Abstract
Parkinson’s disease (PD) mainly affects the dopaminergic neuronal networks of the substantia nigra, which leads to both motor and nonmotor symptoms of the disease. Based on the reports from the previous studies, 95% of the cases are presented along with olfactory dysfunction. The relevant publications from 2002 to 2021 were searched and shortlisted using PubMed and Google Scholar. In this review, we have discussed the correlation between olfactory dysfunction and PD. Olfactory damage presents earlier than the motor symptoms. Because there are no current methodologies for the early detection of PD, olfactory dysfunction can be used as a potential marker for the early detection of PD and hence paving the way for better therapeutic approaches.
Introduction
PD is one of the most common neurodegenerative diseases and currently the most prevalent movement disorder. The ailment was initially explained in 1817 by James Parkinson, who described the disease accompanied by several motor symptoms, including tremors, rigidity, and bradykinesia.1, 2
PD mainly affects the dopaminergic neuronal networks of the substantia nigra, which leads to both motor and nonmotor symptoms of the disease. Based on the reports from the previous studies, the nonmotor manifestations, including loss of smell and depression, sleep abnormalities, hypotension, and gastric problems, will have an early emergence than the other manifestations involving the motor activity. 1 Among the nonmotor symptoms, the major manifestation of PD, which is prevalent in more than 95% of the PD-diagnosed people, was the absence of smell. 2
As the olfactory system is one of the early systems affected by PD, it can be used as a potential marker, which helps in the early detection and treatment of the disease. This article overviews the role of the olfactory system in the etiogenesis of PD.
Methodology
To collect data about olfactory dysfunction in PD, a search was performed with PubMed and Google Scholar from 2002 to 2021. The keywords used include “Parkinson’s disease,” “Olfactory dysfunction,” “olfactory bulb,” “molecular mechanism,” “olfactory epithelium,” “neurodegeneration,” “hyposmia,” and “clinical evidence.” Both research and review articles were included; however, only publications in English were considered.

Etiogenesis of PD
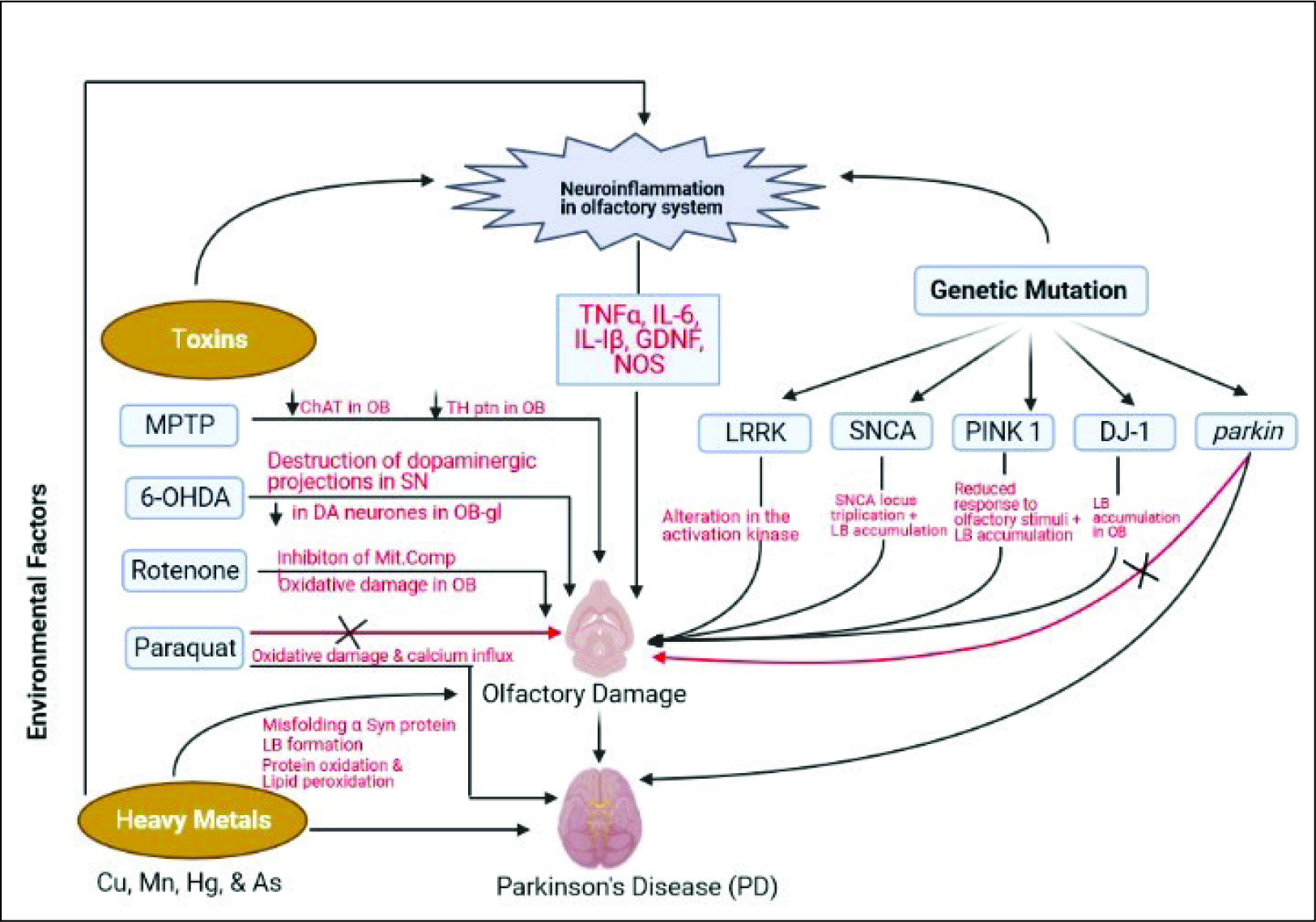
In the majority of the cases of PD, it is idiopathic. Even though it can be caused by various factors, such as genetic mutations, age, environmental toxins, and other comorbidities. Oxidative damage in PD is mainly caused by the generation of free radicals, resulting in the impairment of mitochondrial function, glial cell activation, α-synuclein aggregation, α-synuclein, and alteration in the calcium signaling. 3
Genetic Mutation
Among the reported cases of PD, about 1/10th had a familial history. Mutational changes in cause the LRRK2 and SNCA cause PD with a dominant autosomal inheritance, and mutational changes in th Parkin (PARK 2), PINK1, DJ-1, and PARK 9 genes cause the recessive inheritance of the disease. The onset of the infection happens in a younger period of life in the generations succeeding the familial type of PD.
