Abstract
Objective:
To analyze the cost-effectiveness and side effects of Heparin and Enoxaparin anti-coagulants in patients with COVID-19-associated coagulopathy (CAC).
Materials and Methods:
This was an observational study with a retrospective design of CAC patients from May 2020 to May 2022 in Surabaya, Indonesia, who received Heparin and Enoxaparin. Effectiveness was comprehensively evaluated as clinical outcomes including mortality, length of stay (LOS), laboratory results, and side effects. Pharmacoeconomic evaluation was analyzed by constructing Decision Tree modeling followed by Cost-Effectiveness Analysis (CEA) considering Quality-Adjusted Life Year (QALY), and Incremental Cost-Effectiveness Ratio (ICER). In addition, probability sensitivity analysis was performed to consider the cost-effective intervention.
Results:
A number of 274 samples were included from Medical Records and finance reports. Heparin has a higher mortality rate compared to Enoxaparin (13.9% vs. 23.9; p = 0.040), with no difference in the side effects (p = 0.056). D-Dimer reported a significant change in values after receiving Heparin (2271.01 ± 4595.50 ng/mL) and Enoxaparin (2140.95 ± 5681.98 ng/mL), p = 0.019. Enoxaparin was more cost-effective in pharmacoeconomic analysis, with a US$130.58/QALY ACER value, while Heparin was US$138.67/QALY.
Conclusion:
Enoxaparin therapy has better effectiveness, while side effects and costs are similar to Heparin. However, Enoxaparin is far more cost-effective to use against CAC conditions than Heparin.
Introduction
The most transmitted disease of COVID-19 represents dynamic changes in pathophysiology and post-COVID consequences. Since 2020, various studies and cases have started reporting a high incidence of hypercoagulability, called COVID-19-associated coagulopathy (CAC). The condition is a significant contributor to the mortality rate. The use of anti-coagulant then becomes the leading choice. Heparin and Enoxaparin have been reported by many studies as having the ability to control CAC conditions. However, the outcome reports on anti-coagulant mortality for CAC cases and the cost-effectiveness, especially in low-middle-income countries, still require deepening.
According to the epidemiological data released by the World Health Organization (WHO), on 9 September, 2021, there were 221,648,869 confirmed COVID-19 patients with 4,582,338 deaths. Meanwhile, in Indonesia, there were 137,782 deaths from 4,147,365 patients confirmed with COVID-19. 1 With a rating of 3.3%, Indonesia ranked third in the country with the highest Case Fatality Ratio in the world after Mexico and Myanmar. 2
The clinical signs and symptoms of SARS-CoV-2 infection range from asymptomatic to symptomatic, with pneumonia and severe pneumonia accounting for 5% of symptomatic COVID-19 cases, in addition to ARDS, sepsis, and septic shock. 3 This virus also causes a variety of clinical disorders, one of which is CAC. 4
The rise in fatality rates following ARDS is attributed to hypercoagulability, which leads to microvascular thrombosis in COVID-19 patients. 5 Clinical manifestations of coagulation brought on SARS-CoV-2 virus include Disseminated Intravascular Coagulation (DIC), Venous Thromboembolism (VTE), Myocardial Infarction (MI), and Pulmonary Embolism (PE). 6 According to the IDENTIA registry (a multicenter prospective observational study conducted in Indonesia between January 2016 and November 2017), the incidence of Deep Venous Thrombosis (DVT) was 37.1% and 40.3%, respectively. Given that VTE incidence in Asian populations is rising, particularly in those being treated for serious illnesses, this high finding raises a fresh concern about thrombosis in Indonesia. 7
The International Society on Thrombosis and Hemostasis (ISTH), the American Society of Hematology (ASH), the American College of Cardiology (ACC), and the Ministry of Health of 5 Professional Specialist Doctor Organizations eventually issued guidelines for the management of coagulopathy due to COVID-19. 4 Low Molecular Weight Heparin (LMWH) was then chosen as the therapeutic standard, considering the antithrombotic and anti-inflammatory functions expected to be effective for people with COVID-19. 8 In practice, Heparin and its derivative, Enoxaparin, are the two anti-coagulants that can be given to COVID-19 patients. Both the pharmacokinetics and the pharmacodynamics of the two are different. Additionally, both can treat coagulopathy in COVID-19 patients. 9
