Abstract
Objectives
To evaluate the early and late effects of N-acetyl-
Materials and Methods
Adult male Wistar rats (8 weeks of age) received an HS (8.0% NaCl) or normal sodium (NS; 1.3% NaCl) diet and NAC (600 mg/L) in drinking water or normal water. At 11 weeks of age, the rats underwent a renal I/R procedure. They followed for 10 weeks after I/R, at the 1st, 2nd, 4th, and 10th weeks, in which tail blood pressure (tBP) and renal function were evaluated. And renal renin gene expression was evaluated in the 10th week after I/R.
Results
During the study, it was observed that the tBP remained consistently higher in the HS-I/R+water group compared to the NS-I/R+water group. However, in the early weeks following I/R (1st, 2nd, and 4th weeks), the tBP was lower in the HS-I/R+NAC group than in the HS-I/R+water group. In the 10th week after I/R, the serum creatinine levels were higher in both the HS-I/R+NAC and NS-I/R+NAC groups compared to the HS-I/R+water and NS-I/R+water groups. Conversely, the creatinine clearance was higher in the HS-I/R+NAC group than in the HS-I/R+ group in the 2nd week following I/R. Additionally, the urinary protein levels were higher in the HS-I/R+NAC group than in the NS-I/R+NAC group in the 10th week after I/R. It was also observed that NAC treatment resulted in increased renal renin gene expression in the 10th week following I/R.
Conclusion
After renal I/R in animals given HS, NAC treatment was initially effective in lowering blood pressure or increasing creatinine clearance. However, these positive effects did not persist over the long term, resulting in decreased kidney function and increased blood pressure. Furthermore, the renin-angiotensin-aldosterone system was increased by HS intake, and the benefits of the NS diet were less effective than those of the HS diet. Thus, NAC provides temporary protection only in the early stages following an insult.
Introduction
Renal ischemia and reperfusion (I/R) due to a sudden internal hemorrhage, dehydration, septic shock, and diabetes mellitus, receiving nephrotoxic drugs or contrast media can result in generalized or localized impairment of oxygen and nutrient delivery to and waste product removal from cells of the kidney, which characterize an acute kidney injury (AKI). This imbalance induces the tubular epithelial cells to undergo damage and, if it is severe, death by apoptosis and necrosis (acute tubular necrosis), with functional organ impairment of water and electrolyte homeostasis and reduced excretion of waste products of metabolism. 1 In general, patients who survive an episode of AKI requiring dialytic therapy are at increased risk of chronic kidney disease (CKD), estimated to account for 3% of the overall yearly incidence of CKD. 2
From the evidence in studies, renal function recovery is not considered complete after I/R. Experimental studies have shown permanent renal structure and function alterations after I/R injury in rats. Such changes are associated with developing features indicative of chronic renal disease. Also, the authors observed compromised sodium homeostasis and a predisposition to hypertension when the rats were exposed to a high-sodium (HS) diet.3, 4
Nowadays, the population is subjected to HS intake from highly processed foods, a public health concern. A meta-analysis with cross-sectional, longitudinal observational studies, and intervention trials recommended sodium dietary interventions to prevent CKD and delay its progression or complement traditional strategies, such as controlling blood pressure and diabetes or inhibiting the renin-angiotensin-aldosterone system (RAAS) from decreasing disease-specific mortality. 5 An experimental study showed that AT1 receptor antagonism prevented the AKI–CKD transition before ischemia by improving early renal blood flow recovery, reducing inflammation, and increasing hypoxia-inducible factor 1α activity. This study supposed that activation of the RAAS after AKI triggers maladaptive mechanisms that lead to chronic disease. 6 The sodium intake controls RAAS activity, where low sodium activates, and HS inhibits the system establishing electrolyte homeostasis. This RAAS inhibition by HS intake, or AT1 receptor antagonism study cited above, could change the mechanism of AKI and its recovery. In the previous study, we evaluated the influence of chronic HS intake in rats submitted to I/R. We observed increased blood pressure, albuminuria, and kidney and heart masses, although RAAS inhibition. Besides, we studied the N-acetyl-L-cysteine (NAC) influence on a chronic HS diet. We showed that NAC treatment could not recover kidney function or blood pressure from the effects of I/R associated with a chronic HS diet. However, it promoted structural improvements, such as decreased albuminuria and fibrosis in the kidney and heart. 7 After these observations, we think the follow-up after AKI could be essential to evaluate the kidney recovery conditions and mitigate CKD development through preventative measures.
