Abstract
Commensal microorganisms heavily influence human health and disease pathogenesis, and the impact of the skin microbiome on numerous skin illnesses has recently piqued researchers’ interest, in addition to the gut microbiome. Probiotics are live microbial organisms that are good for the host’s health when given in sufficient proportions. The concept of probiotics has sparked much curiosity and scientific investigation since its inception. Probiotics alter the intestinal microbiota and are employed as a treatment technique for a variety of diseases. Despite several studies, the significance of probiotics in dermatological illnesses such as acne, vaginal infections, and atopic dermatitis has yet to be demonstrated. Evidence pointing to the “gut-skin axis” link has increased, and intestinal microbiota regulation may play a role in dermatological diseases. There is, however, no consensus on the species or the dosage to be utilized for such therapies. This article makes an effort to review recent evidence from the literature.
Introduction
The Greek term “pro bios,” meaning “for life,” was proposed by several sources like Werner Kollath and Ferdinand Vergin, and in 1965, Lilly and Stillwell coined the term “probiotics.” In the early twentieth century, a Russian doctor named Elie Metchnikoff linked the lifespan and health of Bulgarian peasants to the consumption of Lactobacillus-rich yogurt. In ancient Greece and Rome, children and people recovering from illness were given fermented milk products.
According to the United Nations Food and Agriculture Organization, probiotics are live bacteria capable of delivering health benefits to the host when given in sufficient quantities. Lactobacillus and Bifidobacteria are the most commonly utilized bacteria as probiotics and are commonly available as tablets, powders, beverages, and fermented dairy products. 1 The first generation of probiotics is composed of naturally occurring, non-engineered strains, whereas the genetically designed ones belong to the second generation. 1
The probiotic bacteria were initially thought to modulate host immunity by remaining and proliferating in the gastrointestinal tract. However, investigations have demonstrated that swallowed bacteria only persist for a short time after dosage and activate underlying cells in the lamina propria by certain signaling cascades. Probiotics control important signaling pathways such as nucleotide oligomerization domain (NOD)-like receptors, nuclear factor kappa B, and mitogen-activated protein kinase through toll-like receptors (TLR) to achieve the desired impact. 1 They cling to the intestine’s mucosa in a competitive manner, strengthening the gut-epithelial barrier. 1
Prebiotics are indigestible foods that promote the survival and growth of beneficial bacteria in the colon, which benefits the host. Substances like trans-galactooligosaccharides (oligosaccharides in human milk) and inulin meet the prebiotic criteria.
Synbiotics are a type of supplement that combines prebiotics with probiotics. The oligosaccharides in human milk have a prebiotic effect on bifidobacteria. Hence, supplementing formula feeds with comparable oligosaccharides lowers allergy symptoms.
Methodology
We searched the PubMed database and Google Scholar for studies on the use of probiotics using keywords such as atopic dermatitis, acne, psoriasis, rosacea, aging, melasma, and recent advances in probiotics. Original articles, reviews, and meta-analyses on the themes addressed were preferred. Our review included more than 15 articles that were extracted from publications between 2010 and 2022. The findings of these studies were synthesized into a narrative review.
Role of Probiotics in Systemic Immunity
Probiotics can boost overall immunity through immunomodulation. In the process of immunomodulation, gut probiotics interact with macrophages and dendritic cells, which are antigen-presenting cells (APC). The population of T cells is then impacted by these APCs, leading to systemic immunomodulation (lowering sensitivity to various stresses and allergens). This immunomodulation through the gut-brain axis, gut-lung axis, and gut-brain-skin/gut-skin axis enhances the function of the brain, lungs, and skin, respectively (Figure 1 and Table 1).
The Importance of Gut Probiotics in Human Health.