However, there are significant inconsistencies in risk-benefit, making it difficult to determine whether using anti-coagulants, which are often not expensive, will be sufficient to lower the COVID-19 death rate. 10 The side effects of Heparin and Enoxaparin are different and varied. 9 Furthermore, the COVID-19 pandemic is also a worldwide emergency that impacts all aspects, including economy. 11 According to the ICENIS (The 5th International Conference on Energy, Environmental and Information System) 2020 study, COVID-19 is expensive and may put a strain on the Indonesian government’s finance. Indonesia Case Based Groups or INACBGs noted that the average cost needed to treat COVID-19 patients who were treated in the ICU and non-ICU for 14 days ranged from US$8,666 to 8,886, while for patients treated in non-ICU for seven days and in the ICU for eight days, it costs US$5,000–5,100. 12 Developing a pharmacoeconomic analysis is one method for analyzing the cost-effectiveness of the treatments. Given the need for cost-effective comparisons between these two anti-coagulants and the inclusion of the most important clinical criteria to ensure modeling best practices are followed, this research is anticipated to be useful in policy-making.
Based on these deliberations, this study was conducted to evaluate anti-coagulants based on the length of treatment of COVID-19 patients suffering from coagulopathy, to evaluate the side effects that sufferers are likely to get, as well as to evaluate the cost analysis impact framework in the health system in Indonesia. By providing results based on cost-effectiveness, this information can be useful for the health system and the burden of CAC disease due to providing information on the right drug choices and decision-makers in the future. To deploy healthcare resources most effectively, decision-makers will need to calculate the worth of novel therapies. Recent conversations in Indonesia on the value of COVID-19 therapies use cost-effectiveness analyses (CEAs) to determine the relative value and how to best maximize societal welfare through evidence-based price decisions that balance maximizing access and encouraging future innovation. This study compares anti-coagulants Heparin and Enoxaparin for CAC patients regarding effectiveness, side effects, and cost.
Materials and Methods
This is an analytic observational study with a retrospective design with an ethical approval number: 0945/LOE/301.4.2/VI/2022 by using secondary data on COVID-19 patients at Dr. Soetomo Hospital, Surabaya, East Java, Indonesia. This hospital is a referral center for COVID-19 patients from the East Java region, Indonesia, with 1,444 beds. This hospital treats COVID-19 patients in two types of treatment rooms: the Isolation Room (RIK) and the Intensive Care Unit (ICU) for intensive care patients. Heparin and Enoxaparin anti-coagulants will be compared based on their effectiveness, side effects in administration, and the resulting cost impact.
Data Sources
The research samples were collected from COVID-19 patients who were treated from May 2020 to May 2022. Samples must meet the following inclusion criteria: Adult patients ≥18 years with CAC diagnosis that receiving Heparin and Enoxaparin therapy, and must have complete required secondary data (Figure 1). We present the available demographic data (Table 1), with clinical data presented in the Medical Record file as an ICD-10 diagnosis and a series of examinations. Patients who undergone oral anti-coagulant therapy, who had blood disorders, who applied switch therapy to other anti-coagulants, and who were pregnant were excluded based on a manual search in the database system using the ICD-10 code, namely with B34.2 for the diagnosis code of COVID-19 with the keywords “Coronavirus infection, unspecified,” D68.8 for the diagnosis code for “other specified coagulation defects,” and D68.9 for the diagnosis “Coagulation defect, unspecified.”
