Abstract
The coexistence of humans and animals has existed for centuries. Over the past decade, animal research has played a critical role in drug development and discovery. More and more diverse animals, including transgenic animals, are used in basic research than in applied research. Transgenic animals are generated using molecular genetic techniques to add functional genes, alter gene products, delete genes, insert reporter genes into regulatory sequences, replace or repair genes, and make changes in gene expression. These genetically engineered animals are unique tools for studying a wide range of biomedical issues, allowing the exhibition of specific genetic alterations in various biological systems. Over the past two decades, transgenic animal models have played a critical role in improving our understanding of gene regulation and function in biological systems and human disease. This review article aims to highlight the role of transgenic animals in pharmacological, toxicological, and environmental research. The review accounts for various types of transgenic animals and their appropriateness in multiple types of studies.
Introduction
The coexistence of humans and animals has existed for centuries. The use of animals in research goes hand in hand with the development of medicine and modern biological science. Animals are used in scientific research to help us understand human bodies and how they work. Animals are also used to test the toxic effects of chemicals. The anatomical and physiological resemblance between humans and animals, primarily mammals, has led researchers to study various mechanisms and test new therapies in animal models before applying their discoveries to humans. Over the past decade, animal research has played a critical role in nearly all medical breakthroughs. Almost every Nobel Prize winner in physiology or medicine since the beginning of the nineteenth century has based their research on animal data. Animal models have been used to address a wide range of queries, from basic research to developing and evaluating novel biologicals or drugs.1, 2
The difference between man and animals is that man has a mind, a requirement for consciousness, and, thus, the ability to feel pain. The use of animals is not only based on the remarkable similarities in the biology of most mammals but also on the fact that human diseases often affect other animal species. This is especially true for most infectious diseases and also widespread diseases such as type 1 diabetes, hypertension, allergies, cancer, epilepsy, myopathies, and so on. These diseases have similarities, but the mechanisms are often so similar that 90% of veterinary drugs used to treat animals are identical or similar to those used to treat humans.3, 4
Each year, approximately 50–100 million animals, ranging from zebrafish (Danio rerio) to non-human primates, are used in experiments. While experiments on vertebrate animals are regulated in most countries, this is not true for experiments on invertebrate animals, so there are no accurate statistics on their use. The total number of animals used in the United States in 2010 was nearly 1.37 million. This number does not include small animals such as rats, mice (account for about 90% of experimental animals), birds, or fish. Most procedures were performed on mice and rats (96%). Other commonly used animals include guinea pigs (19%), rabbits (18%), and hamsters (13%). Over three million animals were used in the United Kingdom in 2011, primarily including mice (71%), fish (15%), rats (7%), and birds (4%). In 2004, nearly 200,000 fish and 20,000 amphibians were used in the United Kingdom, primarily zebrafish and the African clawed frog (Xenopus laevis). More than 20,000 rabbits were used in animal experiments for eye irritation tests (Draize test) in the United Kingdom in 2004. The animal use statistics in other countries like India and Brazil also show a similar pattern.4, 5
More and more diverse animals are used in basic research than in applied research. These are usually studies of embryogenesis, developmental biology, behavior, and reproduction in fruit flies, nematodes, mice, and rats. Applied research aimed at answering specific questions is usually conducted in the pharmaceutical industry or by universities. Applied research uses animal models of diseases discovered or generated in pure research programs. Preclinical toxicological tests use one million animals yearly in Europe or about 10% of all procedures. About 5,000 animals are used for each chemical test. The most rigorous tests are reserved for pharmaceuticals and food. A series of tests are performed that last from less than a month to years to test general toxicity, eye and skin irritation, mutagenicity, carcinogenicity, and teratogenicity. Rodents such as rats and mice and non-rodents like rabbits are typically used for these types of studies.4, 5
Transgenic animals are generated using molecular genetic techniques to add functional genes, alter gene products, delete genes, insert reporter genes into regulatory sequences, replace or repair genes, and make changes in gene expression. These genetically engineered animals are unique tools for studying a wide range of biomedical issues, allowing the exhibition of specific genetic alterations in various biological systems.
