Abstract
Background
Caffeine citrate (CC)-induced excessive energy expenditure, diuresis, natriuresis, and other CC-associated potential side effects (CC-APSEs) result in lower daily weight gain (WG) in premature neonates. This study aimed to assess higher CC-doses’ effect on the mean daily-WG (MD-WG) and CC-APSE development, considering 5 mg/kg/day as the standard regimen.
Method
This retrospective cohort study included neonates of ≤36 weeks gestational age and received CC-therapy. The same participants were followed for data analysis in two postnatal phases: 15–28 and 29–42 days of life (DOL). Based on daily CC-dose, formed group-I=(5 mg/kg/day), group-II=(>5–7 mg/kg/day), and group-III=(>7 mg/kg/day). Data was analyzed separately for group-II and group-III using group-I as the standard.
Results
The study included 284 neonates. During phase-I, the MD-WG was significantly higher in group-I than group-II (19.9 ± .88 g/kg/d vs 17.5 ± .49, P = .031) and group-III (19.9 ± .88 g/kg/d vs 16.7 ± .71, P < .001). During 29–42 DOL, the MD-WG of group-I was only significantly higher than group-III (21.5 ± .42 g/kg/d vs 18.1 ± .39 g/kg/d, P = .003) and comparable with group-II. During 15–28 DOL, CC-APSEs were significantly higher in group-II and group-III but during 29–42 DOL was significant only in group-III.
Conclusion
Exposure to higher caffeine doses in this study cohort is associated with lower postnatal WG in preterm neonates than standard daily doses may be due to its catabolic effects and CC-APSEs.
Introduction
Caffeine citrate (CC) is now recognized as a standard for treating AOP due to a better therapeutic index, comparable enteral bioavailability, and longer half-life.1-3 Its use in preterm neonates is related to shorter mechanical ventilation (MV) dependence and lower chances of extubation failure (EF). 1 Neonatal studies reported a reduced risk of bronchopulmonary dysplasia and an improved rate of very low birth weight (LBW) infants’ survival with no neurological disabilities at 18–21 months. 4 Apnea of prematurity might persist beyond 37 weeks postmenstrual age (PMA) in preterm neonates born at GA <28 weeks. 5 Hence, CC is often prescribed for preterm neonates until they reach a PMA of 35–37 weeks in doses of 5–10 mg/kg in 24 hrs. 1 In this vulnerable population, the longer duration of CC-therapy and higher cumulative doses with potential catabolic response might affect the initial weight gain (WG). 6
Previous studies reported a strong association between fetal growth restriction and high-dose maternal caffeine intake. 7 It is suggested that this phenomenon was consistent across all trimesters.7,8 A study reported that high-dose maternal caffeine intake resulted in LBW in 7% of neonates and small-for-gestational-age in 10%. Though, weight or head circumference reduction was not reported at the 18–21-month follow-up of participants subjected to CC-therapy for AOP. However, caffeine duration and dose-specific somatic effects through the neonatal phase were not determined. 9 Caffeine acts as a CNS stimulant by readily crossing the blood–brain barrier. It is an adenosine-G protein-coupled receptor antagonist10,11 and competitively inhibits the cAMP-phosphodiesterase enzyme (enzyme which converts cyclic-AMP to its noncyclic-form), therefore letting cAMP build up at the cellular level. Cyclic-AMP activates protein kinase-A to initiate the phosphorylation of glucose synthesizing certain enzymes. Thus, caffeine extends and intensifies the effects of adrenaline and adrenaline-like medications by blocking its removal.10,11 Consequently, caffeine intake results in increased heart rate, oxygen consumption, and metabolic rate. 12 Energy expenditure is increased by methylxanthines independent of physical activities and therefore enhances carbohydrate consumption in exposed neonates. 13 Compromised splanchnic blood flow is reported in the caffeine-exposed neonatal gut, and CC intake is reported to result in diuresis and natriuresis. 14 A study reported a doubling of urine output with caffeine use in respiratory distress premature animal models. 15 The cumulative catabolic effect of caffeine exposure in preterm neonates through the abovementioned mechanisms of action contributes to negatively influencing WG.