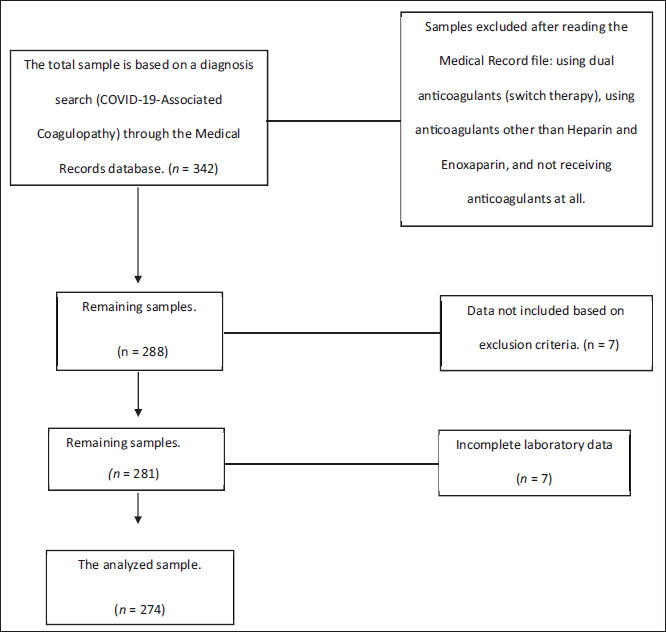
Research Sample Flowchart.
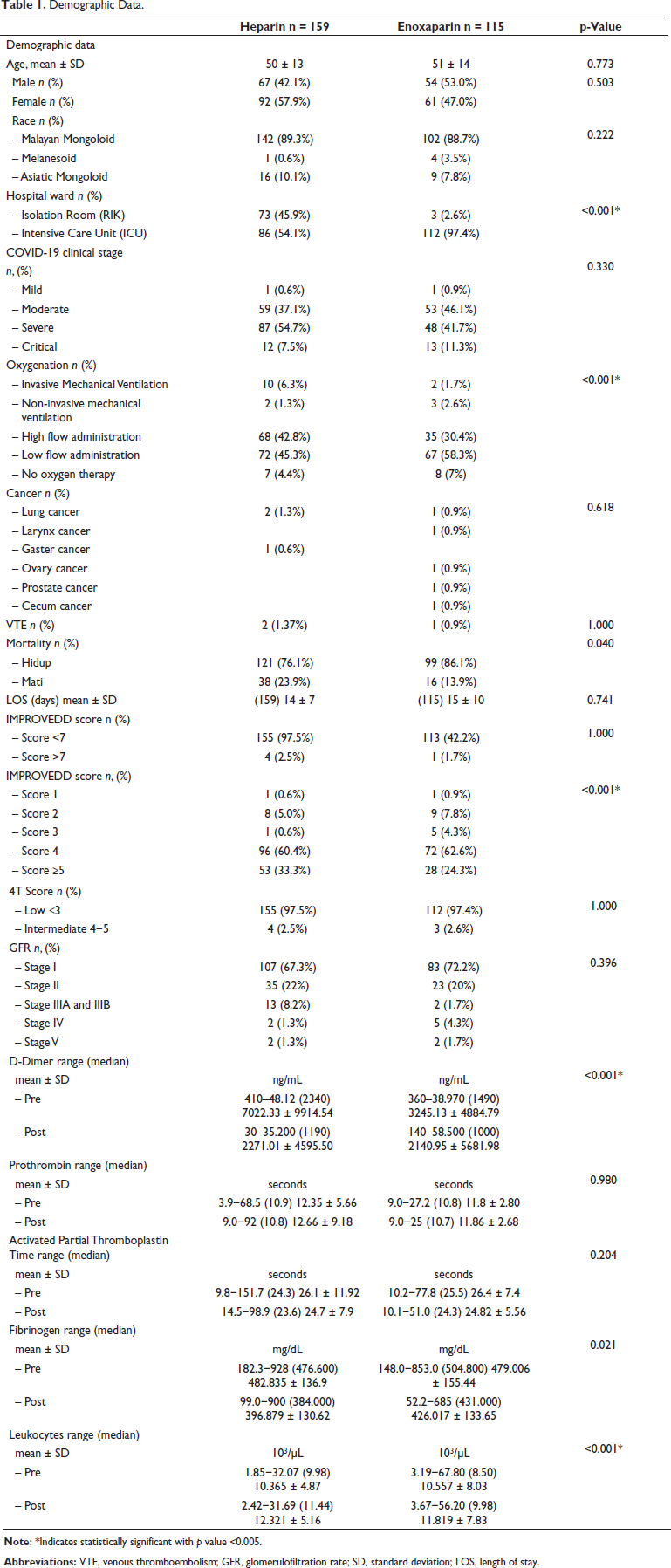
Demographic Data.