Over the past two decades, transgenic animal models have played a critical role in improving our understanding of gene regulation and function in biological systems and human disease. The study of human diseases is greatly facilitated by the generation of transgenic animals that mimic health disorders or enable the evaluation of new drugs. The establishment of stable transgenic animals requires the foreign deoxyribonucleic acid (DNA) to be present in gametes or unicellular embryos to enable its transfer to progeny. To achieve this objective, the foreign gene can be introduced into cells by different methods depending on the species, namely, DNA microinjection, DNA transfer into gametes, the use of transposons, the use of retroviral vectors, gene transfer using embryonic cells, and gene transfer by nuclear transfer.5–7
Pharmacology studies rely mainly on animal systems, and several subjects have been hindered for decades because of the lack of suitable animal models. The established transgenic techniques undoubtedly have facilitated the progress of animal experimentation for some pharmacological questions where potential target molecules have been characterized. Because this technology is evolving rapidly, it is foreseeable that future development of this technology will allow researchers to address some problematic pharmacological questions where no specific targets are identified. However, one should not ignore the complexity of mammalian organ systems in interpreting data collected from these animals because animals do not passively receive the experimenter’s manipulation, and endogenous gene activities may be altered in response to the introduction of exogenous DNA sequences. Therefore, in addition to technical advancement, more basic information about the control of specific gene expression in animals is required to achieve the highest level of specificity and precision in transgenic animal experiments.8, 9
Pharmacology research depends significantly on animal models, and several topics have been unexplored for decades by the lack of appropriate animal models. Validated transgenic techniques have certainly enabled the advance of animal studies for some pharmacological questions in which possible targets have been characterized. As this technology rapidly evolves, researchers can answer some pharmacological issues for which specific targets have not been identified. However, the researchers must consider the complexity of animal organ systems, and they resist experimental manipulations, and endogenous gene activities can be altered in response to the introduction of exogenous DNA sequences. Therefore, in addition to technical advances, more basic information on the control of specific gene expression in animals is needed to achieve the highest level of specificity and precision in transgenic animal experiments.9, 10 In this review, the authors highlight the role of animals, especially transgenic ones, in pharmacological, toxicological, and environmental research. The review accounts for various types of transgenic animals and their appropriateness in various types of studies. This article explains different transgenic animal models used for pharmacological, toxicological, and environmental studies.
Transgenic Animals in Research
Transgenic animals (predominantly mice or rats) are animals whose genomes have been intentionally modified by the irreversible insertion of recombinant or synthetic nucleic acid molecules, or nucleic acids synthesized from them, into the germline. 11 Genetically modified animals have gained prominence as research subjects since 1981 when J.W. Gordon and F.H. Ruddle coined the term “transgenic.” Recently, the widespread use of transgenic animals as human models for various studies in basic biological systems, gene regulation, medical research, toxicology, biotechnology, agriculture, and aquaculture has given us novel insight and enhanced our understanding in these diverse fields. Given the enormous need for studies using human models, the use of transgenic animals is expected to increase in the future. However, parallel to the development of transgenic technology, ethical concerns about its use have arisen. These concerns are diverse, encompassing animal welfare, human health, and environmental issues. They include animal suffering caused by the expression of transgenes that cause tumors or neurodegenerative diseases, as well as the possibility of transgenic animals escaping into the environment, not to mention the possibility of altering the human genome. 12
Transgenic animals are created using three methods, namely DNA microinjection, retrovirus-mediated gene transfer, and embryonic stem (ES) cell-mediated gene transfer. 13 The first method developed is DNA microinjection, the basis for the other two methods. The introduced DNA may cause over- or under-expression of specific genes and the expression of novel genes. Integration of the introduced gene into the host DNA is a random process achieved by the microinjection of DNA into the pronucleus of a fertilized ovum. The introduced gene will not always insert itself into a site that will allow its expression. As a result, other methods, such as vector-mediated gene transfer and homologous recombination, have increased the likelihood of expression. Retroviruses are frequently used as vectors to deliver genetic material into cells. 14 The third method employs homologous DNA recombination to precisely target DNA sites in ES cells. If the homologous sequence to be introduced into the cell contains a mutation or a gene from another species, the new sequence will replace the targeted gene. This procedure, known as the “knock-out” method, is the method of choice for gene inactivation and is especially important for studying the genetic control of developmental processes. 15
Types of Transgenic Animals Used in the Research
The creation of genetically modified animals is aimed at a wide range of species (Table 1).16−59 Cows, pigs, chickens, goats, sheep, dogs, cats, rabbits, fish, rats, and mice are among the safest genetically modified animals. 60 This has shown great importance in assessing therapeutic approaches in human disease models and studying disease progression. As transgenic vertebrae are known to help understand and treat diseases, transgenic invertebrates have proven their value in the agricultural field. In the near future, transgenic animals will greatly impact human life.
