Abstract
Background
Recent pharmacovigilance studies suggested that cluster headache could be a potential adverse effect after coronavirus disease-2019 (COVID-19) vaccination; however, the possibility of coincidence could not be excluded. Detailed case studies might help elucidate their potential link and implicate potential pathogenic mechanisms.
Methods
Patients who developed cluster headache in close temporal relationship to COVID-19 vaccination were identified from two tertiary medical centers in Japan and Taiwan respectively through 2021–2022. Detailed characteristics of the headaches and time between the onset of the index cluster episode and antecedent COVID-19 vaccination were reported. In patients with previous cluster headaches, the duration from previous bout was also recorded.
Results
Six patients with new cluster headache bout 3–17 days after COVID-19 vaccination were identified. Two of them were de novo cases. The others either had been attack-free for a long time or developed new cluster bout in seasons atypical to prior bouts. The vaccines included mRNA, viral vector, or protein subunit vaccines.
Conclusions
COVID-19 vaccines, regardless of vaccine types, may elicit de novo or relapse of cluster headache. Future studies are needed to confirm the potential causality and explore the potential pathogenic mechanism.
Introduction
Cluster headache is the most common type of trigeminal autonomic cephalalgias with a prevalence of around 0.1% among general population (1) and it is considered one of the most painful disorders in humans. As cluster headache exhibits prominent circadian and circannual rhythmicity, the hypothalamus has been considered to play an important role in the pathogenesis (2). In addition, the activation of trigemino-autonomic reflex may explain the cephalic pain and accompanying unilateral autonomic symptoms. Yet, the detailed molecular mechanism of cluster headache remains elusive. Although some triggers such as ingestion of alcohol or infusion of calcitonin gene-related peptide (CGRP) or nitric oxide have been recognized to evoke an attack during the bout of cluster headache, little is known regarding what may trigger the onset of a new bout of cluster headache.
Recent pharmacovigilance study suggested that cluster headache could be a potential adverse effect after the coronavirus disease-2019 (COVID-19) vaccination (3,4). However, because of the lack of detailed clinical information, the causality between COVID-19 vaccination and cluster headache remains unclear and the possibility of coincidence could not be excluded. A recent study reported seven cases with cluster headache, who had been attack free for a long time, experiencing a new unexpected cluster headache bout shortly after vaccination, supporting the speculation that COVID-19 vaccination may be a potential trigger of new attacks in patients with a history of cluster headache (5). It remains unclear whether the COVID-19 vaccination could elicit a de novo bout of cluster headache in patients without history of cluster headache. In this study, we reported six cases with new cluster headache bout in close temporal relationship with the COVID-19 vaccination, with two of them being de novo cases from Japan and Taiwan respectively. These clinical observations may serve as primers for future studies to interrogate the potential mechanisms for the genesis of cluster headache.
Methods
Patients who developed cluster headache in close temporal relationship to COVID-19 vaccination were identified from two tertiary medical centers in Japan and Taiwan respectively through 2021–2022. The inclusion criterion was defined as the onset of cluster headache within three weeks after vaccination. The study protocol was approved by the Institutional Review Board of Taipei Veterans General Hospital (TVGH-IRB 2022-06-007CC) and the participants in Taiwan have provided written informed consent. The Ethics Committee of Keio University School of Medicine determined that this report was not necessary to refer to the Committee because this was a case series and a written informed consent was obtained from the patient. Detailed characteristics of the headaches, types of vaccinations, time between the onset of the index cluster episode and antecedent COVID-19 vaccination, and the duration from previous bout were presented below and summarized in Table 1.
Temporal relationship between the development of cluster headache and COVID-19 vaccination.