Data Collection
To identify the differences in COVID-19 patients who had been given Heparin and Enoxaparin therapy, statistical tests were carried out to assess comparative outcomes: (a) mortality status, (b) length of stay (LOS), and (c) a comparison of D-Dimer, PT, aPTT, and Fibrinogen markers. These examinations are a blood coagulation marker whose value and interpretation are sufficient to pay attention to, in the course of COVID-19 disease related to coagulopathic conditions. All the results were submitted to the normality test, and for better presentation, those were described in mean (amplitude). In addition, side effects and complications after using anti-coagulants were collected with the help of ICD-10 codes, such as cases of bleeding and the calculation of the IMPROVEDD score as a calculation of the VTE score. The total average cost is calculated from drug use (drug costs), doctor visit fees, nurse service fees, laboratory costs, and hospitalization costs (medical costs), as seen in Supplementary Table S4. Data were gathered to analyze the cost-effectiveness: service costs, treatment costs, and total costs. All of the resources are collected through Data Collection Sheets and processed using Microsoft Excel applications, SPSS Statistics, and TreeAge Pro HealthCare 2022.
Statistical Analysis
In this study, the outcomes of mortality, D-Dimer, PT – aPTT, and Fibrinogen levels in the COVID-19 patient group who received Heparin compared to those who received Enoxaparin were tested with Mann–Whitney U test while assessing LOS, and side effects will be carried out by the chi-square test. For the pharmacoeconomic analysis, this study will use CEA with the help of Decision Tree modeling in comparing two types of treatment options with different outcomes and calculating the value of Incremental Cost-Effectiveness Ratio (ICER). The CEA was carried out in several steps, firstly comparing the average total cost with effectiveness. Effectiveness in this study was calculated in units of Quality-Adjusted Life Year (QALY). QALY is a calculation to improve the quality and quantity of patient life with healthcare intervention. QALY is the arithmetic result of life expectancy and remaining quality of life measurement. If in good health, it will be worth 1 score; while dead, it will be worth 0. QALY is calculated based on the multiplication of Life Years Gained (LYG), in which the data are obtained from the average age calculation of Indonesian people based on the United Nations Department of Economic and Social Affairs, Population Division, 2017. In this study of supporting parameters, this number was obtained from the Utility Value (UV) taken based on the results from another study’s supporting parameters, which calculated UV or the value of the quality of life for COVID-19 sufferers based on the conditions at which they were treated. 19 If the UV value is between 1 and 0, this decision analysis is expected to help determine the best therapeutic choices.
The ICER will be known by subtracting the average total cost of Enoxaparin and the average full cost of Heparin and then divided by the effectiveness (QALY) of Enoxaparin minus the QALY of Heparin. A probability sensitivity analysis was then performed to show that the ICER value must be below the Willingness To Pay (WTP) value for cost-effectiveness. WTP is a person’s ability to pay for goods or services. In Indonesia, the term WTP is known as GDP (Gross Domestic Product) or what can be called the GDP per capita. If 1x – 3x GDP is an ICER, then the intervention can be determined to be cost-effective. GDP per capita in 2021 is US$4,322.70. Henceforth, conclusions can be drawn to determine effective therapeutic options for people with CAC.
Results
The 342 patients were then processed manually by opening the Medical Record file for each patient. In this study, a total of 52 patients were not included in the sample because they had received anti-coagulants before being referred to the hospital, were using dual anti-coagulants, did not receive anticoagulant therapy, and used DOAC and anti-coagulants other than Heparin and Enoxaparin. In the further selection stage, by reading the Medical Record files of the remaining 288 samples, it was found that seven samples met the exclusion criteria, and seven other samples did not have complete laboratory data. Thus, 274 samples were obtained to be analyzed and accompanied by the Finance Department of Dr. Soetomo Hospital, Surabaya (Figure 2).