The NAC acts through its antioxidant activity mechanism and through the induction of vasodilation in the renal medulla by stabilizing nitric oxide and inhibiting angiotensin converting enzyme (ACE). 8 NAC is one of the approaches used to prevent contrast-induced AKI.9–13 However, NAC’s lack of protective role has been reported in some studies with chronic renal patients.14, 15 In experimental studies, it is observed that there are mixed results. One study showed that NAC did not improve kidney function in rats during the 1st period after I/R. 16 In another study, it was observed that NAC attenuated the progression of chronic renal failure. 17 These mixed results from different studies made it challenging to understand whether they happened early or after a long time. It is trustworthy that the study could be conducted and observed from the beginning of I/R until the end of the study. Therefore, it drove us to investigate and follow the biochemistry variable’s behavior from beginning to end to evaluate how it led to changes observed at the end of studies.
The objective of this study was to evaluate the parameters followed up to the end of the study to investigate whether the early and late effects of NAC treatment on I/R in adult male Wistar rats influenced by chronic HS intake were similar or not.
Materials and Methods
Research Ethics Coordination from the School of Medicine of São Paulo University approved the experimental study on protocol 037/14. The experimental protocol was elaborated before the study.
Animals
Adult (8 weeks of age) Wistar male rats arriving from the Animal Care Center of the School of Medicine of São Paulo University were kept in a light/dark cycle of 12 h at a room temperature of 22°C. The experimental unit was a cage of animals that kept four animals per cage.
Diets
The diets with HS (8.0% NaCl and 25.0% protein) or normal sodium (NS) content (1.3% NaCl and 25.0% protein) were offered to animals from 8 weeks to 21 weeks of age. The diets were obtained from Harlan (Harlan Laboratories, Madison, WI 53744-4220, USA), and the nutritional composition was the same for both diets, except for the sodium concentration.
Treatment
The animals receiving HS or NS were divided into two groups and subjected from 8 weeks to 21 weeks of age to follow treatment:
Renal I/R
After preconditioning to the NAC treatment and diet intake, I/R was performed at 11 weeks of age, as described previously. 7 Briefly, the animals were anesthetized with a mixture of 2% xylazine and 50 mg/mL ketamine at a ratio of 2:5 vol/vol. After making an abdominal incision, the renal arteries were isolated from both kidneys and clamped for 45 min to induce ischemia. The color change of the kidney from red to brown confirmed the ischemia. After the clamps were removed, the reperfusion could be visualized by the color change of the renal tissue from brown to red. The animals were sutured and received a single dose of 24,000 UI/kg of penicillin G benzathine via intramuscular injection, analgesic treatment with dipyrone (150 mg/kg subcutaneous) for 3 days, 20 and opioid Tramadol intramuscular, 15 mg/kg, 8/8 h for 3 days. The animals were then monitored closely.
Experimental Protocol
The rats started to receive natural water or NAC solution at 8 weeks of age before I/R.
NS-I/R+water: Rats fed an NS submitted to renal I/R and received water (comparator group, n = 16);
NS-I/R+NAC: Rats fed an NS submitted to renal I/R and offered 600 mg/L NAC dissolved in water (comparator group, n = 16);
HS-I/R+water: Rats fed HS submitted to renal I/R and received water (n = 16); and
HS-I/R+NAC: Rats fed HS submitted to renal I/R and offered 600 mg/L NAC dissolved in water (n = 16).
