Abstract
In response to the COVID-19 pandemic, two mRNA vaccines (Comirnaty and Spikevax) received emergency use authorization from the European Medicines Agency. This case report aimed to report a delayed adverse reaction to the mRNA-1273 vaccine against COVID-19 from a Portuguese vaccination center.
A case report was performed with medical observation and reported to the Portuguese Pharmacovigilance System, then investigated based on the WHO-UMC Causality Categories. A 66-year-old female patient with a clinical history of dyslipidemia, essential arterial hypertension, obesity, multinodular goitre and cholecystectomy, who presented delayed large cutaneous hypersensitivity reaction following Spikevax COVID-19 mRNA (mRNA-1273) vaccine administration.
Our clinical findings (time and clinical appearance), along with evidence of previously reported histological findings, are strongly suggestive of T-cell-mediated hypersensitivity. There is no contraindication to the inoculation of subsequent doses in patients developing these clinical conditions, and vaccination should continue to be strongly encouraged.
Background
In response to the coronavirus disease 2019 (COVID-19) pandemic, two mRNA vaccines (Comirnaty and Spikevax) received emergency use authorization from the European Medicines Agency (EMA). This brief report contributes to disseminating safety data, supporting previous reports of delayed local cutaneous reactions following vaccination.1–8
Case Summary
We report a 66-year-old female hairdresser with delayed large cutaneous hypersensitivity reaction following Spikevax COVID-19 mRNA (mRNA-1273) vaccine administration. The patient is autonomous and cognitively whole, a former smoker (40 pack-year), with a clinical history of dyslipidemia, essential arterial hypertension, obesity (body mass index of 31.8), multinodular goitre, and cholecystectomy (bile lithiasis). She is prescribed with olmesartan + hydrochlorothiazide (20 + 12.5 mg) and simvastatin 20 mg, all once a day.
On D7 after receiving the first dose of the vaccine, she developed a clinical condition with severe pain, heat, and redness at the inoculation site, was feverless and self-medicated with ibuprofen 600 mg every 12 h plus applying ice to the reaction site. On D9, at the patient’s initiative, a physician evaluated her and diagnosed erythema with well-defined limits (measuring 6 cm in diameter) with heat to the touch, around the inoculation site located on the external face of the anterolateral region of the upper third of the left upper limb. Based on the patient’s report and the evaluation of the inoculation site, it does not appear to have been an administration error. The case of suspected adverse reaction was reported to the Portuguese Pharmacovigilance System which then assessed based on the WHO-UMC Causality Categories. 9 Moreover, this assessment was also performed using a Bayesian method developed at the Porto Pharmacovigilance Centre, 10 and the causality degree of Probable was attributed. As such, the clinical event occurred with a reasonable time relationship to drug intake and a causal link with concomitant diseases or other drugs is unlikely. 9
Treatment for symptoms relief includes topical steroids, oral antihistamines, and cool compresses and/or ice. The patient was recommended to maintain ibuprofen 600 mg every 12 h for two more days, ice application, and bilastine 20 mg was prescribed t.i.d. Six days after symptom onset, on D13, the patient reports complete resolution of the symptoms. She took the second dose of the vaccine 28 days after the administration of the first dose (D28), having been recommended to start again bilastine 20 mg the day before inoculation (D27) and for three more days after (until D31), which was fulfilled. After the second vaccine dose, she did not report any complications.
