Abstract
Background
Asthma is a very common respiratory disorder, affecting more than 360 million people worldwide. It is a chronic inflammatory disorder of the airways with the symptoms of shortness of breath, coughing, chest tightness, wheezing, and sometimes chest pain. Leukotrienes play an important role in bronchoconstriction during the allergen or exercise-induced acute asthma attack.
Aim
The study aims to predict the interactions between leukotriene antagonist drugs and CysLT receptor-1 (CysLTR1), P2Y12 and peroxisome proliferator-activated receptor gamma (PPAR-γ) on a competitive basis. The study also has the objective of understanding the cardioprotective roles of the drugs.
Introduction
Asthma is strongly linked to the development of acute coronary syndrome by the leukotriene-induced activation of CysLTR1, platelet aggregation and thrombosis by activating P2Y12. PPAR-γ is considered to show benefits against atherosclerosis, diabetes, hypertension, obesity and dyslipidaemia, which are risk factors for the development of cardiovascular disorders. Leukotriene receptor inhibitors act with these three types of receptors to show therapeutic effects.
Materials and Methods
To predict the possible interactions between the drugs and the receptors, the study has used in silico methods.
Results and Discussion
Montelukast, Zafirlukast and Gemilukast are potential antagonists of CysLTR1 and P2Y12. They are also responsible for the upregulation of PPAR-γ. Thus, these drugs show a cardioprotective role in asthma-induced cardiac disorders.
Conclusion
A competitive in silico study of Montelukast, Zafirlukast and Gemilukast to predict their binding to CysLTR1, P2Y12 & PPAR-γ revealed that Montelukast is more effective than the other two drugs for showing a cardioprotective role.
Keywords
Introduction
According to the Global Asthma Report 2018, by 2025 the number of people having asthma will be around 400 million. 1 Without regard to age, gender, race, geographical variation and financial condition, people are affected by asthma worldwide. 2 Common symptoms of asthma include but are not limited to breathing shortness, coughing, chest tightness, wheezing and sometimes chest pain.3–5 Asthma is triggered by sensitization to dust particles, allergens, or pollens. 6 Sensitization leads to the immunoglobulin E-mediated mast cell activation and release of histamine, tryptase, prostaglandin D2, cysteinyl leukotrienes C4 and D4 and adenosine. Mast cells are also a source of cytokines and chemokines like Interleukin (IL)-4, IL-5, IL-6 and TNF-α, which also align with inflammatory mediators to cause the activation of neutrophils, eosinophils and T lymphocytes.6–8 These in the future cause late-phase airway narrowing and airway hyperresponsiveness. 6
Different kinds of leukotrienes such as LTB4, LTC4 and LTD4, being activated cause inflammation and create complications between the body’s coagulation system and innate immunity system. 9 This situation is also known as immunothrombosis, which gradually causes acute coronary syndrome (ACS).9–11 Though immunothrombosis is considered a defence mechanism for the heart to decrease the spreading of pathogens via the bloodstream, abnormal activation of immunothrombosis causes heart attack, stroke and thromboembolism. 10 LT binds with cysteine leukotriene (CysLT) and leads to platelet and clotting factors activation.9, 12 Leukotriene receptor inhibitors like Montelukast, Zafirlukast and Gemilukast are found to be able to block CysLT receptor-1 (CysLTR1) and reduce the risk of ACS.