Inclusion and Exclusion Criteria
The animals were included in the study if they underwent successful renal I/R. The animals were excluded if they died prematurely.
Randomization
After surviving the initial I/R, animals were randomized using a computer-based random order generator (GraphPad by Dotmatics).
Blinding
I, Luzia Furukawa, was aware of the group allocation at the different stages of the experiment (during the allocation, the conduct of the investigation, the outcome assessment, and the data analysis).
Outcome Measures
The serum creatinine was the primary outcome measure to determine the sample size.
Body Weight, Food, and Liquid Intake
The animal body weight was assessed each week using a distinct balance for animals (Model AS5500C, Marte Balanças, São Paulo, SP, Brazil). Food and liquid (water or NAC) consumption were determined before and subsequent I/R (1, 2, 4, and 10 weeks) under a metabolic cage for 24 h (Tecniplast S.p.A. Buguggiate,VA, Italy).
Tail Blood Pressure
Tail blood pressure (tBP) measurement was performed as described previously 7 using a tBP apparatus (Kent Scientific Corporation, USA, model RTBP 2045). For each animal, the tBP measurement was taken five times, and the average was calculated. The tBP measurement was performed every 2 weeks, from 8 until 21 weeks of age.
Renal Function
Urine and blood were analyzed to evaluate renal function. We measured the urinary protein concentration by the Sulfosalicylic acid method, 21 serum and urine creatinine levels (Creatinine kit K-ref: 96; Labtest Diagnostica S.A. Lagoa Santa-MG), serum urea (Ureia UV Liquiform kit, ref: 104; Labtest Diagnostica S.A. Lagoa Santa-MG) with a Cobas Mira analyzer, and urinary sodium by a flame spectrophotometer (model FC 280, CELM®, São Paulo, Brazil). All these parameters were analyzed before and after I/R (1, 2, 4, and 10 weeks).
Fractional excretion of sodium (FENa) and creatinine clearance were calculated using serum and urine analysis data and the 24 h urine volume.
Renin Measurement
Plasma and renal renin were assessed by a radioimmunoassay kit (Angiotensin I RIA Kit, Beckman Coulter, Inc., IMMUNOTECH s.r.o, Prague, Czech Republic); Measurements were performed 10 weeks after I/R.
Euthanasia
The animals underwent analgesia with xylazine (2%)/ketamine (50 mg/mL) (2:5 vol/vol) and total exsanguination 22 by the abdominal aorta to collect the blood sample used for biochemical analyses, and kidney tissues were collected.
Renal Immunohistochemistry and Renal Fibrosis
The renal tissue was fixed in formalin buffer, embedded in paraffin, and 4 µm sectioned by a microtome (2065 Jung Supercut, Leica, Germany) for the immunohistological analysis. Immunohistochemistry was performed to detect the antigens CD68 and transforming growth factor-beta (TGF-β) using mouse anti-rat CD68 (Bio-Rad) and mouse anti-human TGF-β (Bio-Rad) antibodies, respectively. Briefly, the slide of sectioned renal tissue was deparaffinized, rehydrated, and exposed to citrate buffer steam for heat-induced epitope retrieval. The staining protocol for each antibody was followed using a polyvalent HRP-DAB detection system from Spring Bioscience (Pleasanton, CA, USA). Cortical and medullary CD68 and TGF-β were evaluated by counting 30 fields per animal and classified by score (score 1: 0%−5%, score 2: 5%−25%, score 3: 25%−75%, and score 4: 75%−100%) using 200× light microscopy (ocular lens 10×; objective lens 20×).
Masson’s trichrome staining and Panoramic Digital Slide Scanners (3DHISTECH, Budapest, Hungary) were used to assess renal fibrosis in a blinded manner with the Panoramic Viewer program. Renal fibrosis was graded as 0, <5%; I, 5%–25%; II, 26%–50%; III, 51%–75%; or IV, >75%.
