Abstract
This review explores how emerging technologies are reshaping musculoskeletal radiology. It examines recent advances where computer-based algorithms assist in interpreting complex imaging data, aiming to improve diagnostic accuracy and streamline clinical workflows. The discussion centres on practical applications, including automated lesion detection, fracture identification and the quantitative evaluation of degenerative conditions. Drawing on a range of clinical studies and technological evaluations, the review highlights the potential benefits of these tools while also addressing the challenges of data integrity, regulatory compliance and the need for comprehensive training among radiology professionals.
The analysis emphasises that while many of these systems demonstrate significant promise in reducing human error and increasing efficiency, their integration into everyday practice must be approached with caution. Key hurdles include ensuring reliable data quality and balancing technological capabilities with established radiological methods. The review advocates for a measured adoption strategy that combines technological innovation with the expertise of seasoned radiologists.
By assessing both the transformative potential and the inherent limitations of current AI applications, this article provides a balanced perspective on future directions in musculoskeletal imaging. The overall goal is to encourage further collaboration among clinicians, engineers and policymakers to foster innovations that enhance patient care without compromising safety or diagnostic standards.
Keywords
Introduction
Musculoskeletal disorders represent one of the most common and disabling groups of conditions encountered in clinical practice. In particular chronic conditions such as osteoarthritis, rheumatoid arthritis are among the most burdensome disorders, accounting for an increasing number of DALYs, and thus consequently accounting for an increasing burden on scan reads.[1]
With the growing number of cross-sectional images acquired in ever shorter periods of time, the workload for musculoskeletal radiologists is rapidly increasing without a corresponding increase in the number of musculoskeletal radiologists with a high level of expertise available to interpret those images in an efficient manner. In the last two decades, there has been an explosive growth in AI and machine learning (ML), in radiology in general, rendering them with capabilities to assess the nuances involved with image interpretation, previously only available to a radiologist’s reasoning mind. These tools have the potential to enhance diagnostic accuracy, reduce interobserver variability and streamline workflow—benefits that are particularly relevant given the complexity of musculoskeletal disorders.[2,3]
A seminal narrative review by Gitto et al. (2024)[4] has underscored the transformative potential of AI in musculoskeletal imaging. Their work provides a comprehensive overview of emerging applications and highlights how these innovations can be tailored to address the unique challenges posed by musculoskeletal disorders. Building on such insights, this article adopts a modality-wise approach to evaluate the current state of AI applications in musculoskeletal imaging. The review while covering all radiology modalities, provides a commentary on the current applications of AI in musculoskeletal radiology, while also elucidating upon the technical challenges that remain to be addressed. While this is an ever-evolving field, with the algorithms of yesterday, already becoming outmoded and paving the way for further more sophisticated algorithms, our review intends to provide a snapshot of the current capabilities of AI algorithms in musculoskeletal imaging.
Radiography: The First-line Modality in Musculoskeletal Disorders
Radiography continues to serve as the primary imaging modality for the evaluation of musculoskeletal disorders due to its low cost, wide availability and rapid acquisition. It is frequently the first step in diagnosing fractures, degenerative changes, such as joint space narrowing, and osteophyte formation. However, interpreting radiographs can be challenging because of overlapping structures, subtle pathological changes and variability in image quality.
AI-based computer-aided detection (CAD) systems have emerged as valuable adjuncts in radiographic analysis. Recent studies indicate that deep learning algorithms can effectively detect fractures, early signs of osteoarthritis and calcific deposits, even in cases where lesions are subtle or obscured by complex anatomy.[5] For example, in the evaluation of wrist and pelvic fractures, AI models have demonstrated sensitivity levels that rival those of experienced radiologists.[6]
Furthermore, AI applications have been used to assist in the quantification of joint space narrowing and osteophyte formation, which are essential for grading knee osteoarthritis severity. These applications not only provide objective metrics but also help in monitoring disease progression and evaluating treatment efficacy.[7]
In paediatric populations, AI-driven bone age assessment is another promising application that can expedite diagnosis while reducing subjectivity.[8] Bone age assessments have become a major target of the ML community.
