Abstract
Aim:
To determine the concentration of 6-thioguanine nucleotide (6-TGN) and 6-methylmercaptopurine (6-MMP), the interpatient variability, and the relationship with disease activity in patients with Chron’s disease on long-term stable doses of azathioprine (AZA).
Methods:
This is a prospective, tertiary care single-center hospital study in adult Chron’s disease patients treated with AZA. The quantification of phenotypic thiopurine methyltransferase enzyme activity in red blood cells and the estimation of the concentration of 6-TGN and 6-MMP in whole blood was performed using the HPLC-UV detector method. A clinical response was categorized as remission (Harvey-Bradshaw Index [HBI] < 5) or improvement (drop from baseline of at least three points of HBI) based on HBI. Exposure to metabolite concentrations and the clinical response to AZA treatment was observed.
Results:
Study analysis included 30 patients who were initiated on AZA, and they were followed up with an estimation of metabolite concentrations to determine their clinical outcome. At six months, 93% of (n = 28) patients continued to be on AZA and had clinical improvement. All the patients achieved remission of Chron’s disease. Only two patients developed adverse effects such as joint pain and thrombocytopenia.
Conclusion:
AZA is a safe and effective therapy in managing Chron’s disease when administered after determining thiopurine methyltransferase phenotype and with dose optimization performed using therapeutic drug monitoring of 6-TGN and 6-MMP.
Introduction
Inflammatory bowel disease (IBD) includes two clinical conditions, Crohn’s disease and ulcerative colitis, characterized by chronic inflammation of the gastrointestinal tract, and an increase in IBD incidence is observed in the past few years. Pharmacological agents recommended for their treatment include AZA, corticosteroids, mesalazine, biologicals, and antibiotics. Among these drugs, AZA is commonly used to treat IBD and it induces an almost complete inhibition of the production of TNF α, IL-1β, and IL-6, by colonic mucosa. 1
The active metabolite of AZA, 6-thioguanine nucleotide (6-TGN), accumulates in lymphocytes and blocks the expression of cytokines, and finally inhibits the inflammatory response induced by T cells in the intestinal lamina propria. Various studies have shown the efficacy of AZA and 6-mercaptopurine in maintaining remission, 2 and meta-analysis performed by Chatu et al. showed a reduction in chance for surgical resection by 40% in Crohn’s disease. 3
The pharmacokinetics of thiopurine drugs are complex, with high interindividual variability. Multiple pathways such as thiopurine methyltransferase (TPMT), hypoxaine-guanine phosphoribosyltransferase, and xanthine oxidase (XO) are involved in the metabolism of thiopurine drugs. After absorption from gastrointestinal tract, roughly 88% of AZA gets converted to mercaptopurine in red blood cells, which is subsequently metabolized to 6-thiouric acid (XO pathway), 6-methyl mercaptopurine (6-MMP), and 6-TGN Hypoxanthine-guanine phosphoribosyltransferase pathway. TPMT enzyme is involved in converting 6-mercaptopurine to inactive 6-MMP; the enzyme’s activity largely depends on the genetic constitution of the individual. TPMT enzyme activity can be quantified to determine whether thiopurine treatment could be initiated in a patient. The suboptimal activity of this enzyme may lead to increased production of the active metabolite 6-TGN which may lead to myelotoxicity.4, 5 In the Indian population, we observed a wide distribution of TPMT enzyme activity (3.85–35.68 nmol/h/mL erythrocytes), which suggests high interindividual variability in the formation of 6-TGN and, therefore, clinical efficacy and safety of thiopurine drugs. 6
Lack of compliance to AZA is common among IBD patients because of the slow onset of clinical relief and adverse effects.7, 8 To overcome the difficulty in dosing AZA because of its complex pharmacokinetics and to reduce the occurrence of adverse effects, it is recommended to monitor the AZA metabolite concentration (6-TGN and 6-MMP) when treated with AZA. In studies performed on adult and pediatric population, a good association was observed between 6-TGN concentration (>230 pmol/8 × 10 8 red blood cells [RBCs]) and disease remission.7, 9–12
This study evaluated the clinical improvement of patients with Crohn’s disease and their exposure to 6-TGN and 6-MMP concentrations. Dose optimization was performed by the treating clinicians based on exposure to 6-TGN and 6-MMP at their discretion.