Gene Expression
Renal renin gene expression was evaluated by real-time polymerase chain reaction (PCR) analysis (Rotor-Gene Q, Qiagen, Hilden, Germany) using the following primers: renin (sense: GGACACTGGCACATCCTATATC; anti-sense: ACCTGGCTACAGTTCACAAC); reference gene, GAPDH (sense: GTATGACTCTACCCACGGCA; anti-sense: AAGACGCCAGTAGACTCCAC). The REST program performed the analysis and graphics, and the results are expressed as renin gene expression relative to GAPDH expression and correlated to NS as a control group.
Statistical Analysis
The results are presented as the mean ± SE. We used the GraphPad Prism 5 software (GraphPad Software, Inc., San Diego, CA, USA) for all statistical analyses. Primarily, we evaluated the normality of the data by the D’Agostino test and performed the appropriate data analysis. Two-way analysis of variance (ANOVA) performed analyses of the body weight, tBP, liquid (water and NAC) and food intake, FENa, urine volume, serum creatinine and urea, creatinine clearance, and urinary protein, with repeated measures, time, and treatment considered factors. The Student’s t-test analyzed plasma renin activity and renal renin content (Ang I), renal TGF-β, and CD68. The number of animals was calculated based on previous studies from our group, and preliminary experiments suggesting that an n = 16 per group was sufficient to achieve a power (1-beta) of 0.80−0.85 and a probability of a type I error (alpha) of 0.05. p < 0.05 was considered statistically significant.
Results
The body mass from week 8 to week 21 was not different among the groups and was not influenced by diet or NAC treatment (Figure 1).
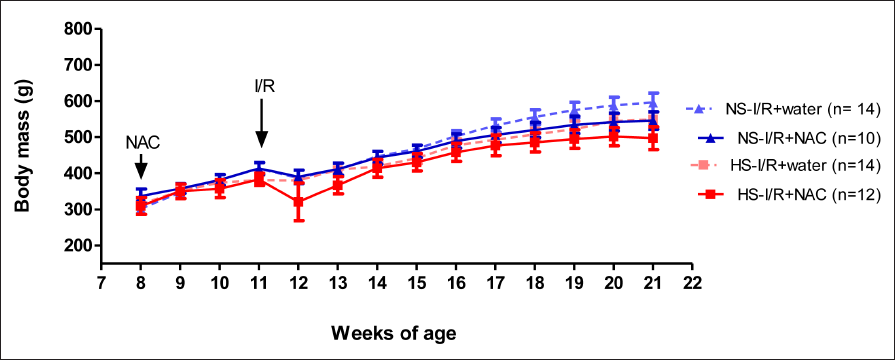
The tBP of the NS-I/R+NAC group was significantly lower than that of the NS-I/R+water group in the 1st week after I/R. In the HS-I/R+NAC group, tBP was lower in the 1st, 2nd, and 4th weeks after I/R than in the HS-I/R+water group. In the HS-I/R+water group, tBP was higher before and in the 2nd, 4th, and 10th weeks after I/R than in the NS-I/R+water group (Figure 2). These results indicated that HS increased blood pressure, and NAC prevented increased blood pressure during I/R associated with HS early but not in the late period. Meanwhile, in the NS group, NAC did not effectively influence the blood pressure in the HS group in the early period.
