one cause of this comeback. Honey made a resurgence in the 20th century as a treatment for several illnesses and as a fantastic wound healer. For example, honey derived from blossoming native New Zealand trees was reported to have the highest antibacterial qualities in 1981. 17 Thus, multiple studies on the application of different varieties of honey in wound healing have been reported in recent years. For this reason, it is necessary to report the bioactivities of honey in wound healing in a systematic approach, which is the primary aim of this review.
Historical Uses of Honey Reported by a Few Ancient Researchers and Systems of Medicines Worldwide Are Arranged in a Chronological Sequence.

Mechanisms of Honey as a Natural Wound Healer
Honey is a naturally occurring, highly complex material with hundreds of components. The composition of honey, which primarily depends on the floral source and other variables like seasonal and ambient conditions along with processing, modification, packing, and storage requirements, will affect honey’s bioactivity and, consequently, its ability to heal wounds. In addition to some inherent properties like acidity and osmotic pressure, honey’s healing properties in various wound types have been related to its ability to target diverse pathways to rebuild the structural integrity of the injured tissues. 18 The different molecular mechanisms followed by honey to exert its wound-healing ability are discussed below, and the working of the immune system in acute wound healing is depicted in Figure 1.
How the Immune System Works in Acute Wound Healing.
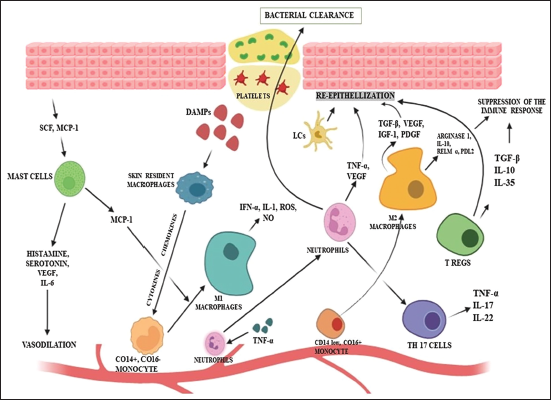
At the initial stage, skin resident macrophages react to the wound site. They produce cytokines and chemokines after being triggered by DAMPs, which draw neutrophils and monocytes to the inflammatory site. Then, M1 macrophages generated from a monocyte, MCP-1(monocyte chemoattractant protein-1), which is secreted by mast cells, also help this particular generation. Mast cells help in vasodilation, which ultimately helps in the recruitment of immune cells. Neutrophils, after activation, promote bacterial clearance tissue reepithelialization. M2 macrophages and Langerhans cells also contribute to tissue reepithelialization. Various T-cells also help in tissue repair and bacterial clearance. 19
Antimicrobial Action
In the past, people have used honey to prevent and cure wound infections. However, the use of honey progressively declined as antibiotics became more widely available. Its inherent properties strongly impact bacterial growth and survival, including high osmolarity, low water activity, acidity, and certain chemicals, including hydrogen peroxide (H2O2), phenolic compounds, methylglyoxal (MGO), and bee defensin-1 peptide. Additionally, honey secondarily exerts antimicrobial activity by stimulating nitric oxide production, cytokines, and immunomodulation through lymphocyte and antibody formation. 18 Although water is the second-largest component of honey, due to the higher concentration of sugar moieties, it has reduced water activity, ranging from 0.562 to 0.62, resulting in osmotic stress in microbes. 20
When honey is added to a sufficiently diluted solution, the enzyme glucose oxidase is activated, which then interacts with glucose to form gluconic acid and H2O2. H2O2 is regarded as a natural antibacterial agent. 20 Recent research has investigated the role of H2O2 in the regenerative effect of honey on the human keratinocyte cell line. This study revealed that specific aquaporin (AQP3) could facilitate the entry of H2O2 through the plasma membrane and induce the extracellular Ca2+ via Orai1 and transient receptor potential melastatin 2 (TRPM2) channel induction of extracellular Ca2+ results in wound healing by activation of tissue regeneration. Figure 2 illustrates the diagrammatic representation of the wound-healing mechanism of honey, which shows H2O2 in the extracellular space entering into the cytosol via AQP3 and activates TRPM2 and Orai1 for cell migration for wound healing. Moreover, inhibition of TRPM2 and Orai1 via catalase, econazole, and siRNA diminishes the Ca2+ concentration, decreasing the response to honey. They have also reported the upregulation of AQP3 in the presence of honey for wound treatment.21, 22
The Enzyme Glucose Oxidase Is Activated in Honey When Diluted.
