Abstract
Dengue is a viral infection caused by one of four serotypes of dengue virus (DENV), which is responsible for causing potentially life-threatening disease and substantial public health as well as economic burden worldwide. The non-structural and structural proteins of DENV are essential to its viral activity and pathogenesis. Unfortunately, despite numerous efforts, there is still no cure for dengue. Most of the available therapeutic options only provide symptomatic treatment. Since there is a lack of cure and dengue-related substantial economic and public health burden, a safe and effective dengue vaccine is urgently required. Currently, there is only one approved dengue vaccine available that provides suboptimal protection; however, a range of other dengue vaccines are under development to lower the infection burden and decrease dengue morbidities. In addition, numerous dengue therapeutics are also being developed, which have great potential in dengue treatment. These therapeutics mostly act by decreasing viral replication, vascular pathologies, and/or inflammation. This review comprehensively summarizes the latest update on dengue vaccines and therapeutics. Moreover, various other important aspects of DENV, including molecular biology, pathogenesis, and control strategies, have also been discussed.
Introduction
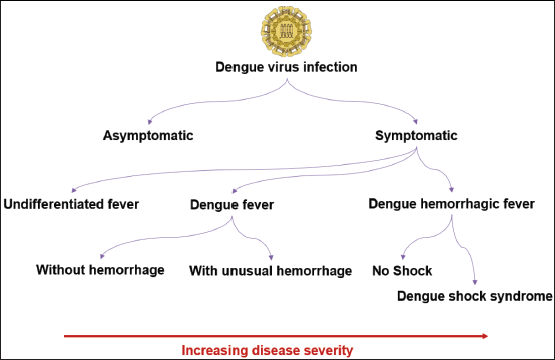
Dengue is a mosquito-borne disease that is transmitted by the bites of Aedes mosquitoes, including Aedes albopictus and Aedes aegypti. Globally, this mosquito-borne disease is regarded as the most common human arboviral infection. 1 More than 2,000 dengue-associated deaths and over 3.7 million dengue cases have already been reported from 70 countries/territories worldwide as of August 23rd, 2023. 2 It has been estimated that around 3.8 billion people in 128 countries are at greater risk of infection caused by dengue viruses (DENVs). In addition, approximately 20,000 deaths due to dengue take place every year worldwide.3, 4 Dengue infections are primarily caused by four antigenically and genetically different serotypes, including DENV 1–4, while the fifth variant, DENV-5, was identified in October 2013. 5 Typically, the DENV incubation period is 4–7 days. As per the World Health Organization (WHO) dengue guideline, the spectrum of DENV infection can vary from undifferentiated febrile illness or asymptomatic infection to more serious manifestations, including dengue shock syndrome (DSS) and dengue hemorrhagic fever (DHF) (Figure 1). DSS is considered the most severe form of clinical syndrome and involves plasma leakage, coagulation irregularities, and elevated vascular fragility. Moreover, elevated levels of capillary permeability can further result in multiple organ failure and hypovolemic shock.4, 6, 7
The Spectrum of Manifestations of Dengue Virus Infection.
The transmission and incidence of dengue are influenced by multiple factors, including a lack of effective vector control, poor waste management systems, urbanization, and uncontrolled population growth. 8 Water storage practices because of inadequate water supply can also play an important role in dengue epidemics. 8 Furthermore, social inequalities, poverty, and illiteracy are also linked with poor dengue management. 4 It has been observed that infection with one DENV serotype can provide prolonged homotypic protection and short-term heterotypic protection. Interestingly, during subsequent heterotypic infections, DENV infection induces immune responses that can either lead to protection or disease augmentation, which further complicates the development of dengue vaccines. Several other factors, including epidemiological context, host immunity and genetics, and viral characteristics, can also have significant contributions in providing protection against or enhancing dengue. 9 Therefore, vaccine developers aim to trigger simultaneous tetravalent immunity against all four types of DENV. 10 Since a higher viral burden can result in a severe form of dengue, studies are required on DENV-specific antivirals in dengue treatment. 11
Up to now, both preclinical and clinical trials have been carried out with repurposed drugs containing antiviral properties to evaluate their potential in dengue treatment, which has been extensively discussed in this review. Moreover, in this review, vaccines that are in advanced stages of clinical testing as well as several other aspects of the dengue virus, including molecular biology, pathogenesis, and control strategies, have also been discussed.
According to the World Health Organization (WHO) dengue guideline, the spectrum of DENV infection can vary from undifferentiated febrile illness or asymptomatic infection to more serious manifestations, including dengue shock syndrome (DSS) and dengue hemorrhagic fever (DHF).
Methodology
In this review, the search strategy involved the identification of all Books, Original Research Articles, Review Articles, and Clinical Trials related to DENV molecular biology as well as recent advancements in control strategies, prevention, management, and therapeutics of DENV. Therefore, various widely used databases including Web of Science, Scopus, ScienceDirect, Google Scholar, and PubMed were searched. A wide range of search terms or combinations of search terms were used while searching the databases including molecular biology of dengue virus, dengue virus life cycle, dengue virus genomic organization, dengue virus proteins, therapeutic targets of dengue virus, molecular pathogenesis of dengue, dengue prevention and control strategies, dengue vaccines, and dengue management.
Genomic Organization, Life Cycle, and Protein Processing of Dengue Virus
DENVs are positive-stranded RNA viruses (containing an 11 kb genome) of the Flaviviridae family. The DENV genome contains a large open reading frame (ORF), which is translated into a single complete polyprotein that subsequently gets processed into various other proteins, including seven non-structural (NS) proteins, an envelope protein (E), a membrane-associated protein (M), a core protein (C), or nucleocapsid, and three structural proteins. 12 These proteins play a range of functions, including several cellular functions and viral replication. DENV NS proteins, including NS4A, NS2A, and NS4B, are responsible for blocking the interferon (IFN) signaling pathways in order to escape the hsost immune responses. Moreover, NS4B also suppresses the IFN signaling pathways via blocking the phosphorylation of STAT-1. 13 DENVs enter the host by receptor-mediated-endocytosis, which subsequently results in the fusion of vesicular and viral membranes (Figure 2). This fusion allows the secretion of genomic RNA into the cytoplasm, which plays a role as mRNA for replication and translation. In the endoplasmic reticulum (ER), the viral mRNA-encoded large polyprotein is then translated in a cap-dependent manner. 14 Subsequently, this polyprotein is co- and post-translationally processed into 7 NS and 3 structural proteins. In the host ER lumen, the amino termini of NS4B, NS1, E, and prM are produced via the cleavage caused by the ER signal peptide peptidase. In the host cytoplasm, various other proteins and the C-termini of the C proteins are processed via the NS2B-NS3 proteases. It has been reported that furin (a cellular endoprotease) localized to the trans-Golgi network causes the cleavage of the precursor prM to form mature M proteins.15, 16
The Life Cycle of Dengue Virus. (Dengue virus enters the host by receptor-mediated endocytosis, which subsequently results in the fusion of vesicular and viral membranes. This fusion allows the secretion of genomic RNA into the cytoplasm, which plays a role as mRNA for replication and translation. The enveloped virions go through ER and trans-Golgi network, which eventually bud through the ER membrane into the cytoplasm in order to obtain a second ER-derived outer membrane. Finally, the ER membrane fuses with the plasma membrane, which results in the secretion of enveloped mature progeny virions into the extracellular space, where they can spread and infect adjacent cells.)