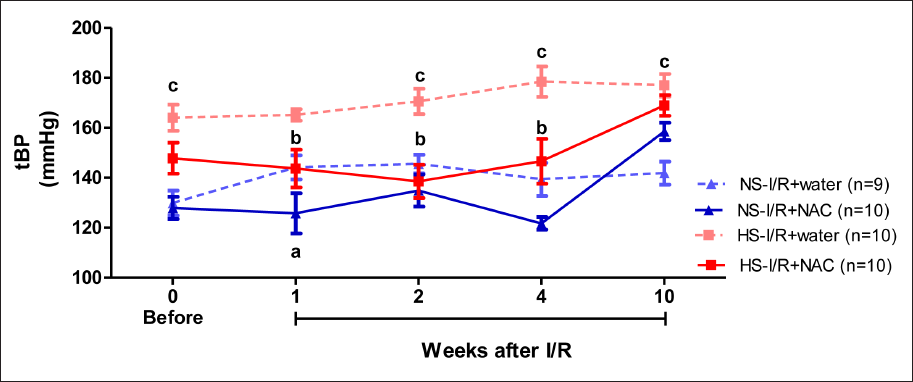
The food intake was not significantly different among the groups, independent of diet and NAC treatment, in rats subjected to I/R (Figure 3A). The liquid intake was significantly higher in the HS-I/R+water than in the NS-I/R+water at the 1st, 2nd, 4th, and 10th weeks after I/R; however, NAC treatment did not change the liquid intake between the diet groups (Figure 3B).
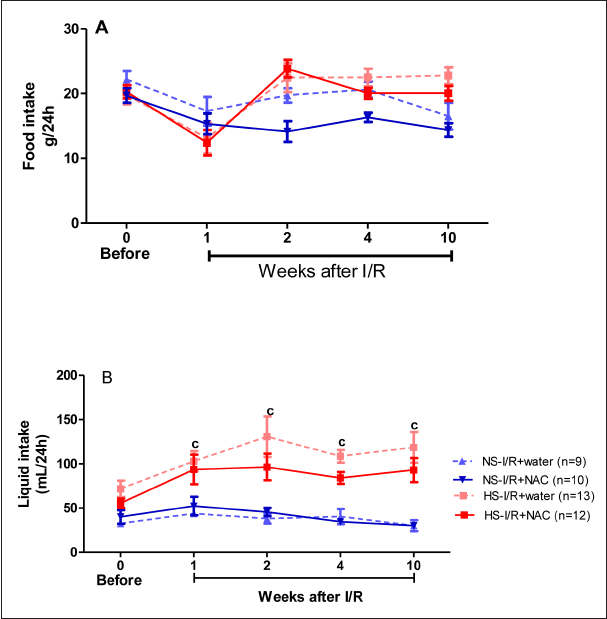
The urine volume was lower in the HS-I/R+NAC group than in the HS-I/R+water group in the 1st and 2nd weeks after I/R, while no difference was observed in the NS groups (Figure 4A). The urine volume was higher in the HS-I/R+water group than in the NS-I/R+water group before and in the 1st, 2nd, 4th, and 10th weeks after I/R. These results indicated that NAC treatment modified the urine volume profile associated with HS.
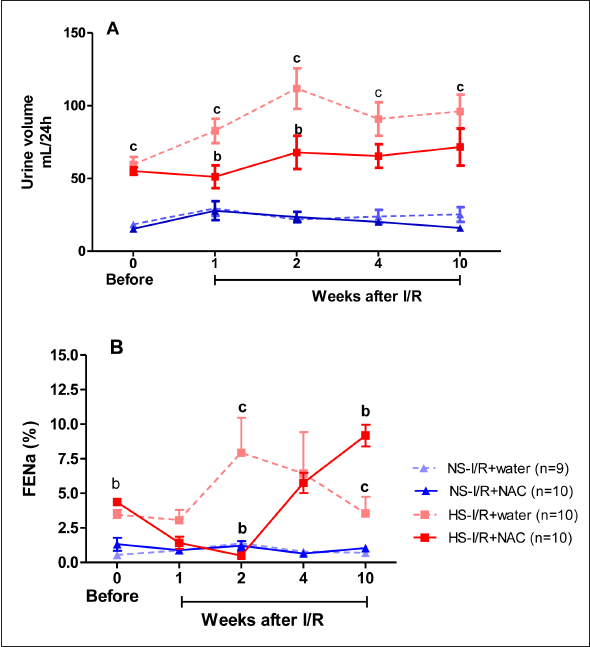
The FENa in the HS-I/R+NAC group was lower in the 2nd week after I/R and higher before and 10th week after I/R than that of the HS-I/R+water group; meanwhile, there was no change in the NS groups. The FENa in the HS-I/R+water group was higher than in the NS-I/R+water group in the 2nd and 10th weeks after I/R (Figure 4B). These results also indicated that NAC affected the FENa associated with HS.