Consequently, this retrospective cohort study aimed to evaluate the potential influence on mean daily-WG (MD-WG) of preterm neonates exposed to three different intravenous or oral dosage regimens of CC-therapy for managing AOP and obtaining other respiratory and neurological benefits in preterm neonates (5, >5–7, and >7–10 mg/kg/day). 16 In addition, the study aimed to evaluate the dose–response relationship with the onset and persistence of caffeine-associated potential side effects (CC-APSEs) and length of hospital stay.
Materials and Methods
Study Design, Setting, and Population
This study was performed in Aga Khan University Hospital (AKUH), a tertiary care setting in Karachi, Pakistan. The AKUH has the facility of 24-bed multispecialty tertiary care NICU. About 1200 neonates are admitted annually with the influx of very preterm high-risk newborns from all over the country. All the preterm neonates (GA ≤36 weeks) were included in the initial cohort who were admitted to NICU and were started prophylactic CC-therapy (intravenous or oral) for managing AOP and obtaining other respiratory and neurological benefits in preterm neonates during the study period (April 2017–December 2018). Neonates were identified from the hospital’s electronic database. All the neonates with fluid restrictions, congenital anomalies, culture-proven infections during CC-therapy, and ≥ grade-III intraventricular hemorrhage (IVH) were excluded. Neonates with NEC (modified Bell’s stage-2b and beyond) were also excluded due to their strong association with several risk factors other than prematurity and the use of caffeine. 17 Additional exclusion criteria were the concurrent prolonged exposure to other WG-influencing medications, such as diuretics, corticosteroids, and ibuprofen. Neonates who died before entering phase-II of the study were also excluded.
Sample Size
The sample size for comparing group-I and group-II: For the outcome of difference between the mean daily weight gain 18 of groups exposed to caffeine daily doses at Confidence Interval (CI) (2-sided) 95%, 80% power, and .5 as the ratio of sample size (group-II/group-I), required total sample size was 126 (84 participants in group-I and 42 in group-II).
The sample size for comparing group-I and group-III: For the outcome of difference between the mean daily weight gain 18 of groups exposed to caffeine daily doses at CI (2-sided) 95%, 80% power, and .4 as the ratio of sample size (group-III/group-I), the required total sample size was 136 (97 participants in group-I and 39 in group-III).
For this retrospective cohort study, we included all the neonates exposed to caffeine therapy during the study period and we had 121 neonates in group-I, 119 in group-II, and 44 in group-III.
Unit Protocol for the Use of Caffeine
Neonates during the study period received licensed neonatal products (Peyona, CC 20 mg/mL solution by Chiesi Ltd.) for IV and oral use. An intravenous loading dose of 20 mg/kg was followed by a once-daily maintenance dose of 5–10 mg/kg CC (equivalent to 2.5–5 mg/kg caffeine base) 24 hours after the loading dose according to unit protocol. Intravenous-CC was switched to the oral route at the achievement of sufficient (>100 mL/kg/day) EN.4,9 The daily dose and duration of CC-therapy were entirely decided by the teams responsible for neonatal care. According to clinical practices, there was the possibility of increasing or reducing the daily CC dose to achieve the clinical outcomes or to minimize the CC-APSEs. Otherwise, for the stable neonates, the dose was adjusted weekly according to the increment in BW with the advancement of age.
We included all the neonates who received CC-therapy, either IV or oral, as the bioavailability of CC is almost similar through oral and IV routes.19,20 Maternal caffeine/tobacco exposure was not an exclusion criterion, and no information was retrieved in this regard. Likewise, neonatal route of nutrition intake (enteral nutrition [EN] vs parenteral nutrition [PN]), type of enteral feed (mother milk vs formula feed), or caloric/protein concentration in formula feed were not considered as exclusion criteria. During the study period, Enfamil® Premature 20 formula feed was used in the unit, where mother/donor milk was not available.