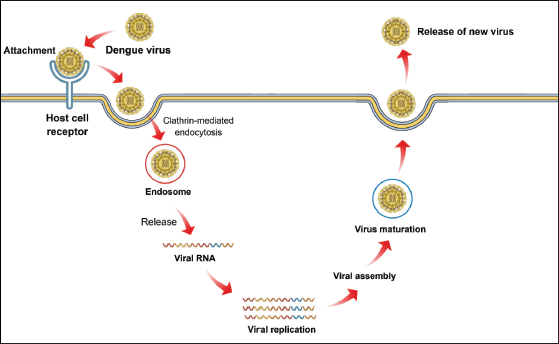
Subsequently, the enveloped virions go through ER and trans-Golgi network, which eventually bud through the ER membrane into the cytoplasm in order to obtain a second ER-derived outer membrane. Finally, the ER membrane fuses with the plasma membrane, which results in the secretion of enveloped mature progeny virions into the extracellular space, where they can spread and infect adjacent cells. 17
Dengue Virus Proteins and Their Importance as Therapeutic Targets
Non-structural (NS) Proteins
NS1 is localized to the ER and has a contribution to viral genome replication. NS1 is expressed in three forms, including a membrane-anchored form, an ER-resident form that was found to colocalize with the viral replication complex, and a secreted form (sNS1). N207 and N130 are the two sites where NS1 glycosylation takes place. It was observed that glycosylation of both of these residues is essential for neurovirulence in mouse models and for viral replication in mosquito cells. It is considered that sNS1 might have an important role in dengue pathogenesis and is an important target of humoral immunity. 18 NS3 (and its cofactor NS2B) and NS5 are the most extensively characterized viral NS proteins. NS3 contains several catalytic domains, which are essential for the cofactor NS2B. Furthermore, NS3 has both nucleoside triphosphatase and helicase properties that are essential for the synthesis of vRNA. NS3 also has a 5′ triphosphatase function, which is required in the 7-methylguanosine capping pathway. As compared to the aforementioned NS proteins, NS4A, NS2A, and NS4B are less well characterized. These small proteins are hydrophobic by nature. NS2A, NS4A, and NS4B have the ability to block IFN-induced signal transduction. These three proteins are most effective in combination; however, NS4B alone is a strong suppressor of IFN-β and IFN-γ signaling. 18 NS5 can play a role as both a methyltransferase and an RNA-dependent RNA polymerase, which is also an important enzyme in the capping pathway. Moreover, the NS5 protein was found to trigger interleukin-8 (IL-8) transcription and translation by activating the CAAT/enhancer binding protein. 18
Structural Proteins
Membrane-associated Protein (M)
M protein helps in enveloping proteins to form mature virions and functions as a part of the nucleocapsid. The precursor membrane protein prM can be utilized to study different antibody responses to different flaviviruses. The C-prM was first detected via immunoprecipitating the DENV-2-infected A. albopictus cells. In addition, these cells were cleaved to generate both non-membrane fragments (pr) and membrane-associated proteins (M). However, this cleavage was found to be less effective in mosquito cells. Proteins comprising the fragments were subsequently fused with staphylococcal protein A. It was observed that the fused proteins were stable and were utilized to produce antisera in rabbits. The interactions between the prM and envelope (E) proteins are facilitated via domains in the carboxy-terminal anchoring domain of E, whereas cell function is facilitated via the trypsin-releasable ectodomain of the E protein. 13
DENV Core Protein (C)
DENV core protein, also known as C protein, is a highly basic protein that can mediate nucleocapsid assembly via interacting with RNA. Structural analysis of the C protein revealed that a high net charge is possessed by the C protein dimer, and there is an unequal distribution of basic residues over the protein surface. Interestingly, it was observed that anti-DENV-2 core protein monoclonal antibodies (MAbs) have the capacity to react with antigens in the nucleus and cytoplasm of DENV-2 as well as DENV-4, but not DENV-1 and DENV-3. In western blot studies, these MAbs reacted with C proteins of DENV-1, DENV-2, and DENV-4. Moreover, all MAbs reacted with the region of DENV-2 C protein, as confirmed by PEPSCAN epitope mapping. 13
Envelope Protein (E)
Viral E has a significant contribution in viral attachment, which contains three domains. Domain I of the E protein is a centrally located β barrel, while domain II possesses the fusion peptide and a dimerization area. On the other hand, domain III of the E protein mediates the receptor-binding activity. E exists as homodimers in a mature virion, while the fusion peptide remains inaccessible. In the case of low-pH conditions, for example, in late endosomes, the E protein goes through irreversible trimerization in order to reveal the fusion peptide and facilitate endosomal fusion in a way in line with the class II fusion proteins of alphaviruses. The kl loop is a flexible hinge area located between domains I and II. This kl loop was found to be essential for the structural alterations that precede fusion peptide exposure. The E proteins of DENV are differentially glycosylated depending on the serotypes and cells in which the virus is propagated. E proteins also play roles in endosomal fusion and receptor binding. E proteins have already been identified as a major target of humoral immunity. 18
Molecular Pathogenesis of Dengue
The DENV serotypes (DENV-1–4) show around 65%–70% similarity in nucleotide sequence. 19 It was observed that primary DENV infection is caused by the first or initial infection with a specific serotype. Interestingly, most of the primary infections are typically asymptomatic or exhibit a mild febrile illness. However, they can result in hemorrhagic fever in some individuals, particularly in infants born to DENV-immune mothers. Following infection with a different DENV serotype is called secondary dengue infection, which might result in severe clinical manifestations, including DSS or DHF.20–22 Following infection with a certain DENV serotype, it is likely that an individual will be immune to re-infection caused by the same serotype. Nonetheless, subsequent infection with a different serotype might take place, as the heterologous immunity is not long-lived. Numerous studies revealed that the heterotypic protective immunity progressively diminishes in 1 or 2 years. 23 Several host and viral factors play roles in dengue pathogenesis, including variation of the DENV genome, subgenomic RNA, NS1 viral antigen, memory cross-reactive T cells, antibody-dependent enhancement (ADE), autoimmunity, and anti-DENV NS1 antibodies. In humans, all these factors exert synergistic effects on severe dengue manifestations.24, 25
Prevention and Control strategies