Autosomal Dominant Forms
The LRRK2 gene mutation is a common observation in the familial PD, and it manifests common symptoms, including tremors, cognitive loss, and behavioral disorder. It was found to have a modification of the Y1699C missense type, which presented all the clinical and pathological manifestations. Further studies revealed a correlation between the LRRK2 gene mutation and olfaction. An olfactory assessment conducted in LRRK2-gene-mutated PD patients showed the presence of Lewy bodies and neuritis in the substantia nigra. Also, the olfactory bulb, cortex, and brain stem were positive for α-synuclein. This further shows that the alteration in the activation of the kinase by the mutated-LRRK2-encoded protein can affect the olfactory system. 4 From the studies conducted to analyze the correlation between PD-related genes and olfactory dysfunction, it was found that 90% of the people with a genetic mutation on the SNCA gene had either total loss of smell or a reduction in smell sensation.
Autosomal Recessive Forms
In the case of PARK2-mutated forms of PD, there was no observed olfactory dysfunction. In some rare cases, the mutated gene carriers having PD showed better olfactory scores. Therefore, the effect of the PARK 2 gene mutation on olfaction is yet to be studied. In the PINK1 mutation studies conducted on fruit flies related to PD, it was found that the olfactory responses were deficient in PINK1-gene-mutated flies. They responded to the olfactory stimuli with higher strength. 5 Similarly, the formation of Lewy bodies is a common observation of PD with monogenic origin because of the mutation in DJ-1. 6 DJ-1 is important in the antioxidant defenses. In the people diagnosed with PD, there is an observed increase in the amount of oxidized DJ-1. Also, oxidized DJ-1 was found in the olfactory bulb; the substantia nigra PARK 9 is also associated with the autosomal recessive type of inherited PD, and it is common in the cases of PD with early onset and is usually responsive to levodopa treatment.
Age
PD etiology is also linked to old-age-related biological damages, which include instability of the genome, damage to mitochondria, and epigenetic alterations. The most important risk factor in age-related neurodegenerative diseases, like PD, is the disruption in the signaling of wingless-type mouse mammary tumor virus integration site (Wnt)/β-catenin (WβC), which plays a part in the neurogenesis of the dopaminergic neurons. Studies conducted on the ageing brain show that the expression of Wnt is affected and that the Wnt ligands are downregulated. 7 Also, in the aging rodent brain, the elements of the Wnt pathway are also downregulated, namely, the Wnt2 and Wnt 4. Along with this, there is a reduction in the signaling in the hippocampus where the downregulation of nuclear beta-catenin is also present.
Environmental Factors
Environmental factors have turned out to be a rising concern of the cause of the disease. The environmental risk factors mainly include elements like heavy metal toxicity, pesticide exposure, and agricultural work hazards.