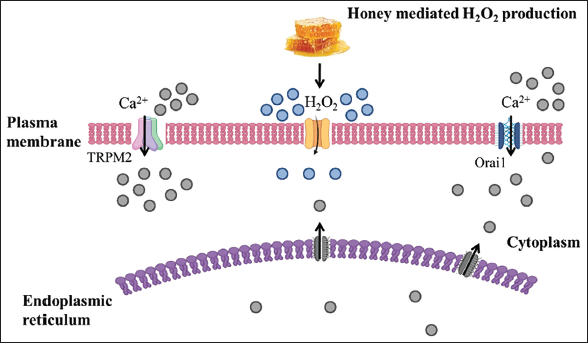
It reacts with glucose, forming gluconic acid and hydrogen peroxide (H2O2), a natural antimicrobial agent. Further, honey also stimulates the entry of Ca2+ ions through transient receptor potential melastatin 2 (TRPM2) and calcium release-activated calcium modulator 1 (Orai1).
Plants primarily produce phenolic compounds, one of the significant families of secondary metabolites, to protect themselves against biotic and abiotic stress and oxidative damage. 23 These substances pass from the nectar to the honey. The phenolic chemicals found in honey mostly come from the phenolic acid and flavonoid categories. 20 In this regard, Biluca and the team investigated the phenolic composition of stingless bee honey along with its anti-inflammatory effect and free radical scavenging activity in RAW264.7 macrophage cell lines. The results revealed that stingless bee honey was cytocompatible, decreased the nitric oxide level, and stimulated the secretion of proinflammatory cytokines. 24
Apart from that, bee defensin-1, an antimicrobial peptide (AMP) generated by the salivary glands of bees and present in bee hemolymph, is added to honey during the primary processing. Defensin is an AMP component of a bee’s immune system, like other AMPs such as apidaecin, abaecin, or hymenoptaecin. Defensin-1 has an action against various pathogens, including gram-positive and gram-negative bacteria; however, at the quantity found in honey, its effectiveness against multidrug resistant bacteria appears constrained. 25 Sojka’s research team investigated. The antimicrobial efficacy of defensin-1 on biofilm formed on wounds. The results revealed that honey significantly reduces wound pathogen viability in polymicrobial biofilm; however, it is unable to eradicate Enterococcus faecalis in biofilm. Interestingly, defensin-1 was effective against Pseudomonas aeruginosa and Staphylococcus aureus after 24 and 48 hours of treatment. 26
Due to the limited availability of antifungal drugs, research on novel chemotherapeutic agents in natural products was sparked by the hunt for new antifungal agents. A study evaluated the antifungal efficacy of various Iranian honey varieties with various botanical sources against certain significant fungi, such as azole-resistant Candida strains. Citrus spp., Thymus vulgaris, Zizyphus spp., Medicago sativa, Astragalus spp., Chamaemelum nobile, and multiflora were among the seven species from which honey samples were collected. Zizyphus and Chamaemelum honey had the lowest antifungal activity, whereas Alfalfa and Thymus honey performed much better than other samples. The two fungi with the highest and lowest susceptibilities to honey’s antifungal properties were Trichophyton rubrum and C. krusei, respectively. 27 Another research study demonstrated that honey flavonoids could resist pathogenic yeast. Candida albicans yeast was inhibited by honey extract, affecting its membrane integrity, DNA behavior, and mitochondrial function. 28
Alteration of Wound Microenvironment
Methicillin-resistant Staphylococcus aureus (MRSA) is a significant pathogen posing a severe threat to human health and calls for innovative therapies because of its resistance to various antibiotics and prevalence in healthcare settings. Proteomic and genomic analyses were conducted to assess the molecular impact of MH on MRSA. The most notable alterations in gene expression were found in the microarray data, which suggested that MH significantly affected the expression of crucial MRSA virulence factors like lipase, leucocidin, hemolysins, and fibronectin-binding protein. The gene that codes for enterotoxin type C3 had the most substantial change. It was shown that MRSA was treated with these honey-repressed genes in the agrABCD and SaeSR global regulatory operons. Both operons in Staphylococcus spp. influence biofilm development in addition to pathogenicity. It was discovered that this variety of honey may enhance levels of autolysins and proteases in MRSA through the increased expression of sarV, a different regulator gene. It has been demonstrated that MH prevents the growth of biofilms and dissolves existing Staphylococcal biofilms. As SaeRS gene mutation has been shown to boost protease activity while restricting biofilm formation, this discovery may be explained by lower expression of the regulator (SaeR) genes and histidine kinase sensor (SaeS) in MH-treated cells. 29 Figure 3 illustrates the possible pathway that affects the gene of MRSA by MH.
