Abstract
Objective
To describe the current scientific evidence of food-derived bioactive peptides and their angiotensin-converting enzyme inhibiting effect.
Methods
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) were followed. We searched MEDLINE (through PubMed) and Science Direct databases to identify studies assessing food-derived bioactive peptides and angiotensin-converting enzyme-inhibiting effects. The evidence was organized and presented using tables and narrative synthesis.
Results
We identified 11 peptides with the best antihypertensive potential: RDGGYCC, LRLESF, FHAPWK, and LVLPG from plants; LSGYGP, ITT, VISDEDGVTH, ATT, and LWHTH from animals; and ALGRV and SPQW from fungi, which demonstrated their antihypertensive potential in vitro and in vivo.
Conclusion
Overall, food-derived bioactive peptides with hypertensive activity were identified, which shows a promising field as a therapeutic alternative to conventional pharmacological treatments.
Introduction
Hypertension (HTN) is a major global health challenge; its prevalence and impact on the cardiovascular system, brain, and kidneys remain the leading preventable risk factors for premature death and disability worldwide.1–4 This disease is characterized by sustained elevation of blood pressure above 120/80 mmHg in subjects aged more than 18 years. 1 Its etiology is unknown in most cases (85%–90%), called primary hypertension. The remaining 10%–15% is secondary to other pathologies such as pheochromocytoma, hyperaldosteronism, alterations in thyroid function, acromegaly, and polycystic kidney disease.1,3,4
HTN is known to significantly affect cardiovascular conditions such as heart failure, myocardial infarction, and stroke. Like other diseases, HTN increases with age, and its prevalence increases from 27% in patients under 60 years of age to 74% in those over 80 years of age. 5
The current pharmacological regimens for the treatment of HTN include the use of Thiazide diuretics (hydrochlorothiazide) and angiotensin-converting enzyme inhibitors such as captopril, enalapril, and lisinopril, angiotensin II receptor blocker (Losartan potassium and valsartan), β-blockers (metoprolol and atenolol), or calcium channel blockers (nifedipine, amlodipine, and diltiazem). 6 However, these synthetic drugs often cause unwanted side effects such as chronic dry cough, taste disturbances, difficulty in swallowing or breathing, headaches, insomnia, diarrhea, allergic reactions, inflammatory responses, angioedema, hyperkalemia, tachycardia, decreased blood pressure, dizziness, ankle swelling, chest discomfort, fatigue, and even leukocytopenia,6–8 which increase therapeutic failures due to lack of adherence to the medication, giving rise to a focus on promoting complementary non-pharmacological alternatives such as physical activity, reduction of alcohol consumption, and a balanced diet in order to mitigate the unwanted effects of antihypertensive drugs. 9
Due to the above, the use of natural alternatives for therapeutic purposes has gained significant interest in recent years for the control of HTN. Foods such as milk, eggs, meat, and corn have presented antihypertensive properties attributed to their significant components, such as proteins; 10 peptides derived from the metabolism of these proteins are increasingly being studied worldwide, including their biological properties on the modulation of human health. 11
Peptides are small, isolated protein fragments, usually consisting of 2–20 amino acids linked by peptide bonds, whose functionality can provide the necessary nutrients for human growth and development. 11 Among them, we can find bioactive peptides or peptides with biological activity produced during gastrointestinal digestion or food processing. They could play an essential role in metabolic regulation and modulation, suggesting their potential use as nutraceuticals and functional food ingredients to promote health and reduce the risk of diseases. 12
In recent years, efforts have been made to study the various potential beneficial activities of these bioactive peptides in the body, including their antihypertensive, hypocholesterolemic, antioxidant, antimicrobial, immunomodulatory, and opiate-like activities. Likewise, research is currently being carried out to detect food sources of bioactive peptides as well as to study their bioavailability, functional properties, and mechanisms of action. 13
Bioactive peptides with antihypertensive activity have explicitly been of great interest to the scientific community, as they are one of the most prevalent chronic pathologies worldwide and the leading cause of death in developed countries.14,15 Most peptides described in the literature with antihypertensive properties can inhibit angiotensin-I converting enzyme (ACE-I), a key regulator in the renin-angiotensin system, which leads to the production of angiotensin II, and a peptide that acts as a vasoconstrictor agent implicated in the exacerbation of HTN. 10 Thus, this review describes the current scientific evidence for food-derived bioactive peptides and their angiotensin-converting enzyme-inhibiting effects.