Polluted Air
The inhalation of air pollution toxins, such as vehicle exhaust gases and ozone, will lead to an increase in the expression of cytokines and oxidative stress in neuronal cells. Also, it can lead to the activation of microglia, which can further contribute to neuroinflammation, all of which results in PD. From the studies conducted on the population exposed to highly polluted air containing ozone and particulate matter, an elevation in the α-synuclein levels, which is a significant marker in PD, was confirmed. Along with this, a few proportions of the population showed the deposition of black particulate matter in the olfactory bulb and the olfactory nerve in the glomerular area, and this can be studied further to show the link between environmental pollution and olfactory damage and PD. 8
Related to occupational hazards, it was found that people involved in farming were found to have a higher incidence of the disease, which is majorly because of the exposure to pesticides and herbicides. Organic solvents, such as toluene, xylene, and isopropanol, and chlorinated solvents are used extensively in many industrial sectors, such as paint industries and dry cleaners, and the exposure to which can lead to many neurodegenerative diseases because of long-term exposure. The same applies for the people who are exposed to welding fumes and the inhalation of welding fumes. Even though there are contradicting results from the studies conducted on the incidence of PD from workplace exposure to welding flames, organic, and chlorinated solvents. 9
Chemicals
The influence of pesticides, specific neurotoxins, and heavy metals play a key role as environmental factors in the etiogenesis of PD. For example, the identification of PD in a younger age group of individuals was first identified in 1983. This was found to be because of the abuse of synthetic heroin, which was contaminated by the chemical MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine), and caused damage in the substantia nigra pars compacta. 10
The common pesticides rotenone and paraquat have been found to induce PD in individuals via long-term environmental exposure. These two pesticides cause a loss of dopaminergic neurons in the nigral region. Paraquat leads to the generation of a series of reactive oxidative species, leading to oxidative stress, which results in increased calcium ion concentration in the cells, causes a drop in the cellular glutamate levels through its efflux, and finally damage of the dopaminergic neurons. 11
The exposure to heavy metals because of pollution or because of workplace hazards can lead to permanent damage to the olfactory system, which is viewed along with PD caused because of environmental factors. A large number of metals, like manganese, copper, and mercury, are associated with the etiopathogenesis of PD. Copper can lead to the misfolding of the PD protein α-synuclein through the acceleration of the aggregation of the PD proteins into fibrillar plaques, which eventually leads to the formation of Lewy bodies. Hence, copper can lead to preliminary events in the pathogenesis of PD. Environmental factors, such as pollution, smoke, and infection, can lead to damage of the olfactory system. 12
Other Factors
Other than the genetic factors and the chemicals, certain diseases or trauma can lead to the progression of PD. Diabetes mellitus (DM) is a risk factor for many neurodegenerative diseases. DM has been found to have a negative impact on the cognitive abilities of the PD patients, thereby exaggerating the disease condition. DM contributes to the cognitive loss by exaggerating the neuronal degeneration. 13
In certain cases, a traumatic injury to the brain and concussions can lead to the initiation and progression of PD. In a study conducted on war veterans suffering from PD who also had traumatic injury in the brain, it was found that the people with a traumatic injury to the head had up to a 24% chance of getting diagnosed with PD. The various factors involved in the etiogenesis of PD and its correlation with olfactory dysfunction are shown in Figure 2.
Correlation of Olfaction with Cognitive and Motor Functions
In the case of people with PD, it was found that there is a marked damage in odor discrimination, identification, and threshold when compared with that of the people without PD. The same population of people with PD was found to show a significantly less score in the Montreal Cognitive Assessment (MoCA) for the cognitive abilities. The olfactory shortfall presented as the early indication of PD does not really cease to progress with the onset of the motor symptoms of the disease, whereas it will go on progressing at least till the motor stages in the earlier stage. 14
When it comes to Alzheimer’s disease (AD), the patients normally suffer from attention and olfactory deficit issues. It is hypothesized to be because of the cholinergic shortcomings in the basal forebrain. The study involving the cognitive assessment using a TMT-A (trail marking test) test, conducted to find the link between the olfactory damage and cognitive deficits, suggests that there is a decline in olfactory ability in the people who were reported with cognitive problems. Also, the condition was found to be minimized in the presence of cholinesterase inhibitors. 15 People who showed olfactory loss were found to have a higher chance of the modification of disease state to show dementia symptoms. The olfactory impairment occurring in the early stages can hence be linked to the involvement of the cerebral cortex and, therefore, is applicable as a biomarker for assessment of patients with PD on the risk of the developing cognitive deficits and conditions like dementia. 16 The cognitive areas, namely, the functioning of execution, attention, and visuoperceptual skills, were found to be more modest compared to the other cognitive functions, which got affected by the deficit in olfactory perception.
