Abstract
Objective:
To develop a physiologically based pharmacokinetic (PBPK) model for individualization of the dosing regimen considering the physiological requirements of these preterm neonates.
Methods:
The study comprised preterm newborns with fewer than 34 weeks of gestation and six apneic episodes in 24 h. A PBPK model was created using PK-SIM (version 9, update 1, GitHub, San Francisco, CA, USA). A PBPK model is built using a typical loading dosage of 5 mg/kg and a maintenance dose of 1.5 mg/kg. Based on the verified base model, a PBPK model representing renal underdevelopment based on nRIFLE/pRIFLE categorization was developed.
Results:
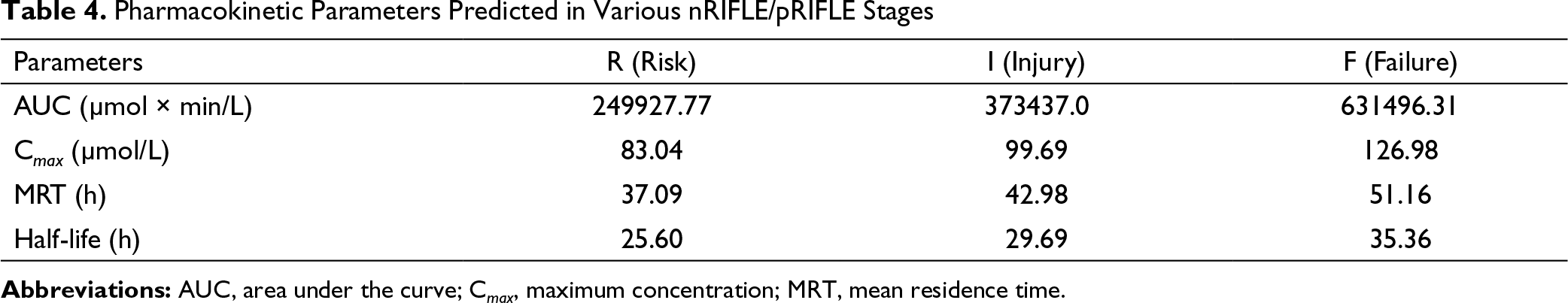
The PK parameters of Aminophylline were computed using the PBPK model. As per the model prediction, T1/2 and area under the curve reduced as postnatal age increased, and in the event of renal underdevelopment, even while C max for patients under R (RISK), I (injury) was within the therapeutic range; it was greater compared to preterm without any renal complications. Mean C max (mol/L) was 59.53 and for R, I, and F (FAILURE) categories the values were 83.04, 99.69, and 126.98, respectively.
Conclusion:
The model was created using appropriate drug, study subject, and dosage protocol inputs. The established PBPK model could help in individualizing aminophylline dose in preterm babies.
Keywords
Introduction
Premature babies struggle with temperature regulation, oral feeding, and breathing control in the first few days of their life. For many premature babies, the resolution of apnea and the establishment of a regular respiratory pattern is a significant developmental milestone. 1 In infants born less than 37 weeks of gestation, apnea of prematurity (AOP) is described as a pause in breathing lasting more than 15 to 20 s, accompanied by oxygen desaturation or bradycardia. 2 The incidence of recurring apnea has been reported with declining gestational age. 3 All infants delivered at less than or equivalent to 28 weeks of gestation presented with apnea. Beyond the 28-week mark, the number of infants with apnea kept reducing, 85% of infants had apnea at 30 weeks of gestation, which declined to 20% at 34 weeks of gestation. 4
Although the pathogenesis of AOP is unknown, the severity of the disorder is exacerbated by immature respiratory reflexes and breathing responses to hypoxia and hypercapnia. Apnea can last for more than 20 s in severe cases and is synonymous with bradycardia or desaturation which can disrupt cerebral hemodynamics and affect neurodevelopmental outcomes. 1
AOP treatment is primarily determined not only by the etiology and nature of the occurrence but also by the sequence of events which led to it. Understanding the progression of events that lead to apnea is crucial for developing treatment regimens for the condition.The treatment of an underlying cause, such as infection, patent ductus arteriosus, epilepsy, maternal or neonatal drug intoxications, or gastroesophageal reflux, can resolve episodes of apnea with an underlying cause (generally referred to as secondary apnea). 5 Generally, treatment options are either pharmacologic or respiratory. The major treatment options include methylxanthine therapy (theophylline or caffeine), doxapram, ampakines, gastric acid suppression, oxygen supplementation, and other respiratory interventions (continuous positive airway pressure, nasal cannula, and synchronized noninvasive positive pressure ventilation). 6
