Abstract
Objective
To evaluate the link between phenotypic traits, antimicrobial resistance, and biofilm-producing capacity of urinary isolates of Citrobacter freundii (C. freundii).
Methods
Both pan-antibiotic-susceptible and -resistant C. freundii isolates (n = 120) obtained from laboratory-confirmed urinary tract infections were analyzed for their link between antimicrobial resistance, phenotypic characteristics, and biofilm production.
Results
Of the total C. freundii isolates (n = 120), 30% (37/120) of them formed large colonies. Among the total large colonies produced (n = 37), they were present in 21.62%, 10.81%, 13.5%, 16.2%, 21.62%, and 16.21% in the control group, CIP-group, FOS-group, COT-group, Gent-group, and ESBL groups, respectively. Compared to the pan-susceptible isolates, the occurrence of large-sized-colony-forming strains was relatively reduced in most of the drug-resistant groups. The overall distribution of mucoid colonies produced (n = 54) includes 9.25%, 18.51%, 16.66%, 18.51%, 20.3%, and 16.66% in the control group, CIP-group, FOS-group, COT-group, Gent-group, and ESBL groups, respectively. Of the total isolates that produced biofilm (n = 51), 11.76% of isolates showed biofilm formation in the control group. Alternatively, the rate was found to be 15.68%, 11.76%, 25.49%, 19.6%, and 15.68% in the CIP-group, FOS-group, SXT-group, Gen-group, and ESBL-groups, respectively.
Conclusion
This study correlates the association between phenotypic characteristics, antimicrobial resistance, and biofilm production, the three main characteristics of C. freundii.
Introduction
Infectious diseases (IDs) threaten humankind, causing high morbidity and mortality rates. 1 Among these IDs, urinary tract infections (UTIs) continue to be a common public health problem. Facultative Gram-negative bacteria are mainly associated with most UTIs. 2 Of these facultative Gram-negative rods, UTIs are usually caused by Escherichia coli (E. coli) (80%), followed by Klebsiella pneumoniae spp. (K. pneumoniae) and other members of Enterobacterales like Citrobacter freundii (C. freundii). 3 C. freundii is becoming a prevalent urinary tract pathogen in hospitalized patients. 4
UTIs may occur in the community or healthcare setting and result in massive economic and public health issues. 5 Anatomically, the UTIs are classified as uncomplicated (which usually occur in patients with healthy premenopausal women and with normal urinary tract) and complicated UTIs (which are seen in both men and women with underlying urological abnormalities). Complicated UTIs may result in fatal florid urosepsis. Greater morbidity, a higher probability of treatment failure, prolonged treatment duration, and frequent hospitalizations are characteristics of complicated UTIs. 6 Patients who have UTIs frequently experience distressing symptoms, which can sometimes result in life-threatening complications. 7 According to the Infectious Diseases Society of America, nitrofurantoin, trimethoprim-sulfamethoxazole, fluoroquinolones, fosfomycin, and mecillinam would be the first-line treatments for uncomplicated UTIs. 8 However, third-generation cephalosporins, carbapenems, and aminoglycosides may also be useful for treating complex UTIs. It is essential to consider serious drug responses and multiple drug resistance before treating challenging UTIs. 9 The rise and proliferation of multidrug-resistant (MDR) strain causes delays in starting proper therapy and results in worse outcomes. Further, in communities with indiscriminate use of antibiotics, higher levels of antimicrobial resistance have been reported. 10 Apart from the emergence of drug resistance; biofilm production is a significant concern that may result in therapeutic failure as well. 11
A “biofilm” is an accumulation of intricate, planktonic microbial communities that are either deeply embedded inside an extracellular matrix or firmly adhered to a surface. 12 Biofilms help the bacteria to adhere to various surfaces of inanimate objects such as implants. It also helps to survive adverse conditions such as antibiotic-mediated killings. 13 Various anti-biofilm molecules have been identified, such as synthetic chemical compounds, herbal agents, peptide molecules, and antibiotics. However, their efficacy has not been clinically evaluated. 14 It is challenging to study the composition of biofilms in experimental conditions since they are inherently heterogeneous. 15 The mechanism of production of biofilm in patients with urinary catheters illustrates that bacteria produce an extracellular matrix that helps in attachment to the urinary epithelium and thereby protects against the host immune mechanism. 16 Various studies are designed to determine the association between parameters such as phenotypes, characteristics of biofilm development, and drug resistance among uropathogenic E. coli.17–18 To the best of our knowledge, however, there are no studies that compare the phenotypic traits, biofilm development, and antibiotic resistance among C. freundii isolated from UTIs. The current study aimed to examine the association between phenotypic traits, antibiotic resistance, and biofilm-producing capacity of urinary isolates of C. freundii.