One of the reasons the motor deficits in PD occur is because of the cholinergic deficits, which can lead to gait disturbances. The same neurotransmitter deficit is a cause of olfactory dysfunction. The olfactory ability is correlated with the motor functions, such as balance, dexterity, and mobility. The olfactory scores were found to be relatively less in people who experienced motor problems. The olfactory system damage in PD is found to correlate with the motor and nonmotor problems, which continues over time along with the disease. 14 Hence, the olfactory function can be observed as a method for the early identification of age-linked neural damage that leads to movement disorders.
Olfactory Dysfunctions in PD: Clinical and Preclinical Evidence
Olfactory damage has been reported in the majority of the confirmed cases of PD and appears before the onset of the motor deficit characteristic of the disease. Hence, this character can be utilized as a possible marker for the disease such that it can be detected earlier, even before the onset of the chronic motor problems. Around 94% of the people with PD have been found to have some type of olfactory dysfunction. Of those, around 40% were found to show loss of smell and 54% were having hyposmia. The people were found to have problems with odor identification when tested with the University of Pennsylvania smell identification test (UPSIT) evaluation of olfaction. The olfactory deficit condition was found to get more severe with the progression of the disease. 14 Earlier, it was considered that the atrophy in the hippocampus, namely, in the parahippocampal gyrus, was associated with the case of olfactory associated with PD. The parahippocampal gyrus is a prominent part of the cortical area and receives input from the olfactory, auditory, gustatory, and somatic cortices. Later studies revealed that the people with PD first show changes in the structure of the parahippocampal gyrus during the initial stages, which later show changes in the orbitofrontal cortex in the final stage of olfactory damage. 17
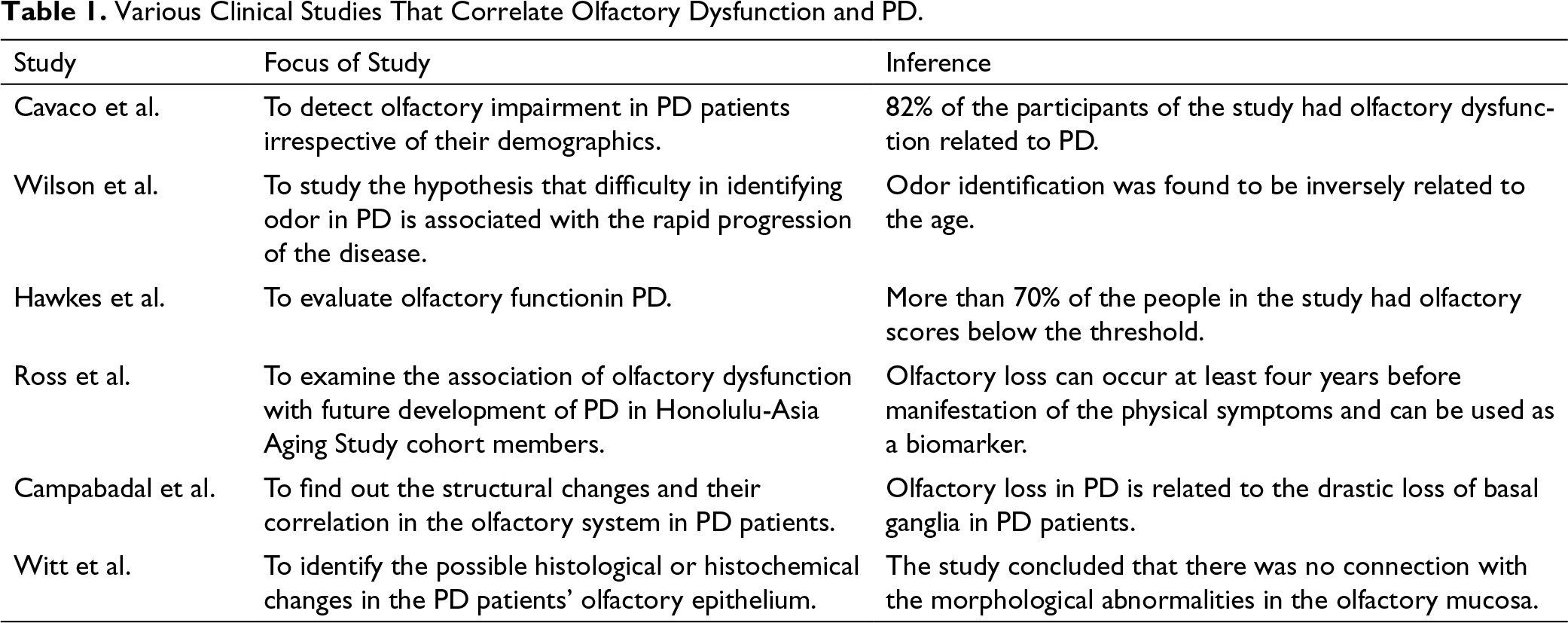
Along with the UPSIT test, patients diagnosed with PD were subjected to the forced odor detection threshold test, which assessed the basal identification sensitivity to the chemical PEA, phenylethyl alcohol. The test is a staircase test, which revealed that the patients diagnosed with PD showed lower sensitivity and higher threshold values of detection. 18 The various clinical studies that correlate olfactory dysfunction and PD are explained in Table 1.