P2Y12 is a chemoreceptor from G-protein coupled purinergic receptor group, that is found in the smooth muscles and immune cells. 13 The receptor is involved with the release of inflammatory cytokines during inflammatory conditions by promoting the interaction between platelets and leukocytes. P2Y12 can promote platelet aggregation and cause thrombus generation.13, 14 LTs activate CysLTR1 and that leads to the activation of P2Y12. Potential CysLTR-1 inhibitors can inhibit the activation of P2Y12 and reduce bone loss. 14 Again, CysLTR-1 inhibitors are found to show cardioprotective effects by the modulation of PPAR-γ (peroxisome proliferator-activated receptor-γ) activities. 15 PPAR-γ activation protects the coronary bed by reducing the spreading of macrophage and pro-inflammatory cytokines activation.16, 17
In this study, we had visualized the possible three dimensional (3D) interactions of Montelukast, Zafirlukast and Gemilukast with CysLTR1, P2Y12 and PPAR-γ to treat several conditions in the body, using a molecular docking process. Molecular docking is considered one of the most important tools of computer-aided drug discovery.18–20 Binding affinity between ligands and proteins can be measured from molecular docking. The potentiality of some drugs against a disease can be predicted based on docking scores obtained from docking software. 21 We used some online websites like PubChem and RCSB Protein Data Bank as well as some software named PyMol, Swiss PDB viewer, BIOVIA Discovery Study, PyRx, etc.
Effects of drugs in the body or a specific organ can be examined using in silico, in vitro, or in vivo methods. Both in vitro and in vivo methods are time-consuming and need special lab set-up, which is costly. Using in silico method prior to in vitro or in vivo methods provides a primary software-generated result on a research proposal, based on binding affinity between drugs and receptors. If a satisfactory result is derived from the in silico method, further in vivo or in vitro methods can be executed with more confidence. So, to make the best use of time and resources on in vitro or in vivo methods for a research proposal, in silico method should be done (if possible) at first to get the primary idea about the outcome.
Materials and Methods
Ligand Preparation
PubChem (
Receptor Preparation
Here target receptors CysLTR1, P2Y12 and PPAR-γ were taken to identify the interaction of the three drugs with these three receptors on a competitive basis, to find out the most effective drug for the treatment of asthma with additional therapeutic benefits. 3D structure of CysLTR1, P2Y12 and PPAR-γ had been downloaded from RCSB Protein Data Bank (
Docking
After the ligands and receptors preparation, AutoDock Vina was used for docking purposes to get the binding score. A better docking complex showed lower binding scores. The less the score, the better the binding. Then, BIOVIA Discovery Studio was utilized for the 2D and 3D visualization of the docked complexes.
Results
Docking Score
Nine docking scores had been obtained for each drug binding with three receptors. Docking scores represent the binding affinity of a drug to the receptor. The lower the value the stronger the bond.18–21 Montelukast, Zafirlukast and Gemilukast were docked individually with three receptors named, CysLTR1, P2Y12 and PPAR-γ.
Montelukast
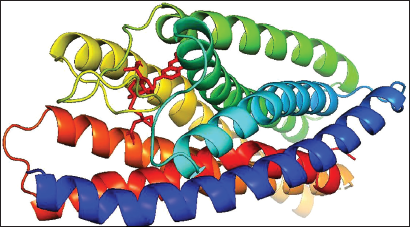
Montelukast and CysLTR1
(a) Docked Complex of Montelukast with CysLTR1.
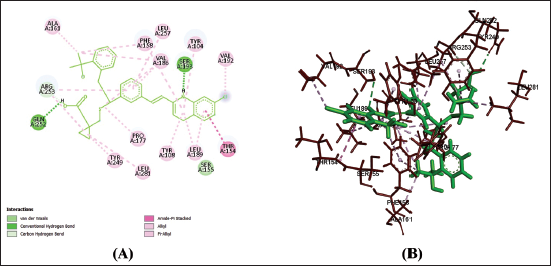
(b) 2D (A) and 3D (B) Structure of the Montelukast-CysLTR1 Complex.
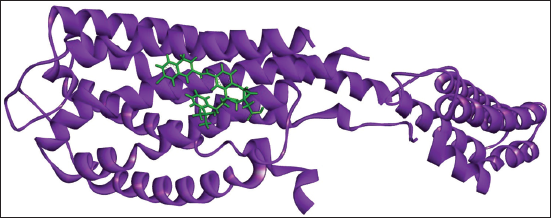
Montelukast and P2Y12
(c) Docked Complex of Montelukast with P2Y12.