Aminophylline, belonging to methylxanthine group, comprises of theophylline and ethylenediamine in a ratio of 2:1. It works in lungs by inhibiting the isoenzymes. It works in lungs by inhibiting the isoenzymes phosphodiesterase (PDE) III and PDE IV, causing smooth muscle relaxation preceded by bronchodilation, and it increases the force of contraction of diaphragmatic muscles by enhancing calcium absorption through adenosine-mediated channels. 7 A 2012 study on prophylactic use of aminophylline in the treatment of high-risk neonatal group apnea found that 7.7% of preterm neonates in the aminophylline group developed apnea, compared to 61.5% in the control group. The benefits of aminophylline in the treatment of apnea in chronically premature babies are shown in the said study. In other words, the younger the baby, the more effective aminophylline is against bradycardia and apnea. 8 The therapeutic index of aminophylline is quite narrow (10–20 µg/mL). As serum concentrations reach 20 µg/mL, theophylline toxicity is more likely to occur and it becomes more extreme as serum concentrations rise further. 7
The fact that preterm newborns’ kidneys are underdeveloped is another key aspect that contributes to treatment problems. In pediatric patients, aminophylline is 50% removed by the renal route, but in adults, most of the elimination happens via the metabolic pathway, particularly CYP1A2. According to published data, 8 to 24% of preterm infants admitted to the NICU experience acute renal failure, which is difficult to identify in preterm 9 and necessitates altering and individualizing the dosage regimen in these patients.
Dose modifications are not possible until the pharmacokinetics (PK) of the drug is understood and physiologically based pharmacokinetic (PBPK) model can give a mechanistic understanding. It is a statistical simulation technique to predict the absorption, distribution, metabolism, and excretion of synthetic or natural chemical substances in human and other animal species. The building blocks for the model are organism parameters, drug properties, formulation and administration procedure, and the predictions obtained in the model depend on the inputs provided. 10 It simulates the concentration-time profiles of the drug and its metabolites in the plasma or any organ of interest. Simulated concentration-time profiles in different study populations, including vulnerable subjects, can assist in the selection of optimal sampling times or dosing strategies. 11 The study aimed to estimate the PK parameters of aminophylline in preterm babies using PBPK model and assess the impact of covariate, postnatal age. This study also focuses on the PK of aminophylline in case of renal underdevelopment in premature babies which will aid individualization of aminophylline dosing.
Materials and Methods
A whole-body PBPK (WB-PBPK) model was developed based on anonymized plasma concentration data of aminophylline obtained from 108 preterm neonates with apnea from a previous study conducted at Kasturba hospital, Manipal. This study was approved by Institutional Ethics Committee (IEC 131/2014), Kasturba Hospital, Manipal. 12 Preterm neonates with less than or equal to 34 weeks of gestational age and greater than six apneic episodes in 24 h were included in the study. 12 The mean gestational age of the population was 30 weeks (26 to 34 weeks) and mean postnatal age (PNA) of the population was 6.85 days (2 to 23 days).
PBPK models were built using the building blocks from PK-Sim (Version 9 Update 1). 13 A mean individual representing the unabridged population (whole population) was created using the individual blocks. This mean individual was used to generate a set of 100 and 50 virtual individuals for building a population representing the unabridged population and subgroup populations. Subgroups were based on the body weights and PNA. The population was built considering the range of demographic data represented in Table 1.
Patient Demographics.
