Abstract
Allergic rhinitis and asthma are common disorders effecting large percentages of the population of Western countries. There are multiple treatment options available for allergic rhinitis and asthma and stepwise approaches to therapy have been recommended. Montelukast is a cysteinyl leukotriene receptor antagonist that has been found to be effective both in the treatment of allergic rhinitis and asthma. This paper will describe the pharmacology, safety, efficacy and tolerability of montelukast. It will examine the comparative efficacy of montelukast to other medications for the treatment of allergic rhinitis and asthma, as well as discuss the recent studies of combination therapy.
Introduction
Allergic rhinitis (AR) is a common disorder that affects up to 50-60 million persons in the U.S, 1 accounting for 10%-40% of the population. 2 There is also strong evidence that the incidence may be increasing. 3 Given this high prevalence and the significant impact on quality of life, AR has been classified as a major chronic respiratory disease 4 and is now the 2nd leading cause of chronic disease in the United States. 1
The characteristic symptoms of allergic rhinitis are nasal congestion, sneezing, anterior and posterior nasal drainage, nasal itching and itchy/red/watery eyes. Patients may also complain of throat clearing, facial discomfort or headache, postnasal drip, or itchy throat and/or palate.1,4–6 Patients may also experience snoring, sleep disturbance, or fatigue and there is evidence that many patients may have decreased sense of sexual function. 7 The treatment of AR can lead to improvements in the specific allergic symptoms, enhancing overall quality of life and potentially reducing the incidence and/or severity of a number of comorbid disorders including asthma, rhinosinusitis, eustacian tube dysfunction and otitis media, allergic conjunctivitis, sleep disorders4,6,8 and may improve sexual function. 7
Asthma is also a very common disorder with 3.5% of the population in the United States having an asthmatic episode annually based on information from the US Department of Health and Human Services and the Center for Disease Control. 9 The annual incidence of an asthmatic episode is increased in lower socio-economic groups. 9 Overall prevalence has been reported at approximately 15 million people or 1 person in 20 in the U.S. 10 These surveys also suggest that asthma is poorly controlled in the population as a whole with 9% of asthmatics requiring a hospitalization and 23% of asthmatic adults and 32% of asthmatic children requiring an emergency room visit. 10
Evidence for the association between rhinitis and asthma has frequently been reported among epidemiologic studies. It has been stated that asthma may affect up to 40% of patients with allergic rhinitis, this percentage is significantly higher than that of the general population.11,12 The prevalence rates of rhinitis among asthmatic patients can vary widely and can be as high as 80% depending on the age of onset. Rhinitis tends to precede the onset of asthma in patients with upper and lower airway disease and tends to be associated with bronchial hyperresponsiveness.11,13 Findings suggest that rhinitis may be the initial manifestation of an allergic airway disease that leads to the onset of asthma.11,13,14
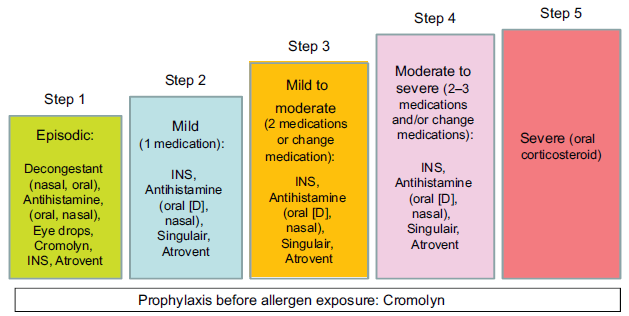
Step-care therapy for allergic rhinitis: US 2008 Updated Practice Parameter recommendations (adapted from the Rhinitis Action Plan). 6
There have been a number of recommendations for a stepwise approach for the management of allergic rhinitis. Two highly regarded and well substantiated recommendations are the Practice Parameter recommendations 6 (Fig. 1) and the ARIA (Allergic Rhinitis and its Impact on Asthma) guidelines. 4 In both of these recommendations there is a stepwise approach depending on the seasonality, duration and/or severity of disease. These also both include multiple different pharmacologic agents including oral antihistamines and decongestants, intranasal decongestants, intranasal corticosteroids, cromolyn sodium, intranasal antihistamines, and ipratoprium bromide. One class of medication that has been found to be effective either by itself or in combination for the management of AR is the leukotriene modifiers. In the United States the leukotriene receptor antagonist (LTRA) approved for the treatment of AR is montelukast. Montelukast is also one of the approved LTRAs for the treatment of asthma. This report will describe the pharmacology and clinical trial data in relationship to the use of montelukast in AR and asthma. The literature was reviewed following a MEDLINE search for “montelukast, leukotriene receptor antagonists, cysteinyl leukotrienes, asthma and allergic rhinitis”. Review articles that describe AR and asthma were also evaluated. Articles were selected that describe key parameters of a review article on montelukast. Although a thorough search was accomplished, a systematic review of the literature was not performed.