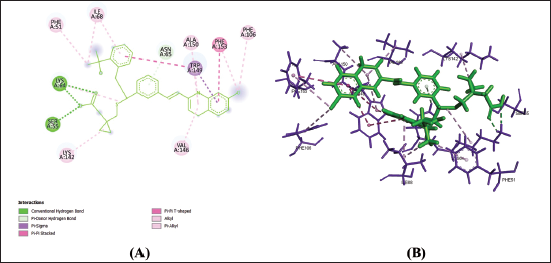
(d) 2D (A) and 3D (B) Structure of the Montelukast-P2Y12 Complex.
Montelukast and PPAR-γ
(e) Docked Complex of Montelukast with PPAR-γ.
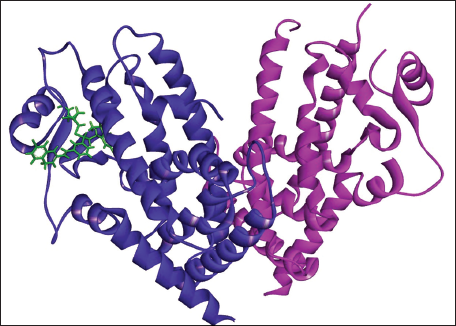
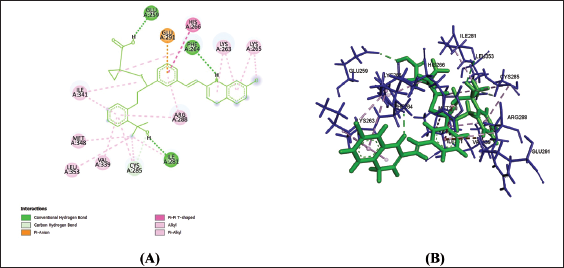
(f) 2D (A) and 3D (B) Structure of the Montelukast-PPAR-γ Complex.
Zafirlukast
Zafirlukast and CysLTR1
(a) Docked Complex of Zafirlukast with CysLTR1.
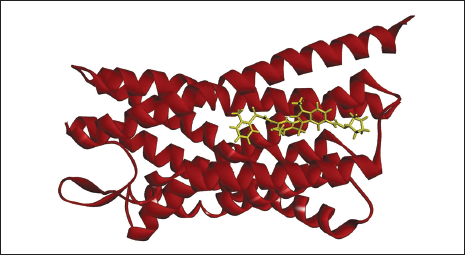
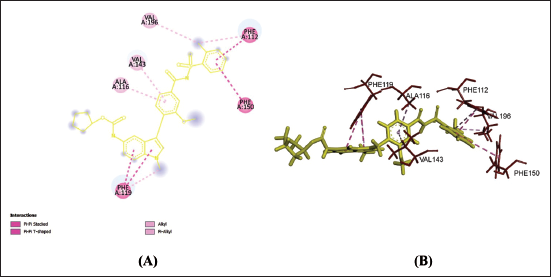
(b) 2D (A) and 3D (B) Structure of the Zafirlukast-CysLTR1 Complex.
Zafirlukast and P2Y12
(c) Docked Complex of Zafirlukast with P2Y12.
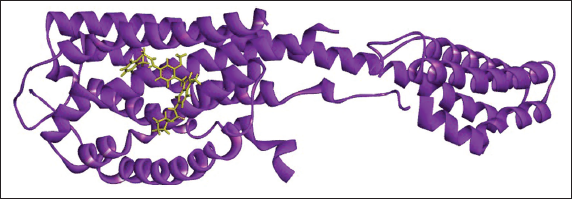
(d) 2D (A) and 3D (B) Structure of the Zafirlukast-P2Y12 Complex.