AI has shown a remarkable performance in the evaluation of orthopaedic implants and implant-related complications, including identification of the body part of interest, orthopaedic implant used, its characterisation, and finally its position and if any implant-related complications.[9]
Despite these advances, radiographic imaging is subject to significant variability. Differences in patient positioning, exposure settings and the quality of X-ray machines can affect the consistency of images, thus impacting the performance of AI systems.[10] Standardisation of radiographic protocols is therefore critical to ensure that AI tools maintain high diagnostic accuracy across diverse clinical settings. However, while understanding that this may not be an executable step due to the diverse nature of centres for healthcare provisions, a way around could be to train the algorithms on multi-centric, transnational data. This would ensure that the algorithm ‘sees’ and ‘learns’ from all possible combinations of data and this would help avoid bias for or against a certain manufacturer, ethnicity and age.
Computed Tomography: Enhancing Detail in Complex Cases
Computed tomography (CT) imaging is indispensable for the detailed evaluation of bony structures and is especially useful in cases of complex fractures, spinal deformities and osseous tumours. The inherent three-dimensional nature of CT allows for precise visualisation of anatomical details, which is crucial in surgical planning and trauma assessment.
Recent advances in AI have significantly improved the automation of CT image processing. Deep learning models are now capable of performing automatic segmentation of bones, enabling rapid identification of fracture lines and dislocations.[11] For instance, AI algorithms have been successfully applied to segment vertebrae and detect compression fractures, providing vital information that can influence treatment decisions in patients with spinal trauma or osteoporosis.[11]
Moreover, quantitative CT analysis powered by AI is playing an important role in the opportunistic screening for osteoporosis. By analysing volumetric bone mineral density from routine CT scans, these systems can identify individuals at risk for fragility fractures without the need for dedicated bone density tests.[12] This opportunistic approach could lead to earlier intervention and improved patient outcomes.
Efforts to reduce patient radiation exposure during diagnostic imaging continue unabated. Machine learning emerges as a promising tool for cutting CT radiation doses. Techniques based on ML work in a way that echoes methods used to quicken MRI scans—they strive to generate high-quality images from either a smaller volume of raw data or data that might be of lower quality (for instance, data that is noisier when acquired at lower tube currents.
CT-based AI applications also extend to the evaluation of bone tumours. In the context of musculoskeletal neoplasms, AI tools assist in delineating tumour margins, characterising lesion heterogeneity and even predicting tumour aggressiveness based on quantitative imaging features. However, the integration of AI into CT imaging is not without challenges. The high radiation dose associated with CT remains a concern, particularly when repeated imaging is required for monitoring chronic conditions. Additionally, the large volume of data produced by CT scans demands robust computational infrastructure to ensure efficient processing and storage.[13]
Magnetic Resonance Imaging (MRI): A Window into Soft Tissue Pathologies
MRI has become the gold standard when it comes to evaluating soft tissue structures. Its exceptional contrast resolution and the ability to acquire images in multiple planes, make it especially valuable for detecting ligament injuries, muscle tears, cartilage degeneration and bone marrow abnormalities. MRI’s unique strength lies in its capacity to visualise fine soft tissue details, that often go unnoticed with other imaging modalities.