Cysteinyl Leukotrienes
Leukotriene LTC4, LTD4, LTE4, collectively termed cysteinyl leukotrienes (CysLTs) are peptide conjugated lipids that are the products of activated eosinophils, basophils, mast cells and macrophages. 15 CysLTs are arachidonic acid derivatives that are synthesized and released by immunocytes in the respiratory mucosa in response to the presence of an allergen.16–18 They are appropriately named as the leukotrienes are produced by leukocytes and their chemical structure contains three double bonds, a triene. 19 The cysteinyl leukotrienes (C4, D4, E4) are generated from the arachidonic pathway via 5-lipoxygenase to intermediate leukotriene A4, then to leukotriene C4 (by leukotriene c synthase), leukotriene D4 (by gamma-glutanyl-transpeptidase) and leukotriene E4 (by dipeptidase). 20 The cysteinyl leukotrienes are highly potent mediators of inflammation that lead to several reactions including contraction of bronchiolar smooth muscle, chemoattraction of eosinophils with stimulation of the release of other mediators of inflammation, increased mucous production, and mucosal swelling brought about through increased vascular permeability and blood flow to the mucosa. 16 Increased CysLTs have been demonstrated in nasal lavage fluid in patients with allergic rhinitis after antigen provocation21,22 and BAL washings in patients with asthma. 23 CysLTs exert their actions through activation of two G protein coupled receptors, termed CysLT subtype 1 receptor (CysLT1) and CysLT subtype 2 (CysLT2) receptor. CysLT1 is most studied and is the target for drugs such as montelukast. 24 Montelukast is a potent and highly selective antagonist of type 1 CysLT receptors, with an affinity two fold greater than the natural ligand.20,25
Pharmacology
Montelukast is an orally active, highly selective LTRA that inhibits the CysLT1 receptor. Montelukast inhibits physiologic actions of LTD4 at the CysLT1 receptor without any agonist activity. It is rapidly absorbed after administration reaching peak plasma concentration (Cmax) in 3 to 4 hours with a mean bioavailability of 64% following a 10 mg oral administration. For the 5 mg chewable tablet, the mean Cmax is achieved in 2 to 2.5 hours with a mean bioavailability of 73% fasting versus 63% with the standard meal. The 4 mg chewable tablet achieves Cmax in 2 hours, with fasting, in the 2 to 5 year old range. 26 More than 99% is bound to plasma proteins with minimal distribution across the blood-brain barrier. Metabolism occurs via liver P450 (CYP) 3A4 and 2CP microsomes, with potent inhibition of P450 2C8. Excretion occurs almost exclusively in bile with a half-life from 2.7 to 5.5 hours in healthy adults. The pharmacokinetic profile is similar in females and males, young and elderly. 26
In patients with mild to moderate hepatic insufficiency, no dosage adjustment is required but data is lacking regarding severe hepatic impairment. Montelukast and its metabolites are almost exclusively excreted in bile and not urine, and it therefore has not been evaluated in patients with renal insufficiency. In drug interaction studies, the recommended clinical dose of montelukast did not have clinically important effects on the pharmacokinetics of the following drugs: theophylline, prednisone, prednisolone, oral contraceptives, digoxin and warfarin. Phenobarbital, which induces hepatic metabolism, decreased the plasma concentration of montelukast approximately 40% following a single 10 mg dose of montelukast, yet no dosage adjustment for montelukast is recommended in conjunction with phenobarbital usage. 26
Safety and Tolerability
Montelukast is well tolerated with a safety profile that is similar in adult and pediatric populations. Studies looking at safety and adverse effects have demonstrated no clinical or laboratory difference in adverse experiences versus placebo.16,27 Adverse effects have been described as mild and most often include headache, gastrointestinal disturbances, fatigue, pharyngitis, upper respiratory tract infection and rash.16,19,27 These events occurred at a frequency greater than or equal to 1% above placebo. 26 In a large phase IV study, 14 of 6158 patients (0.23%) reported 21 drug related adverse events consistent with the events noted above. 28
Isolated reports of Churg-Strauss syndrome (CSS), a rare systemic vasculitis asssociated with asthma, have been described in asthma patients treated with montelukast.29,30 CSS is a rare necrotizing vasculitis with an annual prevalence of approximately 60 per million in asthma patients and 2-7 per million in the general population.16,31 Its precise etiology is not known, yet its clinical onset is most often preceded by discontinuation or tapering of corticosteroid therapy26,32 and consists of eosinophilia, pulmonary infiltrates, cardiomyopathy and other signs of vasculitis. 29 A likely explanation is that inhaled or oral corticosteroid treatment in these patients may mask the underlying vasculitis that develops as the glucocorticosteroid doses are reduced in patients with severe asthma being treated with leukotriene receptor antagonists (LTRAs). Physicians should be aware of signs and symptoms of CSS in patients with moderate to severe asthma with tapering doses of corticosteroids. 29