Methods
Type of Study
A systematic review was carried out as per Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (PRISMA). 16
Search Strategy
A systematic electronic search of the literature was carried out in the MEDLINE (through PubMed) and Science Direct databases to identify studies evaluating the antihypertensive property of bioactive peptides from food, using the filter from 2017 to 2021. Strategies were used for searches adapted to each database and using the following keywords: (“Antihypertensive Agents” [Mesh]) AND (“HTN” [Mesh]) AND (ACE inhibitory) AND (Angiotensin-converting enzyme inhibit*) AND (Antihypertensive activity).
Studies Selection
Studies that met the following inclusion criteria were selected: (i) Type of food from their origin: meat (chicken, pork, fish, beef, etc.), cereals (rice, soy, corn, etc.), dairy products (milk, cheese, etc.), sausages (hams, etc.), eggs, and so on. (ii) Evaluation of antihypertensive activity: we selected bioactive peptides whose antihypertensive activity would have been evaluated in animals or human cells. (iii) Sequence characterization: we selected bioactive peptides with their amino acid sequence established in the literature. All articles where peptide activity was not proven, reviews, book chapters, editorials, case reports, and articles where peptide activity was not antihypertensive were excluded.
All reports obtained were stored on the Rayyan platform, 17 and once duplicates were eliminated, titles and abstracts were examined independently by two reviewers, considering the inclusion and exclusion criteria. Then, the full texts of all relevant articles in the first phase were obtained, reviewed, and determined to meet the eligibility criteria to make a final decision. In case of disagreement between the two reviewers, a third collaborator participated in the selection process until an agreement was reached. For all articles considered relevant for the review and subsequently excluded, the reason for exclusion was clearly stated (Appendix A).
Results
Selection of Studies
The search in the databases yielded 1,009 studies, two of which were duplicates and were eliminated, obtaining 1,007 articles. When examining the titles and abstracts, 78 studies were selected and subsequently examined in full text, applying the inclusion and exclusion criteria. Finally, 23 studies were obtained to write this review (Figure 1).