In recent years, AI has been making waves in the MRI space, particularly in scan acquisition. The appeal is obvious: AI can dramatically reduce scan time and streamline the workflow. However, there is a caveat, unlike traditional imaging methods such as parallel imaging and compressed sensing, where problems such as aliasing and noise are easy to spot, the AI-enhanced scans may actually pass the first quality check, looking as they are supposed to; this despite them misrepresenting the actual anatomy.[14] Another area undergoing rapid change is low-field-strength MRI. Newer systems, such as the 0.55-T whole-body scanner, are opening up possibilities for more cost-effective and widely accessible imaging. This technology integrates several modern advancements—ranging from updated signal transmission and reception methods, superior radiofrequency coils, versatile surface coils, to near-neutral simultaneous multislice acceleration, as well as deep learning-based reconstruction techniques. These combined innovations enhance signal strength markedly, enabling quicker scans with higher image quality and offering the potential to eventually match the performance of higher-strength systems such as 1.5T or 3T.[14]
Looking ahead, further improvements are expected from deep learning-driven image reconstruction. These advancements aim to better mitigate metal-induced artifacts in MRI, with improvements in correcting acceleration errors, sharpening images and reducing distortions caused by implants. Additionally, the diagnostic scope of musculoskeletal MRI could be broadened significantly through deep learning methods that allow for advanced image contrast transformations. Such techniques include the generation of synthetic CT-like images from MRI datasets, the creation of new MRI contrast profiles along with quantitative mapping, and the production of fat-suppressed images from scans that were originally obtained without fat suppression.[14]
AI applications in MRI have rapidly evolved, particularly in the areas of segmentation and classification. Deep learning models are being trained to differentiate between healthy and pathological tissues, identify ligament ruptures and quantify cartilage loss in osteoarthritis.[15] For example, automated segmentation of knee cartilage using AI has shown promise in providing accurate measurements of cartilage thickness and volume, which are key indicators of osteoarthritis progression.[16]
Another critical application of AI in MRI is in the realm of radiomics. Radiomics involves extracting a large number of quantitative features from MRI images, which can be used to build predictive models for disease progression and treatment response. In musculoskeletal oncology, radiomics has been explored for its potential to differentiate benign from malignant soft tissue tumours, offering an additional layer of diagnostic precision.[17]
Similarly, AI is beginning to show promise in analysing dynamic contrast-enhanced MRI to assess inflammatory activity in conditions like rheumatoid arthritis—an area that could one day enable more targeted, personalised therapies.[18]
In musculoskeletal oncology, most AI-based radiomic studies focused on prediction of diagnosis—such as benign versus malignant tumour discrimination or tumour grading—and outcome—such as therapy response, recurrence and survival.
Despite these advancements, MRI-based AI systems face unique challenges. Variability in scanner hardware, imaging protocols and field strengths can lead to inconsistent image quality and limit the generalisability of AI models. This variability necessitates the development of robust models that can adapt to different imaging conditions and emphasises the need for standardised acquisition protocols.[19] In addition, while the promise of radiomics is significant, the integration of such complex quantitative data into routine clinical practice requires careful validation and clear guidelines for interpretation.
Ultrasound: Real-time Applications in Musculoskeletal Disorders