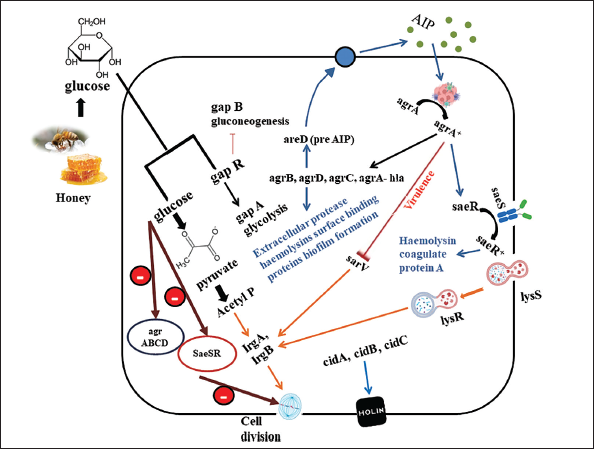
In another research, the results show that the natural extracts of honey had a stimulatory effect on monocytic cells’ production of TNF-α, IL-1β, and IL-6. Both pro- and anti-inflammatory mechanisms include cytokines because several cell types actively involved in tissue healing are modulated by the production of cytokines by monocytic cells in the wound region. The Australian jelly bush honey exhibited the most effect, with significant increases in cytokine levels seen in both New Zealand Manuka and Pasture varieties compared to control groups (sugar solution). These results suggest that components other than sugars are involved in the regulatory effects of honey, although it is unknown which specific component/s are involved in mediating these effects.30, 31
Similarly, Minden-Birkenmaier et al. examined the effects of 0.5% and 3% MH on the release of various cytokines, chemokines, and enzymes that can degrade the matrix from a dHL-60 neutrophil model in the presence of anti-inflammatory stimuli (IL-4, TGF-β, and IL-13). The anti-inflammatory nature of honey stimulated animals to produce much more IL-1Ra, IL-4, FGF-13, CCL2/MCP-1, CCL4/MIP-1β, CCL20/MIP-3α, and CXCL8/IL-8 when used in 0.5% concentration; however, Proteinase 3 synthesis is markedly reduced. Nevertheless, there is a significant increase in the release of TNF-α and CXCL8/IL-8 in the case of 3% honey while reducing the release of all other analytes. The outcome demonstrates that in this inflammatory model, the release of cytokines, chemokines, and matrix-degrading enzymes are all affected differently by MH. This finding highlights the necessity of regulating the MH concentration in a wound or tissue template to produce the desired anti-inflammatory effect during the wound-healing process. 32
Immunomodulatory Effects
Honey has been shown in studies to influence cytokine release (Figure 4). 25 One study discovered that New Zealand honey causes the release of TNF-α from monocytic cell lines. 33 Furthermore, scientists hypothesize that honey can boost the synthesis of proinflammatory cytokines in low-inflammation reactions while decreasing these cytokines (TNF-α and IL-β) in infectious circumstances. In the case of rodents, Gelam honey (GH) has reduced the inducible nitric oxide synthase (iNOS) production along with cyclo-oxygenase (COX-2), indicating that it possesses anti-inflammatory qualities. Despite the lack of infection, the anti-inflammatory impact was still observed in in vivo models.34, 35
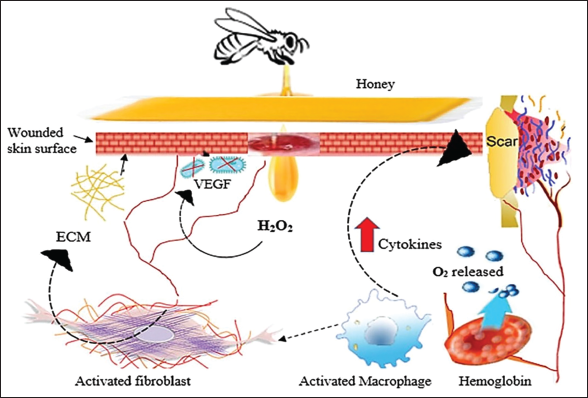
The presumable antioxidant effects of honey include changes in neutrophil reactive oxygen species (ROS) production and the suppression of neutrophilic respiratory bursts. According to studies by Saikaly and Khachemoune, New Zealand honey has been shown to lower human neutrophil superoxide formation due to its phenolic components, known to be a free-radical scavenger. 36 According to another study, increased ROS from neutrophil activation may be the cause of the anticancer impact of Jungle honey. 37 These findings highlight the need for a more thorough investigation of these biological mechanisms. By activating the arabinogalactan proteins in New Zealand MH, Gannabathula et al. altered the immune system by stimulating neutrophils. 33 An unknown molecule of 261 Daltons molecular weight in Jungle honey was discovered, which induced chemotaxis in neutrophils and decreased leukocyte invasion. 36