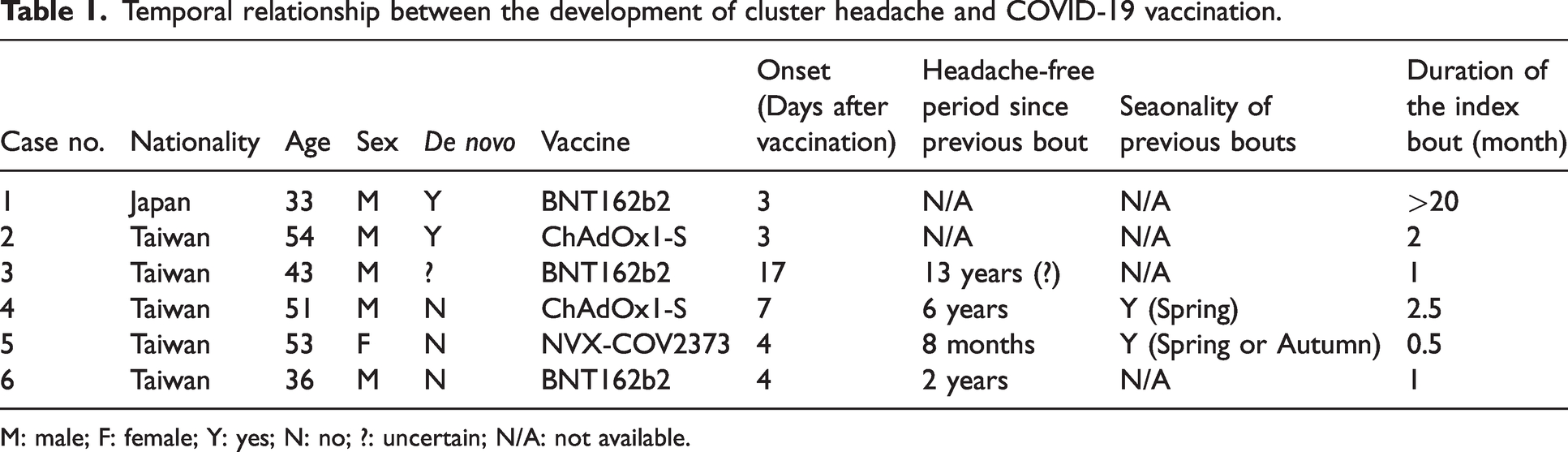
M: male; F: female; Y: yes; N: no; ?: uncertain; N/A: not available.
Case-1
A 33-year-old man with a history of depression, felt severely lightheaded after receiving his first dose of BNT162b2 vaccine. A day after vaccination, on 11 July 2021, he noted a 38°C fever, chills, fatigue, and arthralgia for two days. Three days after his vaccination, he experienced a very severe headache, characterized as being punched from behind over his right eye. The patient rated his headache with a numerical rating scale (NRS) score of 10. During the headache attacks, he was unable to open his right eye. Many times, he felt restless during attacks. The headache lasted for two to three hours, with the next attack starting after one to three hours, occurring approximately four times a day. Conjunctival injection and tearing were noted in the right eye along with nasal discharge on the right side. No eyelid edema or sweating was noted. He consulted our outpatient headache clinic one month after the symptom onset because his symptoms did not improve. He was a 6.5 pack/year smoker and denied regular alcoholic beverage intake. He had past history of migraine without aura which occurred approximately once every month and usually located on the left side. He had no personal or family history of cluster headache.
Neurological examination was normal. Blood tests were non-remarkable. Brain magnetic resonance imaging (MRI) revealed a mild protrusion of the left internal carotid artery C1. His lumbar puncture and electroencephalography were also unremarkable.
Oral sumatriptan was partially effective for his headache, while subcutaneous injection of sumatriptan and home oxygen therapy suppressed the headache attacks. He was subsequently started on oral verapamil (120 mg), causing the severity of his headaches and attacks to be reduced. Prednisolone (up to 20 mg) was used for a few weeks, but it did not show too much of an effect.
The patient was diagnosed with a cluster headache. The response to subcutaneous injection of sumatriptan, oral verapamil, and oxygen inhalation was also suggestive of this disease. He has been followed up for 15 months but the headache had not subsided. Hence, the patient’s cluster headache fulfilled the diagnosis of chronic cluster headache proposed in the International Classification of Headache Disorder, 3rd edition (ICHD-3) and is highly likely a de novo case.
Case-2
A 54-year-old man, who has smoked 0.5 pack of cigarettes per day for 30 years, experienced acute onset of headaches since 10 October 2021, three days after the second dose of ChAdOx1-S vaccination for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). He has had no headache or any systemic disease previously. He also denied family history of cluster headache. The new headache manifested as left nasal soreness initially, with radiation to the left temporal area. The headaches were accompanied by left-sided tearing, conjunctival injection, eyelid edema and ptosis but not nasal congestion, facial flushing or forehead sweating. There was no nausea or vomiting but he was sensitive to light (predominantly on the left eye) and sound and was restless during the attacks. The headaches mostly occurred at 8am and 6pm, with a duration of 1–2 hours. There were two to three attacks per day. Upon presentation, he had experienced daily headache for more than one month. Neurological examinations were unremarkable. Laboratory studies were normal. MRI of sella demonstrated no significant findings except mucus retention in the right maxillary sinus, which was contralateral to the headache side. He was placed on prednisolone (70 mg per day for one week with gradual tapering during the subsequent week) and verapamil (240 mg per day) and his headache remitted within two weeks. Verapamil was discontinued one month later, and his headache did not recur during the subsequent 10 months.