Materials and Methods
Collection of Isolates
For the study, a total of C. freundii isolates (n = 120) (originated from laboratory-confirmed UTI) that were received at the M.M.Institute of Medical Sciences and Research, Ambala, Haryana, India, were used. Laboratory-confirmed UTI is defined as bacterial isolates obtained from clean catch midstream urine samples having culture results of >10 5 colony forming units (CFUs) or >5 × 10 3 CFUs and abnormal urine analysis. 19 Of the total C. freundii strains (n = 120), pan-susceptible strains (control group = 20), ciprofloxacin-resistant (CIP group, n = 20), fosfomycin-resistant (FOS group, n = 20), trimethoprim-sulfamethoxazole-resistant (SXT group, n = 20), gentamycin-resistant (SXT group, n = 20), and extended spectrum lactamase (ESBL) producers (n = 20) were included. For each antibiotic resistance group, different bacterial strains were used irrespective of their resistance pattern. Standard laboratory protocols were used for bacterial identification. 20
Quality Control
To ensure sterility, every new batch of culture media was incubated at 37°C overnight. E. coli ATCC 25922 (pan-susceptible) and Pseudomonas aeruginosa (P. aeruginosa) ATCC 27853 (MDR) were used as quality control strains for antimicrobial susceptibility testing (AST). ESBL screening and phenotypic confirmatory assays were tested using the ESBL-producing K. pneumoniae ATCC 700603, E. coli ATCC 25298 (strong biofilm producer), and E. coli ATCC 35218 as weak biofilm producers. However, other human clinical laboratory isolates of C. freundii resistant to ciprofloxacin (minimum inhibitory concentration [MIC] = 2 µg/l), fosfomycin (MIC = 64 µg/l), trimethoprim-sulfamethoxazole (MIC = 16 µg/l), gentamycin (MIC = 8 µg/l) were used as control strains.
Colony Characteristics
To study the bacterial colony morphology, the bacterial isolates were cultured on MacConkey agar (Himedia, Mumbai, India) and incubated aerobically at 37°C for 24 h. After the incubation period, the culture plates were examined for size (<3mm and >3mm considered as small colonies and large-sized colonies, respectively), consistency of the colonies (mucoid or non-mucoid), and lactose fermentation of the bacterial colonies.
Antibiotic Susceptibility Testing
AST was carried out on Mueller-Hinton agar (Himedia, Mumbai, India) using the Kirby–Bauer disk diffusion method. The results were interpreted as suggested by CLSI 2021 guidelines. 21 ATCC E. coli 25922 was used as the quality control reference strain.
ESBL Phenotypic Screening (Double Disk Synergy Test)
For double disk synergy test, ceftriaxone and amoxicillin-clavulanic acid discs were tested at a distance of 30 mm apart (edge to edge), and enhancement in the zone of inhibition was noted. 22
Phenotypic Confirmatory Test
For the combined disk test, a disc containing ceftazidime (30 µg) alone and a ceftazidime/clavulanic acid (30 mg/10 mg) combination was tested as per CLSI guidelines. An increase in the zone diameter (≥5 mm) for either of these discs tested was considered an ESBL producer. 23
Detection of Biofilm Production
For the tissue culture plate (TCP) method, 10 ml of trypticase soy broth was inoculated with isolates to be tested and incubated under aerobic conditions for 18–24 h at 37°C. This inoculum suspension was diluted in 1:100 with the newly prepared medium. A 96-well TCP was then used to incubate 0.2 ml of this bacterial inoculum (in each well) at 37°C overnight. The un-inoculated broth was added to four wells to check the sterility of the medium and any other nonspecific binding. Immediately after overnight incubation, the plates were tapped to remove free bacteria and washed four times using 0.2 ml of phosphate-buffered saline. Then these plates were air-dried, fixed, and stained using 0.1% crystal violet dye (0.2 ml) for 10 min. After that, these plates were washed using deionized water three to four times to remove added stains, followed by air-drying and the addition of 0.2 ml of 33% glacial acetic acid in each well. The optical density of each well was measured using an enzyme-linked immunosorbent assay reader (wavelength of 570 nm), and the results were interpreted as negative (<0.17), weakly positive (0.17–0.34), moderately positive (0.35–0.68), and strongly positive (>0.68).24, 38
Statistical Analysis
Using Microsoft Excel 365, descriptive statistical data analysis was performed (Microsoft Corp.; Redmond, WA, USA). In addition, the association between colony morphology and biofilm production, antibiotic resistance and biofilm production was analyzed using the Chi-square test (p < 0.05; Chi-square test was considered significant) (GraphPad Prism 9 online version).