Various Clinical Studies That Correlate Olfactory Dysfunction and PD.
The preclinical data obtained from the mice induced with PD using the bilateral injection of α-synuclein fibrils to the olfactory bulb show evidence of the inability of the detection of odor stimuli and its discrimination. This further gives evidence for α-synuclein pathology causing olfactory dysfunction similar to the PD condition observed in human beings. 19 Moreover, a preclinical study regarding PD using the αSynA53T mice shows evidence of partial damage of olfactory perception, and the olfactory damage was observed before the animals showed any signs of motor loss. This further supports the evidence of the early onset of olfactory symptoms and the possibility of using the olfactory parameters as an early detection marker for PD. 20
In the study conducted with the rotenone model of PD in rats, it was found that the animals that underwent the rotenone treatment showed a lower score in the odor discrimination task compared to the sham control group. The mouse model using the intranasal application of MPTP further showed dysfunction in sensory and cognitive abilities similar to the early PD stages. Along with this, there was an observable reduction in the tyrosine hydroxylase levels in the substantia nigra, olfactory bulb, and striatum. 21 The study with the intracerebroventricular (i.c.v) administration of the toxin 6-OHDA was found to induce the disease, which showed motor symptoms identical to those of PD. It was found to cause problems in the olfactory discrimination and the lowering of olfactory sensation. 22 PD-associated pathological changes in the olfactory system are diagrammatically explained in Figure 3.
PD-Associated Pathological Changes in the Olfactory System.23–27
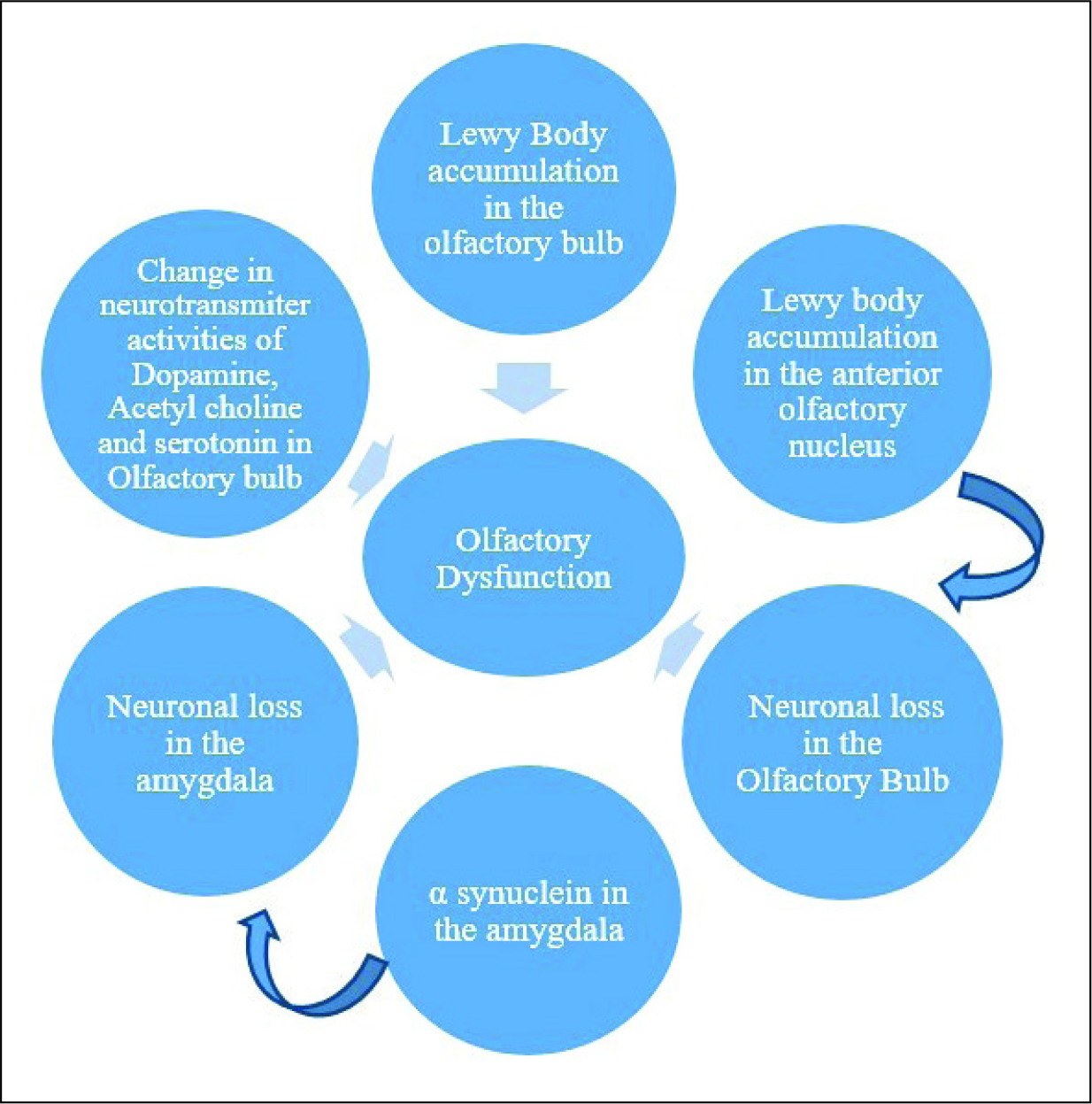