Data Collection
Data were extracted from institutional electronic health records, including antenatal, prenatal, and postnatal clinical characteristics such as gender, GA, and BW. Details about the CC-regimen included route and duration of therapy, loading (mg/kg), daily (mg/kg/day), cumulative doses (mg/kg), and the number of neonates who needed dose reduction or withheld of CC-therapy due to SEs. In addition, data were retrieved about the other concomitant risk factors affecting neonatal WG including exposure to steroids, diuretics, and ibuprofen. Nutritional data included the type and amount of EN and duration of PN. Duration of PN counts for total PN and partial PN days, as in the study center PN is continued till >100 mL/kg/day EN tolerance. 21 Other study variables were obtained from serially recorded weight and clinical progress charts from the nursing notes for CC-APSEs. Laboratory parameters included serum sodium (Na), potassium, and glucose levels. Serum potassium level of 3.5–6.0 mmol/L was considered normal, and hyponatremia was defined as sodium serum levels of <135 mEq/L. Serum glucose greater than 150 mg/dl was considered as hyperglycemia. 22 Any involuntary, rhythmical oscillatory movement of equal amplitude around a fixed axis was defined as tremor, and a resting heart rate of 182 beats was defined as tachycardia.
Intervention and Outcome Measurement
For the current study, MD-WG in g/kg/day was assessed for included neonates during two specified periods: (i) 15–28 days of life (DOL) and (ii) 29–42 DOL. In this study, we used the MD-WG record from the 15th DOL onwards, as up to 14% mean physiological body weight loss is reported in preterm neonates in the first week of life (WOL) and then starts to regain during the second WOL. Based on daily CC-dose/kg of 14th DOL and 28th DOL, all the included neonates were further divided into three groups (Gp) for phase-I and phase-II, respectively.
Gp-I included all the neonates who received 5 mg/kg/day.
Gp-II included neonates who were exposed to >5–7 mg/kg/day.
Gp-III had neonates who were exposed to >7–10 mg/kg/day.
According to clinical practices, there was the possibility of switching neonates from one group to another due to an increment in BW with the advancement of age or dose reduction due to side effects or dose increment to achieve the desired clinical outcomes. During the same phase, neonates facing these scenarios were kept in the initially assigned group but while making groups for phase-II we considered the daily dose of 28th DOL. Therefore, we retrieved data for these SEs, the number of neonates that needed dose adjustments, and the cumulative caffeine dose in each phase. Following the unit practice, experienced neonatal nursing staff measured the neonatal weight from birth to discharge daily and recorded the changes. Less frequent weight measures were done if clinically suggested. 23 Other CC-APSEs were also observed in all groups. Measurement of serum CC-concentrations was not performed in this study. 24
Statistical Analysis
Data were analyzed using STATA version 17.0. To evaluate the effects of different CC daily dose regimens, Gp-II and Gp-III were separately compared with the standard dose group. Descriptive statistics were computed for categorical variables and presented as frequencies with percentages. A Chi-square test was performed to compare the percentage differences between the study groups. Quantitative variables were presented as mean ± SD and median (IQR). T-test was performed for comparison of two group means, whereas the Wilcoxon rank-sum test was performed for non-parametric testing. A P-value <.05 was considered statistically significant.
Ethical Approval and Consent to Participate
Before performing this study, ethical approval was taken from the institutional ethical committee of Aga Khan University Hospital (ERC # 2019-2111-5600), and informed consent from a parent or guardian for participants was waived by ERC as all the data were collected retrospectively.