Indeed, mosquito control is widely used to control dengue cases because of the lack of effective vaccines or therapeutics. It has been observed that Geographic Information System (GIS) mapping can be effective in identifying dengue concentrations. Interestingly, through the identification of dengue seropositive cases within the study region, transmission of dengue can be averted via detecting dengue foci, which can then also be treated with different preventive approaches. In a study, Kittayapong et al. 26 revealed that GIS mapping enabled the estimation of the successful control rate in the mapped regions and also permitted improved surveillance as well as community-based intervention programs to inhibit dengue. 26 These researchers also observed that mapped dengue foci surveillance estimated bath basins and water containers as the main breeding areas of A. aegypti mosquitoes.26, 27 Typically, A. aegypti females lay eggs above the water line in jars or containers, and so on, to improve their survival rate. Indeed, it is important to estimate the behavioral pattern of vectors to detect the density of dengue vectors. In a study, Wong et al. 28 assessed the A. aegypti oviposition pattern and observed that substantial intra-species affinity might be a sign of targeting vector species. Following the selection of the oviposition sites, the implementation of techniques that eradicate the mosquito population at a later developmental stage will elevate the effectiveness of control techniques. 28 In recent times, the use of oviposition-based novel techniques has resulted in better outcomes in increasing control of vector species.27, 29
Unfortunately, the prevention of dengue is becoming more challenging because of the densely populated cities. 30 The global urban population has been growing at a rapid rate since the 1950s. In addition, it is estimated that the incessant population shift will continue to rise by 66% by 2050. Therefore, strong community-based integrated control programs are required, which should aim at teaching the community regarding the techniques for the eradication of mosquito breeding sites. In a community, individuals are categorized into multiple groups according to their level of understanding and education. It has already been demonstrated that community-based programs are effective in the eradication of dengue mosquitoes in Cuba, Mexico, and Kerala districts by increasing awareness amongst the communities. Several techniques can be included for optimum control of the vector population via community engagement; for example, the combination of chemical control and a community-based program of A. aegypti has resulted in substantial outcomes in Cuba.27, 31, 32 So far, dengue control strategies include typical chemical applications and state-of-the-art biological control strategies. 33 For many decades, the use of insecticides has been found to be hard to endure because of their limited effectiveness, high cost, and negative effects on the environment. Currently, different insecticide classes, including pyrethroids and organophosphates, are used in rotation to prevent the development of resistance in A. aegypti mosquitoes. Uncontrolled treatment can lead to resistance that arises from increased detoxifying enzyme functions or structural modification of insecticide target sites; however, the traces of chemical residues might also affect environmental and human health.
Therefore, plant-based insecticides have been developed by researchers as an alternative control method that can sustain and cause less toxicity in the environment as compared to synthetic insecticides. 34 Different parts of the plants (roots, stems, and leaves) and/or herbal extracts are used to formulate these plant-based insecticides, including Asparagus racemosus, Erythrina indica, Callistemon rigidus, and Cipadessa baccifera.35, 36 In addition to their insecticidal properties, these plant derivatives are also effective as repellents against A. aegypti.27, 35, 37 Predatory species play an important role in terms of biological control for mosquitoes. Gambusia affinis, also known as the mosquito fish, exhibited satisfactory agreement with the concurrent usage of other biological or chemical tools. Greater control of mosquitoes can also be achieved if G. affinis is utilized along with other biological control measures. Advantages of using G. affinis include being relatively long-lived, having the capacity to thrive in permanent water bodies, being effective in the transient inhibition or extermination of targeted mosquito species, and having a slower reproduction rate. In Taiwan, various other fish species were also used to control dengue, where fish species such as G. affinis, Sarotherodon niloticus, Tilapia mossambica, and Poecilia reticulata were concurrently introduced in household water containers as a control technique. This technique decreased the container index for the larvae of A. albopictus and A. aegypti. 38
As a biological intervention, there is a growing interest regarding the use of Wolbachia transinfection. It has been reported that these maternally inherited bacteria can cause infection of the testes and ovaries of numerous arthropod species and change the host reproduction rate via parthenogenesis, feminization, and cytoplasmic incompatibility. Transmission of DENV can be inhibited by disrupting the vector breeding cycle, and Wolbachia can block the viral replication in the salivary glands of mosquitoes. 39 Interestingly, a genetically modified and sterile male A. aegypti (OX513A) was used to contain a late-acting, repressible, and dominant lethal transgene insertion, which could be distinguished by the red fluorescence expression. Nevertheless, the aforesaid biological methods are still being studied in various trials to permit long-standing effectiveness. 33
Vaccines
Chimeric Yellow Fever-Dengue-Tetravalent Dengue Vaccine (CYD-TDV)
CYD-TDV is commercialized under the brand name Dengvaxia®, developed by Sanofi-Pasteur, and is the sole licensed dengue vaccine (Table 1). Currently, CYD-TDV is approved for use in 20 countries where dengue is endemic. In addition, this vaccine has also been approved by the United States (US) and European Union (EU) regulatory authorities. 40 It has been observed that CYD-TDV can decrease the overall risk of severe dengue and dengue-related hospitalizations. 41
Current Dengue Vaccine Candidates.
