Abstract
Background and Purpose
In alloxan-induced diabetic rats, this study sought to evaluate the hypoglycemic potential of the hydroalcoholic seed extract of Areca catechu, also referred to as areca nut, both as a stand-alone treatment and in combination with the diabetic drug gliclazide.
Materials and Methods
The hot percolation method was used to produce the hydroalcoholic seed extract of A. catechu. Rats were given alloxan monohydrate and developed type II diabetes. The hydroalcoholic seed extract of A. catechu (1000 mg/kg p.o.) alone or in different combinations of gliclazide (25 mg/kg p.o.) and A. catechu extract (250, 500, and 1000 mg/kg p.o.) were administered to the six diabetic rats. Body weights and blood glucose levels were measured on days 7, 14, 21, and 28 of the treatment. On the 28th day, the kidney, liver, and pancreas underwent histological investigations in addition to an oral glucose tolerance test (OGTT).
Results
Blood glucose levels were significantly lowered upon administration of A. catechu’s hydroalcoholic seed extract, both alone and in conjunction with gliclazide. Comparing the diabetic control group to the A. catechu extract-treated group, histopathological examinations showed that high amounts of the extract, either by itself or in conjunction with gliclazide, caused the active proliferation of pancreatic beta cells, protecting against damage to the liver and kidneys.
Conclusion
The hydroalcoholic seed extract of A. catechu showed promise as a monotherapy for controlling blood glucose levels, and as a co-administer for gliclazide, it showed signs of hypoglycemia. These results indicate A. catechu’s possible therapeutic benefit as an adjuvant diabetic treatment. To investigate the underlying mechanisms of action and evaluate its safety and effectiveness in clinical settings, more study is necessary.
Introduction
The complicated and long-lasting metabolic endocrine syndrome known as diabetes mellitus is characterized by inadequate insulin secretion, which raises blood glucose levels. The use of traditional medicinal herbs has a long historical heritage in the management of diabetes. 1 Herbal medicines have long been acknowledged by patients and medical professionals as an advantageous alternative to conventional pharmaceuticals for the treatment of diabetes. This approach can help reduce the negative effects that come with pharmaceutical interventions. In countries like China and India, the long-standing tradition of fusing herbal therapy with contemporary medicine has been especially common. A significant approach to the complete management of diabetes that highlights the wide range of diabetes treatment approaches is the combination of conventional herbal remedies and pharmaceutical products. Therefore, in order to shed light on herbal therapy’s potential as a supplemental means of managing diabetes, this study will examine the historical significance and current applicability of this treatment. Today, this practice—which takes the form of health supplements has extended to wealthy nations like the United States. In China, it is normal practice to prescribe herbs in addition to medications. In this case, the herb may interact with the medication to change its pharmacodynamics, pharmacokinetics, or both, potentially increasing or decreasing the medication’s effects.2, 3
While oral hypoglycemic medications play a significant role in managing hyperglycemia in individuals with non-insulin-dependent diabetes mellitus (NIDDM), they frequently fail to bring glucose concentrations into the normal range or maintain a normal glucose homeostasis pattern. The use of these medications is limited by their pharmacokinetic characteristics, subsequent failure rates, and unwanted side effects, even if effective glycemic control is obtained. Since the therapy is lifelong, it would be advantageous to utilize therapeutic agents free of adverse effects. Using herbs or herbal products is one approach to this goal, as does using alternative medicine. 4 Areca catechu has been referred to as betel nut or areca nut. An estimated 400 million people chew betel quid, which is primarily made up of lime, areca nut, and betel leaf. It is estimated that 7% of women and 30% of men in India chew areca nuts regularly. 5 Chewing Areca nut is a common and traditional practice among the Indian populace. Many researchers conducted biased and inconclusive population-based studies on people who chew pan masala or betel quid. Without considering the impact of other ingredients in pan masala or betel quid, they solely held the areca nut responsible for the outcomes they observed.6, 7 Conversely, in experimental animals, areca nut has shown hypoglycemic activity.8–10 Hence, in this regard, the aim of the present study was planned to evaluate the effect of hydroalcoholic seed extract of A. catechu alone and in combination with gliclazide on the blood glucose level.