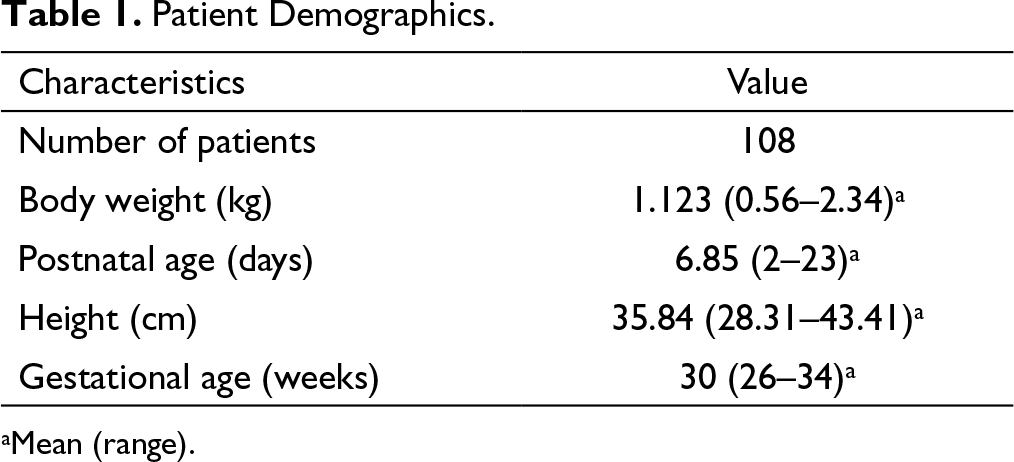
aMean (range).
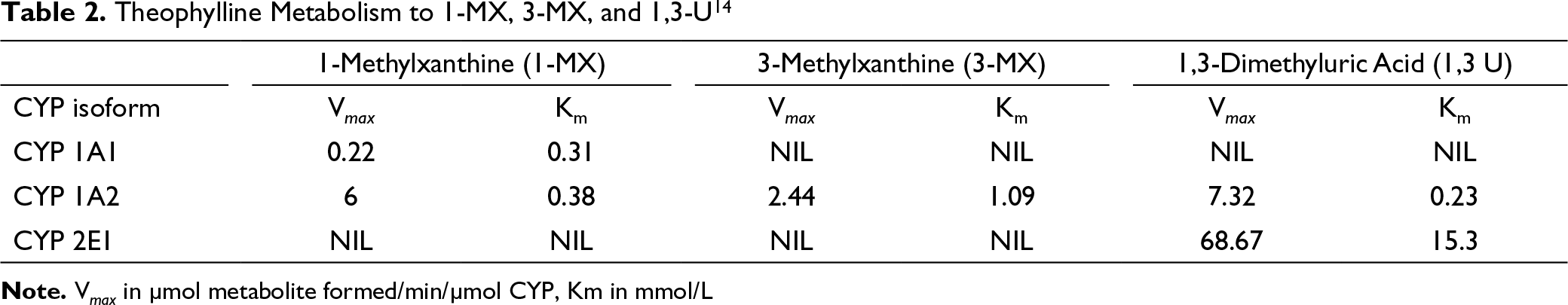
Drug Metabolism and Elimination
Theophylline Metabolism to 1-MX, 3-MX, and 1,3-U 14
Formulation and Drug Characteristics
Aminophylline oral tablets are readily and fully absorbed in adults with a peak serum theophylline concentration of 10 µg/mL (range: 5 to 15 µg/mL), reaching a peak concentration in 1 to 2 h after a single dose of 5 mg/kg. 14 Theophylline, protein-bound by 40% mainly to albumin. 16 Volume of distribution (Vd) ranges from 0.3 L/kg to 0.7 L/kg. 17 As plasma protein binding declines, Vd rises in preterms. Compound parameters used in constructing the compound building blocks were LogP (–0.02), fraction unbound (0.6 Log Units), molecular weight (180.1 g/mol), solubility at pH 7 (7360 mg/L). In adults, liver metabolism is a saturable mechanism that occurs mainly by N-demethylation mediated by CYP1A2. Metabolic clearance is substantially higher compared to renal clearance in adults, while in neonates, because of immaturity of metabolizing enzymes (CYP1A2, CYP2E1, and CYP1A1), 50% of the drug is cleared unchanged by the renal elimination. 14 In premature neonates, 50% of theophylline is eliminated unchanged in the urine.16, 18
Dose and Dosing Protocol for Simulation
A standard protocol of 5 mg/kg loading dose of theophylline infused over 30 min and maintenance dose of 2 mg/kg infused over 10 min was used to build the model. In order to provide an accurate measure of serum theophylline levels, dose changes from aminophylline to theophylline were made using a salt factor of 0.8.16, 18 Further in the validation process, visual comparison was performed using data from patients with the same dosage regimen (loading dose of 5 mg/kg and a maintenance dose of 1.5 mg/kg), and the percentage prediction error was determined.
Percentage Error (PE) = ((Ypred–Yobs)/Yobs) × 100
Ypred = predicted value obtained after simulation; Yobs = observed value obtained from a clinical study. Later, subgroups of populations were built around the mean individual. Fifty virtual individuals in each subgroup were generated within the range of demographic data for PNA and body weights. The simulations were carried out with a dosing interval of 8 h for 424 h.
PBPK Modelling in Premature Babies With Renal Underdevelopment
The features of early renal function in preterm neonates with birth asphyxia (BA) of various gestational ages (GAs) are unknown. Renal insufficiency will occur within 24 h of birth asphyxia because kidneys are susceptible to oxygen deprivation. 19 Renal underdevelopment models are built by scaling down the glomerular filtration rate (GFR) values from the base model based on the acute kidney injury (AKI) classification system. 20 An AKI classification scheme called “Risk, Injury, Failure, Loss, End-Stage Kidney Disease (RIFLE)” was proposed by the Acute Dialysis Quality Initiative (ADQI) to encourage a clear and consensus AKI definition, which was updated to be used in children. 20
Results
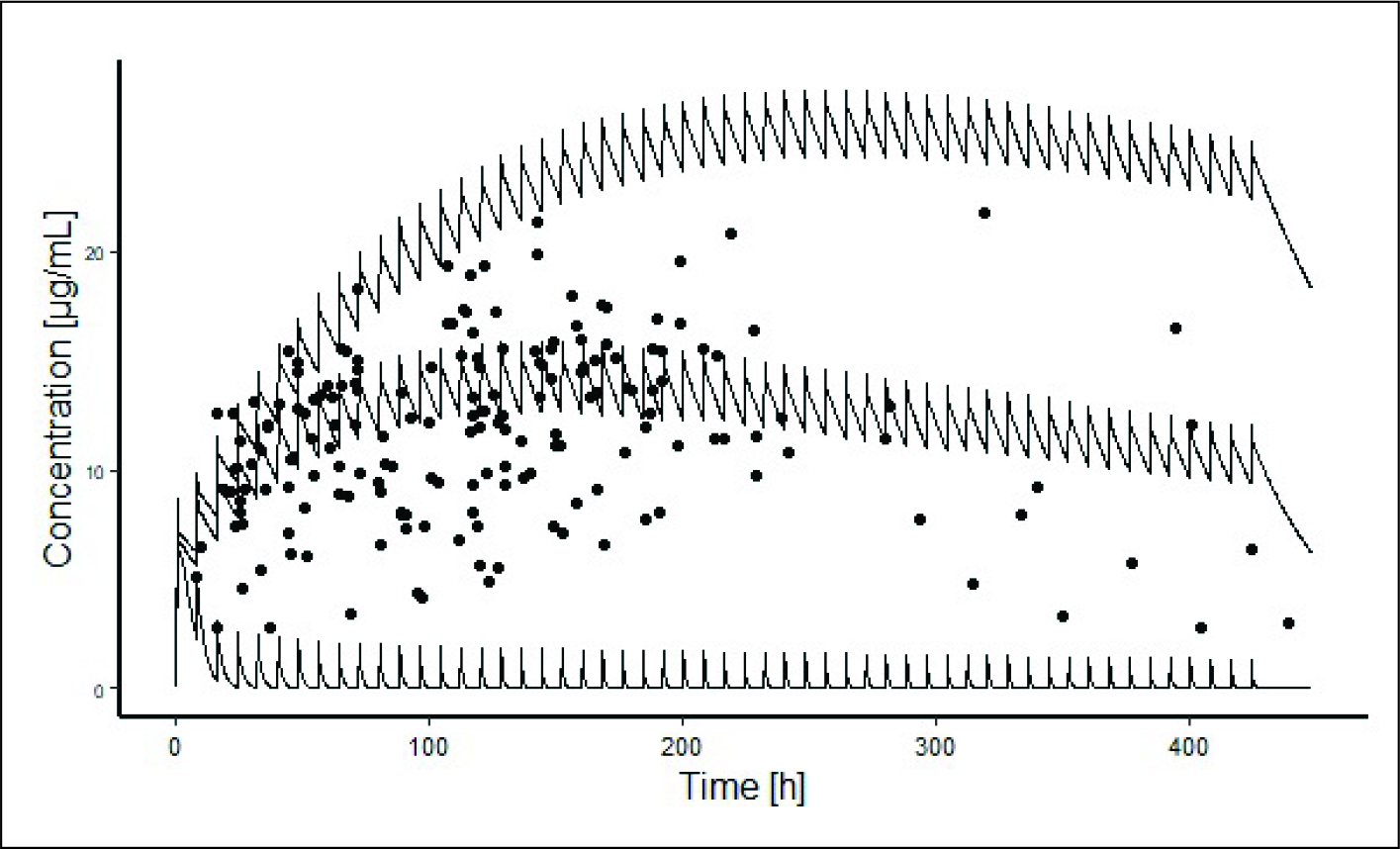
A WB-PBPK model was developed that characterizes both the conventional PK profile and variability of the available data on aminophylline plasma concentrations as depicted in Figure 1. The predicted time-concentration profile showed an increase in clearance with time which could be attributed to the