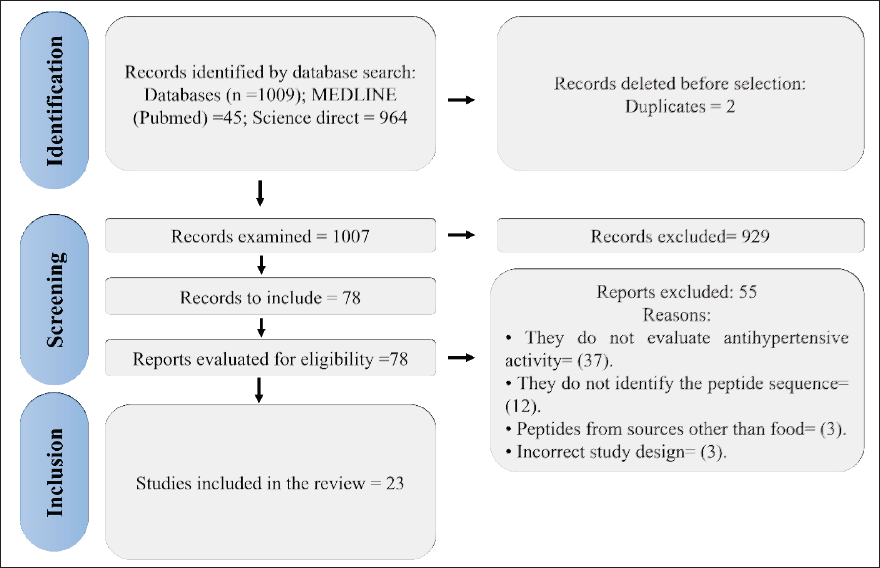
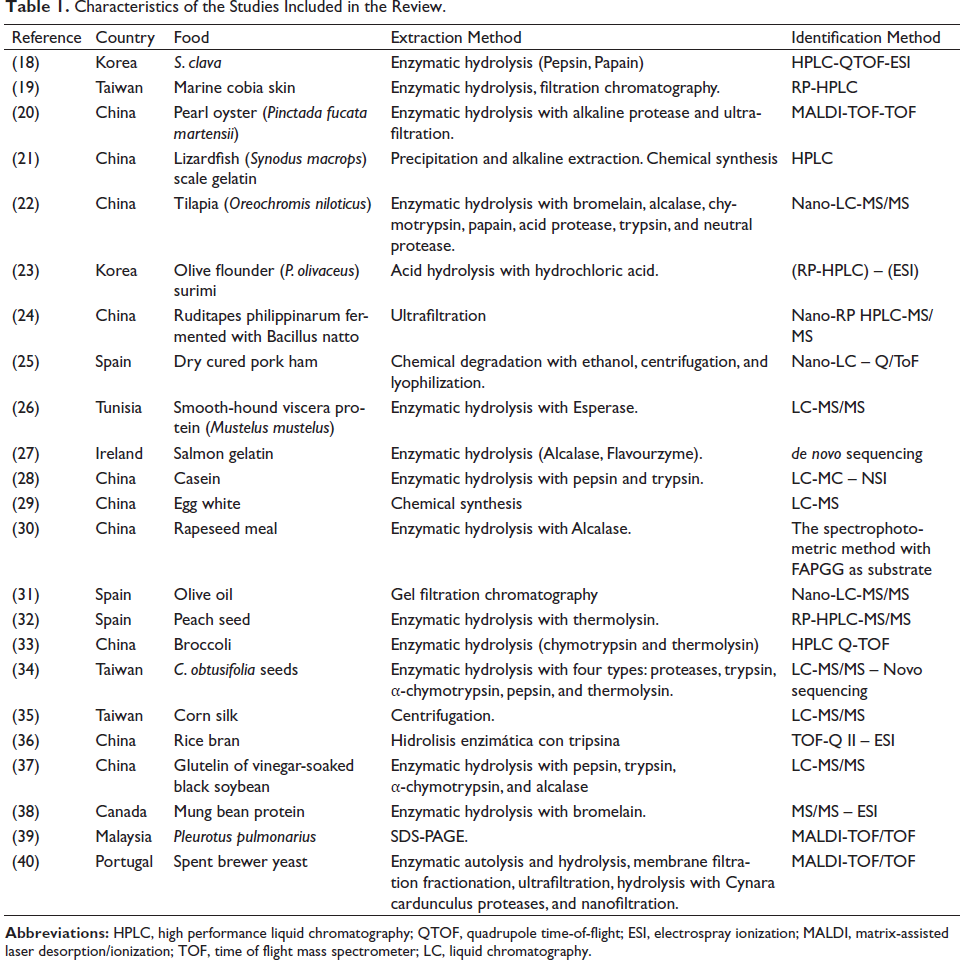
Characteristics of the Selected Studies
The characteristics of included studies are shown in Table 1. The studies were divided according to origin sources: animal,18–29 plant sources,30–38 and fungi.39,40
Characteristics of the Studies Included in the Review.
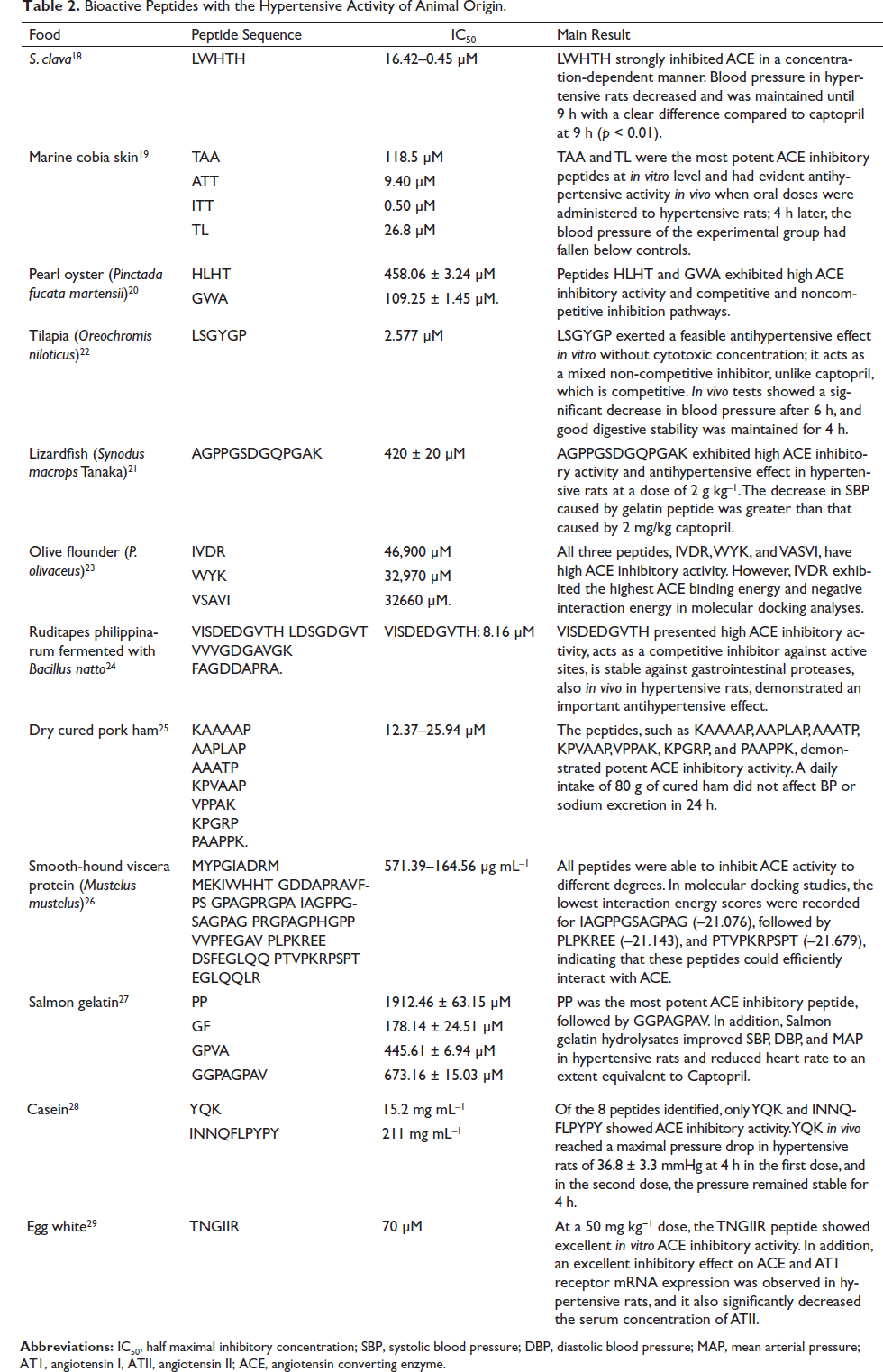
Bioactive Peptides of Animal Origin
There were a total of 12 publications that examined peptides of animal origin, nine of which originated from marine animals,18–24,26,27 one from milk, egg, and pork, respectively (Table 2).25,28,29
Bioactive Peptides with the Hypertensive Activity of Animal Origin.
The substantial contribution of essential amino acids to the diet that comes from the ingestion of proteins like beef, chicken, pork, and even eggs shows how important food of animal origin is to human nutrition. 41 Similar to how they are already commonly used and consumed around the world, these proteins are simple to digest.
In this review, it was found that 52% of the selected studies analyzed peptides of animal origin.18–24,26,27 Most of the animal-derived peptides were obtained from marine sources such as fish, oysters, and clams, which is an interesting fact because these are good sources of protein, and their peptides have shown a wide spectrum of antimicrobial, antiviral, antihypertensive, and antidiabetic activities, among others. 42 For this reason, they have potential applications in the pharmaceutical and nutritional industries. In the case of the marine invertebrate Styela clava (67% protein), the LWHTH peptide was found to regulate blood pressure in hypertensive rats by strongly inhibiting ACE (IC50 = 6.42–0.45 µM). 18 In addition, this peptide was stable against digestion with gastrointestinal enzymes (pepsin, trypsin, and alpha chymotrypsin). It showed better absorption since it is structurally made up of essential nutrients and amino acids, thus favoring its potential use as a natural antihypertensive. 18