Results
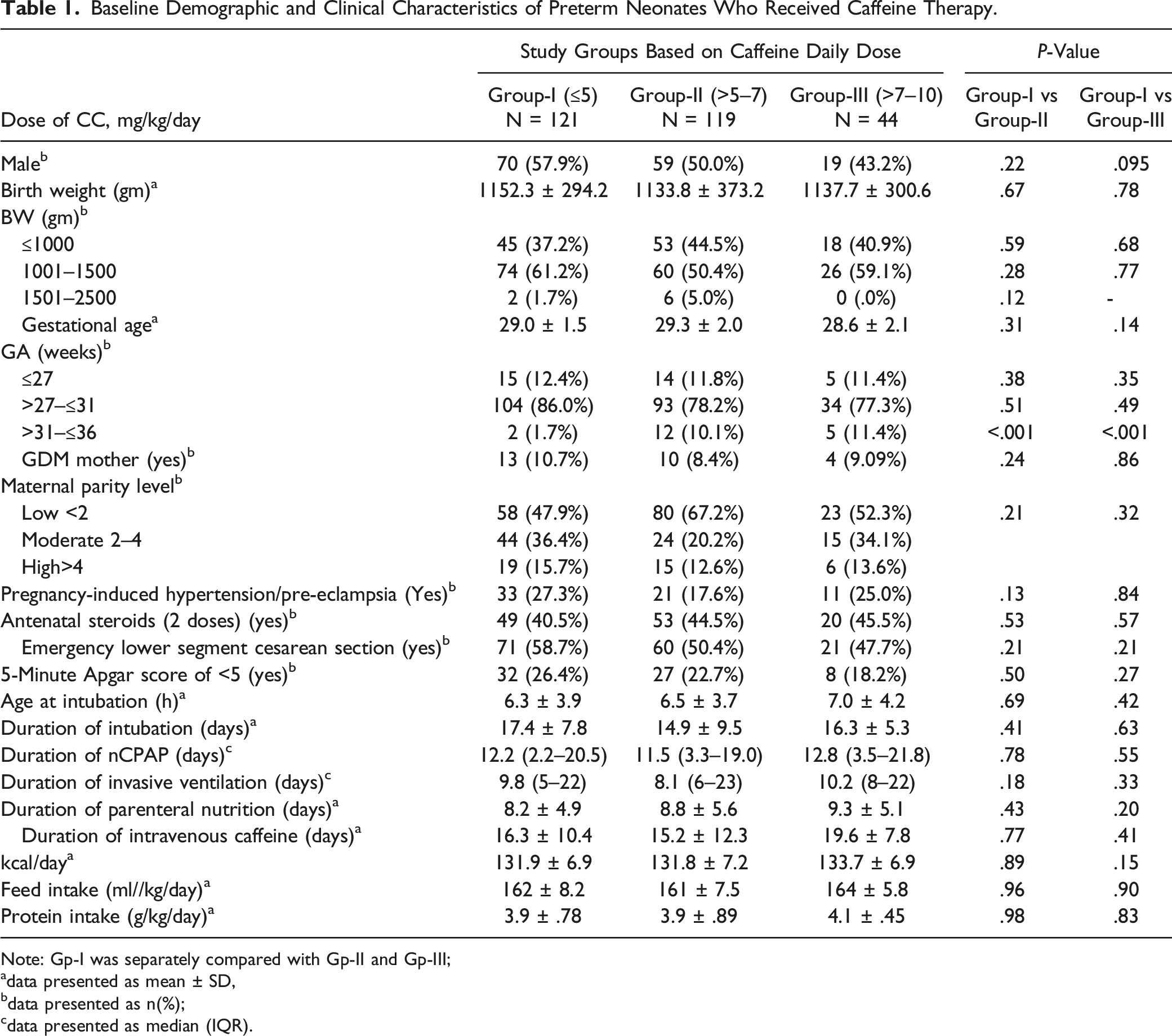
During the study period, a total of 527 preterm neonates received caffeine. Applying exclusion criteria, a total of 243 neonates were excluded and finally, 284 neonates were included in the study (Figure 1). Based on daily CC-dose/kg of 14th DOL, during phase-I 121 neonates received caffeine 5 mg/kg/day (Gp-I), 119 neonates received >5–7 mg/kg/day (Gp-II), and 44 neonates were on >7–10 mg/kg/day (Gp-III). The same 284 participants were followed in the second phase. In this phase based on the daily CC-dose/kg of 28th DOL 183 neonates fell in Gp-I, 73 in group-II, and 28 neonates made Gp-III. Gp-II and Gp-III were separately compared with Gp-I. The demographic and clinical characteristics of all study participants are comparable and statistically no significant differences were found (Table 1). Flow chart depicting recruitment of cohort initially and grouping based on daily CC-dose in two periods (CC = caffeine citrate, Gp = Group, DOL = day of life) (note: each participant could have more than one exclusion criterion). Baseline Demographic and Clinical Characteristics of Preterm Neonates Who Received Caffeine Therapy. Note: Gp-I was separately compared with Gp-II and Gp-III; adata presented as mean ± SD, bdata presented as n(%); cdata presented as median (IQR).