maturation of liver enzymes with age. The predicted half-life of aminophylline, 22.70 h, is in line with previous studies at 24.22 h 21 and C max was predicted to be 59.53 µmol/L. The predicted PK parameters are depicted in Table 3.
Visual Comparison of Predicted Aminophylline Plasma Concentrations With Patient Data of Previous Report. The Middle Solid Line Represents the Median Value of Prediction, and the Upper and Lower Solid Lines Represent Geometric Standard Deviation. Solid Circles Represent Plasma Aminophylline Concentration From the Previous Study Used for Comparison.
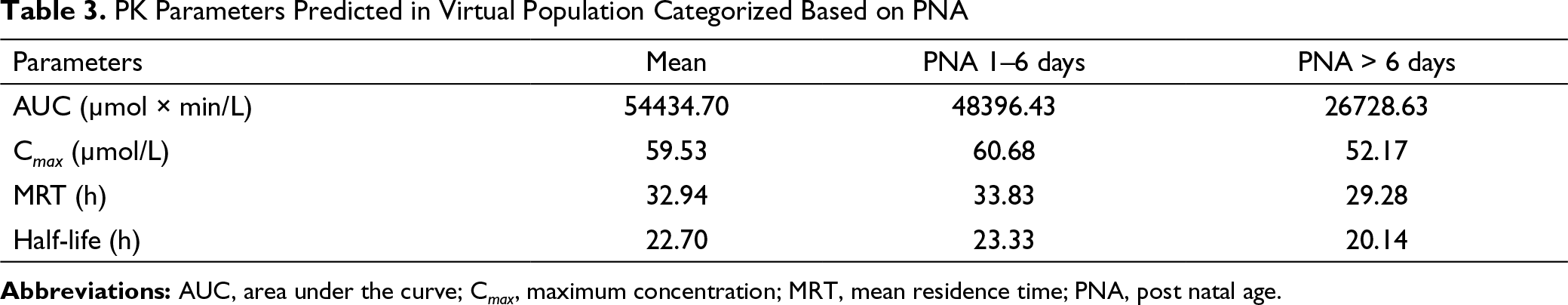
PK Parameters Predicted in Virtual Population Categorized Based on PNA
Impact of Postnatal Age and Body Weight on the Predicted Model
PNA based subgroup simulations provided evidence for the maturation of enzymes with the progression of time; this can be visualized with the decrease in half-life and area under the curve (AUC). In populations with PNA from one to six days and greater than six days, the half-life was roughly 23.33 h and 20.14 h, respectively. AUC fell by almost half in the population with PNA greater than six days, i.e., from 48396.43 µmol × min/L in the population with PNA one to six days to 26728.63 µmol × min/L in the population with PNA greater than six days. With an increase in the PNA, the C max decreases as well. The C max was predicted to be 60.68 µmol/L in the population with PNA one to six days, and 52.17 µmol/L in the population with PNA greater than six days.