Although they are well tolerated, particular attention should be given to the consideration of periodically monitoring liver function tests during treatment with LTRA. There have been reported cases of liver dysfunction going from mild to severe in patients treated with these medications. Most hepatobiliary events occurred in combination with other confounding factors, such as other medications or montelukast administration to patients with underlying potential for liver disease, such as alcohol use or hepatitis. 26 Elevations in alanine aminotransferase/glutamic oxaloacetic transaminase (ALT/GOT) and aspartate aminotransferase/ glutamic piruvic transaminase (AST/GPT) with montelukast occurred at a frequency that was generally comparable with placebo. 26 In patients treated with 10 mg as a single dose or duration up to 2 years, 2.1% had an increase in ALT compared to 2% of placebo. AST elevations were reported as adverse events in 1.6% and 1.2% of montelukast and placebo patients, respectively.26,29 Consideration has been given to the fact that some patients who have demonstrated toxicity to LTRAs, possibly due to the toxic metabolite acyl-glucoronide, may have inherited a defect in the detoxification enzymes,29,33 and therefore a genetic basis for drug-induced liver disease may exist.
Most recently, the FDA has published reports of agitation, aggression, anxiousness, dream abnormalities, hallucinations, depression, insomnia, irritability, restlessness, suicide, suicidal ideation, and tremor associated with the use of montelukast and other LTRAs based upon post marketing reports published by the drug manufacturer. 34 Wallerstedt et al. analyzed all reports of psychiatric disorders during treatment with montelukast in children occurring in Sweden. A total of 48 reports were found and included nightmares, anxiety, aggressiveness, sleep disorders, insomnia, irritability, hallucination, hyperactivity and personality disorder. Forty eight percent of these patients were 3 years of age or younger and in those where time to exposure was reported, 80% of these occurred within 1 week of beginning treatment. 35 Further studies have reported insufficient data to prove a link between montelukast and suicidality. 36 It has been reported that the rates of depression, anxiety, and sleep disturbance which are risk factors for suicide are greater in patients with allergic rhinitis compared to the general population and that patients with a history of allergy may have an increased rate of suicide. 37 Sleep disturbances have additionally been found to be more common in asthmatics and are associated with asthma control and quality of life. 38
Leukotriene Inhibiting Drug
Montelukast (Singulair) is one of the most commonly prescribed LTRAs available and the only LTRA approved for the treatment of both AR and asthma. This LTRA demonstrates rapid oral administration (three hours to peak plasma concentrations), near maximal plasma protein binding (99%) and excretion principally in the bile. The terminal half life of montelukast is 5 hours and is considered a pregnancy category B medication.26,39
Montelukast is indicated for adults and for children two years and above. It is available in tablet, chewable table and cherry flavored tablet form. Ten mg is recommended for ages 15 and older, 5 mg chewable tablets for patients 6 to 14 years of age, 4 mg chewable tablets in patients 2 to 5 years of age, and 4 mg oral granule formulation in pediatrics for asthma in 12 to 23 months and allergic rhinitis in 6 to 23 months of age. 26 Dosing is daily. Adult dosing can be taken with food, pediatric dosing should be avoided 1 hour before and 2 hours after meals. Montelukast is contraindicated in patients with acute asthma or those with severe hepatic impairment. 26 As human model studies in pregnant or nursing women have not been performed, therefore montelukast should only be used in this population if clearly indicated. 26 There are no drug interactions with oral contraceptive pills, warfarin, digoxin or theophylline. Coadministraion with CYP 3A4 hepatic enxyme inducers (phenytoin, phenobarbitonl, rifampin) may decrease montelukast levels. 26
Clinical Trials Comparing Montelukast to Placebo
There have been a number of trials that support the efficacy of montelukast in comparison to placebo. A prospective, randomized, double-blind placebo controlled trial has shown that montelukast was superior to placebo in the management of sneezing, rhinorhea, itching and congestion in spring seasonal AR patients. 40 A study by Chervinsky et al showed similar improvement of symptoms in patients treated with montelukast in comparison to placebo in an evaluation of the results of three prospective clinical trials in fall AR patients. 41 A study in children 7 to 14 years of age showed a reduction in nasal and eye symptom scores and decreased blood esosinophil counts in subjects treated with montelukast in comparison to placebo. 42 A randomized placebo-controlled trial has also shown efficacy of montelukast in comparison to placebo in total nasal symptom scores and a quality of life questionnaire in children 2-6 years of age. 43 Studies have also shown the effectiveness of montelukast in comparison to placebo in both seasonal and perennial AR patients in both adults and children as part of trials that evaluated montelukast in combination with other medications.44,45 This improvement included the control of daily symptoms of nasal congestion, itching, sneezing and rhinorhea as well as other endpoints such as eye symptoms, global evaluations, nighttime symptom scores and quality of life surveys.44,45
The clinical efficacy of montelukast in adults and children with persistent asthma, including patients sensitive to aspirin, has been evaluated in several randomized, double-blind, placebo controlled trials. A study by Reiss et al demonstrated that montelukast improved airway obstruction (FEV1, morning and evening peak expiratory flow rates) and patient reported end points (daytime asthma symptoms, as needed beta-agonist use, nocturnal awakenings) when compared with placebo in asthmatic patients greater than 15 years of age. 46 In over 12 weeks of treatment of patients aged 2 to 5 years, montelukast administered as a 4-mg chewable tablet produced significant improvements compared with placebo in multiple parameters of asthma control including: daytime asthma symptoms (cough, wheeze, trouble breathing, and activity limitation); overnight asthma symptoms (cough); the percentage of days with asthma symptoms; the percentage of days without asthma; the need for beta-agonist or oral corticosteroids; physician global evaluations; and peripheral blood eosinophils. The clinical benefit of montelukast was evident within 1 day of starting therapy. 47 Direct bronchoscopic exam after 6 weeks of montelukast therapy in adults demonstrated significantly reduced numbers of activated eosinophils and mast cells compared to placebo upon examination of bronchial mucosa. 48 Pizzichini et al have reported decreased sputum and blood eosinophils after 4 weeks of montelukast therapy compared to placebo. 49 In one study, montelukast therapy offered significantly greater protection against exercise-induced bronchoconstriction than placebo therapy as evidenced by the improvement in the area under the FEV1 curve, the significant improvement in the maximal decrease in FEV1 after exercise and the time from the maximal decrease in FEV1 to the return of lung function to within 5 percent of pre-exercise. After 12 weeks of treatment, patients in the montelukast group were more likely to rate their asthma control as better and less likely to require rescue therapy with a beta-agonist during or after exercise challenge. 50
Monteleukast in Comparison to other Medications
A number of studies have shown that intranasal corticosteroids are superior to montelukast in the management of symptoms of AR.51–54 A double-blind randomized controlled trial showed that fluticasone propionate (FP) was superior to montelukast in the control of both daytime and nighttime symptoms as well as reduction in eosinophil counts in patients with seasonal AR. 51 In patients with asthma who were being treated with inhaled fluticasone propionate/salmeterol, the addition of FP provided superior rhinitis control to the addition of montelukast. 52 Two additional randomized, double-blind placebo controlled trials have also shown greater improvement in daytime and nighttime nasal symptom scores and individual nasal symptom sores.53,54 A comparison of montelukast to pseudoephedrine showed comparable control of nasal symptom scores and quality of life scores and increasing peak nasal inspiratory flow rates in the treatment of AR. 55
Combination Therapy