Ethical Details
Patients’ clinical, personal, and epidemiological details were not gathered or presented during the study; their serial numbers were used to identify bacterial isolates. Therefore, the current study obtained a waiver of consent from Maharishi Markendeswhar university’s ethical committee.
Results
The phenotypic characteristics (such as size and consistency of the bacterial colonies), antimicrobial resistance, and biofilm production in both antimicrobial-resistant and pan-susceptible clinical isolates of C. freundii are summarized in Table 1. In the current study, of the total C. freundii isolates (n = 120), 30% (37/120) isolates formed large colonies (>3 mm). Of the total large colonies produced (n = 37), 40% (8/20), 20% (4/20), 25% (5/20), 30% (6/20), 40% (8/20), and 30% (6/20) were found in the control group, CIP-group, FOS-group, COT-group, Gent-group, and ESBL groups, respectively. Compared to the pan-susceptible isolates, the occurrence of large-sized colony strains was relatively reduced in most of the antibiotic-resistant groups. However, the difference was not significant (Table 1). In this study, 78.43% of isolates (40/51) produced strong biofilm formation, followed by 7.84% (4/51) moderate positive, and 13.72% (7/51) weak positive biofilm producers. Further, of the total mucoid colonies produced (n = 54), the mucoid colonies were present in 25% (5/20), 45% (9/20), 55% (11/20), 50% (10/20), 45% (9/20), and 50% (10/20) in the control group, CIP-group, FOS-group, COT-group, Gent-group, and ESBL groups, respectively. However, no substantial variation in the mucoid colonies was observed among the other groups of antibiotic-resistant isolates (p > .05, Chi-square test). All the C. freundii isolates were lactose fermenters with an exception (one ciprofloxacin-resistant isolate) that strain neither produced biofilm nor showed large colonies. Control strains such as both E. coli ATCC 25298 and E. coli ATCC 35218 produced small, mucoid colonies. Similarly, the clinical laboratory isolates of C. freundii (control strains) resistant to ciprofloxacin, fosfomycin, trimethoprim-sulfamethoxazole, and gentamycin (MIC = 8 µg/l) showed small, mucoid, lactose fermenting colonies.
The Phenotypic Characteristics (Such as Size and Consistency of the Bacterial Colonies), Antimicrobial Resistance, and Biofilm Production in both Antimicrobial-resistant and Pan-susceptible Clinical Isolates of C. freundii.
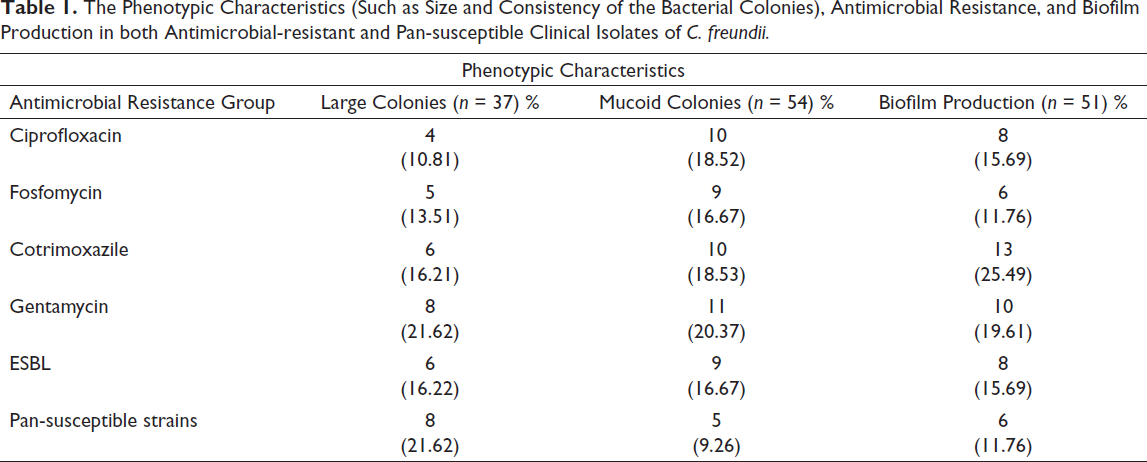
Table 2 shows biofilm production as indicated by phenotypic characteristics and antimicrobial resistance. Out of 120 isolates that were checked for biofilm production, 42.5% of isolates (51/120) tested positive for biofilm formation in the TCP method. Of the total isolates that produced biofilm (n = 51), 30% (6/20) of isolates showed biofilm formation in the control group. Alternatively, this ratio was 40% (8/20), 30% (6/20), 65% (13/20), 50% (10/20), and 40% (8/20), respectively, in the CIP-group, FOS-group, SXT-group, Gen-group, and ESBL-group. Major differences were noted in the FOS resistance group, wherein 30% (6/20) strains formed biofilm (p < 0.05, Chi-square test).
Biofilm Production as Indicated by Phenotypic Characteristics and Antimicrobial Resistance.

Discussion
UTI is a major medical concern that impacts millions of people annually. Patients with the urinary catheter are most likely to suffer a UTI, which occurs in these patients at a rate of 100% within a month of urinary tract catheterization. 25 Most hospital-acquired infections are catheter-associated UTIs, which increase healthcare expenses and also impair patients’ quality of life. 25 Biofilms might potentially serve as an ecosystem where various bacterial species and populations gather at certain locations. Due to concentrated, sequential, and/or synergistic interactions of the existing bacteria, this adversely affects the host cells, causing a significant unresolved problem for physicians and researchers. 26 The growth of bacterial biofilms on foreign body implants can result in potentially fatal infections. It is estimated that 50–70 percent of all nosocomial pathogens, or diseases apprehended in a hospital setting, are implant-associated infections. 27 The capacity to generate the EPS matrix is also markedly correlated with the vulnerability of various bacteria to develop into nosocomial pathogens and thrive in challenging hospital environments. In biofilms, antibiotics cannot permeate and reach the microorganisms in adequate quantities, and thus, biofilm indirectly contributes to bacterial antibiotic resistance. 28 A complicated web of regulatory networks, including quorum sensing (QS) systems, small non-coding RNAs, sigma factors for signal integration (via RpoS or TCS OmpR), and c-di-GMP messengers, are involved in the regulation of biofilms. 29 It is not apparent that QS-mediated genes are engaged in biofilm formation since biofilm formation is mutually beneficial and cooperative to increase the overall viability of the bacterial population. 28 Due to the intrinsic differences in the biomass compositions of biofilm and planktonic cells, it is difficult to compare the two growth phases. These evolutionary transfers explain different resistance patterns in biofilm-producing and non-producing bacterial species. It is known that both Gram-positive and Gram-negative organisms can also produce biofilms on hospital instruments and implants. While the frequent biofilm producers mainly belong to the genus Enterococcus, Staphylococcus, and Streptococcus, most of the Gram-negative organisms belong to the Enterobacteroales family and Pseudomonas aeruginosa. 30 Further, the resistance of uropathogenic C. freundii to fluoroquinolones, co-trimoxazole, and cephalosporin has been increasing in recent years. 31 Researchers have examined the relationship between the phenotypic characteristics, antibiotic resistance, and biofilm development of gram-negative bacterial isolates of E. coli and P. aeruginosa.25, 28 However, these relationships have not been studied for C. freundii isolates. The current study compares phenotypic characteristics, antibiotic resistance, and biofilm production of urinary clinical isolates of antibiotic-resistant and susceptible strains of C. freundii. For this study, five antibiotic-resistant groups (namely, CIP, FOS, COT, GEN-resistant, and ESBL producers) and pan-susceptible groups of C. freundii were formed. Each of these groups was composed of 20 clinical isolates, and the isolates were evaluated for colony characteristics (size and consistency), antibiotic resistance, and their association with biofilm production. Another interesting feature of the current study is that it used clinical isolates of C. freundii resistant to ciprofloxacin, fosfomycin, trimethoprim-sulfamethoxazole, and gentamycin as quality control strains. This was mainly because of their unavailability as standard quality control strains.
Of the total Citrobacter isolates (n = 120), 30.83% (37/120) strains produced large colonies. Out of these eighteen C. freundii isolates that produced biofilm, a link was established between biofilm formation and the size of the bacterial colonies (p < 0.05, Chi-square test). This finding was not in agreement with earlier works, which used E. coli urinary clinical isolates for investigating the relationship between the sizes of the bacterial colony and biofilm formation. 32 Further, to our knowledge, there are no studies that compare the relation between the size of the bacterial colony and the development of biofilm in C. freundii isolates. Of the 42.5% (51/120) isolates that formed biofilm in the TCP method, twenty six strains produced were mucoid colonies, indicating a significant association between the mucoid consistency of the colony and biofilm production (p < .05, Chi-square test). This finding was in association with an earlier study that observed an almost 88% correlation between mucoid consistency and biofilm formation among urinary pathogenic E. coli. 18 In the current study, all the C. freundii isolates were lactose fermenters with the exception of one ciprofloxacin-resistant isolate, indicating the low prevalence of non-lactose fermenting colonies among C. freundii. However, various studies conducted on non-lactose fermenting E. coli indicate that these strains are resistant to fluoroquinolones and tend to harbor various virulence and antibiotic-resistant genes. 33 These findings were similar to the current study, wherein the C. freundii isolates showed resistance to ciprofloxacin. However, further molecular characterization of these strains is essential to rule out the virulence genes found in these strains. On the other hand, this strain neither produced nor showed large mucoid colonies. These may be attributed to the inherent physiological or chemical characteristics of bacterial strains.
Alteration of drug target sites, decreased cell permeability, drug efflux mechanism, and other antibiotic-inactivating or destroying enzymes are examples of resistance mechanisms found in planktonic communities. However, the resistance mechanisms are different in biofilm-forming communities. Therefore, the development of microbial antibiotic resistance in biofilm ecosystems does lead to some chronic infections. 34 Antibiotic resistance is caused by the multicellularity of bacterial communities in biofilms; however, the effectiveness of the antibiotics and the host defenses may be augmented if the multicellular structure of the biofilm formation is interrupted. Further, this can enhance the speedy recovery of these chronic infections. Based on these findings, it is apparent that the understanding of the multicellular biofilm growth process will provide novel targets and methods for developing novel molecules to combat MDR bacterial infections. 35 Compared to non-biofilm-producing isolates, P. aeruginosa isolates that produced biofilms had greater resistance rates to beta-lactams and aminoglycoside antibiotics. A strong relationship was established between the ability to produce biofilms and MDR. 36 Similarly, in the current study, the rate of biofilm production varied between 40%, 30%, 65%, 50%, and 40%, respectively, in the CIP-group, FOS-group, SXT-group, Gen-group, and ESBL-groups, as compared to biofilm formation in the control group (biofilm production was found to be 30%) (Table 1). These findings make it uncertain that the decreased antibiotic resistance and enhanced biofilm formation are controlled by the same mechanisms. On the contrary, it is possible that genes encoding both biofilm production pathways and innate drug resistance are found together on the same genetic element. It is also likely that the genes for both features are present on the same plasmid, assuming that some genes for antibiotic resistance can be found in the same plasmids. However, significant differences were noted for the FOS resistance group, wherein 30% (6/20) strains formed biofilm (p < 0.05, Chi-square test). This may be attributed to the inherent property of fosfomycin-resistant groups, wherein some intrinsic factors may be inhibiting biofilm production. In biofilm-producing uropathogenic E. coli, it is documented that the inhibitors of cell wall synthesis (for instance, beta-lactams and fosfomycin) prevent the development of biofilms and also destroy the already existing biofilm communities. 37
In summary, this study correlates an association among phenotypic characteristics, drug resistance, and biofilm production, the three main characteristics of C. freundii. Moreover, a relation between phenotypic characteristics and drug resistance with biofilm formation was also noted. Conversely, further studies need to be performed to unveil the mechanisms that are accountable for these associations.
Footnotes
Acknowledgment
I thank Dr. Varsha A. Singh for providing support and also Mr. Manoj Sharma for the technical support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Patients’ clinical, personal, and gathered or presented during the study; their serial numbers were used to identify bacterial isolates. Therefore, the current study obtained a waiver of consent from Maharishi Markendeswhar university’s ethical committee.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Informed Consent
The participant has consented to the submission of the article to the journal.