Other sources, such as olive flounder (Paralichthys olivaceus) surimi, also showed important physiological actions in the regulation of blood pressure, in which the IVDR, WYK, and VASVI sequences were identified (IC50: 46.90, 32.97, and 32.66 µM, respectively). 23 This study reinforces that evidenced by Ko et al. (2016), 43 where other peptides with antihypertensive activity from the muscle of P. olivaceus were identified, including MEVFVP (IC50 = 79 µM) and VSQLTR (IC50 = 105 µM), a fact that demonstrates the potential of this animal as a rich source of bioactive peptides, which are maintained even in preparations such as surimi. 43
Regarding fish by-products (skin, scales, visors, and bones), it has been found that in addition to obtaining valuable components such as oil, proteins, collagen, enzymes, and minerals, they also have bioactive compounds with antihypertensive action, as reported by Abdelhedi et al. (2017). 26 In this study, bioactive peptides from fish viscera (IAGPPGSAGPAG, VVPFEGAV, PLPKREE, and PTVPKRPSPT) were identified, which showed in silico inhibitory activity against ACE to different degrees, revealing that they bind to the catalytic site of the enzyme. 26 In this sense, Chen et al. (2020) 22 found the peptide LSGYGP (IC50 = 2.577 µM) in tilapia skin, which showed a significant decrease in blood pressure in hypertensive rats as well as good digestive stability. 22
On the contrary, sources derived from animals, such as milk and eggs, have been the subject of much research in terms of bioactive peptides. In this sense, Xue et al. (2018) 28 identified a new ACE-inhibiting peptide derived from bovine casein (YQK, IC50 = 11.1 µM) and from the YQKFPQYLQY sequence, which was shown to significantly reduce the systolic blood pressure of hypertensive rats. 28 Another source of peptides was egg white, of which Yu et al. (2020) 29 found the TNGIIR hexapeptide (70 µM), whose activity inhibited the expression of ACE and AT1 receptor mRNA in hypertensive rats and significantly decreased the serum concentration of ATII. 29
Regarding cured meats such as ham, Montoro et al. (2017) 25 showed that the peptides KAAAAP, AAPLAP, AAATP, KPVAAP, VPPAK, KPGRP, and PAAPPK (IC50 = 12.37 to 25.94 µM) did not show a decrease in blood pressure in individuals when they were given a daily intake of 80 g of ham. 25 This shows that their in vivo effect may be observed when they are previously digested.