There have been a number of studies that have compared montelukast in combination with non-sedating antihistamines (NSA) in comparison to montelukast alone, NSA alone or intranasal corticosteroids. In one study, a combination of montelukast and loratadine has been found to be superior to either montelukast or loratadine alone in seasonal allergic rhinitis 45 while in another study the combination of montelukast/loratadine was not significantly different from loratadine alone. 44 In patients with persistent AR, the combination of montelukast and either desloratadine or levocetirizine were more effective than monotherapy with either of these three medications. 56 In children 2-6 years old, montelukast was equivalent to cetirizine in the management of nasal symptoms of perennial AR except for nasal itching which was better controlled with cetirizine. 43 Montelukast in combination with cetirizine showed better control of the clinical nasal symptoms and reduced eosinophil counts better than either montelukast or cetirizine alone in seasonal AR patients. 57 When given 6 weeks before the start of the pollen season this combination delayed the onset of allergic seasonal symptoms in comparison to monotherapy. 57 Once daily treatment of fexofenadine (120 mg) was found to be equivalent to montelukast (10 mg) plus loratadine (10 mg) on peak nasal inspiratory flow rates and symptoms in seasonal AR. 58 A study comparing fexofenadine-pseudoephedrine was equivalent to a combination loratadine-montelukast in control of symptoms quality of life questionnaire and peak inspiratory flow rates. 59 Combined montelukast and cetirizine showed a significant change from baseline in nasal symptoms and nasal flow rates. 60 Finally, a combination of montelukast with loratadine has been found to be superior to loratadine alone in the treatment of delayed pressure utricaria. 61
Studies have been performed to evaluate the efficacy of an intranasal corticosteroid and combined montelukast with a NSA. In patients with both AR and asthma, both budesonide nasal spray and combined montelukast/cetirizine improved nasal symptoms and nasal airflow rates although had less substantial impact on laboratory measurements of nasal function such as rhinomanometry. 62 In a similar study comparing mometasone furoate nasal spray and combined montelukast/cetirizine, there was no difference between the groups for nasal symptoms, nasal flow and nasal laboratory tests. 60 When fluticasone propionate nasal spray was evaluated in relationship to a combination of loratadine/montelukast, median total nasal symptom scores were not statistically significantly different but scores in a quality of life questionnaire were better and the eosinophil count and eosinophilic cationic protein counts were lower in the fluticasone group. 63
Combination therapy has also been assessed in patients with asthma. In a study comparing montelukast to budesonide alone (in 400 or 800 μg bid) and to combination montelukast/budesonide (400 μg bid), bronchial reactivity was improved in all of the budesonide groups in comparison to the montelukast group while there were no differences between the budesonide groups. 64 Asthma maintenance and control may be improved with the addition of montelukast. One study demonstrated that the addition of montelukast to inhaled corticosteroid steroid (ICS) therapy alone or in combination with a long acting beta agonist in asthma patients resulted in a significant decrease in asthma attacks, emergency room visits, hospitalizations and oral corticosteroid use. 65 Several studies have shown that the addition of montelukast to ICS improves control of mild to moderate asthma compared to ICS therapy alone. Though salmetrol was clinically as effective, or even more effective than the addition of montelukast in these studies, montelukast may have a better long term safety profile.66,67
There are at least 2 studies that have shown that an intranasal glucorticoid spray is more effective than a combination of montelukast with an NSA.51,68 In a study of fluticasone propionate in comparison to montelukast/cetirizine, the fluticasone patients experienced greater improvement in nasal congestion and total nasal symptom scores. 68 The addition of montelukast or cetirizine to fluticasone propionate only improved nasal itching scores in comparison to fluticasone alone. 68 Similarly, fluticasone propionate and combination of montelukast/loratadine both improved daytime symptom scores in comparison to montelukast alone. 51 Fluticasone was, however, superior to either montelukast alone or the combination of montelukast/loratadine in the control of the nighttime symptom scores and the eosinophil count only was decreased in the fluticasone group. 51
Conclusion
Montelukast is an antileukotriene agent that impacts an important portion of the allergic inflammatory pathway. It has been shown to be an effective treatment for AR and in some extent for asthma. The effectiveness may be potentiated with the addition of a NSA in combination. The bulk of the evidence would suggest that montelukast is less effective than intranasal glucocorticoids for AR. When evaluating the results of studies comparing an INS to a combination of montelukast/NSA, the results are inconsistent, with some showing equivalence of treatment effects while others suggest that the INS may be more effective, particularly in nighttime symptoms.
Disclosures
Dr Benninger
Consultant: GSK, Sanofe-Aventis, Schering, Alcon
Speaker: Sanofe-Aventis, Alcon
Research Grants: GSK.
Dr Waters
No disclosures.