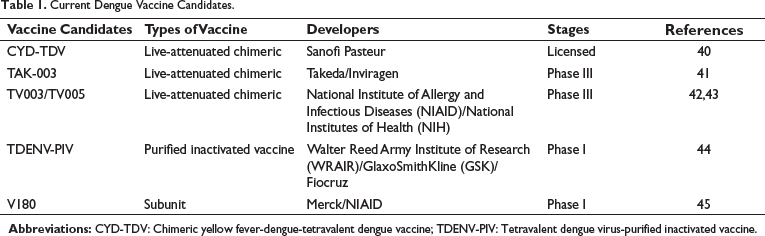
CYD-TDV possesses four recombinant viruses (CYD-1–4), each of which contains the genes encoding the dengue envelope and pre-membrane proteins of one of the four DENV serotypes, along with genes encoding the capsid and NS proteins of the attenuated yellow fever 17D vaccine virus. 48 CYD-TDV is effective in individuals with prior dengue infection, as per the approved indication of the US Food and Drug Administration (FDA). Indeed, CYD-TDV results in rapid activation of immune responses along with an elevated level of expression of innate receptors. 49 It has been demonstrated that CYD-TDV induces robust immune responses in a dengue-immune population, including adults and adolescents.50–52 However, a safety signal was detected in vaccine-receiving individuals who were dengue non-immune during the time of vaccination.53, 54 As compared to the unvaccinated individuals, non-immune and the youngest vaccinated individuals experienced severe dengue and increased hospitalizations. 55 Unfortunately, following a mass immunization program in the Philippines, the vaccine was postponed in 2017 as a precautionary step because of an increased rate of hospitalization in children aged over 9 years with dengue. Nonetheless, the general public health benefit outweighs the negative press regarding the dengue immunization program in the Philippines, which further suggests the unmet need for an alternative, safe, and effective vaccine strategy that would provide robust protective immunity against infection caused by DENV regardless of the serological status or age of the recipients.56, 57
TAK-003
TAK-003 is Takeda’s tetravalent dengue vaccine candidate, which is a live attenuated dengue tetravalent vaccine that contains four DENV serotypes based on a DENV-2 backbone. 58 This tetravalent vaccine comprises a DENV-2 backbone in which the E and prM structural genes are replaced with the chimeric viruses for DENV3, DENV1, and DENV4. 59 In healthy children and teens aged 4–16 years, TAK-003 has already found as effective against virologically confirmed cases of dengue infection in the first part of the phase III trial in endemic regions including Asia and Latin America. 59 In addition, TAK-003 has been studied in a phase III trial in 26 countries, including Thailand, Nicaragua, Brazil, Sri Lanka, the Philippines, Panama, the Dominican Republic, and Colombia. 59 The biologics license application (BLA) of Takeda for TAK-003 to the FDA has been accepted and granted priority review, owing to the recent data obtained from the phase III parts 1–3 tetravalent immunization against dengue efficacy study, suggesting that primary and secondary endpoints were met. 60 At present, TAK-003 is solely permitted to be used in Indonesia for individuals ages 6 to 45 years. 61
TV003/TV005
TV003/TV005 (also referred to as TetraVax-DV or LAV∇30) is a live attenuated recombinant tetravalent dengue vaccine candidate discovered by the U.S. National Institutes of Health (NIH). TV003/TV005 was developed by utilizing three wild-type DENV strains attenuated by deleting several 30 and 31 nucleotides from the 30 untranslated regions, including rDENV4∇30, rDENV3∇30/31, and rDENV1∇30. The TV003 and TV005 are two distinct vaccine preparations that solely vary in the dosage of the rDEN2/4∇30 component. 57 As compared to TV003, TV005 showed a greater tetravalent response in vaccinated participants.62, 63 In a phase I trial, no difference in terms of adverse events (i.e., myalgias, fatigue, headaches, and rashes) was observed in the placebo, TV003, and TV005 groups with healthy adults (18–50 years) from Puerto Rico and the United States. Following the first dose, vaccine viremia was observed in 25.6% and 63.9% of baseline-flavivirus-naïve (BFN) TV005 and TV003 groups, respectively. Moreover, seropositivity was found to be 74.2% and 92.6% after the first dose in BFN-given TV005 and TV003 groups, respectively, as well as 100% in both vaccine groups with flavivirus-experienced (BFE) healthy individuals. 62 Phase II trials (in Taiwan) and phase III trials (in Brazil) are currently ongoing, where immunogenicity against all strains, great safety profiles, and enhanced tolerability in the participants (50–70 years) have been reported so far. 63
Tetravalent Dengue Virus-Purified Inactivated Vaccine (TDENV-PIV)
GlaxoSmithKline (GSK) and Walter Reed Army Institute of Research (WRAIR) have developed the novel dengue vaccine candidate TDENV-PIV. TDENV-PIV contains four DENV serotypes that have been inactivated chemically by using formalin. This inactivation inhibits the infectivity of the DENV strains while maintaining their antigenicity and structures. 64 A monovalent PIV candidate was tested in a Phase I (first-in-man) clinical trial, which revealed that the safety profile of DENV-1 PIV is acceptable along with some mild adverse events, including negligible injection site reactions. In addition, DENV-1 PIV triggered seroconversion in all participants of the study. 65 Although it has a higher safety profile, its immunogenicity was found to be lower than that of live-attenuated vaccines. Nevertheless, the immune response could be increased via the co-administration of an adjuvant. Therefore, two phase I clinical trials were carried out to assess the efficacy and safety of the tetravalent formulation TDENV-PIV along with different adjuvants, including AS03B, AS01E, or aluminum hydroxide. Indeed, all these formulations triggered a balanced immune response against all four serotypes of DENV and were well tolerated by the participants, where AS03B and AS01E induced the highest mean antibody titers.46, 66 The most effective injection schedule (0–3, 0-1-6, or 0–1 months) of TDENV-PIV adjuvanted by AS03B is currently being assessed by a phase II clinical trial. 57
V180
V180 is a recombinant DENV-envelope vaccine developed by MERCK, which is a promising vaccine candidate that has finished phase I clinical trials. 67 In the preclinical study, a mixture of V180 and ISCOMATRIX adjuvant was administered in monkeys and mice to assess the efficacy of stimulating robust neutralizing antibodies against all DENV serotypes and providing protection from viremia.68, 69 MERCK, in a phase I clinical trial, utilized flavivirus-naïve adult volunteers who received V180 adjuvanted by ISCOMATRIX. The participants of the study exhibited a positive and strong immunity; however, the vaccine formulations without adjuvants and with aluminum adjuvants were found to be poorly immunogenic. In addition, the vaccine formulation adjuvanted by ISCOMATRIX was more effective as compared to non-adjuvanted formulations and formulations with an aluminum adjuvant. In general, all these formulations were found to be well-tolerated.70, 71
Management of Dengue
Fluid Therapy
The most dangerous manifestation that is seen in DENV infection is dengue shock syndrome (DSS), which results in a significant rise in vascular permeability. 72 Fluid resuscitation is crucial in severe dengue management. The primary goal of this therapy is to avert the complications associated with hypovolemic shock and vascular leakage. 73 Rapid restoration of circulating plasma volume is the main DSS therapy. Since DSS involves small plasma protein leakage, colloid preparations with larger molecular weights might provide some benefits. It has been observed that colloids are more likely to stay in circulation for a longer time and elevate the colloid oncotic pressure, therefore drawing extravasated fluid back into circulation. 72 Ringer’s lactate and normal (0.9%) saline are most commonly used in the case of dengue-related fluid resuscitation. Normal saline might also be used for initial resuscitation in an individual with normal chloride levels (95–105 mmol/L) as well as with or without hyponatremia. Ringer’s lactate might be a better option if a patient exhibits hypernatremia, hyperchloremic acidosis, or hyperchloremia.