Antioxidant Activity
The presence of various secondary metabolites such as flavonoids (quercetin, apigenin, chrysin, pinocembrin, galangin, kaempferol, and hesperetin), phenolic acids (caffeic, ferulic, p-coumaric, and ellagic acids), ascorbic acid, selenium, Maillard reaction products, peptides, and certain amino acids are responsible for exerting antioxidant activity. The presence of hydroxyl groups in the phenolic constituents of honey and their positions in the ring modulate the compound’s antioxidant potential. Furthermore, the ability of phenolic compounds to decrease and chelate ferric ions, which catalyze lipid peroxidation, might be linked to their activity. The most significant antioxidant in honey is gallic acid. Aqueous and lipophilic antioxidants are also present in honey so that the compound can act as a natural antioxidant at different cellular levels. 38 In this regard, Larsen and Ahmed investigated the antioxidant potential of honey and the phytoconstituent responsible for exerting this activity. They evaluated the change in chemical profile and antioxidant activity of raw honey during the formulation of lozenges and honey drops. Various antioxidant assays revealed that these lozenges and honey drops with high phenolic components had higher antioxidant properties than raw materials. 39
Angiogenic Potential
In the proliferative phase of wound healing, developing new blood vessels supplies the wound with essential oxygen, a crucial step in the healing process. Serum and the surrounding extracellular matrix condition heavily influence this angiogenic mechanism. Hydrogen peroxide (found in raw honey, produced by glucose with the help of the enzyme glucose oxidase) promotes leukocyte migration to wounds via a concentration-dependent mechanism. Macrophages release vascular endothelial growth factors in response to ROS production, which promotes angiogenesis. Furthermore, a local cellular energy source in the moist wound environment is provided by the high sugar content of honey, along with other ingredients like amino acids and trace elements of vitamins, which may increase cellular nutrition and thus promote endothelial cell proliferation. 18 Honey’s effect on angiogenesis was evaluated by Chaudhary et al. using an in vitro (fibroblast cell) model and an in vivo diabetic wound model. The migration or scratch assay findings, where the cell migration and α-SMA expression due to treatment with jamun honey was comparable with the group treated with MH (considered standard due to FDA approval). Jamun honey at 0.1% v/v concentration showed notable wound healing, reepithelialization, and rapid angiogenesis in the in vivo studies. 40
Varieties of Honey in Wound Healing Application
Medical-grade honey (MGH) allowed wound care to overcome the safety risks previously documented in wound therapy with other forms of honey. The medical-grade variety is sterilized by gamma irradiation to destroy Clostridium spores, is manufactured under strict hygiene standards, is free of contaminants and contaminating pesticides, and is standardized according to international guidelines. They are authorized for wound treatment and exhibit significant in vitro bactericidal action, even against antibiotic-resistant bacteria. 18 A few honey varieties currently employed in wound treatments or have shown some prospects in recent wound care research are discussed below.