Serum creatinine was higher before and 10 weeks after I/R in the NS-I/R+NAC group than in the NS-I/R+water group, but was higher in the HS-I/R+NAC group than the HS-I/R+water group only at 10 weeks after I/R (Figure 5A). NAC treatment reduced the serum creatinine to normal levels soon after I/R; however, NAC increased the serum creatinine 10 weeks after I/R in both groups, independent of diet. Diet or NAC treatment did not influence serum urea (Figure 5B).
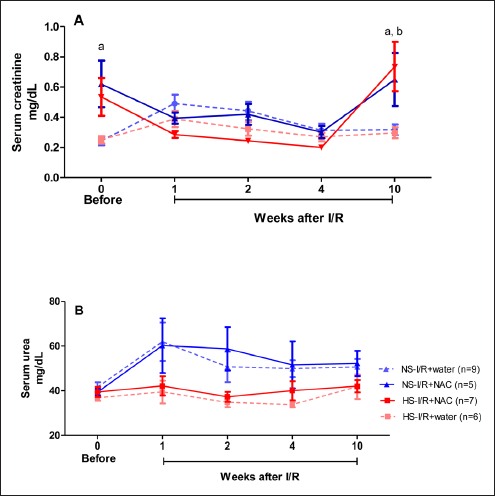
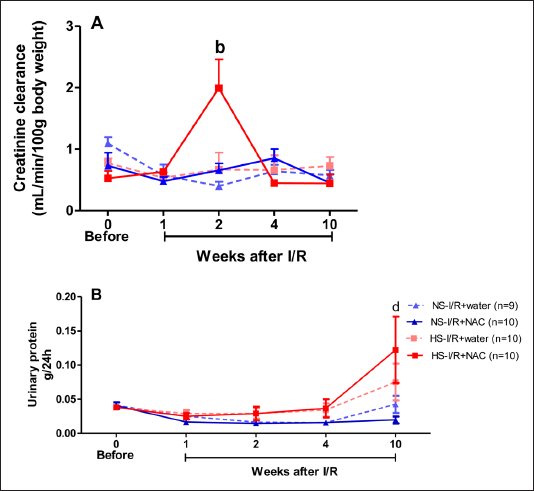
Creatinine clearance was higher in the HS-I/R+NAC group in the 2nd week after I/R than in the HS-I/R+water group (Figure 6A). Urinary protein was higher in the HS-I/R+NAC group than in the NS-I/R+NAC group at 10 weeks after I/R (Figure 6B).
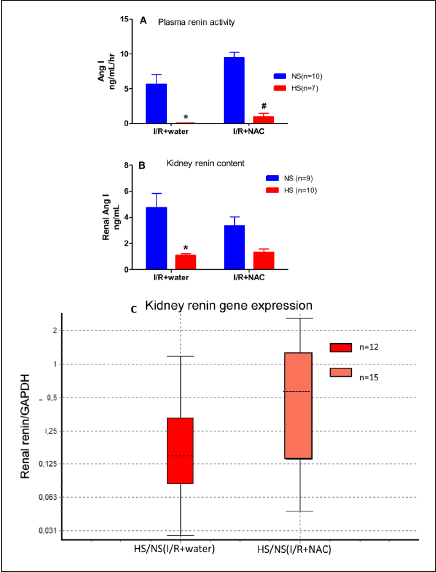
The plasma renin activity (Figure 7A) was significantly decreased in the HS-I/R+water and HS-I/R+NAC groups compared with the NS groups; however, we observed that the plasma renin activity was higher in both diet groups receiving NAC. The kidney renin content (Figure 7B) was lower in the HS-I/R+water group than in the NS-I/R+water group but was not significantly different in the HS-I/R+NAC and NS-I/R+NAC groups. The kidney renin gene expression was decreased in the HS-I/R group compared to the NS-I/R+water group; however, it was not altered in the HS-I/R+NAC group compared to the NS-I/R+NAC group (Figure 7C). These results suggest that NAC treatment increased renin activity in animals given HS.