Materials and Methods
Collection of Plant Material
The seeds of A. catechu (Areca nut) were collected in November 2022 from Kundapura, Karnataka. The taxonomic identification was conducted by Dr. V. Rama Rao, Research Officer (Botany), Central Ayurveda Research Institute, Bangalore, Karnataka, India. A voucher specimen has been deposited under the number RRCBI-mus114.
Preliminary Phytochemical Screening 11
The preliminary phytochemical screening revealed the presence of alkaloids, glycosides, cardiac glycosides, proteins and amino acids, flavonoids, tannins, saponins, and phytosterols.
Preparation of Hydroalcoholic Seed Extract of A. catechu 12
The Soxhlet method of hot percolation was used to carry out the extraction. 200g of areca powder was extracted using a 50:50 ethanol-water mixture. A concentrated thick mass was obtained by evaporating the solvent in a rotary evaporator. It was then dried using a freeze-dryer. Before being used, the dried extract was kept at 4°C. The percentage yield of the extract was found to be 10%w/w.
Chemicals
Alloxan monohydrate [Loba Chemie Pvt. Ltd. Maharashtra, India (Batch No- L3355262101)], Gliclazide [Gift sample from Bal Pharma, Karnataka, India (Batch No- 1001092301004)], Tween 80 [Spectrum PVT. Ltd. Mumbai, India (Batch No- G304508)], Thiopental sodium injection I.P. [Neon Laboratories Limited (Batch No- 173263)].
Experimental Animal Study
The male Albino Wistar rats (200–250g) were approved by the ‘Committee for the Control and Supervision of Experiments on Animals’ (CCSEA) with a proposal number VIPS/IAEC/2909-2022/12-AKV. They were housed at room temperature, in a well-ventilated animal house under 12 hours light/dark cycle. Institutional Animal Ethics Committee (IAEC) approval was obtained before carrying out this experiment. The animals were maintained under standard conditions in an animal house as per the guidelines of CCSEA for one week before use. The rats had free access to standard rat chow and water ad libitum during this period.
Acute Oral Toxicity 13
The acute oral toxicity of hydroalcoholic seed extract of A. catechu was performed by Bhandare A M et al. It was carried out in healthy nulliparous and non-pregnant adult female albino Swiss mice and albino Wistar rats as per the OECD guidelines- 423. The mice and rats were treated with 2000 mg/kg, orally. In the acute oral toxicity test, the dose of 2000 mg/kg did not cause any mortality and did not show any signs of toxicity or change in general behavior or other physiological activities in mice and rats during the 14-day observation. Hence, 1/2th, 1/4th and 1/8th doses were selected.
Treatment
The animals were divided into eight groups consisting of six animals in each group.
Group 1: 1% Tween 80 in control animals
Group 2: 1% Tween 80 in diabetic control animals
Group 3: Hydroalcoholic seed extract of A. catechu (1000 mg/kg) in Normal animals
Group 4: Hydroalcoholic seed extract of A. catechu (1000 mg/kg) in Diabetic animals
Group 5: Gliclazide (25 mg/kg) in Diabetic animals
Group 6: Hydroalcoholic seed extract of A. catechu (250 mg/kg) + Gliclazide (25 mg/kg) in Diabetic animals
Group 7: Hydroalcoholic seed extract of A. catechu (500 mg/kg) + Gliclazide (25 mg/kg) in Diabetic animals
Group 8: Hydroalcoholic seed extract of A. catechu (1000 mg/kg) + Gliclazide (25 mg/kg) in Diabetic animals
The treatment for the respective groups was carried out for 28 days.