Intravenous Immunoglobulins (IVIG)
IVIG is widely used in the treatment of idiopathic thrombocytopenia purpura (ITP). ITP-associated thrombocytopenia is accompanied by autoantibodies known as platelet-associated IgG (PAIgG). It was observed that PAIgG-coated platelets go through increased clearance via Fcγ receptors expressed on mononuclear phagocytic cells. The mechanism of action of IVIGs involves the ligation of inhibitory receptors or competitive suppression of Fcγ receptors. The effectiveness of IVIG treatment was assessed in a clinical trial with 36 dengue patients for 4 days. Unfortunately, the study was unsuccessful in demonstrating the effectiveness of IVIG treatment in mediating platelet recovery. 73
Blood Products
Thrombocytopenia denotes a low platelet count (below 150 × 10 9 /L), which is a DENV infection hallmark and is typically observed between days 3 and 8 after the onset of illness. In the case of dengue, platelet count drops rapidly in association with an increased hematocrit level, which suggests the development of the critical phase of dengue. Platelet count usually reaches its lowest point during defervescence (days 3–6); after that, gradual spontaneous recovery occurs. On the other hand, minor bleeding (petechiae and mucosal) also occurs without hemodynamic instability, which usually goes away spontaneously. 73 Prophylactic platelet transfusion is usually used in individuals without bleeding, particularly when the platelet count falls below 10–20 × 109/L. It is extensively used in cases of sepsis; 74 however, this is not indicated based on evidence in dengue management. Moreover, the benefits of prophylactic platelet transfusion are outweighed by substantial risks such as allergic reactions, blood-borne infections, transfusion-associated lung injury, and fluid overload.75, 76
Immature Platelet Fraction
Immature platelet fraction (IPF) is an indicator of the reticulated platelets present in peripheral blood. Analogous to the estimation of red cell production by using a reticulocyte count, IPF also reflects the recovery of bone marrow. Therefore, an increased IPF level suggests an elevated thrombopoiesis rate in the bone marrow. As compared to mature platelets, freshly secreted platelets are physiologically more effective and larger in size. An IPF value of 10% forecasts platelet recovery within the next 72 h.72, 77
Corticosteroids
Corticosteroids exert suppressive actions on various native immune responses of phagocytes. These corticosteroid-mediated suppressive actions are also observed in the case of B- and T-cell-mediated immune responses. Higher doses of corticosteroids can be beneficial in numerous conditions with immune aberrations, particularly autoimmune diseases, including systemic lupus erythematous. 78 In the early acute phase of DENV infection, the use of corticosteroids did not exert any effects on platelet count recovery, plasma leakage, or shock in patients with dengue. Moreover, this use did not alter the kinetics of plasma cytokine levels or dengue virological markers.79, 80
Platelet Microparticles (MPs)
Microparticles (MPs) are small membrane vesicles, and platelet microparticles (PMPs) function like platelets and are derived from activated platelets. A study conducted in India revealed markedly higher levels of PMPs in patients with dengue as well as thrombocytopenia and without bleeding in comparison with patients with bleeding manifestations. 81 Based on this observation, it was concluded that these dengue patients might be protected because of the procoagulant effect of PMPs; thus, PMPs might play a role as a biomarker to determine prophylactic platelet transfusion. 82
Mast Cell Inhibitors
Several studies have reported that mast cell (MC) activation plays a role in the pathogenesis of hemorrhage and dengue-vascular leakage.83, 84 In mice, activated MC was found to secrete various proteases, including tryptase and chymase, in the serum that cause vascular integrity loss in cases of DENV infection. In addition, serum chymase concentrations were associated with severity in dengue-infected individuals who were enrolled in a prospective study. Researchers also reported that various MC-stabilizing compounds such as ketotifen, montelukast, and cromolyn decreased vascular leakage in wild-type mouse models of DENV challenge in spite of the small and nonsignificant rise in mouse viremia. 83 Collectively, these results suggest the prospects of MCs as therapeutic targets to reduce DENV pathogenesis. In Singapore, a clinical trial is currently ongoing to evaluate the effectiveness of ketotifen to decrease the extent of vascular leakage. 85
Therapeutics for Dengue
Suramin
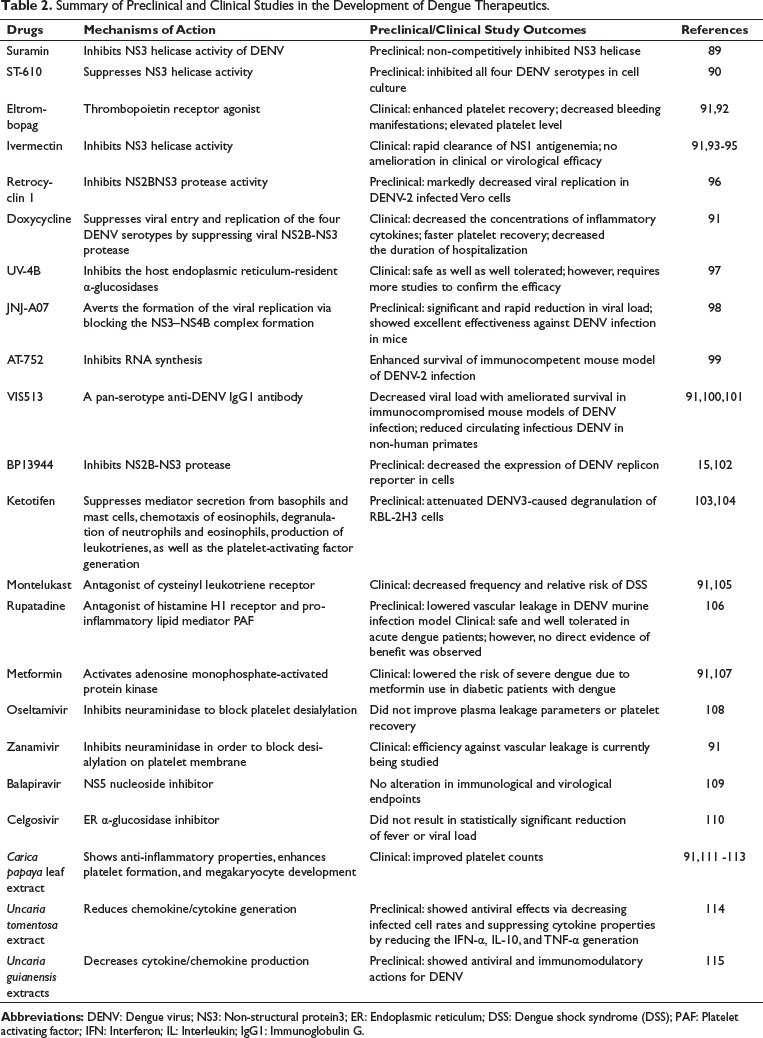
Suramin is a synthetic polysulfonate that is used in the treatment of river blindness and African sleeping sickness. 86 Suramin inhibits multiple viruses, including dengue, Zika, chikungunya, hepatitis, herpes simplex, and Ebola viruses.87, 88 In a study, Basavannacharya et al. 89 reported that suramin significantly inhibited the helicase activity of NS3 (Table 2). In addition, suramin suppressed the NS3 helicase activity of DENV as a non-competitive inhibitor. 89
Summary of Preclinical and Clinical Studies in the Development of Dengue Therapeutics.