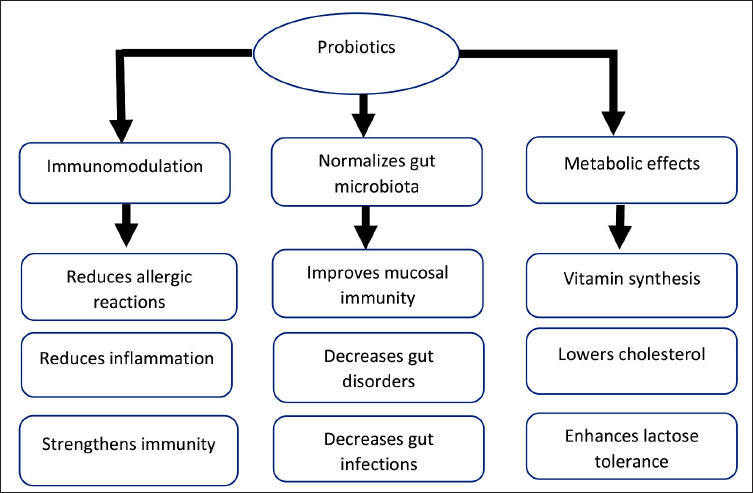
Benefits of Gastrointestinal Flora.

Dermatological Conditions in which Probiotics Play an Important Role
Seborrheic dermatitis.
Acne vulgaris.
Psoriasis vulgaris.
Rosacea.
Atopic dermatitis.
Bacterial vaginosis.
Vulvovaginal candidiasis.
Aging.
Oral mucositis.
Wound healing.
Psoriasis
Psoriasis is a hereditary immune-mediated papulosquamous disorder affecting the skin, joints, and nails. It has a variety of clinical manifestations, some of which can be more severe than others, but all of which share common symptoms like scaly plaques, inflammation, and tissue stiffness.
Cytokines produced by Th17 cells, such as interleukin-17, interleukin-22, and interleukin-23, are thought to play a crucial role in psoriasis pathogenesis, despite their unknown pathophysiology. The microbiome of the gut is believed to play a role in the activation of pro-inflammatory Th17 cells and the development of psoriasis. 2
Patients with inflammatory bowel disease and psoriasis have similar dysbiosis patterns, indicating a link between the gut microbiome and the skin axis. This dysbiosis is characterized by the presence of a smaller number of symbiotic bacteria, such as Bifidobacterium species, Lactobacillus species, and Faecalibacterium prausnitzii, as well as the colonization by pathobionts like Salmonella species, Escherichia coli, Helicobacter species, Mycobacterium species, Campylobacter species, and Alcaligenes species. In addition, Staphylococcus aureus colonizes the skin of psoriasis patients in greater numbers than that of people who do not have dermatoses.
Reduced quantities of helpful bacteria can have negative implications, such as alterations in specific inflammatory mediators, which can have an impact on distant organs. Thus, probiotic supplements can be quite helpful in the treatment of psoriasis. Oral delivery of Lactobacillus pentosus (GMNL-77) reduced epidermal thickness and erythema in a mouse model of imiquimod-induced psoriasis compared to a placebo. Inflammatory cytokines like tumor necrosis factor-alpha (TNF-α), interleukin-6, interleukin-17A, and interleukin-23 have their mRNA levels dramatically lowered by the therapy. L. pentosus (GMNL-77) also reduced the weight of the spleen in the imiquimod group and the count of CD4 T cells producing interleukin-17 and interleukin-22 in the spleen, according to the study. 3
Furthermore, compared to a placebo, supplementation with Bifidobacterium infantis 35624 resulted in a drop in inflammatory markers like C-reactive protein (CRP) and TNF-α in individuals with psoriasis.
Szántó et al. in their article, in a nutshell, described the effects of oral probiotics on psoriasis and found that they have been shown to improve the condition significantly. 4 Navarro-López et al., in their randomized controlled clinical trial, studied the effects of probiotics in 90 patients with psoriasis randomized into two groups (probiotics and placebo) for a period of 12 weeks and reported that probiotics significantly reduced the physician global assessment index and, in addition, showed a reduced risk of relapse six months after the end of the study. 5 On the contrary, another older randomized controlled trial by Kaur et al. studied the effects of a Lactobacillus nutritional supplement on 40 patients (18 receiving the supplement and 22 on placebo) for a period of 12 weeks with no significant improvement. 6 Vijayashankar et al. reported a case of pustular psoriasis resistant to steroids, methotrexate, and dapsone, wherein supplementing with Lactobacillus sporogens thrice daily and Biotin 10 mg once daily showed considerable improvement. 7 However, the data are limited and uneven, with few human trials, thus hindering a proper supplementation protocol.