Mean Daily Weight Gain of Premature Infants Exposed to Caffeine Citrate at Three Different Doses During Two Distinct Time Frames.
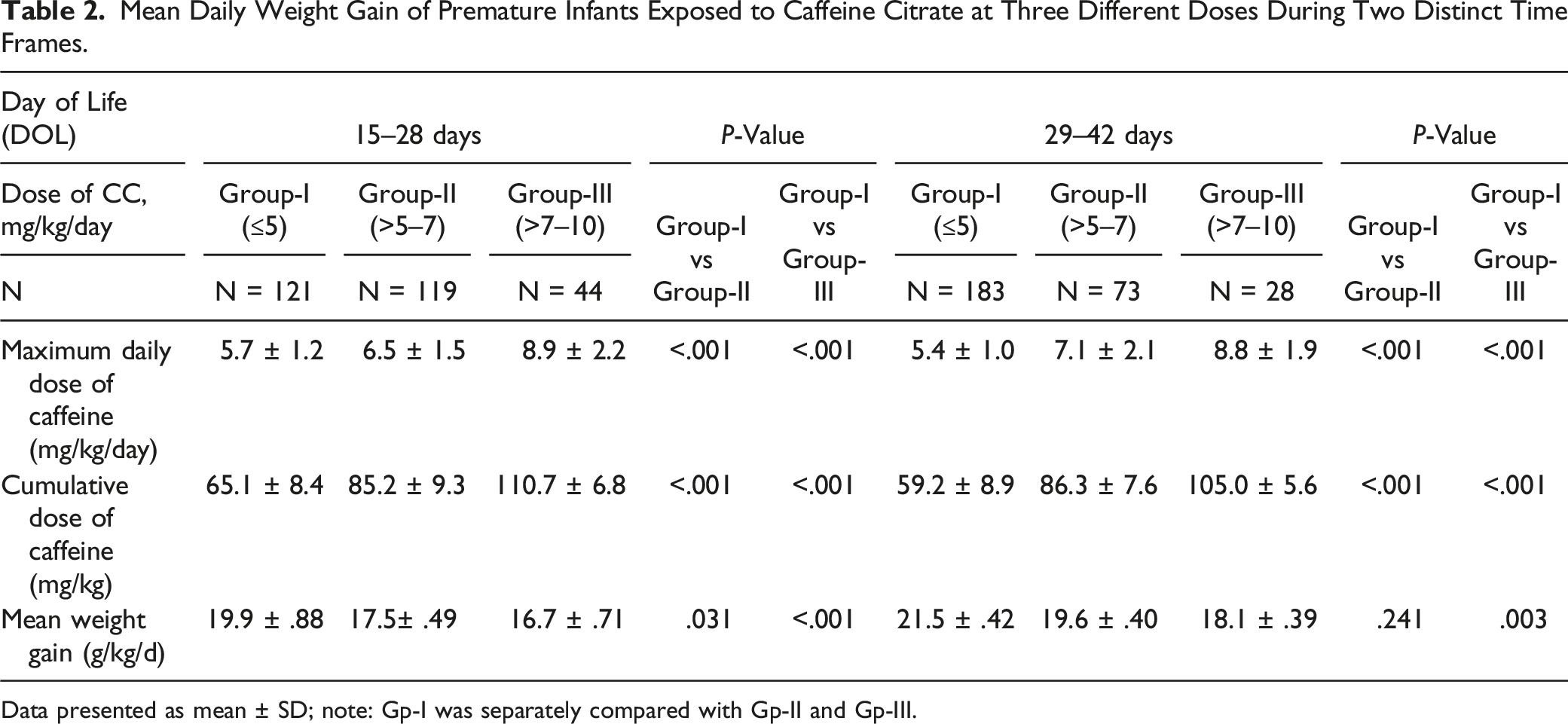
Data presented as mean ± SD; note: Gp-I was separately compared with Gp-II and Gp-III.