ST-610
ST-610 is a novel small-molecule DENV inhibitor that selectively and strongly suppresses all four DENV serotypes replication (Figure 3) in vitro. In a study, Byrd et al. 90 revealed through a molecular-beacon-based helicase assay that ST-610 suppressed DENV NS3 helicase RNA unwinding action. However, it did not suppress nucleoside triphosphatase action in a malachite green ATPase assay. ST-610 was found to be nontoxic, well tolerated, and nonmutagenic in mouse models and showed effectiveness in a sublethal DENV-infected murine model with the capacity to markedly decrease viral load and viremia in comparison with the vehicle controls. 90
Molecular Mechanism of various Dengue Therapeutics.
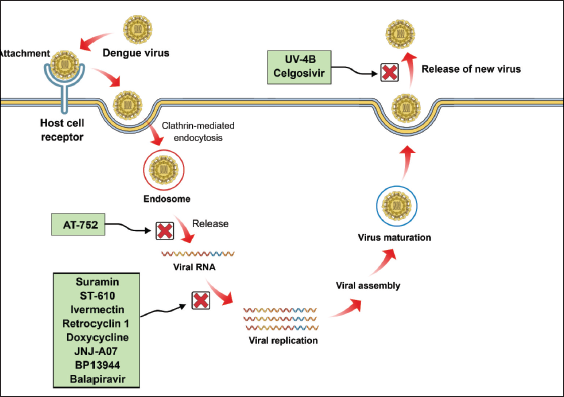
Eltrombopag
Eltrombopag is a thrombopoietin receptor agonist that is effective in treating thrombocytopenia in individuals with a range of pathological conditions, including severe aplastic anemia, chronic liver disease, and immune thrombocytopenia.92, 116 In a clinical trial, the potential of eltrombopag was found to be beneficial in treating thrombocytopenia in patients with moderate to severe dengue (platelet count <100 × 109/L). 92 In addition, treatment with eltrombopag at a dose of 25 mg induced megakaryopoiesis, and when used for 3 days, it markedly increased the platelet count above the lower normal limit (150 × 109/L) and enhanced platelet recovery in 91% of participants on day 7 post-enrollment in comparison with the 55% in the control group. This thrombopoietin receptor agonist also decreased the bleeding manifestations in 93% of grade II patients with DHF by day 7. Whereas, in the control group, sporadic bleeding with low platelet counts was still detected in 40% of participants. Collectively, these data indicate that eltrombopag might be beneficial in the treatment of dengue patients with thrombocytopenia and bleeding manifestations. 91
Ivermectin
Ivermectin is a broad-spectrum antiparasitic drug that is typically used in the treatment of helminth infections. Interestingly, ivermectin was found to suppress all 4 DENV serotypes in vitro via suppressing the host nuclear import proteins that are crucial for nuclear localization of the dengue virus-encoded RNA-dependent RNA polymerase, NS5.118, 119 A clinical trial (NCT02045069) was carried out to evaluate the efficacy of ivermectin (once daily at the dose of 400 µg/kg for 2–3 days) in adult dengue patients. 120 Ivermectin treatment rapidly cleared the NS1 antigenemia; however, no difference was observed in viral clearance, viremia, or other useful clinical outcomes such as fluid requirements, hemoconcentration, pleural effusion, hospitalization, DHF incidence, or fever. 120 More studies are required to determine the pharmacodynamics of this drug and the NS1-related mechanism of action. 91
Retrocyclin-1
Retrocylin-1 (RC-1) is a synthetic peptide that acts against the DENV NS2B-NS3 protease. RC-1 and its analogues have been utilized in several studies to estimate its suppressive capacity against replication of the herpes simplex virus, the avian influenza H5N1 virus, HIV (human immunodeficiency virus), and various other micro-organisms, including Staphylococcus aureus and Bacillus anthracis. In a study, Rothan et al. 96 reported that recombinant RC-1 suppresses the replication of DENV-2 in Vero cells via interfering with its serine protease activity. They observed that RC-1 markedly decreased the viral replication in DENV-2-infected Vero cells at simultaneous treatment after 75 h (85%) and 48 h (70%). Moreover, a moderate reduction was observed in viral replication in pre-treatment mode after 72 h (38%) and 48 h (70%), as well as post-treatment at 72 h (45%) and 48 h (30%). 96
Doxycycline
Doxycycline can act both as an antimalarial agent and an antibiotic. It has been observed that doxycycline exerted antiviral properties against all four DENV serotypes in vitro via suppressing NS2B-NS3 protease activity, which further resulted in decreased replication and viral entry. 121 In Brazil, it was reported in a clinical trial that doxycycline reduced the levels of pro-inflammatory cytokines (TNF-α and IL-6) in the treatment-receiving group in comparison with the individuals receiving standard supportive and symptomatic care. 122 On the other hand, in India, it was observed that dengue patients who received doxycycline treatment exhibited reduced hospital stays as well as faster recovery of leukocyte and platelet counts. 123 Another study in India is currently ongoing to assess the antiviral potential of doxycycline in a pediatric population. 91
UV-4B
UV-4B is a small molecule and an iminosugar that inhibits alpha-glucosidase activity, which resulted in an antiviral property in vitro against all four DENV serotypes. 124 Furthermore, in a lethal ADE model of dengue, UV-4B also reduced viral burden and viremia as well as ameliorated survival. 125 It was also reported in a clinical trial that a single dose of up to 1000 mg of UV-4B was well tolerated and safe. 97 However, the efficacy of UV-4B in dengue treatment has not been determined yet. 91
JNJ-A07
Janssen Pharmaceuticals has developed an NS4B inhibitor called JNJ-A07. 98 JNJ-A07 was screened from 2000 related molecules generated via altering the ketoindole compound structure that suppressed DENV2-caused infection. 126 In addition, JNJ-A07 suppressed the formation of the DENV replication complex by suppressing the interaction between NS3 and NS4B. 127 The drug also exerted antiviral properties against the 21 clinical isolates and all 4 DENV serotypes in vitro. Interestingly, irrespective of whether JNJ-A07 treatment was introduced at a later stage or at the onset of infection, it rapidly cleared viremia and viral burden in organs of immunocompromised murine models infected with sub-lethal and lethal DENV2 doses and in the ADE model of dengue. In the JNJ-A07-treated infected mouse models, a decrease in various pro-inflammatory cytokines (IL-18, TNF-α, IL-6, and IFN-γ) as well as a rise in survival percentage were also reported. An analog of JNJ-A07, JNJ-64281802, is being studied in two phase II clinical trials to assess its effectiveness as dengue therapeutics in individuals with confirmed dengue fever and as dengue prophylaxis in healthy people. 91
AT-752
AT-752 is a guanosine nucleotide analog synthesized by Atea Pharmaceuticals that exerts its activities by targeting the NS5 RdRp activity. 99 AT-752 plays roles via metabolizing into its active triphosphate metabolite known as AT-9010, which is a GTP analog and is incorporated into RNA through RdRp, which eventually suppresses viral replication. It was reported that the prodrug AT-752, AT-281, suppressed DENV3 and DENV2 serotypes in vitro, while AT-752 improved survival and decreased viremia in immunocompromised DENV2-infected immunocompromised mouse models. Clinical trials are ongoing to assess the antiviral activity and safety of these drugs in dengue patients and the dengue human challenge model, respectively. 91