Acne
Acne affects the pilosebaceous unit and appears as an inflammatory eruption of papules and pustules or as non-inflammatory eruptions such as open or closed comedones. The condition is characterized by excessive production of sebum, hyperkeratinization of follicles, and elevated pro-inflammatory cytokines.
The beginning of the disease could be linked to a number of circumstances. Acne is well-known to be associated with a high carbohydrate diet, and high glucose levels produce insulin and insulin-like growth factor-1 (IGF-1) in the sebaceous glands, boosting sebocyte, keratinocyte proliferation, and lipid production. The significance of the bacteria Cutibacterium acnes in the pathogenesis of acne has been extensively investigated, though not understood. C. acnes has been demonstrated to stimulate the skin’s IGF-1 system/IGF-1 receptors, just as the intestinal flora can. As a result, it is possible that an imbalance in the gut flora can lead to increased sebum production and skin colonization by C. acnes, disrupting the skin flora’s delicate balance and thus triggering the disease.
Supplementation with probiotics can be used as an adjuvant therapy for treating acne. In vitro investigations revealed that certain strains like Lactobacillus salivarius LS03, Streptococcus salivarius, and Lactococcus produces bacteriocins that inhibit the growth of C. acnes. 8
Bifidobacterium adolescentis SPM0308, due to its antimicrobial activity, was found to be effective in controlling the growth of both C. acnes and S. aureus. 8
Gueniche et al. wrote about the beneficial effects of Lactobacillus paracasei CNCM I-2116 on the release of TNF-α induced by substance P, a key mechanism associated with the skin barrier function. 9
Streptococcus thermophiles were found to boost beneficial lipid production, like ceramides in the stratum corneum, which helps the skin maintain moisture, and phytosphingosine, which helps fight C. acnes. 8
In addition, certain strains like Lactococcus lactis NCIMB 66, Lactobacillus casei NCFB 161, Lactobacillus gasseri NCFB 2233, Lactobacillus acidophilus NCFB 1748, Lactobacillus plantarum DSM 12028, and glucomannan hydrolysates of Amorphophallus konjac can also inhibit the growth of C. acnes. 10