From the studies conducted on the PD based on the autopsy data, it was found that the disease progression has different stages, namely, Braak stages. In this case, olfactory damage is one of the markers considered in order to group the conditions into different stages; the appearance of lesions was used to stage the different phases of the disease, and the damage to the anterior olfactory nucleus was observed in the fourth stage of the six stages of the disease according to the Braak hypothesis in sporadic PD. 23
Molecular Mechanism of Olfactory Dysfunction in PD
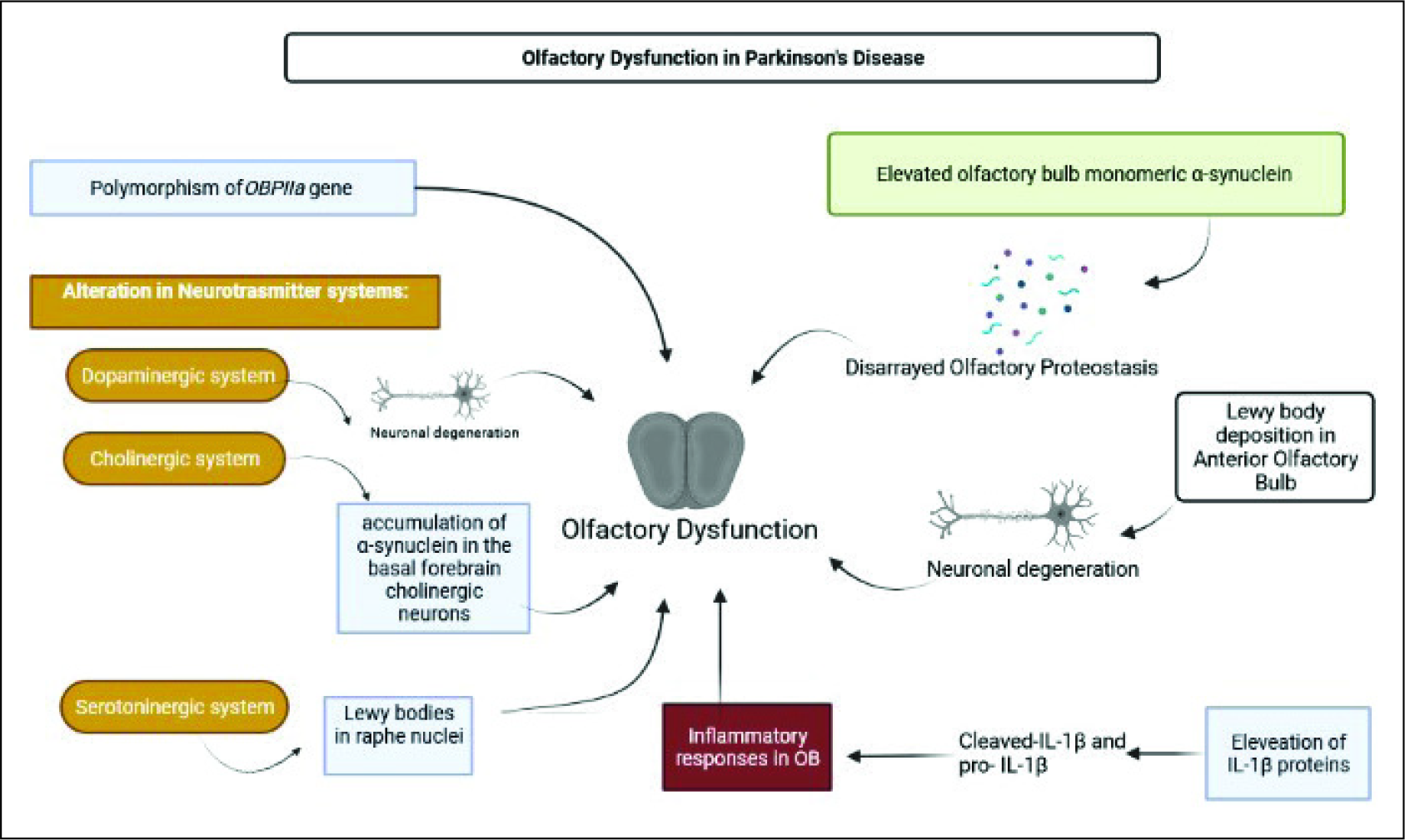
The α-synuclein pathology of olfactory dysfunction has been observed mainly within the central olfactory system, comprising the cortical as well as the anterior olfactory nucleus and the orbitofrontal cortex. But the Lewy body presence in the anterior olfactory nucleus is deemed to cause loss of neurons.28, 29 One of the reasons for olfactory dysfunction in PD is because of the misfolding of α-synuclein and its deposition occurring in the olfactory bulb and the enteric nervous system. 30 The disarrayed olfactory proteostasis affects cell survival. From the observation of proteostasis, a gradual elevation in the olfactory bulb monomeric α-synuclein was evident in different Lewy-type α-synucleinopathy (LTS) stages of proteostasis. The 139, 197, and 148 proteins in the olfactory bulb get expressed differentially in LBDE, LBDN, and the LBDL stages, respectively. The dysregulation of certain clusters of proteins was found to be related to cell death, movement disorder, and the dysfunction of basal ganglia. Also, the disarrangement in transcription in the olfactory bulb adds to the olfactory dysfunction, which is evident in the LBDE stage because of the alteration in various interactors of nucleophosmin, whereas in the LBDN phase, the functional interactors of survival routes get damaged, leading to a drastic drop in the survival potential of the neurons in the olfactory bulb. Moreover, around 268 proteins were found to be expressed differentially in the PD models studied compared to the normal groups. 31
The alteration in various neurotransmitter systems, such as dopaminergic, serotonergic, and cholinergic systems, has been associated with the olfactory loss in PD. 32 Dopaminergic neuronal loss is also a hallmark of PD, and the level of acetylcholine is also drastically varied in PD, which is important for the detection as well as discrimination of odor. Nevertheless, the accumulation of α-synuclein in the basal forebrain cholinergic neurons also contributes to the olfactory loss. 33 Lewy pathology is also found in the raphe nuclei, which keeps projections toward the olfactory bulb and shows the involvement of serotonergic system dysfunction leading to olfactory loss. 34 Even though there are no conclusive evidence, the studies conducted so far link the dysfunction of certain neurotransmitter systems to olfactory loss in PD.
Apart from the α-synuclein aggregation in the olfactory bulb, inflammatory proteins have also been found in the olfactory bulb. The levels of IL-1β proteins, viz. cleaved-IL-1β and pro-IL-1β, were found to be higher in MPTP-treated mice, along with evidence of elevated expression of procaspase. Also, the levels of TL4 proteins were found to be elevated, which led to inflammatory responses in the olfactory bulb. 35
The genetic polymorphism of the genes, which code for odorant-binding proteins (OBP), can be considered as another mechanism that leads to the alteration in olfactory functions in PD. The major genetic polymorphism rs2590498 (A/G) of the OBPIIa gene affects the perception of olfaction, and this particular mutation leads to the reduced expression of OBPIIa present in the olfactory epithelium.16, 36
Mechanism of Olfactory Dysfunction in PD.
Olfaction as a New Tool for Treatment in PD
As mentioned earlier, olfaction and its absence play a key role in PD in both clinical and preclinical phases. And currently, there are no disease-modifying treatments available for PD.