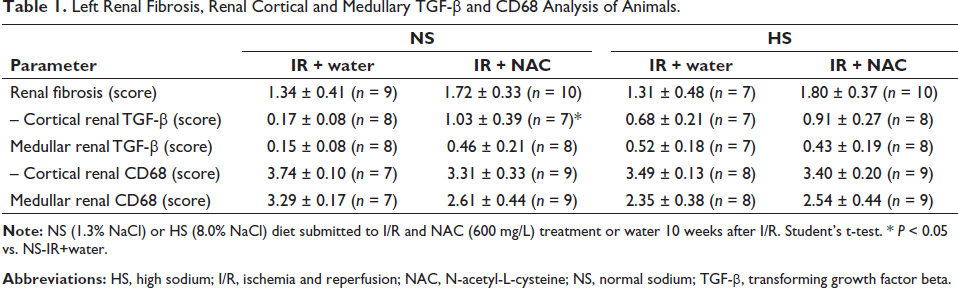
The renal cortical TGF-β score was higher in the NS-I/R+NAC group than in the NS-I/R+water group but was not different in the HS group. Renal medullary TGF-β was not different in either diet group. Renal CD68 was not different in the cortex and medulla of either diet group (Table 1).
Left Renal Fibrosis, Renal Cortical and Medullary TGF-β and CD68 Analysis of Animals.
Discussion
In our previous study, we found that long-term NAC treatment combined with HS intake did not prevent kidney dysfunction. 7 However, to gain a deeper understanding, we conducted an evaluation of various parameters every week from before I/R up until the 10th week after I/R. We aimed to determine the point at which the harmful effects of NAC treatment became apparent.
Based on the study findings, it was observed that NAC effectively managed the biochemical parameters and hemodynamic levels during the initial period. However, there was a decline in these parameters during the later period. Furthermore, NAC could not reverse the tissue damage that resulted from I/R.
The animals’ body weight was unaffected by orally administered NAC, even with varying sodium concentrations in each diet group throughout the study period.
During the early 1st, 2nd, and 4th weeks after I/R, rats who were fed HS and given NAC did not observe an increase in blood pressure, while the NS group observed a decrease only in the 1st week. In the HS group, NAC intervention helped maintain low blood pressure for a longer period, which is crucial for kidney recovery post I/R. I/R-induced kidney injury is linked to hypoxia, oxidative stress, and apoptosis.
1
NAC, an acetylated precursor of
There was no significant effect of NAC treatment on the food and liquid intakes in both the NS and HS groups. This was confirmed by the similar body mass shown in Figure 1. Although the HS+water group had a higher water intake compared to the NS+water group, NAC treatment did not affect the liquid intake in either group. The HS-I/R+NAC group had a lower urine volume and FENa in the early period, but FENa increased 10 weeks after I/R. The significant fluctuation in urine volume and FENa among rats in the HS-I/R+NAC group may be due to their HS intake. We noted that these parameters remained consistent in rats fed an NS, regardless of NAC treatment, throughout the study period. The current findings align with the study conducted by Conessa et al., 26 indicating that NAC can provide partial prevention against the rise in FENa levels during acute renal failure.