VIS513
VIS513 is a humanized pan-serotype reactive anti-DENV monoclonal antibody (mAb) discovered by Visterra (Cambridge, Massachusetts), which binds with E protein EDIII of all four DENV serotypes. 101 In non-human primates, VIS513 treatment for 24 h or 5 days post-dengue infection diminished circulating DENV without changing the endogenous antibody response. 100 VIS513 enhanced the protection of immunocompromised mouse models from lethal primary and secondary infections in the murine model of antibody-enhanced DENV infection. 101 Currently, in India, VIS513 is being studied in a phase II clinical trial to evaluate its efficacy and safety in adult individuals with dengue fever (CTRI/2021/07/035290). Phase I clinical trials with other human monoclonal antibodies, including Dengushield (developed by the Serum Institute of India) (NCT03883620) and AV-1 (under clinical development by AbViro) (NCT04273217), are also ongoing to estimate the pharmacokinetics and safety in healthy adults. Nonetheless, details of these therapeutics, including their efficacy against DENV, target epitopes, and preclinical development, are yet to be revealed. 91
BP13944
BP13944 is a potential small-molecule inhibitor identified by high-throughput screening of 60,000 compounds by means of a stable cell line containing an effective luciferase replicon of DENV-2. 102 This small-molecule inhibitor decreased the expression of the DENV replicon reporter in cells. In addition, BP13944 suppressed viral RNA synthesis or replication in all 4 DENV serotypes, with no noticeable cytotoxicity. Researchers observed that BP13944 has the capacity to target the NS3 protease of DENV, which might prove beneficial in suppressing DENV. 102 However, more studies are required in this regard.
Montelukast and Ketotifen
It has been reported that activation of mast cells (MC) plays a role in the pathogenesis of hemorrhage and dengue-vascular leakage.128, 129 During DENV infection, MC activation has also been identified in mice, which results in the secretion of several proteases, including tryptase and chymase, into the serum and causes vascular integrity loss. In addition, it was also observed that serum chymase concentrations are associated with the severity of dengue in individuals who participated in a prospective study. Researchers observed in the study that various MC-stabilizing compounds, including ketotifen and montelukast, decreased the level of vascular leakage in wild-type mouse models of the DENV challenge; however, there was a minor (insignificant) rise in mouse viremia. 128 Collectively, these results suggest the role of MCs as therapeutic targets to control DENV pathogenesis. In Singapore, a clinical trial is ongoing to evaluate the effectiveness of ketotifen to decrease the extent of vascular leakage. 73
Rupatadine
Rupatadine is a second-generation antihistamine that shows sustained histamine-1-receptor and PAF receptor blocking properties and is commonly used in the treatment of chronic urticaria and allergic diseases. 130 In Sri Lanka, a clinical trial with 183 adult participants with acute dengue was carried out, and it was observed that rupatadine up to 40 mg is well-tolerated and safe. 106 In addition, a similar level of adverse events with placebo and rupatadine was reported in that study. The primary end-point of a substantial decrease in fluid leakage (development of ascites or pleural effusions) was not met in that clinical trial; however, post hoc analyses showed minor but noteworthy differences in various parameters on individual illness days: lower concentrations of aspartate-aminotransferase and greater platelet counts on day 7 in the rupatadine group in comparison with the placebo group, and minor effusions on day 8 in the subgroup of participants with pleural effusions. Nonetheless, owing to several factors of that clinical trial, including the range of recruitment time and the small sample size, the therapeutic potentials of rupatadine need further assessment in large studies focusing on patient recruitment during the early febrile phase. 106
Metformin
Since severe dengue risk factors include metabolic disorders including diabetes and obesity,131–134 the potential of metabolic drugs in dengue treatment has also been evaluated. Metformin is a widely used anti-hyperglycemic agent in the treatment of type 2 diabetes mellitus. Metformin causes the activation of adenosine monophosphate (AMP)-activated protein kinase (AMPK), which is a crucial sensor for cellular energy that is activated during low cellular energy levels to preserve homeostasis via increasing glucose, protein, and lipid metabolism. 135 DENV was found to impair the phosphorylation of AMPK, which results in downstream upregulation of HMG-CoA reductase action, the rate-limiting step in cholesterol synthesis. 136 This resultant lipid-enriched environment mediates DENV proteins, including NS4A and NS3, to colocalize, which eventually results in increased generation of the DENV replication complex.136, 137 Metformin exerted in vitro antiviral effects in DENV-infected cells in a significant dose-dependent manner. 136 Interestingly, it was observed in diabetic patients with confirmed dengue that the use of metformin was linked with a reduced risk of experiencing severe dengue. Furthermore, there was a dose-dependent inverse relationship between dengue severity and metformin intake. 107 Nonetheless, it is not clear whether the activities of metformin on DENV replication or on diabetic control might be associated with the usage of metformin. In Vietnam, a clinical trial with metformin (NCT04377451) is currently ongoing in dengue patients who have obesity to identify the therapeutic potential of metformin against acute DENV infection. 138
Oseltamivir and Zanamivir
Oseltamivir is a neuraminidase inhibitor that is typically used in influenza treatment. In a clinical trial in Indonesia, the efficacy of oseltamivir was evaluated in the treatment of thrombocytopenia associated with acute DENV infection. 108 It was observed that oseltamivir is not effective in preventing plasma leakage and platelet recovery in individuals with acute dengue with severe to moderate thrombocytopenia. 91 Zanamivir is a neuraminidase inhibitor that suppresses DENV2 NS1-induced endothelial hyperpermeability in vitro via suppressing endogenous sialidase. Currently, the assessment of the efficacy and safety of 5 days of zanamivir treatment in the treatment of vascular permeability syndrome is ongoing. 91
Balapiravir
Balapiravir is a prodrug of the nucleoside analogue R1479 (4′-azidocytidine). Balapiravir was developed by Hoffmann-La Roche to treat chronic hepatitis C virus infection.140, 141 In a study, Nguyen et al. 109 revealed that balapiravir treatment at a dose of 1500 mg for 5 days was well tolerated. Nonetheless, daily evaluation of NS1 antigenemia and twice-daily evaluation of viremia showed balapiravir did not significantly reduce the fever clearance time or change the kinetics of the virological markers. Moreover, the whole blood transcriptional profile and kinetics of plasma cytokine levels were not decreased by treatment with balapiravir. 109