Because of the fluctuation in body weight in the early weeks of preterm, models based on PNA would accurately predict the PK parameters in this cohort compared to models based on body weight, as well as it would provide us an indication of the amount of enzyme maturity as a function of age. Because of the fluctuating body weight, the model with bodyweight could not explain the concentrations of observed data and the model was unstable.
Pharmacokinetic Parameters Predicted in Various nRIFLE/pRIFLE Stages
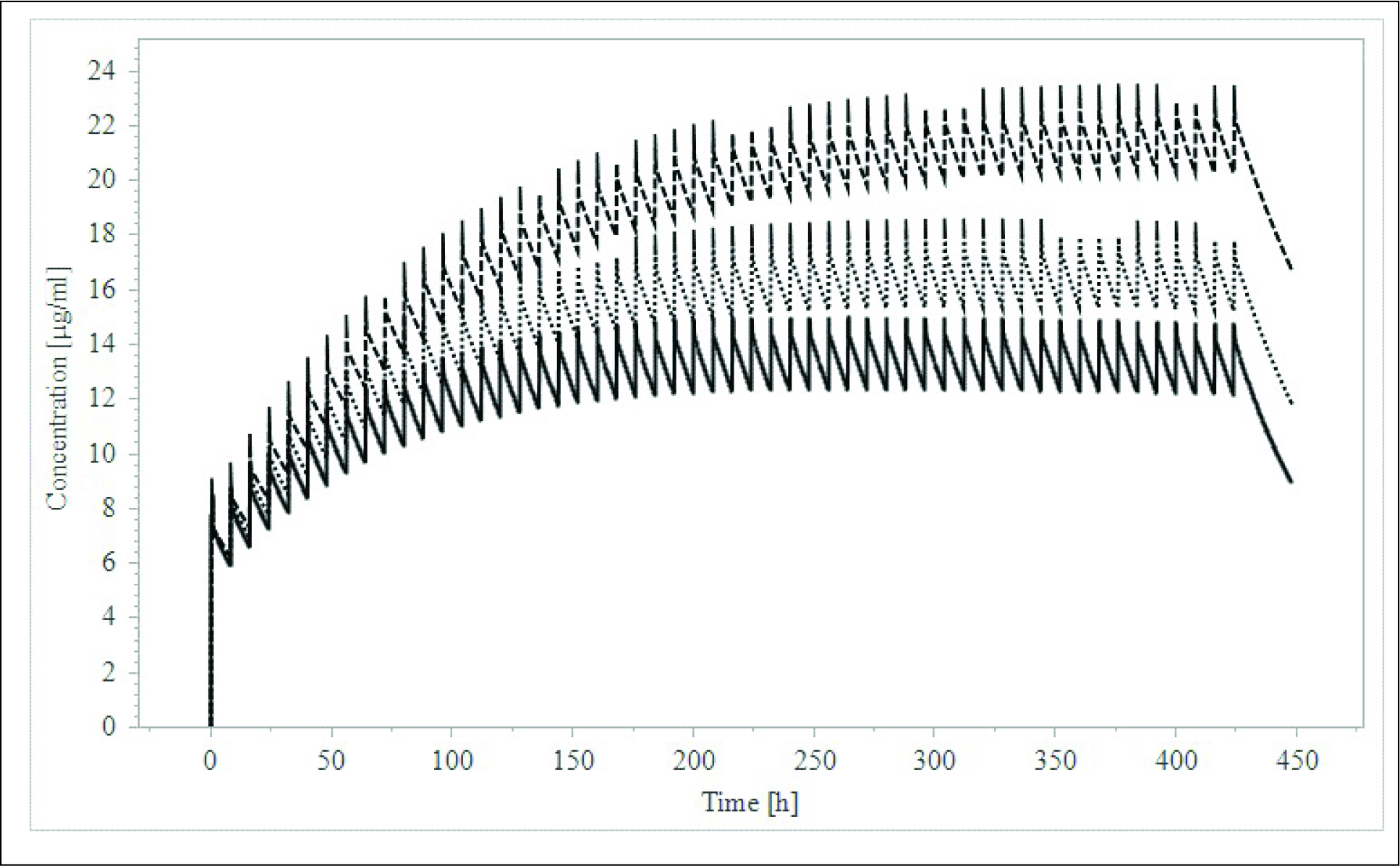
Plasma Concentrations of Aminophylline With Renal Failure at Various Stages of nRIFLE/pRIFLE Category. The Lower Solid Line Represents the Predicted Aminophylline Concentration of the Group With Risk Category of Renal Function as Per RIFLE Category. Dotted Line Represents the Injury Category and Dashed Line on the Top Represents the Aminophylline Concentration of the Failure Category. One Can Clearly Observe Increasing Aminophylline Concentrations With Increasing Severity of Renal Dysfunction.