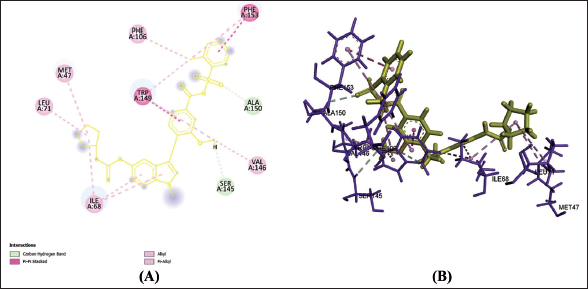
Zafirlukast and PPAR-γ
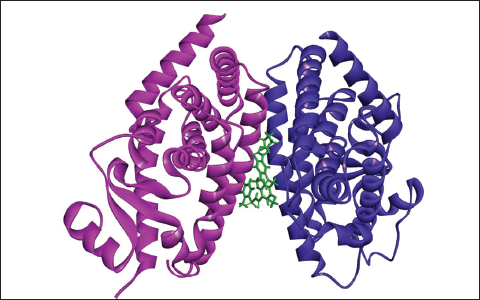
(e) Docked Complex of Zafirlukast with PPAR-γ.
(f) 2D (A) and 3D (B) Structure of the Zafirlukast-PPAR-γ Complex.
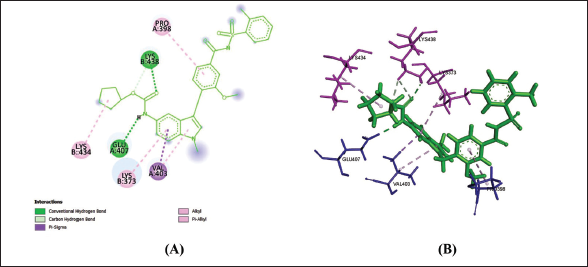
Gemilukast
Gemilukast and CysLTR1
(a) Docked Complex of Gemilukast with CysLTR1.
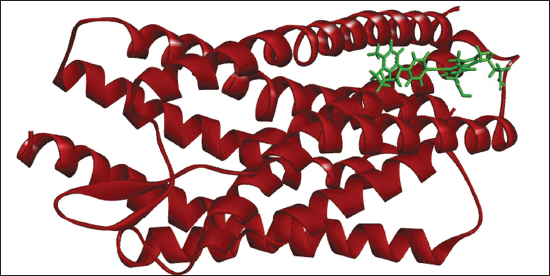
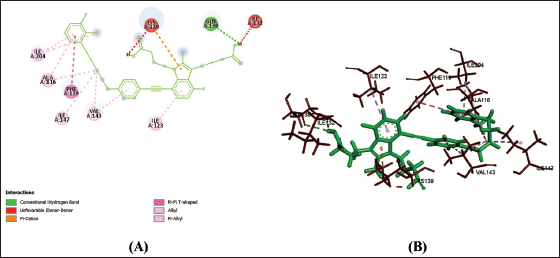
(b) 2D (A) and 3D (B) Structure of the Gemilukast-CysLTR1 Complex.
Gemilukast and P2Y12
(c) Docked Complex of Gemilukast with P2Y12.
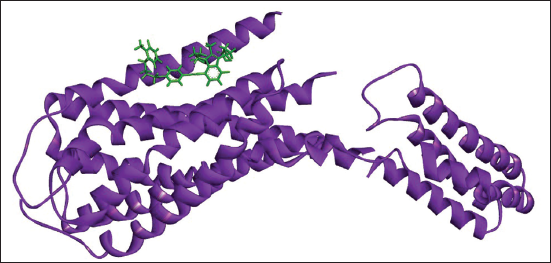
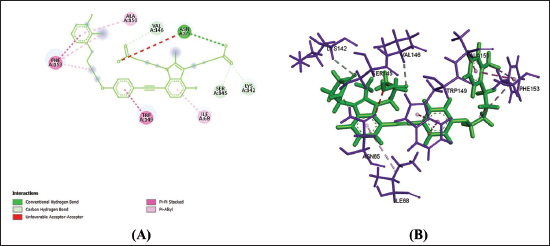
(d) 2D (A) and 3D (B) Structure of the Gemilukast-P2Y12 Complex.
Gemilukast and PPAR-γ
(e) Docked Complex of Gemilukast with PPAR-γ.