Celgosivir
Celgosivir is an alpha-glucosidase inhibitor that reduced NS1 folding and trapping and showed marked antiviral properties in dengue-infected mouse models. 142 Unfortunately, the previous clinical trial did not meet its primary outcomes of decreasing fever or viremia; however, subsequent assessment of the pharmacokinetic endpoints of the clinical trial concluded that a revised dose regimen of celgosivir might elevate the steady-state through drug concentrations in the serum of patients, which may prove beneficial.110, 143
Phytochemicals
Carica papaya Leaf Extract (CPLE)
CPLE was found to increase the platelet count in both adult and pediatric dengue patients. 144 Clinical studies have revealed that CPLE administration three times a day increased the rate of platelet recovery, along with a statistically significant rise in platelet counts in the CPLE-receiving group from day 3. The exact mechanism of CPLE is yet to be uncovered; however, it has been observed that CPLE can elevate the expression of CD110 (known as the thrombopoietin receptor) on megakaryocytes and platelets, which is essential for platelet formation and megakaryocyte proliferation. 145
Uncaria tomentosa and Uncaria guianensis
Uncaria tomentosa and Uncaria guianensis belong to the family Rubiaceae, which is also called cat’s claw. These large woody vines are traditionally used in the treatment of various degenerative and inflammatory disorders, including hemorrhages, fevers, gastric ulcers, cancer, diabetes, and arthritis. 115 In a study, Reis et al. 114 reported in vitro inhibitory properties of U. tomentosa, which resulted in the reduction of DENV-Ag+ cell rates in treated monocytes. Furthermore, the alkaloidal fraction of U. tomentosa mediated a robust immunomodulation, which markedly reduced the levels of IFN-α and TNF-α, along with an IL-10 modulation. These researchers also reported that the alkaloidal fraction was most effective in decreasing the levels of cytokines as well as the rates of monocyte infection. The immunomodulating and antiviral effects in vitro were mainly exerted by the pentacyclic oxindole alkaloids of U. tomentosa, which need to be further studied to demonstrate their potential in dengue treatment. 114 In a different study, Mello et al. 115 observed that hydro-alcoholic extracts derived from U. guianensis leaves (UGL) primarily differ from U. guianensis bark (UGB) by selectively possessing kaempferitrin (a natural flavonoid glycoside). Interestingly, both extracts ameliorated hepatocyte viability. Moreover, UGL and UGB suppressed the NS1 release and decreased the level of intracellular viral antigens. It was also noticed that UGB decreased the release of macrophage migration inhibitory factors, whereas UGL reduced the level of IL-6. On the other hand, both UGL and UGB decreased the level of IL-8. More studies are required in the future to demonstrate their potential as dengue therapeutics.
Monoclonal Antibodies
Therapeutic human serum polyclonal antibodies (IgG) formulated from pools of plasma derived from several healthy blood donors are supposed to comprise a range of antibodies that would be present in normal human serum and against common viral pathogens. Disadvantages of polyclonal antibodies include the risk of blood-borne pathogens, batch-to-batch variations, and various side effects, particularly allergic reactions. Moreover, batches should be screened to confirm neutralization action against all four DENV serotypes. 146 On the other hand, therapeutic monoclonal antibodies (mAbs) can be in large quantities and have high consistency and specificity, with markedly lower levels of adverse events linked with polyclonal IgGs. 147 It was reported that antibodies that target the lateral ridge or A-strand of E protein EDIII can strongly neutralize some, but in rare cases, all four DENV serotypes. 148 4E11, an A-strand-specific mAb, was redesigned by using computational chemistry to counteract all four DENV serotypes, both in mouse models of the DENV challenge and in vitro. 149 This humanized antibody was further developed by means of both X-ray crystallography and computational protein chemistry to further ameliorate this antibody for therapeutic use. 150 Currently, this antibody is going through preclinical development. It is likely that mAbs derived from convalescent patients with dengue that neutralize all DENV serotypes with robust potency might have therapeutic activity. Human mAbs typically bind with quaternary epitopes, which are not different from mouse-derived antibodies. It was observed that a potent serotype-specific mAb has the capacity to bind to the hinge area between the EDI and EDII. 151
Another strong serotype-specific mAb, HM14c10, has been identified that has the capacity to bind with a discontinuous epitope covering adjacent surfaces of E-protein dimers on DENV-1. 152 Indeed, a strong mAb against DENV-3 should have the capacity to bind with the functionally significant domains across DI–III proteins. 153 Another DENV2-specific human mAb, 2D22, has also been discovered that can bind across E proteins and block the E-protein structural reorganization essential for viral fusion. 154 In addition, various human mAbs have been detected that bind epitopes that link across two E proteins (known as envelope dimer epitopes). 155 These antibodies were found to exhibit pan-serotype neutralizing function and could be beneficial therapeutically. There is a great therapeutic potential for antibodies; however, only in vitro and animal models have been used so far to assess their potential. Therefore, these therapeutics need to be studied in clinical trials to identify their potential in dengue treatment. 73
Conclusion
Dengue is increasingly becoming life-threatening for humans. DSS and DHF pathogenesis are multifactorial and complicated and include both host and viral factors. The lack of an effective vaccine to control dengue is making it more difficult to prevent the disease. Moreover, dengue vaccines ought to be cheaper, as most of the countries facing outbreaks are economically challenged. Even though there are some live-attenuated tetravalent vaccines that are approved for dengue and some are in the clinical trial stages, the unique DENV pathogenesis complexity and its relationship to immune enhancement are hindering safe and effective dengue vaccine development. Due to the limited number of effective dengue vaccines and therapeutics, there should also be a focus on control strategies to counter DENV infection.
Footnotes
Acknowledgments
The author would like to extend his regards to the University of Jeddah, Kingdom of Saudi Arabia, for supporting the research.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