However, there might be a significant variation in the biological activities of different forms of honey due to its geographical distribution or its botanical origin, which is governed by the pollen origin or distribution. The presence of phenolic constituents in a specific variety of honey depends mainly on its floral origin and can be determined via floral and geographical evaluation, as well as spectrophotometric techniques. Apart from these, the abundance of flowering in a particular season also influences the polyphenol constituent of honey. 41 For instance, Kavanagh et al. evaluated that the total phenolic constituent (TPC) of Irish honey found in the town region was 28.26 mg GAE/100 g of honey, while those found in the rural areas had a TPC of 20.32 mg GAE/100 g of honey. Due to Ireland’s floral diversity and landscape, the TPC ranged significantly between 2.59 and 81.10 mg GAE/100 g of honey. 42 On the other hand, Muñoz et al. observed that Peru’s multi-floral wild varieties of eucalyptus honey had the highest TPC with the most negligible variation (207.89 ± 2.18 mg GAE/100 g) in comparison with the monofloral types (83.15 ± 4.09 mg GAE/100 g). 43 However, among the monofloral varieties, MH found in Australia and New Zealand has the highest reported TPC with a value of 217–203 mg GAE/100 g. 44 In a study based on the variation of TPCs based on altitude, Nepalese honey showed the lowest TPC value of 61.77 mg GAE/100 g among honey harvested at >1,500 m from sea level compared to the honey harvested at 800–1,500 m from sea level, which showed a TPC of 118.65 mg GAE/100 g. 45 The TPC of honey also varies according to the climatic conditions. For example, in the case of Kenyan honey, the TPC of the honey was found to be 98.3777 mg GAE/100 g in the semiarid regions, 116.1777 mg GAE/100 g in the hot and humid area, and 141.7177 mg GAE/100 g in the high precipitation region. 46
Manuka Honey in Wound Care Application
MH is produced in Australia and New Zealand by bees pollinating the native Leptospermum scoparium shrub (Tea tree). The word “Manuka” comes from Maori, New Zealand’s original language, and others contend that only approved New Zealand MH should carry the title. Bees collect nectar from Manuka flowers during the summer to generate this variety of honey. Nectar is a sweet liquid and a rich source of nutrition that flowers produce, which attracts bees. The bees return the nectar to the hive and seal it in wax cells, which grow naturally into honey. MH is then collected from the honeycombs. A compound known as MGO is one of the essential antibacterial constituents of MH. MGO is produced by modification of dihydroxyacetone (DHA) present in MH found abundantly in the nectar of Manuka flowers. MGO concentration is directly related to the antibacterial action of this variety. Apart from MGO, MH can contain over 200 secondary metabolites and is made up of mainly carbohydrates (80–85%) along with some water (15%–17%), proteins (0.1–0.4%), ash (0.2%), and trace amounts of different vitamins, minerals, amino acids, organic acids along with enzymes. Honey producers rate MH’s potency on a scale known as UMFTM, which stands for the unique Manuka factor. 47 The concentration of three characteristic chemical constituents in authentic MH depends on MGO, DHA, and leptosperin, influencing the UMFTM grade. The UMF score range of 0 to 4 indicates an insignificant quality, 5 to 9 indicates modest quality, 10 to 15 indicates relevant quality, and >16 indicates high-grade quality. MH, with a high UMF, is required for formulation purposes to treat chronic wounds. Copper-doped borate bioactive glass particles embedded in methylcellulose foams cross-linked with MH increased the wound-healing potential of the honey due to improved angiogenic and antibacterial capabilities. This formulation’s physicochemical characterization showed foam formation for ease of application with enhanced porosity. 47 A biocompatible and antibacterial nanofibrous mat made of cellulose acetate and different concentrations of Manuka was produced via an electrospinning process having good tensile properties. The ATR-FTIR spectra confirmed the presence of honey in the nanofiber mat matrix. Findings suggested that the fiber diameter increased gradually with the concentration of MH. The electrospun composite contents limited the growth of microbes in the wound site and exhibited potent antioxidant ability due to their ability to scavenge DPPH free radicals. This property was influenced by the mat’s honey content and the time it was kept in the DPPH solution. 49
Combining the antibacterial MH with gelatin-based hydrogel in a 3D patch can improve printing efficiency and produce positive biological results useful in regenerative wound treatment. An extrusion-based printing approach created antibacterial Manuka-gelatin 3D patches with optimum porosity, good form accuracy, and structural stability. The gram-positive microorganisms (S. epidermidis and S. aureus) and gram-negative microbes (E. coli), frequently found in infected ulcer sites, were successfully eradicated with Manuka-gelatin 3D patches. These patches also increased human epidermal keratinocyte and dermal fibroblast proliferative rates and encouraged angiogenesis. 50 Numerous research studies on using honey for wound treatment in animal and clinical instances exist. Fresh wounds treated with topical honey accelerated wound contraction and enhanced granulation tissue formation.