When comparing the inhibitory activities of peptides of animal origin against ACE (Table 2), it was found that the best in decreasing order were: LSGYGP (tilapia skin gelatin), 22 ITT (marine cobia skin), 19 VISDEDGVTH (Ruditapes philippinarum), 24 ATT (marine cobia skin), 19 and LWHTH (S. clava). 18
Bioactive Peptides of Plant Origin
A total of nine articles identified bioactive peptides of plant origin, which were derived from cereals,30,35,36 seeds, 34 legumes,37,38 fruits, 32 vegetables, 33 and oils 31 (Table 3).
Bioactive Peptides with the Hypertensive Activity of Plant Origin.
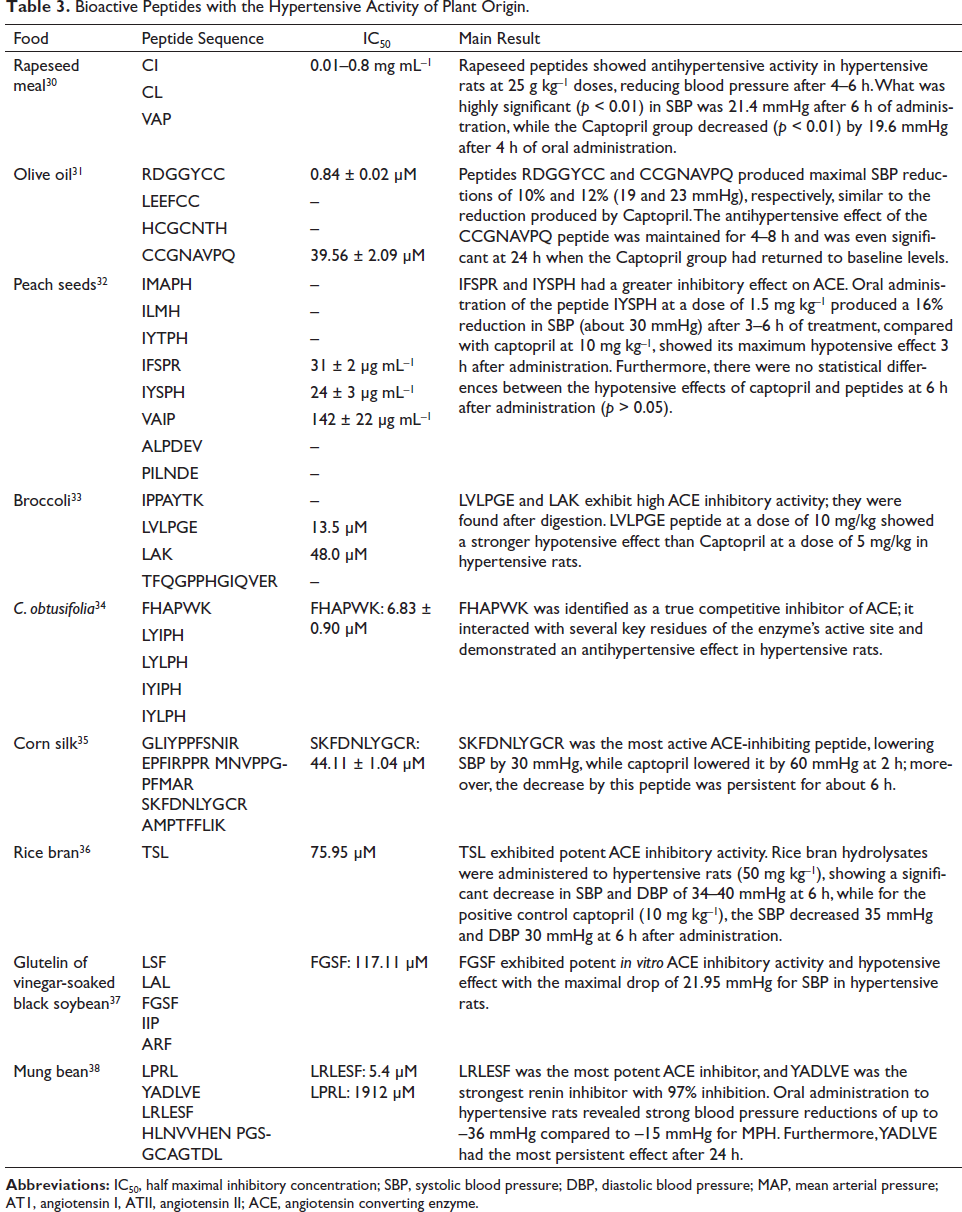
Plant-based proteins also play an important role in meeting dietary protein needs; 39% of the studies examined peptides from this source.30–38 Plant-derived peptides have potential ACE inhibitory activities and are also a sustainable option for global consumption. 41
Despite having fewer essential amino acids than foods of animal origin and also having reduced digestibility due to their storage in granules that limit the accessibility of proteolytic enzymes, vegetables are regarded as good sources of protein. 44 One of the reasons why some plants are used as a source for the extraction of bioactive peptides is because they contain enzyme inhibitors.
Among other plant-based foods, we find rice, one of the main cereals that serve as a staple food for almost half of the human population, and its by-product, rice bran, is rich in proteins that are efficient for body growth.45,46 Wang et al. (2017) 36 isolated a peptide from the rice bran protein, TSL, that exhibits potent ACE inhibitory activity (IC50 = 75.95 µM), like the study reported by Shobako et al. (2018), 46 which identified two peptides, namely, LRA and YY, derived from this protein and which also demonstrated inhibitory activity of the enzyme (IC50 = 62 and 16.5 µM, respectively).36,46
Peptides from rapeseed meals reported by Wang et al. (2021) 30 (CI, CL, and VAP; IC50 = 0.01–0.8 mg mL–1) showed relatively good ACE inhibitory activity in vitro and in vivo (hypertensive rats). 30 Additionally, these authors indicated that when administered concomitantly with captopril, no synergistic effect was observed in the inhibition of the enzyme; however, they found an improvement in nitric oxide levels in hypertensive rats.