Blumenthal et al. 1 reported delayed site reactions in 12 patients who had received the Spikevax vaccine, with a median onset on day 8, similar to the case we report here. Five of the reactions were grade 3 plaques (≥10 cm in diameter), and the symptoms resolved a median of 6 days after onset, which is consistent with the clinical findings identified in our patient. Of the 12 patients who took the second dose, only three reported recurrent reactions similar to the first dose. Still, most patients had no complications, as did our patient, eventually because she has received prophylaxis with bilastine. Later, Baeck et al. 2 reported a case of a 38-year-old woman who received the Comirnaty vaccine and developed a clinical condition with erythema of the upper arm associated with numbness of the fingers, which appeared six days after inoculation. Johnston et al. 5 reported a series of 16 cases with delayed localized cutaneous reactions developed in a median of 7 days after receiving the Spikevax vaccine, with pruritic, painful, and edematous pink plaques. Of 15 participants who reacted to the first vaccine dose, 11 developed a similar reaction in the second dose. Of the latter, 10 developed a reaction earlier than the first dose. No prophylactic treatment has been described for these patients (Figure 1). Jacobson et al 6 reported the case series of 13 individuals, all females aged 28–55 years, with delayed injection site reaction that involved erythema and pruritus that began 2 or more days after vaccination. Pain or tenderness was reported by 7, swelling or induration by 9, and lymphadenopathy by 2, in which three individuals reported using an over-the-counter medication to relieve pruritus symptoms. Only 5 of these 13 individuals had a delayed reaction to the second dose, which started 2–3 days after injection and was less severe in 4 and of equal severity in 1. Reactions to the second dose were of similar duration to initial dose reactions (2–7 days).
Delayed Large Reaction Around the Vaccination Site on D7 (Onset of the Symptoms) Post-immunization with Spikevax Vaccine.
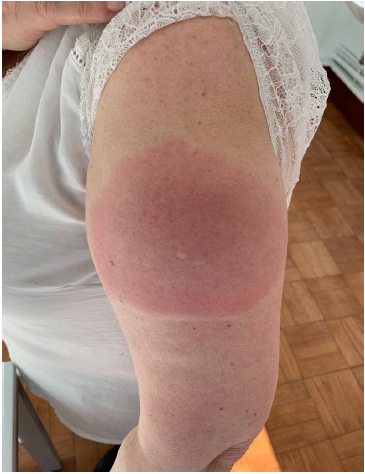
Unlike previously reported cases, we did not perform biopsy or skin tests. Although the pathophysiological mechanism underlying the disease is unknown, our clinical findings (time and clinical appearance), along with evidence of previously reported histological findings, are strongly suggestive of T-cell-mediated hypersensitivity. Even so, this hypothesis must be studied carefully due to the low incidence after the reexposure. We can also hypothesize that the patient reacted to the Spikevax vaccine owing to cross-reactivity between excipients, such as Lipid SM-102, PEG2000 DMG, and DSPC. 11
Practice Implications
Although our case report lacks histological findings, the clinical findings identified are very similar to those associated with both mRNA vaccines available to date. So, our findings strongly support previously reported evidence that self-limited, benign and localized delayed hypersensitivity reactions to the mRNA vaccines may occur. Still, in clinical practice, there is no contraindication to the inoculation of subsequent doses in patients developing these clinical conditions, and vaccination should continue to be strongly encouraged. In cases of late adverse reactions with an exacerbated profile compared to what is expected, medical evaluation and prescription of pharmacological therapy are necessary, particularly in patients with a history of allergies.
Footnotes
Authors’ Contributions
RFS submitted the work to the ethics committee. RFS and IRV conceived the first draft and critical discussion of the manuscript. FN and PG were responsible for collecting data from the patient. All authors contributed to data analysis and writing, reading, and approval of the final version of the manuscript.
Availability of Data and Material
Not applicable.
Code Availability
Not applicable.
Consent to Participate
Informed consent was obtained from the patient for being included in the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All procedures followed the ethical standards of the ethics committee of the Centro Hospitalar e Universitário de São João and the Helsinki Declaration of 1964 and its later amendments. This ethics committee approved the study on September 24, 2021.
Funding
Renato Ferreira da Silva is grateful for the PhD scholarship 2020.10231.BD (DOCTORATES 4 COVID-19), funded by Portuguese national funds and community funds from the European Social Fund (ESF) through FCT – Fundação para a Ciência e a Tecnologia (Portugal). The Porto Pharmacovigilance Centre is a regional centre of the Portuguese Pharmacovigilance System, funded by INFARMED - National Authority for Medicines and Health Products of Portugal, I.P.
Informed Consent
Informed consent was obtained from the patient for the publication of the case report.