Chae et al. and Espinoza-Monje et al., in their in vitro studies, investigated the benefits of Lactiplantibacillus plantarum and Weisella viridescens in acne.11, 12 While L. plantarum, derived from green tea, inhibited different skin pathogenic strains (five pathogenic skin strains, such as S. aureus, C. acnes, Candia albicans, Malassezia globosa, and Malassezia restricta), W. viridescens, in both oral and topical formulations, reduced the replication of C. acnes and reduced the adhesion of the organism to the keratinocytes.11, 12
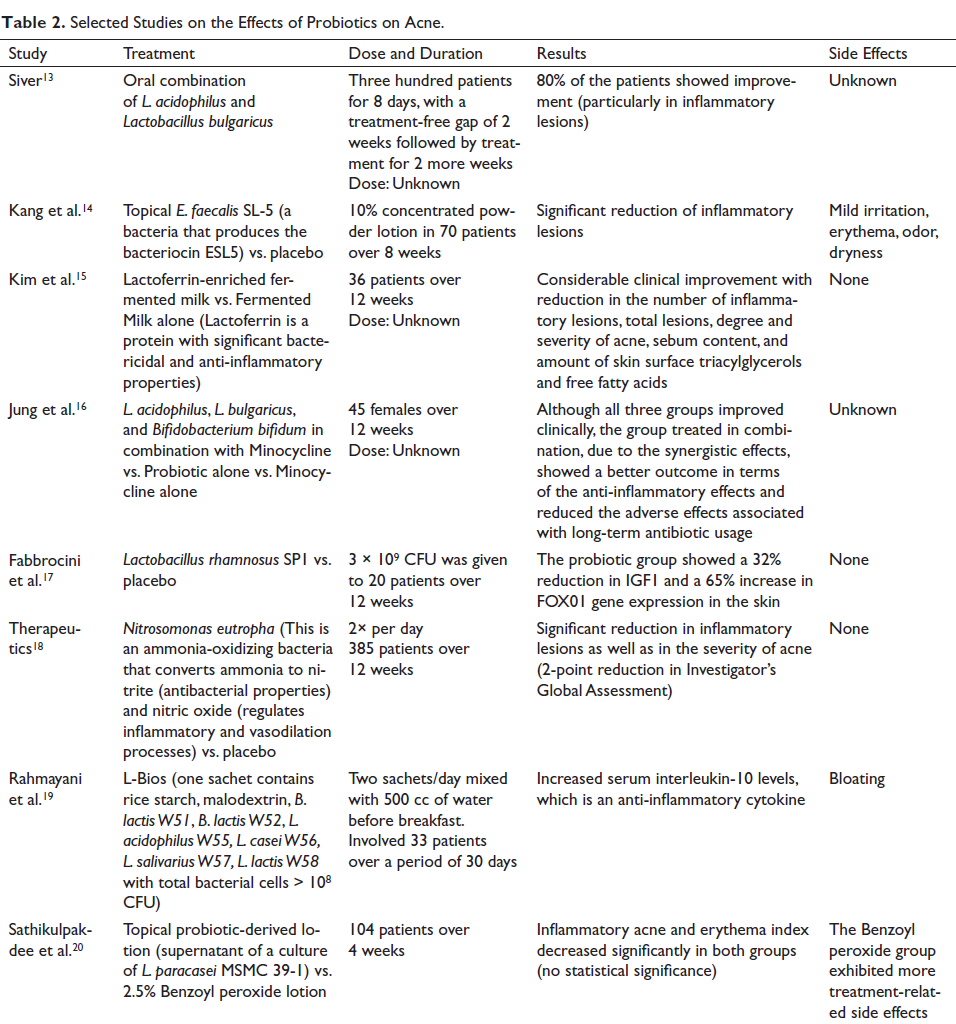
There are several clinical studies, as mentioned in Table 2, on the effects of probiotics on acne, with the first study started in 1961 by Siver. 13
Selected Studies on the Effects of Probiotics on Acne.
Atopic Dermatitis
Atopic dermatitis is characterized by excessive colonization of harmful microorganisms on the skin due to epidermal barrier malfunction (Figure 2) and immunological dysregulation. 21 High colonization by bacteria such as S. aureus is directly associated with the severity of the disease and a loss in the diversity of the microbiome during episodes of the disease.22, 23 Effective therapies have been linked to microbiota recolonization and a greater diversity of bacteria in the same direction. 23 Because the intestinal microbiota is linked to the clinical outcome of atopic dermatitis, it has emerged as a potential treatment option.
Epidermal Barrier Malfunction.
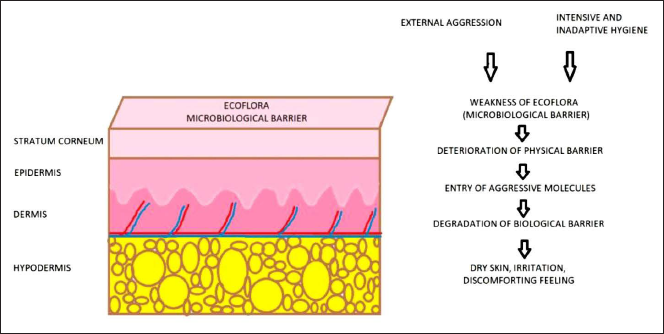
It is important to note that patients with atopic dermatitis had a higher skin surface pH than individuals with normal skin. Whether the cutaneous dysbiosis of atopic dermatitis (AD) patients is a cause or consequence of elevated skin pH is yet unclarified, but the application of a topical probiotic, Lactobacillus johnsonii, for 3 weeks was shown to be highly effective in bringing down colonization of S. aureus and providing symptomatic relief to these patients. 24 In addition, the metabolites of Lactobacilli can also play a role in reducing skin surface pH.