Alcaide-Hidalgo et al. (2020) identified olive oil’s bioactive peptides as RDGGYCC, LEEFCC, HCGCNTH, and CCGNAVPQ. Of these, RDGGYCC (IC50 = 0.84 ± 0.02 µM) and CCGNAVPQ (IC50 = 39.56 ± 2.09 µM) showed the highest inhibitory activity against ACE; in addition, their hypotensive effect was like that produced by captopril under in vivo conditions. 31
As for the by-products of food processing, such as peach pits, Vásquez et al. (2019) 32 identified the peptides IFSPR (IC50 = 31 ± 2 µg mL–1), IYSPH (IC50 = 24 ± 3 µg mL–1), and VAIP (IC50 = 142 ± 22 µg mL–1) with antihypertensive activities. IYSPH showed the best ACE inhibitory capacity (IC50 = 24 µg mL–1); it was less susceptible to intestinal peptidases, and its oral administration produced a 16% reduction in its oral administration SBP (around 30 mmHg) in hypertensive rats after 3–6 h of treatment. 32 Shih et al. (2019) 34 identified the peptide FHAPWK (IC50 = 16.83 ± 0.90 µM) from the seeds of Cassia obtusifolia. 34 This peptide proved to be a competitive inhibitor of ACE, whose action managed to reduce at 17 mmHg the SBP of hypertensive rats. 34 Sonklin et al. (2020) 38 reported five bioactive peptides derived from mung beans; among them, only LRLESF (IC50 = 5.4 µM) exhibited significant inhibitory activity against ACE. However, despite not showing in vitro inhibitory activity against ACE, the YADLVE peptide did show a 97% inhibition of renin; it also reduced the SBP of hypertensive rats between 36 and 15 mmHg, and its effect was persistent after 24 h. 38
On the contrary, Dang et al. (2019) 33 reported that peptides derived from broccoli, such as LVLPGE (IC50 = 13.5 µM) and LAK: (IC50 = 48.0 µM), showed inhibitory activity against ACE. However, only LVLPGE exhibited a significant antihypertensive effect against ACE in hypertensive rats. 33 Li et al. (2019) 35 identified the bioactive peptides GLIYPPFSNIR, EPFIRPPR, MNVPPGPFMAR, SKFDNLYGCR, and AMPTFFLIK from corn silk. However, only SKFDNLYGCR (IC50 = 44.11 ± 1.04 µM) showed potent in vivo activity against ACE. 35 Zhang et al. (2019) 37 reported the bioactive peptides such as LSF, LAL, FGSF, IIP, and ARF from black soybean glutelin soaked in vinegar. Despite the number of peptides obtained, only FGSF (IC50 = 117.11 µM) showed a significant hypotensive effect in hypertensive rats. 37