Gelam Honey in Wound Care Application
GH is a Malaysian wild mono-floral honey made by Apis dorsata. These bees’ principal nectar and pollen come from the shrub Melaleucacajupati powell, often known locally as the “Gelam tree.” 51 The plant is a species of the Myrtaceae family. GH is plentiful in Terengganu, peninsular Malaysia’s eastern coast, where these mangrove trees grow abundantly in the swamp. GH is typically produced by base hydrolysis and extracted with ethyl acetate. In Malaysia, Gelam wood is known as “Kayu Putih,” a 24-meter-tall evergreen tree. Melaleuca species occur naturally in Kedah, Negeri Sembilan, Melaka, Kelantan, and Terengganu. The Malaysian swamp woods are behind these states’ mangroves and sandy beaches. The trees are easily identified by their distinctive thick, papery, flaky bark. The famous English name for the Gelam tree varies by location, although it is commonly known as the “swamp tea tree,” as well as the “paperback tea tree” and the “cajeput” tree. 52
The effectiveness of GH in promoting wound healing was studied in Sprague-Dawley rats by Tan and team by making a 2/2 cm full-thickness excisional lesion on the neck region. Compared to the untreated and saline-treated groups, wounds treated with the formulated GH in situ gel showed faster wound contraction till day 13 with minor scabs and thin scar production. With these findings, the authors concluded that GH benefits wound healing, as evidenced by the histological findings. 53 Concurrently, GH has also been explored to evaluate its effect on burn wounds. Here, the researchers investigated the physical features of the synthesized honey hydrogel and its wound-healing efficiency on severe, partially thickened burn wounds in rats. Skin samples were collected for histological and molecular analysis. Compared to the control hydrogel and OpSite film dressing treated animal groups, honey hydrogel dressings showed substantial improvement in wound healing and accelerated the reepithelialization phase. Honey hydrogel-treated groups substantially reduced the inflammatory response as early as seven days after the burn wound induction. A semiquantitative real-time reverse transcriptase-polymerase chain reaction (RT-PCR) study demonstrated that GH hydrogel treatment significantly reduced the production of proinflammatory cytokines. 54 Research findings showed evidence that GH hydrogel dressing is an extremely promising alternative wound care component. Similarly, various hydrogels have been utilized in the fabrication of certain wound dressings as discussed elsehwere. 55
Honeydew Honey in Wound Care Application
Bees collect the phloem sap from the leaves, bark, and stems of trees to make honeydew honey (HDH). 56 HDH is a polyfloral variety because it originates from various species; not a single species account for more than 10% of the total HDH production. It has a robust taste profile with a slight sweetness, and its coloring is often darker. The aroma of this honey is distinctive and potent; it may be resinous, aromatic, piney, or malty, which may consist of several monosaccharides (fructose and glucose), disaccharides (maltose, trehalose, and sucrose), and trisaccharide (raffinose, erlose, and melezitose). 56 Honey bees enthusiastically collect the honeydew nectar because of its high sugar content and mix it with an enzyme Invertase produced in its mouth by specific glands before returning to the hive. In this process, the honeydew’s water content will fall, turning it into honey, and infusing wax cells. 