Type of Transgenic Animals.

Transgenic mammals such as cows, sheep, goats, pigs, rabbits, and monkeys are preferred in research studies because they phylogenetically mimic the human biological and homeostatic systems. They do, however, necessitate a significant investment of money, time, and resources. The choice of use for these animals has not been encouraging among researchers, owing to the ethical issues that have arisen as a result. 61
Mice and rats have long served as the preferred species for research purposes as animal models due to their anatomical, physiological, and genetic similarity to humans. The significant advantages of mice include their small size, ease of maintenance, short life cycle, fast breeding, and abundant genetic resources. 62 However, the average rate has only limited scope for human study. On the other hand, transgenic mice can be engineered to specifically focus on essential aspects of the disease. 62 The first transgenic mice were created by microinjecting Simian virus 40 into explanted mouse blastocysts and exposing early embryonic cells to retroviruses. As a result, other methods for producing transgenic animals have evolved. 63
Transgenic Rodents in Research
Transgenic rodents are predominantly mice, and they were used to produce human antibodies for therapeutic purposes. In 1992, United States Food and Drug Administration (US-FDA) approved the first therapeutic monoclonal antibody (mAb) (muromonab-CD3) an immunosuppressant. As of December 31, 2018, US-FDA has approved 90 therapeutic mAbs, including two diagnostic mAb. 64 Recently (December 24, 2021) US-FDA approved mAb (EVUSHELD™ [tixagevimab co-packaged with cilgavimab]) for preexposure prophylaxis (prevention) of COVID-19. 65 Mouse monoclonal antibodies can neutralize coronavirus and have been produced in mouse milk at a concentration of up to several grams per liter. 66
Transgenic Rodents in Toxicological Studies
Toxicological data are essential for any pharmaceutical and investigational drugs to understand the effects on the biological system. Also, these data will help to titrate the dose for further experimental and clinical pharmacological studies. The currently available toxicological assay methods (in vivo) are designed using healthy rodent/non-rodent species. But most pharmaceutical formulations are indented for therapeutic purposes. If, further toxicological studies are extended to explore the effect on the diseased animal model, it may be helpful to understand the interaction between the investigational drugs and disease. Developing animal models with the desired pathological conditions is a complex process, and sometimes it would not mimic human disease conditions. Also, maintaining the animals with the desired pathological conditions is a tedious process and requires additional care to minimize the experimental hazards. On the other hand, developing animal models with the desired pathological conditions using genetic engineering techniques will help fulfill the experimental requirement effectively.
The severity of the toxicity of drugs mainly depends on physiological, disease, and genetic factors. In the last few decades, more attention has been given to genetic factors that play a major role in drug metabolism. The drugs are chemical in nature, and they may cause at least oxidative changes that influence other messengers’ activities if it does not follow the biotransformation process. The biotransformation process or metabolic activity occupies a critical role in the toxicity of many drugs. 67 Cytochrome P450 enzymes are key enzymes and are responsible for drug metabolism. This class has more than 50 enzymes, and six (CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP3A4, and CYP3A5) of them metabolize 90% of the drugs. 68 Any mutations in this/these metabolizing enzyme(s) alter the severity of the toxicity of drugs. In recent years, studies identified polymorphisms in human genes that alter the drug metabolism and severity of the toxicity. Using genetically engineered rodent models influence of polymorphisms on drug metabolism and toxicity can be explored. The transgenic animal models are best suited for these purposes.