Discussion
Preterm birth leads to various complications, one of which is AOP. Methylxanthine therapy is used to manage this condition and concentrations within therapeutic range are necessary to attain therapeutic benefits. The benefits of methylxanthine treatment for premature apnea outweigh potential short-term risks. 22 In a controlled study on the prophylactic use of aminophylline in the prevention of AOP in high-risk neonatal group, apnea symptoms were seen in 7.7% cases in the aminophylline group and in 61.5% cases in the control group (P < 0.001). This study also showed that younger the infant, greater the protective impact of aminophylline on bradycardia and apnea. 8
In adults, about 53 to 65% of theophylline is bound reversibly to circulating plasma proteins; however, because this plasma protein is missing in neonates, free drug availability in the blood increases, resulting in enhanced bioavailability, toxicity, and side effects. 23 Because of immaturity in the development of the physiological system in preterm, the prescribed medicines may induce toxicity or end up with subtherapeutic amounts. Because of the narrow therapeutic range of aminophylline/theophylline, standard doses may lead to unintended consequences. 2
In the current study, a standard dosing protocol of 5 mg/kg loading dose and 2 mg/kg maintenance dose with a dosing interval of 8 h was compared in preterm neonates with different PNA and its effect with the progression of time, that is, for about 424 h. Serum concentration ranging from 15 to 20 µg/mL is considered effective and anything greater than 20 µg/mL leads to toxicity. The predicted mean concentration for the unabridged population was found to be 15.27 µg/mL. Preterm neonates have lower levels of hepatic enzymes and renal function, which leads to reduced clearance and higher bioavailability of the compound after release. The maturation of CYP1A2 begins from day eight which reaches the adult levels by the age of 12 to 18 months. 24 This explains the observation of higher clearance in subgroup with PNA more than six days compared to group with PNA one to six days. The key finding of the present developed model was that PNA has a notable influence on clearance as reported elsewhere. 25
Another important challenge with preterm neonatal dosing is renal underdevelopment, acute kidney insufficiency and, lower clearance of. Bulk of the aminophylline is removed by the renal route in neonates due to the immaturity of the metabolizing enzymes. Preterm newborns account for approximately 31% of all documented instances of acute renal failure in the NICU, with a mean age for diagnosis of six days. 9 Hypoxia and ischemia may impair nearly every tissue and organ in the body, with the kidneys being implicated in 50% of instances, followed by other organs. 26 In this situation, dosing should be conservative in case the neonate has renal underdevelopment, as the simulations showed that drug concentration increases corresponding to the stages of renal failure. Predicted change in PK parameters with the developed WB-PBPK model can potentially guide dosage regimen in such cases of renal underdevelopment to attain appropriate therapeutic range. Aminophylline is a useful but possibly toxic drug that, if dosed inappropriately, can induce significant complications or even death. 27 Therefore, it is important to use appropriate approaches and tools to individualize the doses. The physiochemical data of drugs were obtained from different sources and any uncertainty of those could potentially impact the validity of a PBPK model.
Conclusion
In the present study, a WB-PBPK model was developed using PK-Sim platform for aminophylline in premature infants. The model was developed with appropriate inputs on drugs, study subjects, and dosing protocols. The developed model was validated using previous Aminophylline data from a similar cohort. The developed model was used to assess the impact of renal underdevelopment on aminophylline pharmacokinetics. The developed WB-PBPK model has the potential for dosage individualization of aminophylline in premature infants.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