Ultrasound is widely used in musculoskeletal imaging due to its ability to provide real-time, dynamic assessments without ionising radiation. It is particularly valuable in evaluating tendon injuries, muscle tears, joint effusions and guiding interventional procedures such as injections.
The integration of AI in ultrasound imaging has focused on overcoming the inherent operator dependency and variability in image acquisition. Recent AI tools have been developed for real-time segmentation and classification of musculoskeletal structures, which can assist less experienced operators in obtaining diagnostically useful images.[20] For example, AI algorithms that automatically delineate tendon margins or identify signs of tendinopathy have shown potential in reducing interobserver variability and improving diagnostic confidence.[21]
In addition to structural assessment, AI is being applied to elastography—a technique used to evaluate tissue stiffness—which can be particularly useful in conditions like muscle injuries or inflammatory disorders. By quantifying elasticity parameters, AI-enhanced elastography may aid in differentiating between acute and chronic soft tissue injuries, thereby influencing treatment planning.[22]
Nevertheless, ultrasound imaging remains a challenging domain for AI. Variability in operator technique, patient anatomy and the inherent limitations of ultrasound image resolution can all impact algorithm performance. Efforts to standardise image acquisition protocols and develop adaptive AI models are essential for maximising the utility of AI in ultrasound applications.[23]
Challenges and Considerations for Clinical Integration
While AI applications offer significant promise in improving the diagnosis and management of musculoskeletal disorders, several challenges [Figure 1] must be addressed for their successful clinical integration:
Data Quality and Annotation: High-quality, annotated datasets are critical for training robust AI models. The diversity of musculoskeletal disorders and the variability in imaging protocols across institutions make data curation a complex task.[24] Collaborative initiatives and multicentre data sharing are essential to overcome these limitations. Standardisation and Generalisability: AI models must be trained and validated on diverse datasets to ensure they are applicable across different populations and imaging systems. Standardised imaging protocols can help improve model consistency and clinical reliability.[25] Interpretability and Clinician Trust: The ‘black-box’ nature of many AI models can hinder clinician acceptance. Providing explainable outputs and integrating decision support that complements radiologist expertise are crucial for fostering trust in these technologies.[26] Regulatory and Ethical Issues: The integration of AI in clinical practice raises important regulatory and ethical considerations. Ensuring data privacy, addressing liability concerns and establishing clear guidelines for AI performance are necessary steps before widespread adoption.[27] The liability for harm caused by a medical AI error could fall on the radiologist, the health care provider or the AI system developer. The radiologist may be responsible for any misdiagnosis or missed diagnosis linked to the AI’s use, the health care provider could be liable for injuries stemming from inadequate staff training or oversight, and the developer might face liability for any system defects or for not supplying sufficient usage instructions. Training and Workflow Integration: Successful implementation of AI requires changes in clinical workflow and radiologist training. Education programs that emphasise both the technical and clinical aspects of AI can help bridge the gap between technology and patient care.[28]
Challenges and considerations for clinical integration

Future Directions
Looking to the future, several avenues show promise for further integrating AI into the imaging of musculoskeletal disorders:
Multimodal Data Integration: Combining imaging data from radiography, CT, MRI and ultrasound with clinical and laboratory data can yield more comprehensive diagnostic models. Multimodal integration holds the potential to personalise diagnosis and treatment strategies in musculoskeletal disorders.[2,29] Federated Learning Approaches: By enabling AI models to learn from distributed datasets without compromising patient privacy, federated learning may help address data scarcity and improve model generalisability across institutions.[25] Real-Time and Interactive AI Systems: Advances in hardware and software will likely facilitate the development of real-time AI systems, particularly in ultrasound imaging. Such systems could provide immediate feedback during examinations, improving image quality and diagnostic yield.[21,27] Enhanced Radiomics and Predictive Analytics: Continued research into radiomics could lead to robust predictive models for disease progression, treatment response and even risk stratification in conditions like osteoarthritis and musculoskeletal tumours.[17] This could pave the way for more personalised treatment plans. Regulatory Frameworks and Clinical Guidelines: Establishing standardised regulatory pathways and clinical guidelines for the use of AI in musculoskeletal imaging is essential. As regulatory bodies develop frameworks tailored to these technologies, clinicians will have clearer benchmarks for safe and effective implementation.[27] Human-AI Collaboration: Rather than viewing AI as a competitor, the future of musculoskeletal imaging lies in collaborative systems where AI augments the capabilities of radiologists. This synergy can lead to more accurate diagnoses, improved workflow efficiency and ultimately better patient outcomes.[26,29]
Conclusion
The application of AI in musculoskeletal disorders imaging represents a significant advancement in the field. Across radiography, CT, MRI and ultrasound, AI technologies are increasingly being harnessed to enhance the detection, segmentation and classification of pathological features [Figure 2]. As demonstrated in recent literature—including the narrative review by Gitto et al. (2024)[4]—these computational tools offer the potential to improve diagnostic accuracy, streamline workflows and support personalised treatment strategies for a broad range of musculoskeletal conditions.
Modality-based summary of artificial intelligence in MSK imaging
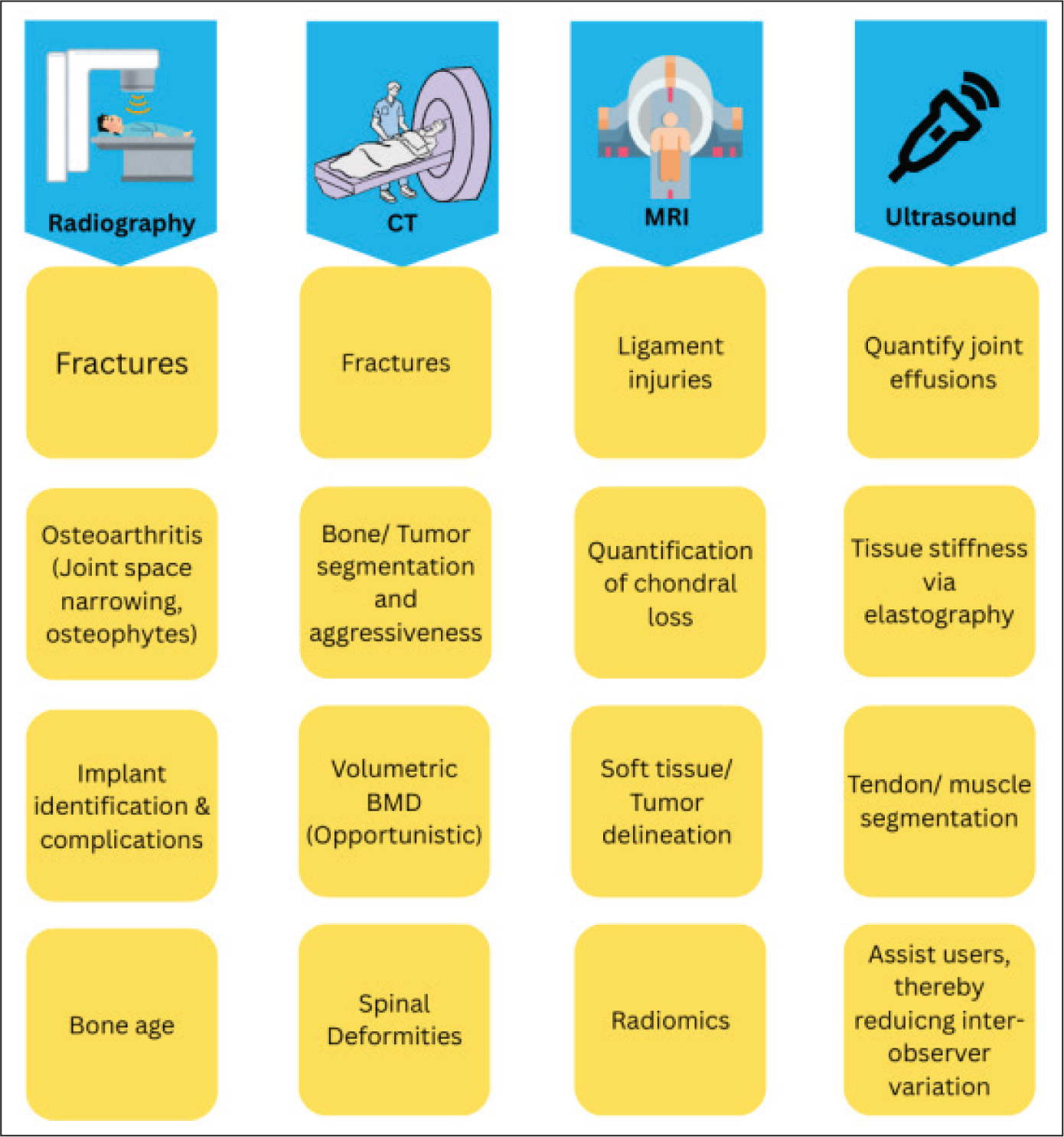
However, several challenges remain. Variability in imaging protocols, data quality issues, the need for standardisation and the requirement for explainable AI outputs must be addressed to ensure these tools are clinically reliable. Moreover, ethical, regulatory and educational hurdles will need careful navigation before AI can be fully integrated into routine clinical practice.
The future of musculoskeletal imaging is likely to be shaped by a collaborative approach where AI serves as an indispensable adjunct to radiologist expertise rather than a replacement. With continued innovation, rigorous validation and the development of robust regulatory frameworks, AI is poised to play a pivotal role in advancing the diagnosis and management of musculoskeletal disorders.
Ultimately, as research and clinical trials continue to refine these technologies, the integration of AI into musculoskeletal imaging will lead to a more precise, efficient and patient-centred approach to healthcare. By embracing these advances, the radiology community can ensure that the benefits of AI are fully realised while maintaining the highest standards of clinical care.
Footnotes
Acknowledgements
The authors would like to thank my colleagues, juniors and seniors for their support and contribution to this work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional Ethical Committee Approval Number
No ethical committee was involved in the review article.
Informed consent
No informed consent was needed as this is a review article.
Credit author statement
Nimisha Lohiya: Conceptualisation, manuscript writing and final approval.
Pranav Ajmera: Imaging evaluation and figure preparation and final approval.
Sanjay Desai: Literature review, manuscript writing and final approval.
Vijay Kumar Jain: Literature review and final approval.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Use of artificial intelligence
Artificial Intelligence tools were not used in the generation or analysis of data in this manuscript.