Mean daily weight gain of neonates in three different caffeine daily dose regimens during 15–28 days of life (DOL = days of life).

Mean daily weight gain of neonates in three different caffeine daily dose regimens during 29–42 days of life (DOL = days of life).
Frequency of Caffeine-Associated Potential Side Effects in Study Neonates During Two Distinct Time Frames (15–28 and 29–42 days of life).

Data presented as n (%); note: Gp-I was separately compared with Gp-II and Gp-III. All the bold p values present statistically significant difference.
During 29–42 DOL, CC-APSEs were comparable between Gp-II and Gp-I. However, a significantly higher number of neonates in Gp-III reported tachycardia (P = .043), tachypnea (P = .005), hyponatremia (P = .011), polyuria (.009), and feed intolerance (P= <.001) (Table 3). In phase-I and phase-II, no dose reduction was required in Gp-I. During this period, 5/73 neonates of Gp-II and 11/28 neonates of Gp-III needed dose reduction due to CC-APSEs. Not a single participant’s daily caffeine dose was withheld in any phase.
Clinical Outcomes in Daily Caffeine Dose-Based Neonatal Groups.

Hospital stay = NICU stay + step-down unit stay; note: Gp-I was separately compared with Gp-II and Gp-III; NEC = necrotizing enterocolitis.
IVH = intraventricular hemorrhage; ROP = retinopathy of prematurity; BPD = bronchopulmonary dysplasia.
aData presented as n (%).
bData presented as mean ± SD
Discussion
Our study suggests a clinically and statistically significant lower MD-WG in neonates treated with higher daily and cumulative CC-doses. Neonates exposed to a higher dose of >5 mg/kg/d reported lower MD-WG not only during the 15–28 DOL but also during 29–42 DOL. But the major finding in our study is that the neonates exposed to 5–7 mg/kg/d initially had lower MD-WG, but during 29–42 DOL their MD-WG was comparable with neonates exposed to a standard daily dose of 5 mg/kg/day. On the other hand, significantly lower MD-WG was observed in neonates exposed to >7 mg/kg/d till the sixth WOL than in neonates exposed to standard doses.
Previous recent studies reported initial higher daily weight loss in caffeine-treated premature neonates through the first 21 DOL and then a gradual reduction in daily weight loss in subsequent DOL.4,25 However, in our study, the higher daily and cumulative CC-doses were linked with lower MD-WG till 6th WOL in neonates who received > 7 mg/kg/day. Maybe the exclusion of more sick and premature neonates in this study could explain this finding. Similar results are reported in another recent study, 18 but they compared only two dose regimens that are 5 and 10 mg/kg/d.