57 However, the water concentration might be too high at this point. This causes the honeybees to move their wings over the wax cells, leading to the evaporation of excess water in them. 58 When the process is complete, the HDH will have a less than 20% water percentage, which is optimum for their consistency. The honey bees will then cap it and repeat the procedure. Farmers who collect HDH undertake electrical conductivity tests to see if it fits the standards that define the product for labeling purposes. The composition of this HDH differs between the hives created by the different species of bees, the sap from which it has been collected, the condition of the environment, ant–aphid interactions, and the season of its collection also result in variations of the quality.59–62 HDH was also explored for wound healing activity. The research investigated the therapeutic effectiveness of sterile HDH in treating lower leg ulcers in 25 individuals suffering from persistent venous leg ulcers. In this process, 100% irradiated sterile HDH was administered to the cleansed wounds, and each wound was examined at least twice during the first six weeks of treatment. Surprisingly, the average wound area of all patients was reported to be reduced considerably due to the therapy. In total, 18 patients reported decreased pain and discomfort, whereas five individuals maintained the same discomfort throughout the research. In 80% of patients, the overall satisfaction with the honey therapy was favorable. 63 Alternatively, Martinotti et al. conducted a wound healing assay using HDH in an in vitro wound healing model using keratinocytes and fibroblasts. According to the results, HDH accelerated wound closure by functioning on fibroblasts and keratinocytes. The findings indicated that the healing process involved multiple Ca2+-dependent cell signaling pathways. 22
Revamil Honey in Wound Care Application
Revamil honey (RH) is an MGH that is Conformité Européenne (CE) marked, standardized, and produced in greenhouses by Bfactory Health Products, Rhenen, Netherlands. RH is manufactured in the Netherlands under well-controlled circumstances involving healthy bee colonies. Because of this carefully monitored production process, RH has a predictable, highly antibacterial activity. Each item complies with all the CE-registered safety standards for professional wound care.64, 65
US Food and Drug Administration (USFDA) approved several honey-based wound-care commercial products, such as wound dressings, gels, and ointments, registered as medical devices for clinical use (Table 2). Most of these products consist of MH, notably the most researched honey variations in the world, and was the first variety of honey to be designated as MGH. There are, however, options using different types of honey, like multi-floral, buckwheat, and Revamil. A honey-based gel formulation, for example, can be made entirely of 100% MGH or blended with other agents, namely natural emollients (glycerine, polyethylene glycol, lanolin, myristyl myristate), plant waxes, and gelling agents.64, 65 In the UK, RH gel, a hydrophilic wound gel comprising RH of the highest purity, is marketed as enzyme-rich (pH 3.5). Wound dressing and poly acetate sheet dressings coated with pure RH are readily accessible. Apart from an allergy to honey or bee products, there are no obvious contraindications to their use, and they may be used on various wound types. 66 It has been demonstrated in vitro that RH significantly reduces the number of antibiotic-resistant gram-positive and gram-negative bacteria. As a result, this MGH can be used to treat secondarily infected wounds comprising antibiotic-resistant strains. 65 An infant male, aged eight months, with severe burns on his left hand due to hot water, received treatment with RH dressing. The dressings could be conveniently changed every 3–4 days. The results indicated wound recovery within 14 days with minimal wound exudates and intact peri-wound skin. 66
Few Marketed Formulations, Including Gel, Cream, Paste, and Balm, Are Based on Different Honey Varieties.