Case-3
A 43-year-old man experienced a newly developed cluster headache since 13 December 2021. He had one bout of daily headaches lasting seven weeks at age 30, but he could not remember the detailed characteristics well so whether there was pre-existing history of cluster headache could not be ascertained. He had not had other headache episodes. He did not have history of systemic diseases. He denied family history of cluster headache. He received two doses of vaccination with BNT162b2 on 26 October, and 26 November 2021, respectively. The headache was right sided at the temporal and orbital region, with unilateral tearing, rhinorrhea, conjunctival injection, ptosis, nasal congestion, facial flushing and ear fullness. He had a sense of restlessness during the attacks. There was no nausea or vomiting but photophobia and phonophobia were noted. He had taken multiple over-the-counter compound analgesics with nearly no response. The attacks mostly occurred at 9am and lasted for 1–2 hours. Diagnostic workups were unrevealing except that the MRI of the sella demonstrated a 4 mm microadenoma in right aspect of pituitary gland. He was placed on Verapamil 240 mg per day and Prednisolone 60 mg for one week with gradual tapering for another week. His headache completely remitted nine days after treatment. No recurrence was noted during the subsequent 10-month follow-up.
Case-4
A 51-year-old man, with no systemic diseases, experienced cluster headache since age 20. He did not have family history of cluster headache. He is a non-smoker and drinks alcohol only socially. The headache usually lasted one hour per attack, up to three attacks per day, with each bout lasting for around two months. Remarkably, the headache was always unilateral but side shifting. There was accompanying unilateral conjunctival injection, tearing, rhinorrhea, nasal congestion, ptosis, and restlessness. He also has nausea, unilateral photophobia, and phonophobia during the attacks. The last bout was in April 2015. However, on 29 July 2021, he experienced a new bout, one week after ChAdOx1-S vaccination for SARS-CoV-2. The headaches were daily, with one or two attacks per day. The headaches were initially located on the left side for two months and then shifted to the right side. All the cluster headache features were similar to his previous attacks. Laboratory studies including sella MRI showed normal results. He was placed on verapamil 240 mg per day and his headache had not recurred after 11-month follow-up.
Case-5
A 53-year-old otherwise healthy woman has had cluster headache since age 34. She denied history of other headaches and had no family history of cluster headache. She has experienced one to two bouts of cluster headache per year, which lasted for more than one month per bout. The bouts mostly occurred in March to April or October to November. During the past five years the headache had improved in that the duration of each bout was shortened to less than one month. The latest bout ended in November 2021. On 16 July 2021, she received ChAdOx1-S vaccination. There was no significant adverse effect. On 11 July 2022, she received NVX-COV2373 vaccine as a booster vaccination against SARS-CoV-2. Four days later, she experienced a new bout of cluster headache. She has never experienced cluster headache in similar season previously. The headache was located at the right orbital region, with unilateral tearing, rhinorrhea, ptosis, eyelid edema and right-sided photophobia. She was restless during the attacks. Each attack lasted for 2–3 hours. Sella MRI demonstrated a 3 mm less-enhancing nodule in right aspect of pituitary gland, suspected to be a microadenoma. However, hormonal studies were normal. She was placed on verapamil 240 mg per day. The headache remitted gradually and has not recurred during the following two months.
Case-6
A 36-year-old man who smoked 1/3 packs of cigarettes per day for years has experienced two bouts of cluster headache since age 28. He did not have history of systemic diseases or other headaches. He denied family history of cluster headache. The latest bout of his cluster headache occurred two years ago. He developed a new bout of cluster headache since 30 January 2022, four days after receiving BNT162b2 as a booster vaccination. He had received two doses of MVC-COV1901 vaccines on 28 August and 2 October 2021, respectively, without noticing significant adverse effects. This new bout was similar to his previous bouts in that his headache was located at the right temple and orbit, with accompanying unilateral tearing, rhinorrhea, conjunctival injection, ptosis and facial flushing. He was agitated and required pacing during the attacks. He also had nausea, vomiting, photophobia on the affected side, and phonophobia during the attacks. The headache was responsive to sumatriptan, rizatriptan and oxygen inhalation. Upon presentation, he had experienced daily headache for 1.5 months. He was placed on the regimens that worked for his prior bouts, i.e., Verapamil 240 mg, lithium 300 mg, and topiramate 25 mg twice a day as preventive therapy, with bridging therapy with Prednisolone for two weeks. The latest attack occurred on 9 March 2022. He had no further headache during the subsequent six months.