Materials and Methods
Materials
Recruitment of Patients
Patients diagnosed with Crohn’s disease and on a constant dose of AZA for a minimum of eight weeks were included in the study. The institutional ethics committee approval was obtained before the initiation of the study and written informed consents were obtained from the study participants after elaborating study details. The baseline HBI, a validated disease activity score, was calculated, and TPMT enzyme activity was measured before the treatment with AZA was initiated. After administering stable doses of AZA for a minimum period of eight weeks, 2 mL of blood specimen was collected in a K2EDTA tube. As the patients were from long distances, telephonic consultations were done. Dose optimization by the treating clinician was performed based on clinical parameters only. The factors considered at this time included the clinical condition (as determined by the improvement in HBI with minimal toxicity) and inflammatory markers. After which, during the patient’s next visit to the hospital, it was confirmed that they were on the clinician recommended stable dose of AZA for a minimum of eight weeks. During this visit, an additional blood specimen (2 mL) was collected. Whole blood was aliquoted and stored at –20°C till analysis to quantify 6-TGN and 6-MMP and the blood concentration of the metabolite. Clinical parameters such as HBI and inflammatory markers were repeated during this visit.
Preparation of Calibration Standards
Primary stock solutions of 1 mg/mL of 6-TGN in 0.1 mol NaOH, 1 mg/mL of 6-MMP in HCL, and 1 mg/mL of internal standard (aminoacetophenone) in methanol were prepared and stored at –70°C. Further dilutions of the stock solutions in water were made to prepare final calibrators of QC concentrations. Calibrators for 6-TGN were 996.5, 597.9, 498.25, 299.1, and 199.4, and for QC were 249.2 and 697.9 pmol/0.8 × 10 8 erythrocytes. Calibrators for 6-MMP were 8026.5, 6019.8, 5016.5, 4013.2, 2006.6, 1003.3, 501.65, and for QC were 3511.55, 7023.1 pmol/0.8 × 10 8 erythrocytes.
Measurement of 6-TGN and 6-MMP Concentrations in Whole Blood of Patients
On the day of analysis, 20 µL of internal standard (aminoacetophenone) and 100 µL of D, L-dithiothreitol were added to 200 µL of thawed whole blood, and vortex mixed for 1 min. Proteins were precipitated with 50 µL of 70% HCLO4. After centrifugation at 12,300*g for 10 min, 250 µL of supernatant was kept in the heating block at 100°C for 60 min to hydrolyze 6-TGN to corresponding bases (6-TG) and to convert 6-MMP to 4 amino-5-(methylthio) carbonyl imidazole. These samples were placed in an ice bath for 10 min, followed by 50 µL di-potassium hydrogen phosphate, and 80 µL supernatant was injected into the HPLC. The concentrations were expressed in pmol/8 × 10 8 RBCs.
High-Performance Liquid Chromatography Conditions
Waters e2695 Separations Module quaternary pump with symmetry C18 column (150 × 3.9 mm; 5 µm particle size) and 2489 UV/Vis detector system was used for separation and quantification of analytes. The mobile phase used was a mixture of 20 mM phosphate buffer of pH 3.5 and 7% methanol at a constant flow rate of 1.2 mL/min. The column was maintained at a temperature of 35°C and autosampler at 5°C, respectively. The retention time for 6-TGN, 6-MMP, and internal standard were approximately 6 min, 11 min, and 15 min, respectively. Chromatographic response for 6-TGN and internal standard was measured at 355 nm, and that of 6-MMP and internal standard was measured at 302 nm.