One of the major concerns regarding PD is the early detection before the onset of the motor complications. The PD pathology begins at the olfactory bulb, which can be attributed to the early olfactory loss; the disease then progresses in an upward fashion, which later causes motor dysfunctions in the late stages. Hence, the olfactory dysfunction can be used as a tool for early detection of the disease. Because olfactory dysfunction happens years before the onset of the motor symptoms, this method of detection can be used as a diagnostic tool for the early initiation of treatment. 37
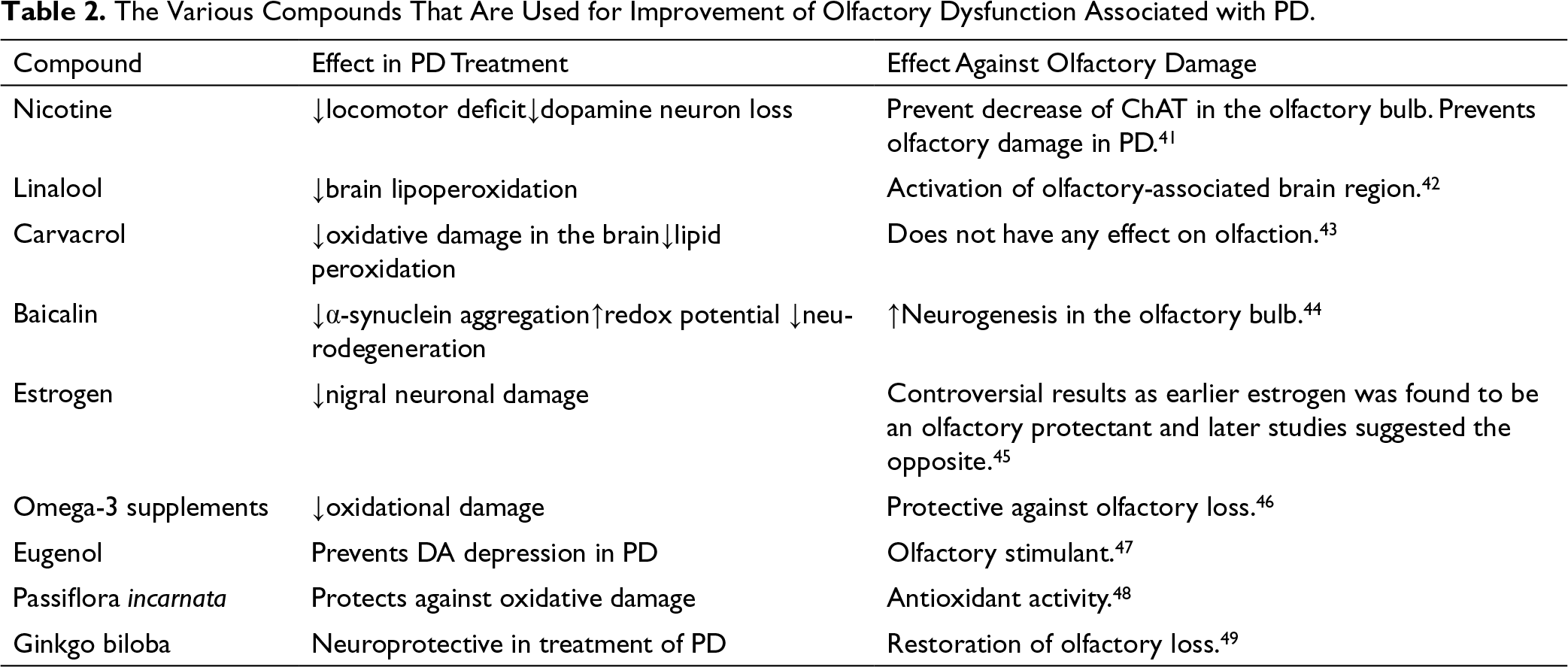
The use of olfactory dysfunction as a biomarker for early detection is confirmed by the identification of Lewy pathology in the olfactory bulb in the early stages of PD. The same Lewy pathology can be observed in the anterior olfactory nucleus along with the neuronal damage in the olfactory bulb, which leads to the early emergence of olfactory dysfunction. 38 Sensitivity to certain odors will be impaired in the case of PD, and this was further proved through cohort studies. Hence, a diagnostic protocol meant for the detection of complete or partial loss of smell comprised of specific odors which the PD patients will be less sensitive to can be employed for the early detection of the disease. 39 Moreover, olfactory damage is a much frequent early symptom compared to other nonmotor disturbances, which can be considered as an early symptom of the disease. 40 The various compounds that are used for the improvement of olfactory dysfunction associated with PD are listed in Table 2.
The Various Compounds That Are Used for Improvement of Olfactory Dysfunction Associated with PD.
Conclusion
From previous studies conducted in the past, it is evident that the role of the olfactory system in PD is important. Clinical and preclinical data regarding the link between olfaction and PD show a clear correlation as olfactory damage has been noticed in 90% of the cases. The pathological changes occurring in the substantial nigra of PD patients are also found in the olfactory bulb and the associated structures that affect olfaction. Therefore, olfactory damage being a relatively very early nonsymptom can be used as a diagnostic marker in the rapid and timely detection of the disease. Once the disease is identified at an early stage, it is easier to manage the disease to avoid or at least slow down the progress of the disease. This early nonmotor symptom, which is very evident, can be used for therapeutic benefits, which in turn can be useful in improving the quality of life of PD patients.
Hence, the olfactory damage and its impact on PD are topics to be studied as they will have an impact on disease management. Because PD is a progressive disorder, the absence of disease-modifying drugs will worsen the disease. So, currently, the strategies of exploring the disease have shifted their focus to more intrusive characteristics of the disease, which were initially considered minor. The olfactory problems associated with the disease are a good target for the development of new treatment strategies.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