Discussion
We report six cases of patients who developed a new bout of cluster headache shortly after COVID-19 vaccination in Japan and Taiwan respectively. Two of them never had cluster headache previously, one had a bout of uncharacterized daily headache lasting for seven weeks a decade ago, and the other three either had been attack-free for a long time or developed new cluster bout in seasons atypical to their prior bouts. Remarkably, the vaccines included mRNA vaccine (BNT162b2), viral vector vaccine (ChAdOx1-S), and protein subunit vaccine (NVX-COV2373), suggesting this link might not be due to certain vaccine class. Except for the first case who developed cluster headache after the first dose of COVID-19 vaccine, most cases had a relatively benign course and responded to preventive medications well except for Case 1 which was thought to be de novo chronic cluster headache. Because Case 1 is the only case who experienced constitutional symptoms such as fever and chills after vaccination, this observation may suggest that a more robust inflammation following vaccination may cause a longer lasting activation of trigeminovascular system. Of note, none of our cases had been diagnosed with COVID-19 before vaccination. However, we did not have evidence excluding concomitant SARS-CoV-2 infection in these patients at headache onset, although the incidence of COVID-19 at the time we recruited these patients was very low in Taiwan and Japan.
Consistent with the signals noted from the pharmacovigilance studies (3,4), our findings supported the possibility of COVID-19 vaccines being a potential trigger of cluster headache. A recent Dutch study reported seven cases of patients who had been free from cluster headache for a long time, experiencing new bouts of cluster headaches after both vector based and mRNA vaccines against COVID-19, suggesting that COVID-19 vaccines may elicit cluster episodes in patients with history of cluster headache (5). Our de novo cases further suggest that COVID-19 vaccines could elicit cluster headache in subjects who have never had cluster headache. Because of limited case numbers, we could not completely exclude the possibility of coincidence. On the other hand, we could not exclude the possibility of under-reporting or under-diagnosis because cluster headache may be missed by clinicians who are unfamiliar with this rare headache disorder. Together with previously reported cases and the signals suggested from pharmacovigilance studies, our report support further prospective studies to look for the potential causal link between COVID-19 vaccination and cluster headache.
In a survey using the Vaccine Adverse Events Reporting System (VAERS) database during a 30-year period prior to the COVID-19 pandemic, headache presented in 8.1% (43,218/536,120) of all reports; however, cluster headache was reported in only 14 cases (6). Given the scarcity of prior cases, a causality could not be determined. Nevertheless, although this vaccine-cluster headache association may not be restricted to vaccines against SARS-CoV-2 alone, the potential link between COVID-19 vaccination and cluster headache may provide pathological implications. As the link was not specifically restricted to a certain vaccine type, it is possible that the inflammation induced by COVID-19 vaccination or the antigen associated with a structural subunit of SARS-CoV-2, e.g., the spike protein, may induce a cluster headache by activating the trigeminovascular system. However, the antigenic cross-reactivity between the spike protein of the SARS-CoV-2 and self-antigens in selected subjects could not be validated yet as no report has indicated an increased incidence of cluster headache during the COVID-19 pandemic. In fact, post-COVID-19 vaccination headaches are quite common (around 70%) in patients with migraine or non-migrainous headache in comparison with those without migraine or headache history (37.9%) (7), supporting the possibility that the inflammatory response evoked by COVID-19 vaccination may activate trigeminovascular system in susceptible subjects. However, because the threshold to evoke a cluster headache when the patient is not in-bout or in subjects who have never had history of cluster headache may be very high (in comparison with migraine), there has been no large case series reporting post-COVID-19 vaccination cluster headache. Nevertheless, since we did not systematically examine inflammatory markers in these patients, we were unable to validate this speculation.
As in the post-pandemic era, the general population will still require vaccinations against new variants of SARS-CoV-2 or other emerging infectious diseases, we may expect more cases being identified if there is a causal link between COVID-19 vaccination and cluster headache. Although a firm conclusion could not be drawn now, our report suggests that clinicians could consider including COVID-19 vaccination history in their checklists when encountering patients with new attacks of cluster headaches.
Clinical implications
COVID-19 vaccine may trigger de novo or recurrent cluster headache in susceptible subjects. The relation between COVID-19 vaccine and cluster headache was unrelated to vaccine types.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Brain Research Center, National Yang Ming Chiao Tung University, from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan (to SJW & SPC); the Ministry of Science and Technology, Taiwan [111-2321-B-A49-004 (to SJW)], and JSPS KAKENHI Grant-In-Aid for Early-Career Scientists (Grant number 19K16989) (to TT). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