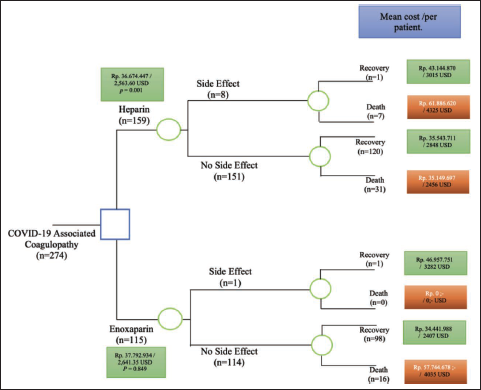
Model Structure of Decision Tree. The Comparison Between Heparin and Enoxaparin Therapy.
Mortality Ratio of Heparin and Enoxaparin Therapy
Based on the distribution, it was found that mortality in Heparin was 23.9%, while that in Enoxaparin was 13.9% (p = 0.040). The companion data in Supplementary Table S1 also show the mortality percentage based on the dose of each therapeutic agent.
Length of Stay Ratio of Heparin and Enoxaparin Therapy
Patients with Heparin and Enoxaparin have almost the same average LOS, between 14 and 15 days (p = 0.741). Although patients taking Enoxaparin therapy had a more extended hospital stay of 90 days when compared to those taking Heparin, the most prolonged treatment period was 41 days.
Laboratory Evaluation of Heparin and Enoxaparin Therapy
Based on the laboratory values of the research samples, it was found that there were statistically significant differences in the comparison of D-Dimer levels (p = 0.019). The mean value of D-Dimer before and after receiving Heparin therapy was 7022.33 to 2271.01 µg/mL, and Enoxaparin 3245.13 to 2140.95 µg/mL (p = 0.019).
Side Effect Ratio of Heparin and Enoxaparin Therapy
During administration, nine samples received side effects from using anti-coagulants. These patients suffered side effects of bleeding, such as epistaxis, melena, and hematochezia, which accompany hematoma, VTE, and stroke (3.3%) (p = 0.056). The presentation data of side effects are available if reviewed based on the dosage of Heparin and Enoxaparin used as shown in Supplementary Table S2.
Cost Analysis of Heparin and Enoxaparin Therapy
The cost data in Rupiah are also converted into US Dollar exchange. It uses an exchange rate based on nominal reimbursement in 2021 from The World Bank of ₹. 14,308.14 per 1 US$ are listed in Supplementary Table S3. In the results of the Decision Tree, it can be seen that of the 274 research samples with CAC condition, patients who received Heparin therapy tended to have more side effect complications when compared to Enoxaparin recipients. The total cost of treatment and medical expenses that must be paid by a COVID-19 patient who enters a CAC using Heparin is US$2563.60 (p = 0.001). Meanwhile, the total cost for patients who receive Enoxaparin is US$2641.35 (p = 0.849).
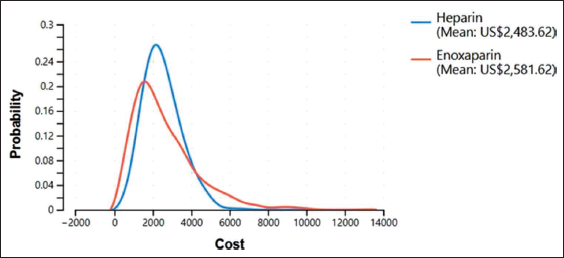
It was found that the QALY of therapy from the use of the drug Heparin was 17.91 QALY, while the effectiveness of Enoxaparin was 19.77 QALY (Figure 3). From the calculation, the probabilistic average total cost of CAC therapy in patients using the anticoagulant Heparin is US$2,483.62, and the average total cost of CAC therapy in those using the anticoagulant Enoxaparin is US$2,581.62 (Figure 4).
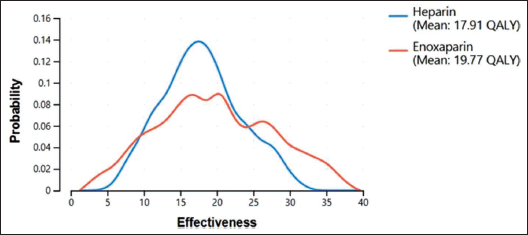
Average Total Cost of Heparin and Enoxaparin Based on a Probability Curve.