Ten weeks after I/R in the NS-I/R+NAC and HS-I/R+NAC groups, there was an increase in plasma creatinine levels and high urinary protein in the HS-I/R+NAC group. This suggests that long-term NAC treatment with HS can be harmful and contribute to deteriorating kidney function. However, in the early period after I/R, there was an increase in creatinine clearance, indicating the renoprotective effect of NAC through its vasodilation and antioxidant properties. 27 Additionally, lower blood pressure and urinary volume in the 2nd week after I/R likely contributed to this punctual increase in creatinine clearance. Previous studies on contrast-induced nephropathy have shown that NAC administration can decrease creatinine levels and reduce the risk of developing this condition.8, 28 Furthermore, NAC administration with nitroglycerin and streptokinase has been found to improve cardiac left ventricle function 29 and reduce oxidative damage in patients with myocardial infarction. 30 However, these studies were mostly conducted in acute or short-term settings. Long-term clinical outcome studies are scarce. Our study highlights that the effects of NAC are dependent on the treatment time.
There was no significant difference in the macrophages evaluated by renal CD68 between the different diet groups. The expression of TGF-β, which is also produced by macrophages, showed an increase in the NS group with NAC treatment but not in the HS group. The specific function of macrophages in prolonged kidney damage following I/R injury remains unclear. However, it is believed that various subsets of macrophages help to maintain a balance between renal injury, inflammation, repair, and fibrosis. During the restoration and fibrosis phase of I/R, macrophages are thought to have an anti-inflammatory effect by releasing cytokines like IL-10 and TGF-β, known for their anti-inflammatory properties. 31 Our analysis of kidney histology focused on the repair and fibrosis phase of I/R. The NS group treated with NAC showed increased TGF-β, which could contribute to the anti-inflammatory effect of macrophages. However, this effect was not observed in the HS group treated with NAC, suggesting that it may not be related to an anti-inflammatory effect of macrophages. In another study that involved uninephrectomy and I/R, the experimental suppression of macrophages through liposome clodronate injection decreased fibrosis and protected kidney function. 32 While it is important to understand the mechanism of macrophage suppression through experiments, investigating the role of macrophages after I/R is equally necessary.
After 10 weeks of I/R, it was observed that the HS+NAC group had increased plasma and renal renin activity and gene expression. This suggests that NAC can activate the RAAS system even when animals are given HS, which leads to the inhibition of the system for promoting sodium excretion. The activation of RAAS could cause changes in blood pressure and kidney function in rats treated with NAC and HS. The study indicates that NAC-mediated vasodilation effects could activate the renin-angiotensin system, 27 leading to renin activation, which may contribute to long-term blood pressure increases.
Conclusion
After renal I/R in animals given HS, NAC treatment was initially effective in lowering blood pressure or increasing creatinine clearance. However, these positive effects did not persist over the long term, resulting in decreased kidney function and increased blood pressure. Furthermore, the RAAS was increased by HS intake, and the benefits of the NS diet were less effective than those of the HS diet. Thus, NAC provides temporary protection only in the early stages following an insult.
Footnotes
Acknowledgments
The authors express their gratitude to Mirela Coutinho, Marcia Ribalta, and Kelly G Lehns for conducting the biochemical determinations.
Author Contributions
Rafael Canavel Pereira and Carolina Martinez Romão equally performed all protocol experiments. Beatriz Santos Geoffroy Corrêa was responsible for the renal immunohistochemistry and microscopy analysis; Wagner Vasques Dominguez handled the statistical analysis; Luzia Naoko Shinohara Furukawa played a significant role in planning, performing the study, and writing the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Research Ethics Coordination from the School of Medicine of São Paulo University approved the experimental study on protocol 037/14.
Funding
The Fundação de Amparo à Pesquisa supported this work do Estado de São Paulo (FAPESP) (grant number 2013/25909-1) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