Oral Glucose Tolerance Test (OGTT) 14
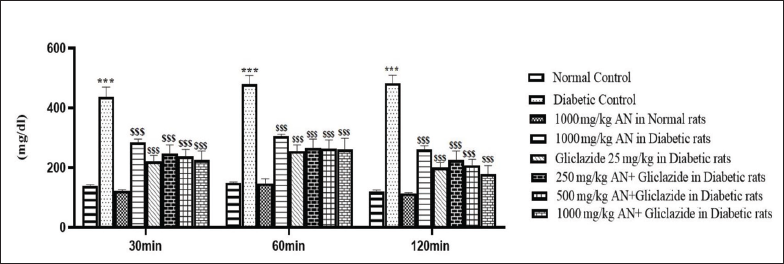
The OGTT was performed in overnight fasted (16 hours) rats. Rats were divided into eight groups and treatment was given as mentioned above. Glucose (2 g/kg) was fed 30 min after the administration of drugs. Blood was withdrawn from the tail vein puncture at 30, 60, and 120 min of glucose administration and blood glucose levels were determined using a glucometer.
Alloxan-induced Diabetes in Adult Rats 3
The fasting blood sugar was determined in Albino Wistar rats after an overnight fast with free access to water. Diabetes was induced by a single intraperitoneal injection of freshly prepared Alloxan monohydrate (150 mg/kg dissolved in 0.9% w/v normal saline). After that, the animals were left aside for four hours and then 10% glucose solution was given to the animals for 24 hours. The diabetes was confirmed by estimation of blood glucose level on the fifth day. Rats having blood glucose levels >250 mg/dl were used for the study. Animals received drug treatment for 28 days after induction of diabetes. The fasting serum glucose levels were determined on the 7th, 14th, 21st, and 28th day of drug treatment by the withdrawal of blood (0.1 ml) through the retro-orbital sinus under thiopental sodium anesthesia (40 mg/kg) I.P. The body weight of the animals was monitored every seven days during the drug treatment. After the last blood glucose estimation (GOD-POD method), the rats were sacrificed, pancreas, liver, and kidney were excised and subjected to histopathological studies to determine the inflammatory and necrotic changes. The tissues were stained using H&E stain and observed under 100X magnification.
Statistical Analysis
Results are expressed as mean ± S.E.M. The statistical significance was determined using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test. p < 0.01 and p < 0.001 were considered statistically significant.
Results
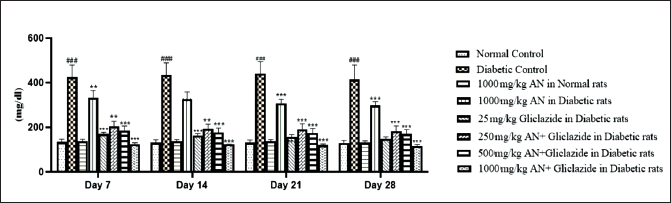
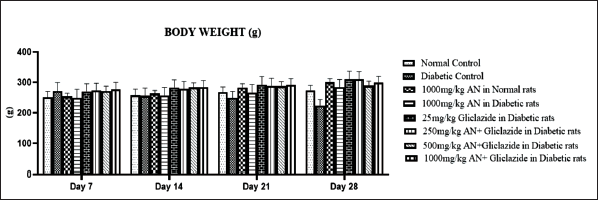