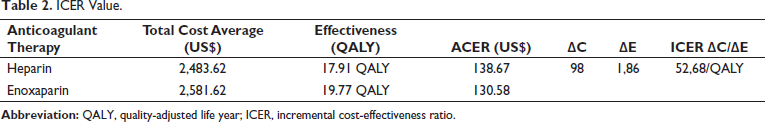
It can be seen in Table 2 that the ICER value of Heparin therapy against Enoxaparin is US$52.68 per QALY.
ICER Value.
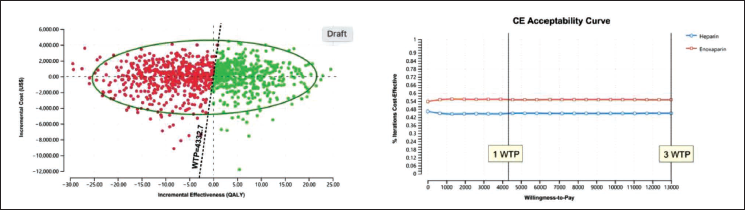
After carrying out all the steps above, a probability sensitivity analysis was carried out to show that the ICER value must be at least below the WTP value. Based on the Scatter Plot diagram, Enoxaparin recipients are on the acceptable line below the WTP, which is cost-effective. Then, the probability of acceptance of various WTP numbers was also analyzed. The black line is the Indonesian people’s WTP value (US$4,322.70). The acceptability value of Enoxaparin users is at a value of 55%, so Enoxaparin is a practical choice. Results will be cost-effective if one to three times the GDP is ICER (Figure 5B). Then, a straight line can be drawn where three times the WTP is US$12,998.10 as the limit of the government’s ability to finance the CAC case. This can also refer to the INACBGs data where the Indonesian government’s capacity to cover COVID-19 costs is US$5,755.28 (for class D JKN/BPJS; national health insurance) to US$9,905.93 (for class A JKN/BPJS).
Discussion
In addition to the clinical signs and symptoms, COVID-19 is also associated with abnormalities in the kidneys, heart, mediastinal lymphadenopathy, nervous system, and blood or hematological systems. Numerous pathological investigations have demonstrated that the abnormalities caused by COVID-19 are more likely to lead to hypersensitivity pneumonitis than viral pneumonia. The SARS-CoV-2 virus raises cytokine levels in the blood circulation and triggers a cytokine storm, an overactive immune response potentially followed by multiple organ systemic symptoms. When COVID-19 patients have pneumonia, it reduces oxygen then leads to multi-organ dysfunction due to the overproduction of pro-inflammatory cytokines accompanied by reduced oxygen capacity in the patient’s blood. It was also reported that one of the symptoms that COVID-19 patients may encounter is septic shock, a condition where it is challenging to correct metabolic acidosis and coagulation dysfunction. Due to many symptoms produced by COVID-19 patients, multi-therapy must also be applied. It starts with giving antivirals, anti-influenza, antibiotics, and vitamins to anti-coagulants. 13
Anti-Coagulants are given as one of the therapies for COVID-19 caused by an underlying condition, CAC. This condition is a life-threatening complication. Many factors initiate the formation of coagulation. The underlying pathological mechanisms include vascular endothelial dysfunction, hyperinflammatory immune response, and hypercoagulability. This condition can cause micro- and macrothrombi, eventually damaging essential organs such as the lungs, heart, brain, and kidneys. Compared to other viral infections, COVID-19 has a frequency and severity of blood clots associated with increased D-Dimer, C-reactive Protein, P-selectin, and Fibrinogen. 14 In a study conducted by Conway et al., CAC was said to be most likely caused by a dysregulation that is not yet understood. They started with the attachment of SARS-CoV-2 and ACE2, which can reduce ACE2 expression. Such adhesions can indirectly damage the alveolar epithelium, which triggers a hyperactive innate immune response. Epithelial damage can also trigger an increase in Bradykinin/Angiotensin II. ACE2 is a regulatory tool for the renin-angiotensin-aldosterone and Kallikrein-Kinin systems. Kinin is a protein that acts as a promoter of vascular smooth muscle relaxation and increases vascular permeability. Dysregulation of degradation can increase the activity of kinin, which results in angioedema. This condition triggers increased vasodilation, which results in leakage of blood vessels. Simultaneously, the imbalanced fibrinolysis associated with vascular endothelial damage and glycocalyx leads to coagulopathy and platelet hyperactivation. The thrombotic formation then results in multi-organ damage and failure.
Conditions of platelet dysfunction also contribute to the formation of an immunothrombotic contribution. This condition in COVID-19 triggers the appearance of abnormalities in coagulation and complement factors, as well as abnormalities in endothelial cells and neutrophils. Hypercoagulability conditions can change the procoagulant plasma conditions and fibrinolytic activity, especially in COVID-19 patients who are entering a state of sepsis (critical condition). This critical condition triggers an increase in plasma levels and increases endothelial cell activation. 14 Management guidelines issued by the ISTH make coagulation markers (D-Dimer, PT, Platelet, and Fibrinogen) an initial consideration for the management of CAK. It is said that D-Dimer is associated with increased mortality in COVID-19. Studies also state that multi-organ failure and sepsis can trigger CAK conditions. 15 The D-Dimer level measures blood clot formation and lysis resulting from cross-linked fibrin degradation. D-Dimer is often used to indicate coagulation disorders, including DIC. D-Dimer is produced only from cross-linked fibrin. The presence of D-Dimer indicates that clot formation (thrombosis) and subsequent fibrinolysis have occurred. 16
One study stated that D-Dimer indicates the severity and poor outcome of COVID-19 sufferers. D-Dimer levels are reported to be potent coagulation activating agents, so D-Dimer levels are one of the markers of the most attention in the case of COVID-19, especially those who enter the CAC condition. 10 Heparin does not necessarily reduce D-Dimer levels directly, but D-Dimer levels can decrease due to the prevention of thrombus formation initiated by anti-coagulants, such as Heparin and Enoxaparin. In a study conducted by Billett et al. (2020), it was proven that anti-coagulants could reduce D-Dimer levels in people with COVID-19 (p = 0.017), with other alleged benefits that anti-coagulants have in people with COVID-19, which are assumed, one of them is as an anti-inflammatory. In this study, it can be proven by the decrease in D-Dimer values just before and after administration of anti-coagulants, with both Heparin (mean D-Dimer; pre: 7022.33 µg/mL, post: 2771.01 µg/mL) and Enoxaparin (mean D-Dimer; pre: 3245.13 µg/mL, post: 2140.95 µg/mL), p = 0.019.
Meanwhile, based on pharmacodynamics, Enoxaparin is said to be capable of inhibiting factor Xa to become factor IIa. If viewed based on the effect on aPTT values, this study shows that both Enoxaparin (mean aPTT; pre: 26.15 sec, post: 24.7 sec) and Heparin (mean aPTT; pre: 26.4 sec, post: 24.8 sec) were also able to reduce the value of aPTT time.
A meta-analysis shows that high fibrinogen levels also trigger poor outcomes for COVID-19 sufferers. Fibrin is formed when intravascular clotting occurs. 17 Based on the FDA, Heparin works and can inhibit the conversion of Fibrinogen to Fibrin. 18 This study shows that Fibrinogen was reported to experience a significant decrease when assessed before and after receiving Heparin (mean Fibrinogen; pre: 482.83 mg/dL, post: 396.87 mg/dL) and Enoxaparin agents (mean Fibrinogen; pre: 479 mg/dL, post: 426.01 mg/dL), p = 0.021.