IgE-linked eczema in children has a reduced proportion of Bifidobacterium species and less microbiota diversity, according to a study. Another study found that early colonization of E. coli in the intestine during the second month of life may have long-term health benefits, as these children had a decreased incidence of atopic dermatitis when they were six years old. Recent meta-analyses demonstrated that intestinal modulation in children could lower atopic dermatitis scoring values (SCORAD). 24 However, details on the most effective dose, the optimum time to administer it, and the most efficient strains for treatment are yet unknown. 25
A study of the literature looked at various complementary and integrative therapies (CIT) for treating kids with atopic dermatitis. 26 The analysis found that probiotic supplementation is still the best CIT for treating pediatric Alzheimer’s disease, as evidenced by trials. Treatment of children aged 12 months and older with Lactobacillus fermentum and L. plantarum had the most solid evidence. Each probiotic was studied over the course of 12 months in two separate randomized clinical studies. When taken alone, without adding other probiotic strains, the studies revealed a decrease in atopic dermatitis scoring values (SCORAD), that is, the improvement was clinically significant. Another investigation of the treatment using L. plantarum, however, found that it was ineffective.
Vaginal Infections
Lactobacilli predominate in the vaginal flora in good health. These commensals are thought to play an important role in preventing vaginal infections by creating hydrogen peroxide, acetic acid, and lactic acid that keep the vaginal pH at 4.50 or below. At this pH, harmful bacteria, including C. albicans, cannot grow, limiting the spread of diseases. The vaginal microbiota, in addition to lactobacilli, also contains modest quantities of Clostridium, Bacteroides, Peptococcus, Gardnerella, Streptococcus, Enterococcus, and Staphylococcus epidermidis.
Bacterial vaginosis (BV) is one of the most common vaginal infections that can have serious gynecological and obstetric consequences. Mycoplasma hominis, Gardnerella vaginalis, Peptostreptococcus, Prevotella, Mobiluncus, and Bacteroides species prevail in the vaginal microbiota of BV patients, but lactobacilli are significantly less common than expected. It has been proven that lactobacilli suppress the growth of the pathogenic bacteria causing BV, create hydrogen peroxide, lactic acid, and bacteriocins, and inhibit the adhesion of G. vaginalis to the vaginal epithelium.
According to a review of probiotics in the treatment of BV, increasing the number of lactobacilli in the vagina can cure or reduce the recurrence of BV. However, the same study uncovered other trials that found no meaningful benefit for BV when lactobacilli were instilled intravaginally. A meta-analysis of randomized controlled trials comparing the efficacy of metronidazole combined with probiotics to metronidazole alone in the treatment of BV published in 2017 found only a small amount of evidence, indicating that more research is needed. 27
In 2017, a Cochrane evaluation of probiotics in the treatment of vulvovaginal candidiasis in women who were not pregnant found a rise in short-term mycological and clinical cure rates and a decrease in relapse rates after one month. Also, it was discovered that this did not improve mycological or long-term clinical cures. 28 The potential of L. plantarum (P17630) in restoring the vaginal microbiome was discovered in a study, and given the possibility of antifungal resistance in vulvovaginal candidiasis, its role needs to be investigated further. 29
Photoaging and Melasma
Ultraviolet radiation is the most significant extrinsic aging inducer. Ultraviolet radiation exposure has been proven to cause major changes in the immune system of humans, including a reduction in the number of Langerhans cells as well as a change in the shape and ability of these cells to deliver antigens. Additionally, an increase in immunosuppressive cytokines such as interleukin-10 has been seen. Lactobacilli could be used as an alternative to ultraviolet radiation for skin protection.
A mouse study found that supplementing L. johnsonii (La1) could protect skin against the detrimental effects of ultraviolet radiation. 30 Human research studied the effects of giving L. johnsonii (La1) along with 7.2 mg of carotenoids to healthy women who had been exposed to simulated or natural sunlight for ten weeks. 31 The outcome was that dietary supplementation, compared to a placebo, hastened immune system homeostasis recovery after ultraviolet radiation exposure. The minimal erythema dose (MED) increased by 20% in those who received supplementation. The study demonstrated the advantage of the connection despite necessitating a comparison between carotenoids and isolated probiotic therapy. 31
A human study looked at how efficient a supplement combining L. johnsonii, beta-carotene, and lycopene was at treating melasma. 32 When the Melasma Area and Severity Index (MASI) scale and Taylor scale were used to analyze the outcome, considerable improvement in melasma was noted in the treated group. When compared to mice given a placebo, oral Bifidobacterium breve administration reduced ultraviolet-induced transepidermal water loss in hairless mice. B. breve also inhibited the ultraviolet-induced increase in levels of hydrogen peroxide, xanthine oxidase, and protein oxidation activity in the skin of mice. 33 Another study with mice found that oral L. acidophilus prevented the appearance of fine wrinkles caused by ultraviolet B exposure. 34 This protection was attributable to a decrease in matrix metalloproteinase (MMP) expression, including MMPs one and nine.