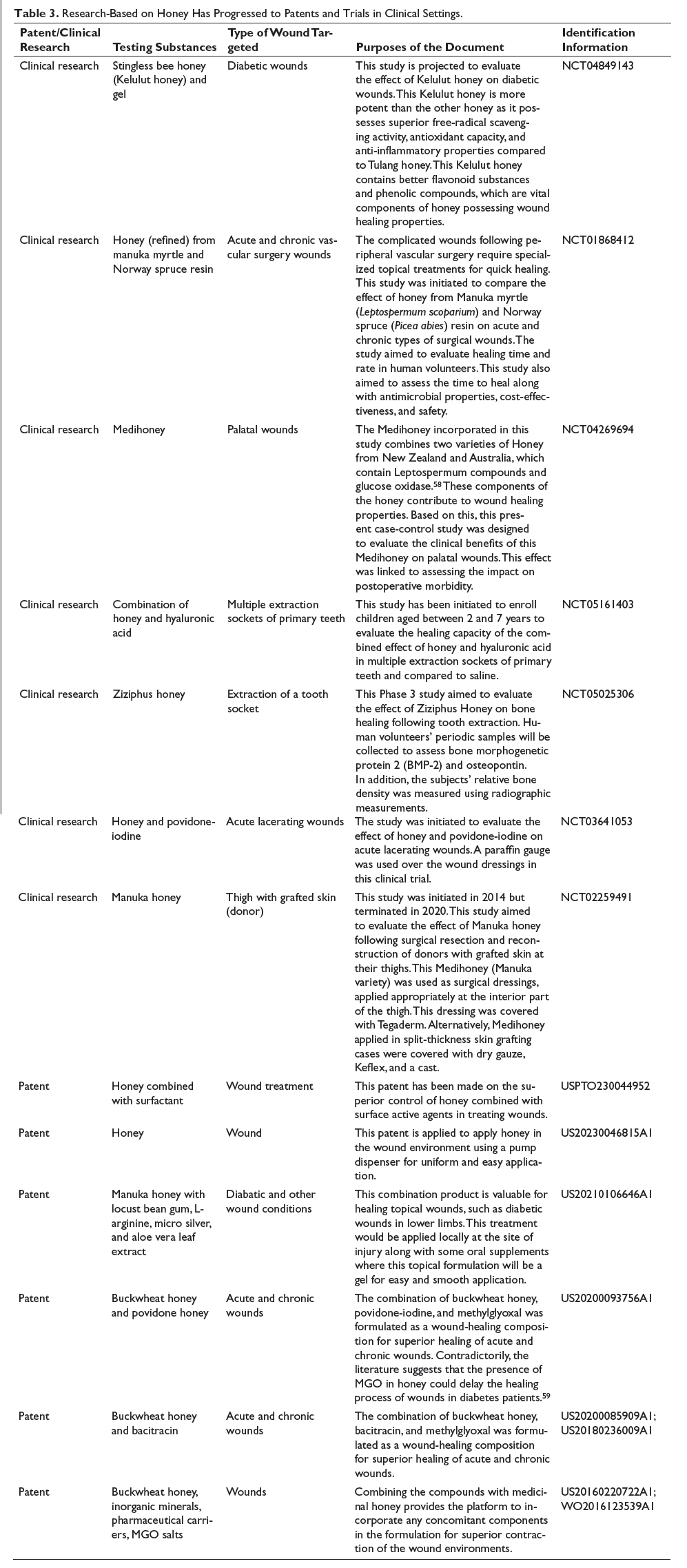
Clinical Trials and Intellectual Properties with Different Varieties of Honey
From the discussions above, it is clear that different varieties of honey or formulations containing MGH have been explored in wounds and burn conditions. These have shown potential in generating extracellular matrix, reducing inflammation, improving angiogenesis, and controlling secondary infections. Applying honey at the wound site can stimulate the production of monocytes and macrophages, thereby increasing the production of cytokines, including TNF-α.30, 67 Overall, honey accelerates the healing process, reflected by its favorable influences at the different phases of healing (either in traumatic or burn wound conditions). Various components of honey have a potential role in the healing process, which has converted numerous researches with honey into successful patents.68, 69
A registered clinical trial (NCT02259491) on MH is under investigation on patients undergoing reconstructive surgeries in a randomized, single-blinded, and controlled study. The study aims to evaluate the surgically removed skin reconstruction from the donor’s thigh area, where MH-based formulation was applied with a transparent covering. So far, no results have been reported yet; however, superior healing of the surgically created wound is expected. Similarly, several research works have moved into clinical settings, as summarized in Table 3. The application of honey is also known to promote wound healing through faster epithelialization, clearance of the wound exudates, dead and infected tissue, formed callus, and replacement with granulating tissue for the sloughs. 18 With the application of honey in wounds and ulcers, researchers are trying to establish suitable formulations so that the problems associated with raw honey can be overcome (US5980875A).
Research-Based on Honey Has Progressed to Patents and Trials in Clinical Settings.
Conclusion
Since the ancient past, honey has been employed in treating different types of wounds, but not until recently was the exact mechanism behind honey’s bioactivity evaluated. 70 The advent of antibiotics suppressed the researcher’s interest in honey; however, due to the recent antibiotic resistance developments, the focus of modern-day research is shifting towards natural alternatives. The multifunctional mechanism of action, including peroxide generation, inducing proinflammatory cytokines, decreasing biofilm formation, decreasing wound pH, inhibiting the bacterial cell cycle, modulating pain perception, and exertion of antioxidant and anti-inflammatory action makes honey a potential candidate for formulation development required for wound healing applications. 71 Honey for therapeutic applications is quite different from culinary honey based on its quality and secondary metabolite content, and it is known as MGH. This grade is currently employed in various marketed topical dressings for treating multiple types of wounds. Despite such extensive application in wound healing, modern-day medicine still lacks interest in applying honey due to the presence of multiple-drug treatment regimens for wound healing. This scepticism of physicians and patients can only be removed through extensive safety profiling and larger randomized controlled clinical trials of formulations involving honey.
Abbreviations
Footnotes
Acknowledgments
The authors sincerely acknowledge Birla Institute of Technology for the support to accomplish the work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not Applicable.
Funding
The authors are thankful for the financial support received from the Science and Engineering Research Board, Government of India, for the Start-up Research Grant (File No. SRG/2022/001211).
Informed Consent
Not Applicable.