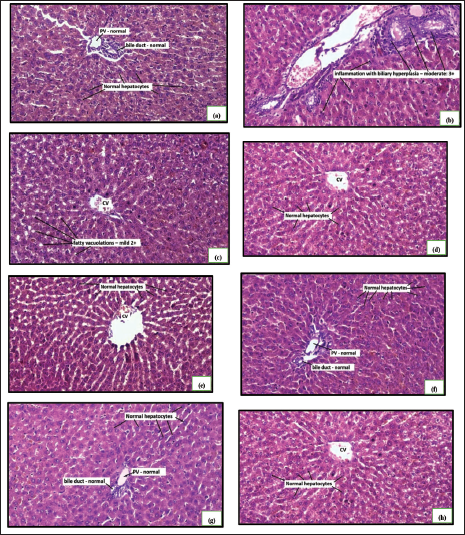
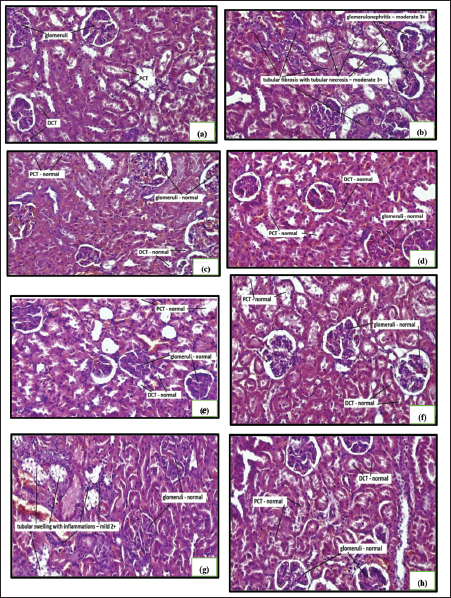
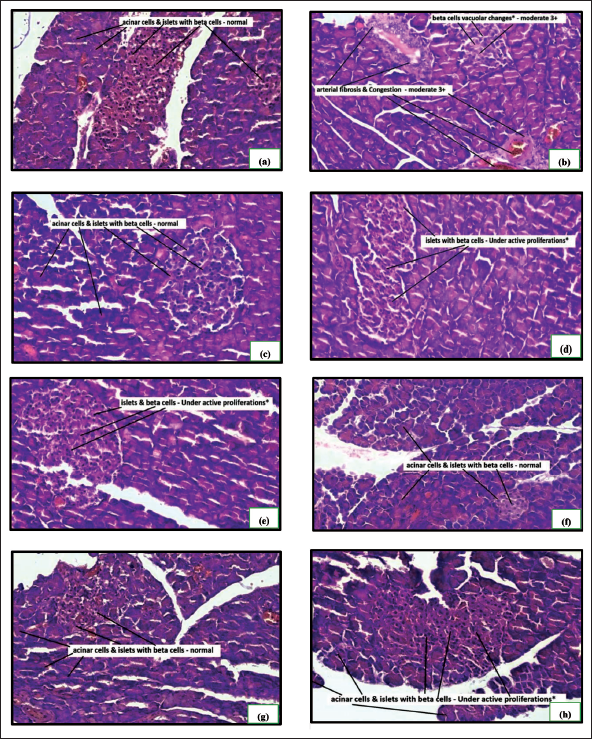
The alloxan monohydrate-induced hyperglycemia was significantly attenuated by all the treatments, as shown in Figure 1. The combination of a high dose of hydroalcoholic seed extract of A. catechu and gliclazide was more effective than individual treatments. The combination of a low dose (250 mg/kg) of hydroalcoholic seed extract of A. catechu (AN) and gliclazide, mid-dose (500 mg/kg) of AN and gliclazide produced a significant reduction in blood glucose level. In OGTT, all the treatments showed a significant decrease in blood glucose levels compared to diabetic control. The combination of a high dose of AN with gliclazide was as effective as that of gliclazide in reducing the blood glucose level. The maximum effect was seen with the combination of a high dose of AN with gliclazide. The histopathological studies on the liver (Figure 4) revealed inflammation with biliary hyperplasia in diabetic animals. The liver sections of all the other groups showed normal hepatocytes, normal portal vein with bile duct and normal central vein. The kidney section (Figure 5) showed arterial fibrosis and tubular necrosis with a collection of inflammatory PMN cells in the diabetic untreated group. The other groups showed normal glomeruli with normal tubules and vessels. The histopathological studies on the pancreas (Figure 6) showed arterial fibrosis and congestion with vacuolar changes in the diabetic control group. Individual treatment with a high dose of AN and a combination of a high dose of AN with gliclazide showed normal architecture of acinar cells and islets of Langerhans which were under active proliferation, whereas the other groups showed normal architecture of acinar cells and islets of Langerhans. Figure 2 showed a decrease in body weight in the alloxan-induced diabetic control group when compared to the normal control group. There was a non-significant increase in the body weights of rats of all other groups.