Rosacea
It has also been looked into to see if gut bacteria and their products play an essential role in developing skin lesions like rosacea. Patients with inflammatory skin disorders had an unbalanced gut microbiota, according to a clinical investigation. Small intestinal bacterial overgrowth (SIBO) was shown to be substantially more common in patients with rosacea than in people without the condition. Furthermore, and more importantly, SIBO removal caused rosacea to almost completely disappear. In a study, treating a rosacea patient with B. breve BR03 and low-dose doxycycline in combination with L. salivarius LS01 improved skin and eye symptoms. 35 Patients should be advised to keep their intestinal microbiome healthy by eating a fiber-rich diet (prebiotics) or taking oral probiotics to modulate their intestinal microbiota.
Rejuvenation
Other aspects of skin aging may benefit from probiotic use since probiotics can affect both internal and extrinsic elements, such as genetics, hormone status, and oxidative metabolic processes, as well as extrinsic ones like sun exposure, cigarettes, and psychological stress. Normal, healthy skin has a slightly acidic pH that ranges between 4.2 and 5.6, which helps to prevent pathogenic bacterial colonization, control enzyme activity, and maintain moisture. The pH of the skin increases dramatically after the age of 70, boosting the action of proteases. The metabolism of probiotics typically produces certain acidic molecules, which lower the pH; hence, utilizing probiotics could help restore the skin’s normal pH.
A placebo-controlled clinical trial evaluated people between the ages of 41 and 59 years who had wrinkles and dry skin. After 3 months of administration of 1 × 1,010 colony-forming units (CFU) per day of HY7714 (L. plantarum), there was a dramatic reduction in transepidermal water loss and wrinkle depth, as well as enhanced skin brightness when compared to day zero. 36 Furthermore, the group treated with probiotics had an increase in skin elasticity of 21.733%, a significant difference from the placebo group. These findings show that probiotics could be used as a nutricosmetic.
Probiotics in Wound Healing
The topical use of probiotics has been studied for the prevention of infection, inflammatory management, and healing enhancement since wounds are linked to changes in local microflora due to injury and immune response activation. A few studies using both human and animal models have shown they are helpful; however, there is a lot of variation between the investigations. 37
The effects of L. plantarum, kefir, L. fermentum, and Saccharomyces cerevisiae have been studied in animal models with thermal wound injuries, diabetic ulcers, and uninfected wounds with topical antimicrobial activity against Shigella, Salmonella, Helicobacter, and Staphylococcus, and E. coli. 38 It was discovered by Valdéz et al. that when L. plantarum was used as a therapeutic agent for the local treatment of Pseudomonas aeruginosa burn infections, it altered interleukin-8 production; promoted wound healing; decreased bacterial load, neutrophils, necrotic and apoptotic cells; and resulted in positive outcomes. 39
Other Uses
Probiotic use was found to be inversely related to the occurrence of skin lesions in patients suffering from inflammatory bowel disease, suggesting that probiotics could be used as a therapy tool. Probiotics have been shown to improve skin suppleness and hydration and reverse ultraviolet-induced aging; therefore, research into their anti-aging potential is ongoing.
Potential Adverse Effects of Probiotics
In a joint report published in 2002 by the United Nations Food and Agriculture Organization and World Health Organization, a few theoretical concerns were raised: the risk of various systemic infections, harmful metabolic activities such as D-lactate production, deconjugation of bile salts, excess immune stimulation, and the risk of gene transfer to certain other bacteria. Although incidences of fungemia and bacteremia have been documented following probiotic consumption, research from Finland and Sweden has found no change in the prevalence of Lactobacillus bacteremia despite increased Lactobacillus probiotic consumption over a prolonged study period. Gastrointestinal side effects like nausea, diarrhea, flatulence, abdominal cramping, and taste disturbances have been reported.
Patients can be advised to eat a fiber-rich diet (prebiotics) or use oral probiotics to modulate the intestinal microbiota to maintain a healthy intestinal microbiome.