When comparing the inhibitory activities of plant-derived peptides against ACE (Table 3), it was found that the best in decreasing order were: RDGGYCC (olive oil), 31 LRLESF (mung bean), 38 FHAPWK (C. obtusifolia seeds), 34 and LVLPGE (broccoli). 33
Fungus-derived Bioactive Peptides
Two studies identified bioactive peptides derived from fungi.39,40 The fungus has been the subject of recent studies thanks to its beneficial effects on health. 47 Although the protein content and quality of mushrooms rank below most animal proteins, they rank above most other foods,47,48 making them a good starting material to produce peptides with bioactive activities, including ACE inhibitory activity.
Manoharan et al. (2017) identified the bioactive peptides ALGVR, VTVGLVVR, VVLRNNK, and ATGNLNPR from the fungus Pleurotus pulmonarius among this group; only the ALGVR pentapeptide (IC50 = 55 mg mL–1) was considered a strong competitive ACE inhibitor. 39 Amorim et al. (2019) 40 observed a high percentage (85%) of ACE inhibition in brewer’s yeast extracts, which can be easily absorbed due to their size and hydrophobicity. 38 The peptides identified in these extracts were SPQW, PWW, and RYW, of which SQPW showed the highest inhibitory activity against ACE (IC50 = 12 µM). 40
Both studies indicated that the peptides with the highest inhibitory activities (ALGVR and SQPW) were resistant to the action of gastrointestinal enzymes, which favors their absorption in the active form, thus potentiating their antihypertensive effect.39,40
Discussion
To summarize the available evidence about the antihypertensive activity of bioactive peptides from foods, this systematic review was carried out, in which the currently available evidence of bioactive peptides with antihypertensive activity from foods of plant, animal, and fungi origin was unified.
As mentioned above, ACE inhibition blocks the transformation of angiotensin I into angiotensin II, thus regulating the renin-angiotensin-aldosterone system and preventing an increase in blood pressure. The potency of the peptides to inhibit ACE has been expressed as IC50 (half-maximal inhibitory concentration), which, at a lower value, expresses a greater potential for inhibition of this enzyme (Tables 2 and 3).