The recommended daily CC-dose ranges from 5 to 10 mg/kg/d, which gives clinicians a wide range to prescribe for LBW and VLBW neonates. This wide range, however, needs to be evaluated in terms of its useful and undesirable effects on these vulnerable populations. To the best of our knowledge, this is the first study that compared two different high-dose caffeine regimens with standard daily dose regimes in preterm neonates. Previous studies have shown similar respiratory advantages from 2.5 to 5 mg/kg/d of caffeine base and therefore throughout higher maintenance daily dose is debatable.4,18,25 A prospective cohort study proposed that earlier initiation of CC-therapy reduces the incidence of IVH and invasive ventilation requirements. 26 In addition, a recent randomized trial (RT) preferred the use of twice-daily caffeine dosage. 27 Another trial suggested that a higher daily CC-dose regimen possibly decreases the risk of EF. 28 Though, a more recent RT has not demonstrated earlier extubation from earlier CC-therapy initiation. 29
A European trial reported about twenty-one CC-APSEs in premature neonates. 30 The CC-APSEs of the current study are the same as reported in previous studies, including tremors, vomiting, hyperglycemia, and hypokalemia. 31 Other SEs involve tachycardia, tachypnea, hypertension, 32 increased urinary output, and 33 sodium and calcium excretion. 34 It is reported that the onset of CC-APSEs normally takes place at higher doses leading to higher serum concentration. 35
Romagnoli et al 36 tested the frequency of CC-APSEs of two different dose regimens for preventing idiopathic AOP in premature neonates. They reported a greater number of neonates exposed to higher daily maintenance doses who experienced CC-APSEs including vomiting, feed intolerance, regurgitation, hyperglycemia, and tachycardia. A higher daily CC-dose could promote a faster CC-metabolism into theophylline and other methylxanthine metabolites. These were transient manifestations and mostly reverted or resolved during CC-therapy in the following weeks. Our results are similar and even more specific in terms of daily dose. In our study, most of the SEs resolved in subsequent weeks in neonates exposed to ⩽ 7 mg/kg/day, but neonates exposed to a higher dose of >7 mg/kg/d experienced several SEs till the sixth WOL.
Currently recommended daily CC-doses expose premature neonates to a significantly higher caffeine dose than at any other age. A daily maintenance CC-dose of 5–10 mg/kg is frequently given to neonates born at 25–32 weeks gestation and have a body weight of 500–1500 g. The CARE and other recent studies demonstrated that this caffeine amount is more than the amount received from dietary sources during the antenatal period.7,37 According to a recent systematic review that highlighted the dose–response relationship between caffeine exposure in pregnancy and suboptimal fetal WG, results are suggestive of causation and do not indicate a threshold effect. 38
Caffeine has psychoactive properties, thus influencing the sleep cycle and WG pattern through its somatic effects by reducing the overall time and worsening the quality of sleep. 39 Along with these effects, dose-dependent SEs and their intensity and duration can affect the overall clinical outcomes. These observations advocate the vigilant use of CC in premature neonates with a customized regimen. Our study aimed to analyze the clinical advantages of CC based on different dose regimens including length of NICU and hospital stays. Higher daily dose exposure not only restrained the WG but also worsened the clinical condition by exposing the neonates to undesirable toxic effects, resulting in longer NICU and hospital stays.
Dose-dependent polyuria, hyponatremia, and feed intolerance in neonates exposed to higher daily doses are other major contributing factors for suboptimal WG. A study established a dose–response relationship between CC-doses and electrolyte concentrations in serum and urine. 40 The author stated a substantially higher sodium (Na) excretion accompanying diuresis at higher caffeine doses. The probable causes of caffeine-induced natriuresis are caffeine A1-adenosine receptor antagonism and proximal tubular Na-reabsorption inhibition.41-43 Secondly, through caffeine A2-adenosine receptor antagonism and reduced distal Na-reabsorption.19,44 However, the underlying mechanisms are still unexplained. The mean MD-WG during the 15–28 DOL is higher than the 29–42 DOL. This might be related to the fact that most of the neonates received optimal nutrition through total parenteral nutrition (PN) or partial PN during 15–28 DOL. However, during the study duration, we did not have breast milk fortifier practices in our NICU.