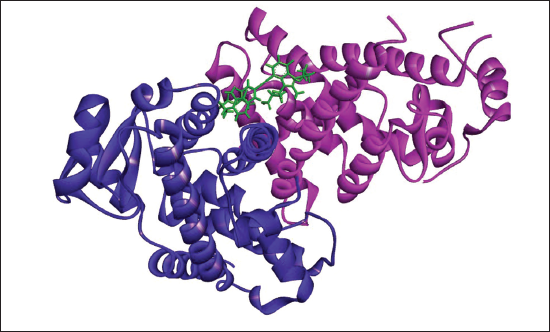
(f) 2D (A) and 3D (B) Structure of the Gemilukast-PPAR-γ Complex.
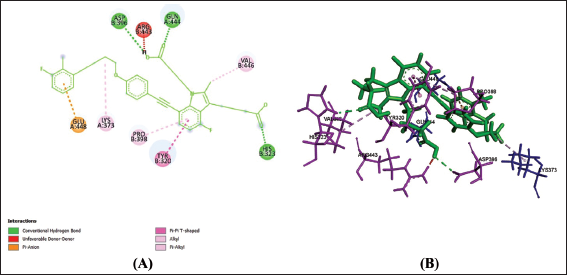
Discussion
The docking scores are discussed in the following:
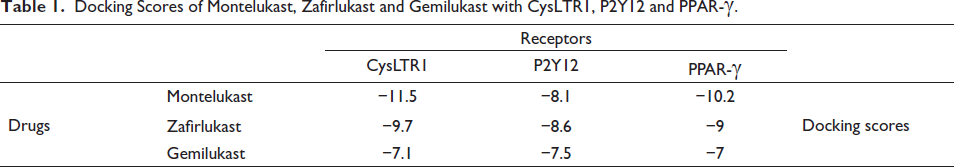
Montelukast binds to CysLTR1 and PPAR-γ more strongly than the other two drugs. On the other hand, Zafirlukast binds to P2Y12 with a lower most value than others, representing the strongest bond (Table 1).
Docking Scores of Montelukast, Zafirlukast and Gemilukast with CysLTR1, P2Y12 and PPAR-γ.
Amino acids are building blocks of proteins or receptors. The amino acids involved in CysLTR1, P2Y12 and PPAR-γ share different kinds of chemical bonds with the drugs (Table 2).
Bonds Between Amino Acids of Receptors (CysLTR1, P2Y12 and PPAR-γ) and Drugs.

From Table 2, Figures 1a–f, 2a–f and 3a–f, it is seen that all three drugs have the same binding points with P2Y12 by Pi bonds with PHE153 and TRP149 amino acids. Montelukast and Gemilukast have an H bond with P2Y12 by ASN65 amino acid in common. According to the docking scores, Montelukast binds to CysLTR1 and PPAR-γ more strongly than the other two drugs, by H bonds with SER193 and GLN252, Pi bond with THR154 for CysLTR1 and by H bonds with ILE281, PHE264 and GLU259, Pi bond with HIS266 for PPAR-γ. So, by blocking CysLTR1 and activating PPAR-γ, Montelukast shows a cardioprotective role by reducing the risk of ACS.
Conclusion
Competitive in silico study of Montelukast, Zafirlukast and Gemilukast to predict their binding to CysLTR1, P2Y12 & PPAR-γ had revealed that Montelukast is more effective than the other two drugs for showing cardioprotective role. Though Montelukast is highly used for the treatment of asthma, it also shows some beneficial roles for the heart. Asthma can lead to the development of ACS, so treating asthma with Montelukast can not only treat asthma effectively but also reduce the risk of ACS. As derived from several articles, this study also found that Montelukast, Zafirlukast and Gemilukast, all interact with CysLTR1, P2Y12 & PPAR-γ with different affinities, indicating all show beneficial effects to the heart.9, 22, 23 However, according to vina binding affinity, Montelukast is the most prominent drug among all three drugs. The cardioprotective effect provided by the drugs could be more efficiently claimed if an in vivo or biological model had been utilized. In the future, the result of the study will be extrapolated into the biological model. So far this is the limitation of the study.
Abbreviations
Footnotes
Acknowledgments
This study was self-funded. The author would like to thank Dr Sukalyan Kumar Kundu for his kind supervision during the research work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Not applicable
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Not applicable