Peptides of vegetable origin proved to be more active, with an IC50 of 0.84 µM, standing out RDGGYCC (0.84 ± 0.02 µM) from olive oil 31 and LRLESF (5.4 µM) from mung bean. 38 On the contrary, within the peptides of animal origin, LSGYGP (2.577 µM) from tilapia skin gelatin 22 and VISDEDGVTH (8.16 µM) from the clam Ruditapes philippinarum stand out. 24 Other foods, such as fungi, showed a marked inhibition of ACE, as occurs with SPQW (12 ± 3 µM) from brewer’s yeast. 40 This is also in contrast to that reported in similar reviews, such as the one reported by Ngoh and Gan (2018), which identified biologically active peptides from pinto beans with significant ACE inhibitory activity (PPHMLP and PLPTGAGF) with IC50 values of 1.52 and 1.84 µM, respectively. 49 Likewise, peptides with antihypertensive activity have been isolated from many other food sources, such as chicken, red meat, wheat gluten, soybeans, sunflower, and spinach. Plant sources are mainly attractive for the extraction of these peptides in the industrial sector (food and pharmaceuticals), since they could be included in various food systems and medicines to achieve their effect in the body of the final consumer.50,51
Several authors attribute the antihypertensive effectiveness of peptides to their structure, which is generally reported to be of low molecular weight (less than 3 kDa), short length (2–20 amino acids), and the presence of hydrophobic residues in their C-segment positively charged terminal, which have been associated with increased binding affinity with the active site of ACE.40,51 However, Sonklin et al. (2020) found that long peptide chains (LRLESF, PGSGCAGTDL, and HLNVVHEN) had strong ACE inhibitory activity, demonstrating that not only short-chain peptides are active inhibitors of the enzyme. 38
On the contrary, in addition to demonstrating the ACE inhibitory activity, it is important to study the in vivo effect of the peptides to ensure that they can exert their physiological action since they must overcome the action of proteases present in the gastrointestinal digestion process. For this reason, some studies also show the in vivo effect of peptides,18,19,21,30,33 either through an animal model in hypertensive rats, in simulated gastrointestinal digestion, or even in human studies.
In this way, it was found that peptides such as LWHTH from the marine invertebrate S. clava, 18 presenting powerful ACE inhibitory activity, presented a strong suppressive effect on systolic and diastolic blood pressure in an animal model. Likewise, the peptides (CI, CL, and VAP) from rapeseed meal showed a high inhibition of ACE (IC50 = 0.01 and 0.8 mg mL–1) and showed a decrease in blood pressure in hypertensive rats, even higher in a group treated with captopril, a commercial antihypertensive drug.
However, the in vitro inhibitory activity does not always correlate at the in vivo level, as occurs with the mung bean peptide YADLVE, which had no detectable ACE inhibitory activity but was found to be a potent inhibitor of renin activity in 97.06% of cases. When performing in vivo studies, this peptide presented the most persistent reductions in blood pressure during a period of 24 h after oral administration in hypertensive rats. 38
In this sense, it makes it clear that there are other mechanisms other than ACE inhibition through which bioactive peptides can act to reduce high blood pressure, such as direct inhibition of the enzyme renin, inhibition of endothelin converting enzyme, and antioxidant properties.
Amorim et al. (2019) demonstrated the antioxidant activity of spent brewer’s yeast peptides and suggested a possible synergistic antihypertensive and antioxidant effect since the deficiency of antioxidant activity has also been involved in the appearance of cardiovascular diseases, 40 like Neves et al. (2017) in salmon gelatin 27 and Wang et al. (2017) in rice bran. 36
Regarding the practical implications, our results contribute to making known to the scientific and health communities about the potential benefits offered by bioactive peptides from food in the management of HTN, considering that these promise to be an excellent pharmacological alternative to conventional medications and being able to reduce the side effects that these can cause. Likewise, identifying the presence of bioactive peptides in foods as natural sources of ACE inhibitors makes them functional ingredients applicable both in the food and drug industries to positively reduce the risk of cardiovascular diseases, maintain body health, and prevent disease.
Some limitations of this review are the language barrier due to all the evidence found being published in English, which eliminated the inclusion of available evidence published in any other language. However, it is highlighted that no restrictions about languages were performed. In addition, only some of the selected articles evaluated the safety of peptides. However, this is a window for conducting studies that include this phase.
Among the strengths of this review, we highlight that all methods were described in a protocol beforehand. Furthermore, a sensitive search strategy was carried out, so it is unlikely that any relevant evidence was missed. In addition, two reviewers independently performed all selection, methodological quality assessment, and data extraction processes. All these processes provide reasonable confidence in our results.
Conclusion
Bioactive peptides present in foods have potential health benefits and can be considered an option for developing functional products useful in the prevention and treatment of arterial HTN. Thus, there has been increased interest in identifying foods that may be natural sources of antihypertensive peptides in recent years. Among the peptides described in this review with the best antihypertensive potential were the sequences RDGGYCC, LRLESF, FHAPWK, and LVLPG from plants; LSGYGP, ITT, VISDEDGVTH, ATT, and LWHTH from animals; and ALGRV and SPQW from fungi, which demonstrated their antihypertensive potential in vitro and in vivo. However, studies are required to demonstrate the usefulness of these peptides in daily clinical practice in humans.
Footnotes
Abbreviations
ACE-I, angiotensin-I converting enzyme; HTN, Hypertension; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses;
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