Caffeine is a relatively safer drug to treat AOP in preterm neonates which may make practitioners less cautious about individualizing the caffeine regimen, thus prescribing unnecessary higher doses for a prolonged period resulting in possible SEs.21,35 We need to explore all recent respiratory support developments including nasal high-flow humidified O2-therapy or low-flow systems, continuous positive nasal pressure accommodated with synchronization/backup breaths, neonatal postural adjustment, and advancements in neonatal care procedures, to circumvent unnecessary dependence on CC-usage. Schmidt et al reported that CC-therapy used for treating AOP was not related to significantly reducing the collective rate of motor, behavioral, and academic impairments nonetheless had an association to reduce the risk of motor impairment. 45 Recent literature supports the positive role of CC-therapy in managing AOP and might be associated with reducing incidences of chronic lung disease, however, may not be to the extent it is practiced. 46
Some inherent limitations were there in the present study because of its retrospective observational nature and not an RT to make it more conclusive. The selection of CC-dose for any neonate was the clinicians’ decision ranging from 5 to 10 mg/kg/d, and there was a likelihood of treating sicker neonates with higher doses, who were not responding to the standard dose. There was no practice of testing serum CC-concentrations in our NICU. Although the MD-WG estimation was obtained from daily weight measurement, in a few neonates, thrice-weekly weight was measured due to some clinical limitation; however, this was reflected across all neonatal groups. Some CC-APSEs such as hyperglycemia, tachycardia, tachypnea, and vomiting could be due to underlying neonatal clinical conditions in the ICU.
Despite a few limitations, our study has several strengths. This is the first study that evaluated the caffeine dose–response relationship and compared two different high-dose caffeine regimens with standard daily CC-dose regimens in preterm neonates and evaluated the effect on MD-WG and onset of CC-APSEs. During the study period, all the eligible neonates were included in the study. Hence, with a large number of participants study was completed on the same neonates in 15–28 DOL and 29–42 DOL. During the study period, no major changes took place in the NICU practice guidelines. All the participants received similar feeding regimens. Days on PN and total caloric intake were comparable in all groups. Throughout the study period, the same caffeine pharmaceutical brand was used for intravenous and oral dosages for all participants. The first two WOL were excluded for all participants when the possibility of relatively physiological weight loss is greater. Neonates were observed from the third WOL onwards, which is the more stable and growing phase, hence increasing more clinical significance and relevance to the results of our study.
Conclusion
Exposure to higher daily and cumulative doses of caffeine in this study cohort is associated with lower postnatal weight gain in preterm neonates than standard daily doses may be due to its catabolic effects in higher doses. Neonates exposed to 5–7 mg/kg/d initially had lower MD-WG, but during 29–42 DOL their MD-WG was comparable with neonates exposed to a standard daily dose of 5 mg/kg/day. On the other hand, the MD-WG in neonates exposed to >7 mg/kg/d was significantly lower till the sixth WOL than in neonates exposed to standard doses.
This study also concludes that neonates exposed to higher doses have more frequently experienced potentially undesirable SEs and they persist for several weeks, a contributing factor for lower weight gain and longer hospital stay. Our results warrant a vigilant approach from neonatal practitioners for the use of higher caffeine doses in the preterm neonate. Further RCTs are needed for the validation of these outcomes and to establish the most effective and safest caffeine regimens for premature neonates.
Footnotes
Acknowledgments
Authors would like to acknowledge the support of all the neonatal faculty and team for facilitating. We are grateful to Abdul Moiz Hussain for his expert guidance and facilitation of manuscript edits and submission.
Author Contributions
GA has substantial contributions to conceptualizing and designing the project. GA, IH, MK, AA, FN, SR, and WAK were involved in data collection and data entry. GA wrote the manuscript. KH and MSS were involved in supervising the project, interpreting data, and substantively revising the manuscript. GA, IH, MK, AA, and SR analyzed the data. GA, KH, and MSS completed the final draft. All authors were equally involved in manuscript writing and critical revising.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