Results
Characteristics of Patients with Crohn’s Disease on Treatment with Azathioprine

*p Value < 0.001; #p-Value < 0.05.
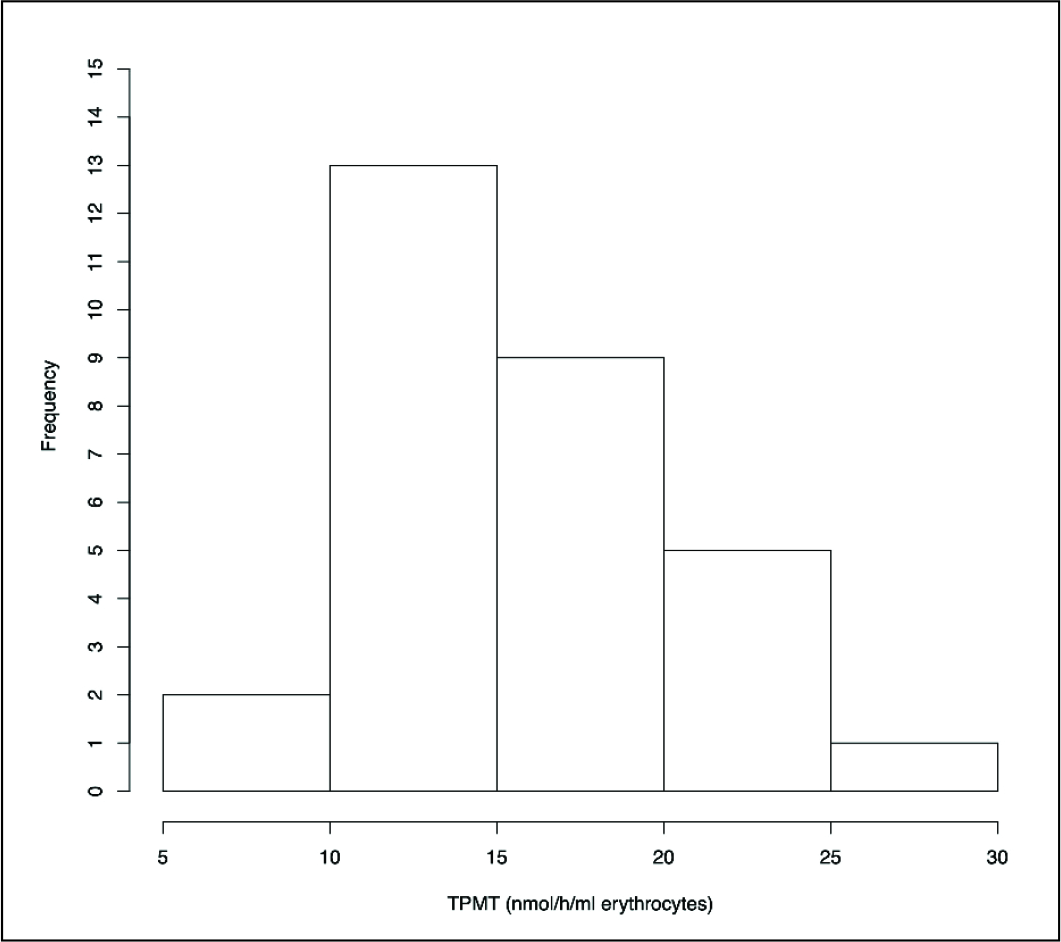
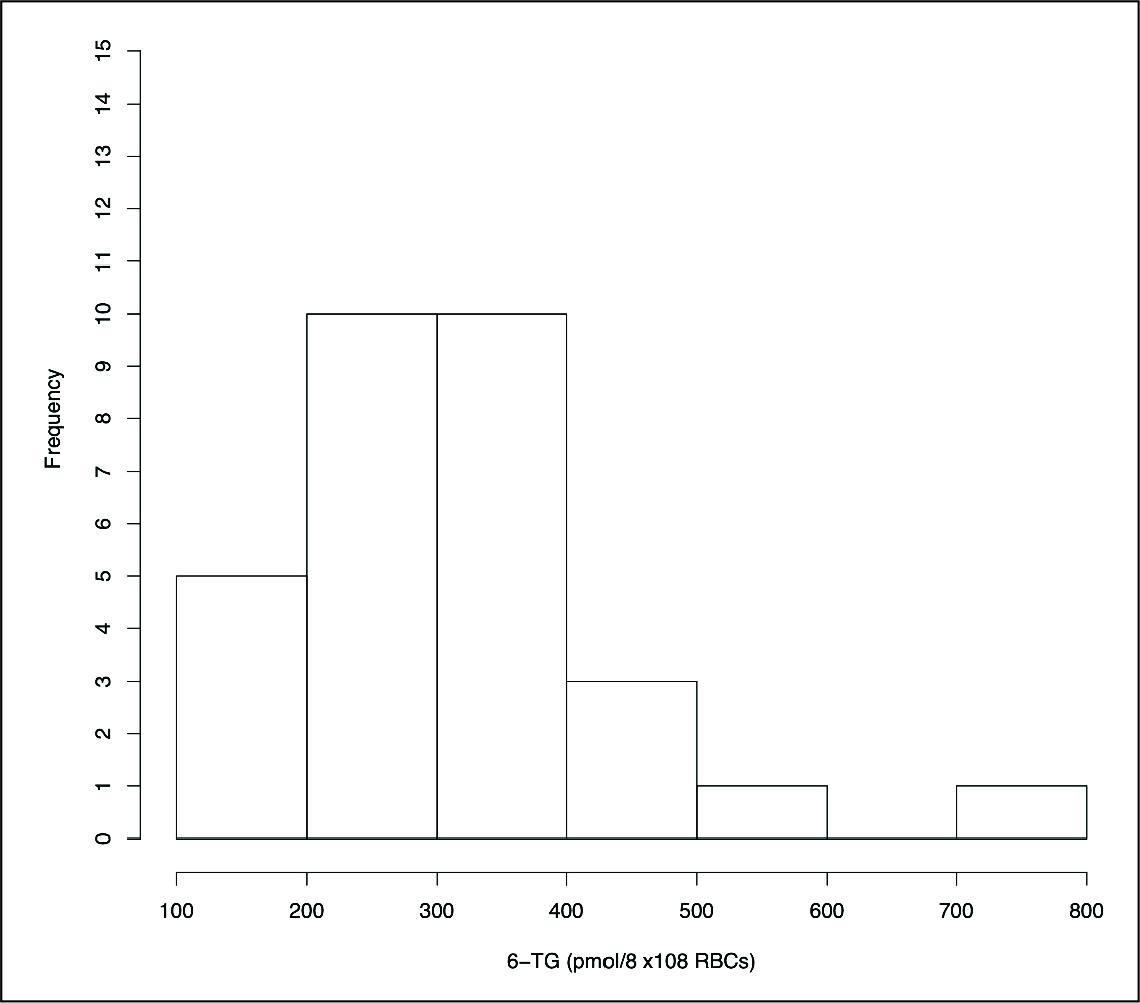
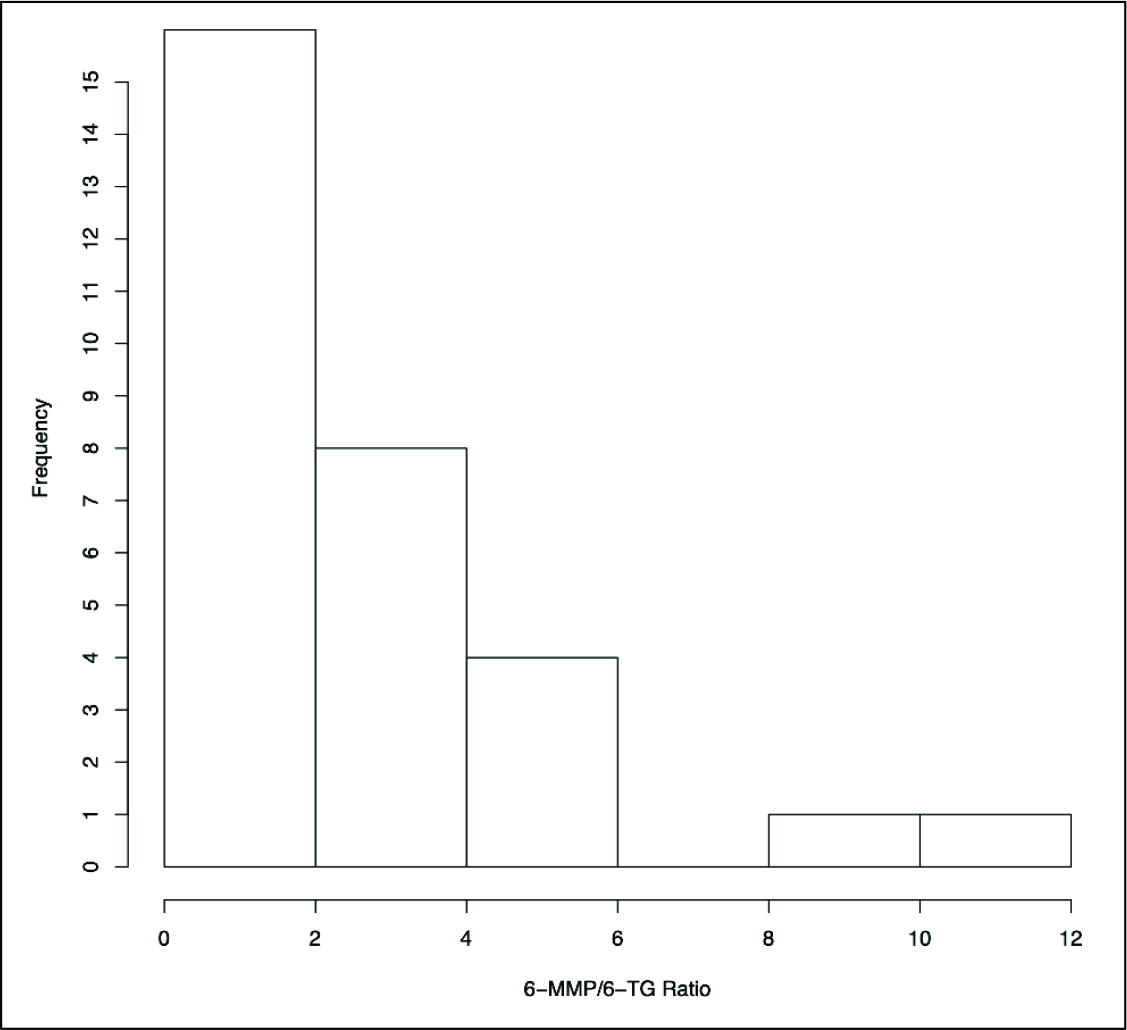
In all the patients, the minimum starting daily dose of AZA was 50 mg because TPMT activity was >8 nmol/h/mL erythrocytes (Figure 1). In six patients, the dose of AZA was increased to maximize clinical improvement after determining the metabolite concentrations in steady state. The interindividual variation (CV%) in a weight-adjusted dose of AZA (range: 0.7–2.5) was 36.7%. According to the treating clinician’s discretion, all patients recruited into the study achieved remission after dose optimizationas as patients were doing well clinically and steroids has side effects clinicians stoppped the steroids. The range of 6-TG concentrations was 120 to 798 pmol/8 × 10 8 RBCs (median [IQR] = 301 [247.3–380]) and the range of 6-MMP/6-TG ratio was 0 to 10.4 (median [IQR] = 1.74 [0–3.2]; Figure 2 and Figure 3). After AZA dose optimization, 80% of patients had 6-TG concentrations above 235 pmol/8 × 10 8 RBCs. The range of final stable doses of AZA was 50 mg to 100 mg (median [IQR] = 50 [50–93.8]). All the patients who had mild to moderate IBD achieved remission after the AZA therapy and dose optimization. After initiating guided treatment with AZA, there was a clinically significant improvement in serum albumin, C-reactive protein, and HBI (Table 1).
Distribution of TPMT Phenotypic Enzyme Activity in Indian Population.
Distribution of 6-TGN Concentration in Indian Population.
Distribution of 6-MMP/6-TGN Ratio in Activity in Indian Population.
The interindividual variability of AZA doses that normalized 6-TG and 6-MMP were 47.6 and 107.9, respectively.
There was no correlation between the final HBS score and 6-TG concentrations or 6-MMP/6-TG ratio (spearman, rho = 0.06 and 0.08, respectively). There was no good correlation between TPMT activity and the 6-MMP/6-TG ratio (r = –0.17).
The 6-TGN concentration among those who received 5-aminosalicylic acid (293 pmol/8 × 10 8 RBCs), was not significantly different when compared to those who did not receive (352 pmol/8 × 10 8 RBCs) 5-aminosalicylic acid (Wilcoxon rank sum test, P = .3).
Discussion
In this study, we did not observe any significant correlation between TPMT activity, 6-TGN concentration, 6-MMP/6-TG ratio, and clinical efficacy. This may be because of the relative low disease activity at baseline (HBI: 0–9). There was a median HBI reduction of 66.7% from baseline, and all patients had remission of disease with the final optimized dose of AZA. Most of the patients (interquartile range) had TPMT activity in 13.1 nmol/h/mL to 18.4 nmol/h/mL erythrocytes. Only two patients had TPMT activity lower than 10 nmol/h/mL erythrocytes; nevertheless, the 6-MMP/6-TG ratio was more than 1.5 in both these patients. Lowest TPMT enzyme activity observed in this cohort of patients was 8.4 nmol/h/mL erythrocytes, considered normal TPMT activity. The lowest dose of AZA given was 50 mg (0.73 mg/kg), and majority of the patients did not experience any serious adverse event during AZA therapy over one year of follow-up.
Concomitant treatment with 5-aminosalicylic acid did not affect the 6-TGN concentration in our study population, and this is in agreement with the study performed by Armstrong et al. 13 The poor correlation between the metabolite concentration and TPMT activity agrees with the analysis performed by Jonason et al. 14 This may be because of the involvement of multiple pathways in the metabolism of AZA. In this study, we have not quantified the activity of XO, which contributes to over 50% of elimination of AZA. 15
The median dose of AZA was 1.3 mg/kg, which was adequate to achieve remission in all the patients with mild to moderate Crohn’s disease. Most of the patients (80%) had a 6-TGN concentration above 235 pmol/8 × 10 8 RBCs, and this observation is similar to the study performed by Dubinsky et al. 10 There was a high interindividual difference in final stable weight-adjusted dose of AZA received by patients (CV% = 36.7). The patient who received the lowest dose of AZA (0.73 mg/kg) with normal TPMT activity (14 nmol/h/mL erythrocytes) had a 6-TGN concentration (284 pmol/8 × 10 8 RBCs) close to the median 6-TGN concentration (301 pmol/8 × 10 8 RBCs). The maximum concentration of 6-TGN achieved in patients recruited for the study is 700 pmol/8 × 10 8 RBCs, and this patient did not develop any adverse effects. A few case reports were published where patients achieved 6-TGN concentrations of around 800 pmol/8 × 10 8 RBCs and did not develop adverse effects. 16 After determining metabolite concentrations, the dose of AZA was increased in 20% of patients to optimize clinical outcomes. It is advisable to perform dose optimization using therapeutic drug monitoring in patients with Crohn’s disease. Further clinical studies are required in patients with moderate to severe Crohn’s disease to establish the role of TDM in AZA dose optimization.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Not applicable
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable