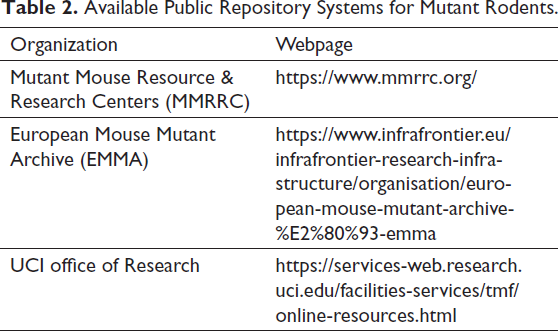
In the last few decades, transgenic animal models have been widely used in biomedical research. These animals play an important role in understanding gene regulation and functions in the biological system and human disease. Transgenic animal models are the tool to examine the role of individual genes in various toxicological outcomes. 69 Transgenic animals have extra or exogenous fragments of DNA in their genome by inserting the gene of interest into ES cells or into a zygote. 70 This DNA is randomly incorporated into the mouse genome by homologous or by non-homologous recombination. This endogenous process allows the integration of foreign DNA into the host genome. The ES cells technique is preferred over zygotic injection because controlling the integration of foreign DNA into the host genome is difficult in zygotic injection. 69 These genetically modified rodents/transgenic rodents are most commonly utilized in cancer studies and carcinogenicity and mutagenicity tastings. Before the discovery of transgenic animals, comet assay and unscheduled DNA synthesis assays are frequently used to detect premutagenic lesions of investigational compounds. 71 OECD TG 488 is recommending lacZ bacteriophage mouse, lacZ plasmid mouse, gpt delta mouse, and rat and lacI mouse and rats for the mutation assay. 71 In COVID-19 research, both transgenic hACE2 mice and nongenetically engineered mouse models (GEMMs) are used. 72 Genetically modified rodents are largely used in genotoxicity and carcinogenicity tests. The available public repository systems for mutant rodents are listed in Table 2. The animal strains such as Big blue mice/rats, gpt delta rats, Human c-Ha-ras proto-oncogene transgenic rats, Probasin-SV40 T antigen transgenic rats, and Connexin 32 dominant-negative transgenic rats are used in the toxicological research, and their importance is summarized in Table 3.73−79
Available Public Repository Systems for Mutant Rodents.
Importance of a Few Transgenic Strains in Preclinical Research.

Transgenic Rodents in Environmental Research
Environmental mutagens and carcinogens such as cigarette smoke play a vital role in the development of many diseases, including cancer. International Agency for Research on Cancer (IARC) categorized more than 100 chemicals, including biological agents, as carcinogenic to humans. 80 Transgenic rodents are a unique tool for understanding how individual genes and the environment affect human health. These animal models help to identify and characterize dietary, occupational, environmental, genetic, and other factors that induce the neoplastic process. 81 The use of transgenic fish in ecotoxicology is a relatively recent development. It has the potential to provide advanced and integrated systems for assessing risks of waterborne and sediment-associated contaminants. 82 This transgenic fish may contribute to emerging issues concerning environmentally induced, reproductive, developmental, and heritable diseases. 83
Future Perspective
“Gain of function [Knock-in’s, altered regulation, new expression, over-expression, antisense Ribonucleic acid (RNA), Insertional mutations]” or “loss of function [Knock-out’s, altered regulation, lost expression, over-expression, antisense RNA, Insertional mutations]” is the two main strategies used in producing transgenic animal. These transgenic animals can be used to study the basic mechanisms of genetics, role of genes in the progression of tumor (oncogenes), interaction between cells and immune system, health and genetic disease model, and mechanism of growth control.
84
Many monoclonal antibodies, which are now derived from transgenic mice, are commonly indicated for the treatment of cancer and immunological disorder. Cancer is a multifactorial disease influenced by genetic and environmental factors. Developing such disease models in normal animals is the default. Hence using modern technologies to establish disease models in animals that mimic the human physiological condition may help to develop a drug for rare diseases/disorders. Still, we are fighting old enemies such as tuberculosis. Every year, we are added with new health care tasks (e.g., developing drugs for RNA viruses), which increases the health care burden. The major reason for the delay in the drug development process is limitations in the availability of preclinical models. The available disease models are used to develop a model with similar clinical symptoms, but these models are not genetically identical to human diseases. Hence developing transgenic animal models with genetic similarities may help reduce the gap between drug development and clinical utilities.
Conclusion
Transgenic animal and animal models have large applications in biomedical research. Currently, these animals are largely used in the production of monoclonal antibodies and genotoxicity testings. Further exploration may lead to the development of new models for rare genetic disorders that may be used in pharmacological assays and toxicological studies.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Not applicable
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable
