Abstract
Hypoglycemic drugs in the dipeptidyl peptidase-4 (DPP-4) inhibitor class are used as a second-line treatment for type 2 diabetes mellitus. With DPP-4 inhibitors, there have been a few reports of cutaneous side effects such as bullous response, fixed drug eruption, and photosensitivity. There is no definitive pathophysiology for the above mentioned allergic reactions. Sitagliptin phosphate belongs to the DPP-4 inhibitor class. This is a case report of a sitagliptin-induced bullous drug reaction manifesting three weeks after starting therapy. He had bullous pemphigoid-like eruptions all over the body. The patient showed improvement once sitagliptin was discontinued alon with oral and topical steroid treatment.
Introduction
Bullous pemphigoid (BP) is an autoimmune blistering skin disease mostly affecting elderly people and is related to a high rate of morbidity and mortality. It is associated with autoantibodies produced against BP180 and BP230 antigens, hemidesmosomal components of basal keratinocytes. Various factors such as trauma, burns, radiation therapy, and medication can trigger BP. 1 The most commonly involved drugs are spironolactone, furosemide, captopril, and phenacetin, and nonsteroidal anti-inflammatory drugs such as ibuprofen, d-penicillamine, etanercept, and systemic antibiotics. According to recent reports, it is more common in diabetic patients who are taking DPP-4 inhibitors. A significant level of suspicion and careful analysis are often vital in evaluating drug-induced BP, as it is difficult to differentiate it from a classical autoimmune BP.
Clinically, it may have a subacute or acute onset. The typical skin lesion is a large tense subepidermal blister that appears on erythematous or normal skin. It may be preceded by itchy urticarial or eczema-like lesions. Although they can occur anywhere, these lesions are most common in the lower abdomen, inner or anterior thighs, and flexor forearms. The bullae are filled with clear fluid or hemorrhagic fluid and are associated with significant pruritus. Involvement of the oral and ocular mucosa rarely occurs. The bullae typically heal with post-inflammatory pigment. Histopathology and immunofluorescence studies are mainly used for the diagnosis of BP. Localized BP is frequently treated with topical steroids alone. For extensive disease, systemic anti-inflammatory drugs like corticosteroids and concomitant immunosuppressants such as azathioprine, cyclophosphamide, methotrexate, cyclosporine, and tetracycline/minocycline with nicotinamide or mycophenolate mofetil can be used. For resistant cases, biologics (anti-TNF drugs, rituximab), IVIG, or plasma exchange are rarely used. 2
Case Report
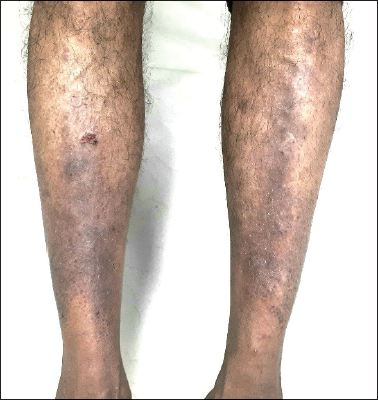
A 54-year-old male presented to our dermatology OPD with bullous lesions all over the body. He was diagnosed with diabetes type 2 recently and had no other comorbidities. His blood sugar levels were poorly controlled on conventional antidiabetics. So, he was started on oral sitagliptin therapy. After three weeks of sitagliptin therapy, he developed generalized itching with fluid-filled lesions over the shin, forearms, and trunk. On examination, there were bullous lesions and erythematous hyperpigmented, excoriated cutaneous eruption confined to the extensor aspect of the legs and forearms (Figure 1). The bullae were tense and Nikolsky’s sign was negative. The lesions had involved the whole body gradually. Oral and genital mucosae showed mild erosion. Hair and nail examination were normal. A routine blood test and a skin biopsy were performed. The preliminary diagnosis remained as gliptin-induced bullous pemphigoid. Blood tests showed an increase in white blood cell counts with increased eosinophils and increased HbA1c. The rest of the reports were normal. Skin biopsy showed subepidermal bullae with numerous neutrophils and eosinophils. Direct immunofluorescence for IgGs or complements (C3) was negative. Indirect immunofluorescence revealed linear deposition of serum IgG1. The patient was advised topical clobetasol propionate and oral doxycycline 100 mg once daily and an oral antihistamine. Oral prednisolone was started with 30 mg dosage, which was tapered gradually and stopped at four weeks when the lesions resolved completely. The patient was given an alternate oral hypoglycemic agent. After stopping sitagliptin, there was a marked improvement in the skin lesions. There was no recurrence of symptoms during the follow-up period for three months.
Sitagliptin Induced Bullous Drug Reaction.
Discussion
Sitagliptin phosphate belongs to the DPP-4 inhibitor class for hypoglycemic drugs and was approved by the US Food and Drug Administration in 2006. It is used as a second-line drug for the management of type 2 diabetes mellitus. There are a few reports of cutaneous adverse effects such as bullous reaction, fixed drug eruption, and photosensitivity with sitagliptin. 3 Definite pathogenesis of allergic reactions in the patients taking sitagliptin is unknown. DPP-4 inhibitors are incretin-based drugs used in diabetes mellitus type 2, which act by inhibiting the degradation of incretins and cause increased insulin secretion with alteration of blood glucose levels. The drugs approved by FDA under this category include sitagliptin, vildagliptin, linagliptin, saxagliptin, and alogliptin. DPP-4 (also known as CD26) is a cell-surface glycoprotein with enzymatic activity present in various cell types all over the body like keratinocytes. A DPP-4 inhibitor results in the release of many pro-inflammatory cytokines, leading to cutaneous eosinophil activation and blister formation. 4 DPP-4 inhibitor is associated with eosinophil activation by exotoxin-mediated mechanism and increased TGF beta-1 production through Th3 regulatory cells which are responsible for bullous pemphigoid-like reaction. DPP-4 plays a role in plasmin formation, which cleaves the major BP autoantigen BP180. Thus, DPP-4 inhibitor causes inappropriate cleavage and immunotolerance. Lymphocyte activation leads to upregulation of DPP-4 enzyme, and DPP-4 inhibitors could modulate the immune function. 5
The time period between the exposure of gliptin and the development of BP was variable from 8 days to 36 months. 3 In patients with gliptin-induced BP, most of these autoantibodies have been reported to react to LAD-1 and/or the terminal carboxyl domain of BP180 protein. The analysis of studies based on pharmacovigilance databases suggests an association between DPP-4 inhibitors and bullous pemphigoid, with the majority of affected patients being over 70 years of age. The association would be stronger in the case of vildagliptin, followed by linagliptin and sitagliptin. 6
We must be able to differentiate the autoimmune bullous pemphigoid from the drug-induced BP, as mostly they have similar clinical and histopathological features. Therefore, when treating patients, we must keep an adequate medical history, considering all medications, if the patient is receiving polypharmacy. 7 This will avoid losing a drug-induced condition.
Patients with DPP-4 inhibitor-induced bullous reaction may present with inflammatory or a non-inflammatory phenotype of bullous pemphigoid. Clinical improvement after withdrawal of the suspected drug suggests the diagnosis of drug-induced BP in our patient. Discontinuation of DPP-4 inhibitor treatment in patients with diabetes should be done when bullous reactions occur.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