Kang et al., in their study involving topical Enterococcus faecalis SL-5, reported minor side effects like irritation, erythema, odor, and skin dryness. 14
Recent Advances
Fecal Microbiota Transplantation
Fecal microbiota transplantation (FMT), also known as fecal/stool transplantation or fecal bacteriotherapy, is a treatment for a specific condition that involves infusing or engrafting filtrate excrement from any healthy donor into a recipient’s stomach. FMT now plays a part in the treatment of inflammatory and infectious skin disorders. Recent case reports, including two cases of alopecia universalis, expressed regrowth of hair on various body parts following FMT. Additionally, after FMT, patients with psoriatic arthritis and psoriasis exhibited clinical improvement. Additionally, several mouse studies have tried to comprehend the function of FMT in the treatment of melanoma. 40
Using GI upper endoscopy and colonoscopy, a patient who had been diagnosed with severe plaque psoriasis for a period of 10 years and irritable bowel syndrome (IBS) for a period of 15 years was given FMT twice with a 5-week interval. Psoriasis area and severity index (PASI), body surface area (BSA), dermatology life quality index (DLQI), intestinal symptoms, histological examination, adverse reactions, and serum TNF levels were utilized to evaluate responses. The aforementioned elements significantly improved after the second FMT treatment compared to before. Additionally, the IBS was fully resolved, and neither the treatment nor the follow-up had adverse effects. Finally, FMT might be a cutting-edge treatment for psoriasis. Additional clinical studies are required to provide reliable evidence. 41
Topical Probiotics
The past few years have seen a rise in the commercially available topical probiotics, considered safer alternatives to conventional oral/topical therapies, and a dearth of studies reporting the efficacy and safety of the same. Topical probiotics may help enhance the skin’s natural barrier by having a direct effect at the site of application. This may be due to the resident bacteria and the probiotic bacteria that produce certain antimicrobial amino peptides that benefit the immune responses in the skin and help eliminate pathogens.
Topical probiotics can prevent pathogenic flora by binding to receptor sites and consuming nutrients in a competitive manner. They also trap infections by increasing mucin and ceramide synthesis and altering the immunological responses of the host. In addition, they also help with tissue healing and reduce biofilm formation. As a result, they can be utilized to compensate for the changes in cutaneous flora in acne, wound healing, atopic dermatitis, seborrheic dermatitis, and heat-damaged skin. Topical Bifidobacterium longum lysate treatment reduced skin sensitivity, dryness, and aging, sparking interest in topical probiotics for cosmetic purposes.
Although antimicrobials have been used in washing liquids to kill the pathogens in clothes, a newer trend is the incorporation of probiotics in the form of a paste in the lining of hospital apparel, which stays on for many washes. This evolving use of probiotics could prevent nosocomial infections and keep pathogens at bay. 38
Commercially, these probiotics are present in every cosmeceutical over-the-counter product like sunscreens, eye serums, day serums, and moisturizers, with the drawback being that these commercial probiotics are unsterile with a high bacterial load, and the preservatives used can inadvertently affect the cutaneous flora, negatively.
Conclusion
This review highlights the dermatologist’s viewpoint and the therapeutic effects of helpful species of probiotics as one of the alternatives and secure techniques for treating patients suffering from cutaneous ailments. Our understanding of how commensal bacteria can be used to treat various illnesses using high-throughput genomic technology and the selection of bacterial strains is particularly crucial because the effects of probiotics may vary greatly depending on the strain. Probiotics can also affect the gut-skin axis and systemic health in patients with a variety of disorders. Given the growing body of information on probiotics and the crucial role they play in maintaining human health, their use as an integrative therapy can open up new therapeutic options that can be used by dermatologists.
Footnotes
Abbreviations
APC, antigen presenting cells; TNF-α, tumor necrosis factor-alpha; IGF, insulin-like growth factor; CIT, complementary and integrative therapies; FMT, fecal microbiota transplantation; CFU, colony-forming unit; MMP, matrix metalloproteinase; SIBO, Small intestinal bacterial overgrowth; IBS, irritable bowel syndrome; PASI, Psoriasis area and severity index; BSA, body surface area; DLQI, dermatology life quality index.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Ethical committee approval and consent forms were not required as this is a review article and did not include any patients.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