Histopathology of Liver.
Histopathology of Kidney.
Discussion
The present study was undertaken to evaluate the influence of the hydroalcoholic seed extract of A. catechu on the pharmacodynamics of gliclazide in Wistar rats in order to evaluate the safety of the combination concerning blood glucose. In the present study, Alloxan-induced diabetic rats showed a significant rise in serum glucose level when compared to the normal control rats. This happens as a result of a partial insulin deficiency that causes derangement in the metabolism of carbohydrates. 15 The metabolism and absorption of dietary carbohydrates are mainly carried out by the intestinal glycosidase enzymes. 4 Treatment with hydroalcoholic seed extract of areca nut alone and along with gliclazide significantly decreased the serum glucose level by effectively overcoming the beta cell degradation induced by alloxan. The histopathological studies on the pancreas revealed, that the diabetic rats administered with hydroalcoholic seed extract of A. catechu showed normal acinar cells along with normal beta cells of the islets which were under active proliferation justifying the statement of earlier researchers.8, 16–30 Previous studies conducted by researchers proved the potency of areca nut in inhibiting the action of α-glucosidase which is responsible for the absorption of carbohydrates and hence, decreased the blood glucose level. The results observed suggest that the combination of a high dose of AN (1000 mg/kg) with gliclazide (25 mg/kg) showed a maximum hypoglycemic effect and the effect produced was similar to the effect produced by gliclazide alone.8, 9, 29
The hepatic morphology was normal with normal hepatocytes, normal central vein, and normal portal vein with bile duct in the normal control group. In the diabetic control group, it showed distension of sinusoidal spaces and inflammation with biliary hyperplasia. Reactive oxygen species (ROS) are more concentrated in the tissue, and their antioxidant defenses are significantly weakened as a result. 16 Normal rats treated with high doses (1000 mg/kg) of hydroalcoholic seed extract of areca nut showed mild fatty vacuolations. In diabetic rats treated with high dose (1000 mg/kg) of hydroalcoholic seed extract of areca nut and in gliclazide (25 mg/kg) treated diabetic rats, rats treated with low dose of hydroalcoholic seed extract of areca nut (250 mg/kg), mid-dose (500 mg/kg) of hydroalcoholic seed extract of areca nut, and high dose (1000 mg/kg) of hydroalcoholic seed extract of areca nut with gliclazide (25 mg/kg) showed normal hepatocytes with normal central vein with normal portal vein and normal bile duct. The potency of the treatment might be due to the presence of condensed tannins and procyanidins, which are reported to possess free radical scavenging activity, anti-inflammatory activity, anti-hepatotoxin as well as inhibitors of enzymes like phospholipase A2, cyclooxygenase and lipoxygenase and hence, it provides effective anti-oxidant activity, and potentially confer protection against the oxidative stress-induced liver injury. 17
The histopathological observation of the kidneys of normal control animals showed normal glomeruli and normal tubules. The diabetic control group showed glomerulonephritis with arterial fibrosis, tubular necrosis, and fibrosis with collection of inflammatory polymorphonuclear neutrophils (PMNs) cells. This may result from impaired PMN phagocytosis, adhesion, and chemotaxis caused by altered phagocyte function. This could destroy islet cells by causing the release of cytokines, ROS, extracellular traps, and degranulation products during neutrophil maturation or activation, which further increases the systemic inflammatory load. Through their interactions with other immune and non-immune cells, these further trigger the diabetogenic T-cell response and encourage the development of type I diabetes. Because of this, the glycemic control deteriorated.18, 19 The remaining groups showed normal glomeruli, normal tubules and vessels which were evident except for rats treated with a mid-dose of hydroalcoholic seed extract of areca nut along with gliclazide (25 mg/kg), which showed normal glomeruli with mild tubular swelling with inflammations. This might be due to the presence of polyphenols which are responsible for protection against mitochondrial damage.20–22 The ability of polyphenols to mitigate damage and reduce oxidative stress may be the cause of their protective nature. There are three likely mechanisms: scavenging ROS, upregulating antioxidative mechanisms, and inhibiting the production of ROS by chelating metal ions or suppressing enzymatic activity that can produce free radicals. Research has demonstrated that lipoxygenase, cyclooxygenase, NADH oxidase, and other proteins involved in ROS production can be inhibited by specific polyphenols. 23
The histopathological observation of the pancreas of the normal control group showed normal acinar cells and normal beta cells of islets, whereas the diabetic control group showed arterial fibrosis and congestion of beta cells of islets with vacuolar changes, which is a degenerative response. Normal rats treated with high dose of hydroalcoholic extract of areca nut, low dose (250 mg/kg) of hydroalcoholic seed extract of areca nut with gliclazide (25 mg/kg) treated in diabetic rats and rats treated with mid-dose (500 mg/kg) of hydroalcoholic seed extract of areca nut along with gliclazide (25 mg/kg) showed normal acinar cells and normal beta cells of islets. Diabetic rats treated with gliclazide (25 mg/kg); diabetic rats treated with a high dose of hydroalcoholic seed extract of areca nut (1000 mg/kg) alone and when given along with gliclazide (25 mg/kg) showed normal acinar cells along with normal beta cells of the islets which were under active proliferation. This may be attributed to the potency of the polyphenols, which can stimulate insulin secretion in the pancreas, attenuate oxidative stress, activate insulin signaling, and encourage the proliferation of β cells. They can also act in an insulin-independent manner by inhibiting the absorption of glucose. 24 The potency of polyphenols mentioned above may be related to their ability to increase the activity of antioxidant enzymes such as glutathione-S-transferase. 25 Additionally, by inhibiting caspase-3, polyphenols may inhibit beta cell apoptosis and reverse beta cell degeneration in diabetic rats. The level of plasma insulin rises as a result. 24
In alloxan-induced diabetes, where beta cells are destroyed, impaired glucose tolerance results from a lack of insulin. The present study indicated that hydroalcoholic seed extract of A. catechu when given alone and when given along with gliclazide improved the glucose tolerance. In comparison to glibenclamide, areca nut extract at doses of 250 mg/kg and 500 mg/kg improved glucose tolerance the most at 60 minutes, and the decline peaked at 120 minutes, as observed in a study by Anthikat et al. (2014), which supports our findings. 26
In diabetic rats, muscle atrophy and weight loss are caused by the excess protein being catabolized to produce amino acids for gluconeogenesis during insulin deficit. 27 When compared to the normal control rats, the diabetic rats had a significant decrease in body weight, which could have been caused by insufficient glycemic control. The body weight of the rats in all the other groups that received gliclazide (25 mg/kg) and AN at different doses increased significantly. This could be because gliclazide and AN prevent muscle atrophy by reversing gluconeogenesis. It could also be because they improve insulin secretion and glycemic control. Drug toxicity in diabetic rats is directly reflected by a histopathological examination. 28
Conclusion
Studies revealed that the hydroalcoholic seed extract of A. catechu augments the hypoglycemic action of gliclazide. A high dose of hydroalcoholic seed extract of A. catechu when given as an individual treatment and in combination with gliclazide showed maximum hypoglycemic activity when compared to other groups.
Future Perspective
The information obtained from the present study might help carry out a pharmacokinetic study of areca nut alone and in combination with gliclazide, which would further help in determining the concentration-response relationship. It is possible to determine the molecular pathway by which arecanut and Gliclazide interact. In vivo studies can be carried out as well.
Footnotes
Acknowledgments
The authors are thankful to Mrs. Viba L, Drugs Inspector, Drugs Control Department, who helped us procure a gift sample of Gliclazide from Bal Pharma, Bengaluru, Karnataka, India.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia [Project No. GRANT6088].