This study focuses on comparing effectiveness, side effects, and cost. Effectiveness is assessed based on the mortality outcomes of Heparin and Enoxaparin anticoagulant users. The conclusion that can be drawn from this study is that the chance of mortality with Heparin tends to be higher than that with Enoxaparin. This is evidenced by many Heparin users who died while being treated in the particular infection isolation treatment room and the ICU. Another factor contributing to this study’s high mortality rate was the admitted patients’ poor condition. A total of 73.9% (p = 0.164) of patients who received Heparin therapy moved from isolation rooms due to worsening conditions. Based on the modality of oxygen support, a total of 6.3% (p = 0.009) of Heparin sufferers eventually had to use Invasive Mechanical Ventilation. A total of 64.4% of Heparin users are patients suffering from moderate to severe COVID-19 degree. This condition is worse than Enoxaparin users (35.6%). The results of a study conducted by Pawlowski et al. in the United States in 2021 suggested that the shortest LOS was found in patients receiving Enoxaparin therapy (5.7 days), followed by Heparin (6.5 days). However, this study found no significant difference in LOS between Heparin and Enoxaparin, with an average of 14−15 days. However, there is a considerable distance between the LOS, which is 90 days for Enoxaparin, compared to Heparin, which has the most extended treatment period of 41 days.
In addition, by calculating the IMPROVEDD scoring results, Heparin sufferers (60.4%) tend to be at score 4 with a 1.6% chance of VTE occurring on days 35−42 and 1.9% on day 77 of the treatment period.
A pharmacoeconomic analysis is then being proceeded. Service fees include administrative costs, hospitalization costs, doctor and nurse service fees, laboratory fees, and radiology fees. At the same time, treatment costs include drug prices and accompanied medical equipments (such as abbocath and injections). These costs are arranged in Supplementary Table S4 for data in the Rupiah and Dollar exchange rate.
The CEA was carried out. Based on the first-stage analysis results, the average cost of CAC therapy in Enoxaparin patients was higher. It shows that based on the method of use or application to patients, which tends to be more accessible, the side effects tend to be lower. 9 The average total cost in CAC patients who use Enoxaparin is higher than that in those who use Heparin due to the higher price of the drug Enoxaparin. In addition, treatment class and use of breathing apparatus are also factors that affect treatment costs.
The results of determining the alternative positions for CAC treatment based on the cost-effectiveness diagram (Figure 5A) show that the position of Enoxaparin is in column I, which means that Enoxaparin has high effectiveness and cost compared to Heparin. After knowing the position of Enoxaparin in column I, it is necessary to calculate the ICER. The ICER is used to determine the additional cost for each increase in the effectiveness of therapy. The effectiveness referred to in this study is QALY. The ICER value obtained was US$52.68 per QALY. The ICER value shows that additional costs are required if a therapy transfer is made from Heparin to Enoxaparin. If the hospital wants to increase the effectiveness of improving the condition of CAC per patient using Enoxaparin, then the hospital must pay a fee of US$52.68. However, when deciding to choose anti-coagulants, it can be returned to the hospital’s policy, which is adjusted to the hospital’s budget.
Conclusion
Heparin therapy has a higher mortality risk for CAC when compared to Enoxaparin. The fact that taking Heparin had more adverse effects than using Enoxaparin further supports this. Therefore, the modality of Enoxaparin can be an option for CAC therapy. These two therapies can also reduce D-Dimer, PT, aPTT, and Fibrinogen levels. Based on the pharmacoeconomic analysis, nevertheless, Enoxaparin is more expensive and is considered more cost-effective when compared to Heparin.
Limitations
In this study, sample selection was carried out using a retrospective method. Unmeasured confounding variables, such as socioeconomic characteristics, that are not included in this dataset may have an impact on the study’s findings. Additionally, if the anticoagulant drugs for some of these patients were put improperly into the Medical Records, there may be a misclassification bias. The prescription codes used to identify the cohorts of patients who received Enoxaparin and unfractionated Heparin during the duration of the trial have stayed consistent; therefore, bias resulting from evolving eligibility requirements is unlikely given the study’s brief time frame.
Footnotes
Abbreviations
CAC: COVID-19-associated coagulopathy; QALY: Quality-adjusted life year; ICER: Incremental cost-effectiveness ratio; PT: Prothrombin time; aPTT: Activated partial thromboplastin time.
Acknowledgments
All authors thank the Faculty of Medicine, Master Program of Basic Medical Science, Laboratorium of Pharmacology Department, Airlangga University, for supporting the research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from the Institutional Ethical Committee of Dr. Soetomo Hospital (0945/LOE/301.4.2/VI/2022), Surabaya, East Java, Indonesia.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
